
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Matteo Tassetto | + 2017 word(s) | 2017 | 2021-07-05 08:52:48 | | | |
| 2 | Vivi Li | Meta information modification | 2017 | 2021-07-13 11:19:03 | | |
Video Upload Options
Diabetes is a worldwide emergency. Its chronic complications impose a heavy burden on patients, health systems, and on society as a whole. Diabetic retinopathy is one of the most common and serious complications of diabetes, and an established risk factor for blindness in adults. Over 15 years of investigation led to the identification of vascular endothelial growth factor (VEGF) as a main pathogenic factor in diabetic retinopathy and to the introduction of highly effective anti-VEGF-based therapies, such as the monoclonal antibody bevacizumab or its fragment ranibizumab, which helped to prevent diabetes-related blindness in millions of patients. Recently, a pathogenic role for uncontrolled increases in the extracellular ATP concentration (eATP) and for overactivation of the purinergic receptor P2X7 (P2X7R) has been suggested. The P2X7R is an eATP-gated plasma membrane channel expressed in multiple tissues and organs, with a pleiotropic function in inflammation, immunity, cancer, and hormone and growth factor release. P2X7R stimulation or overexpression positively regulate the secretion and buildup of VEGF, thus promoting neo-angiogenesis in a wide variety of disease processes.
1. Introduction
2. Role of the P2X7R in Diabetic Retinopathy and Its Potential Role as a Therapeutic Target
2.1. The P2X7 Receptor as a Target to Restore the Blood–Retinal Barrier and Reduce Inflammation
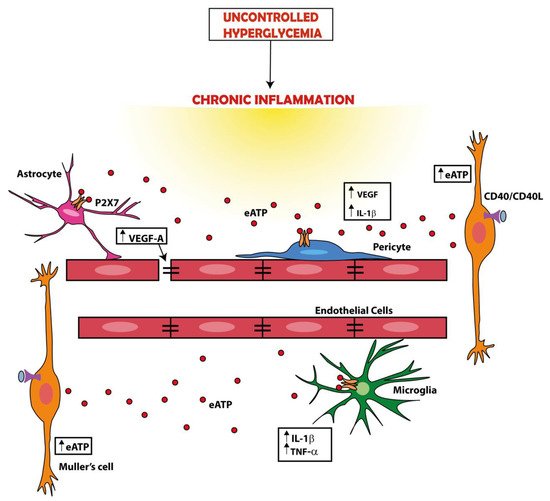
2.2. Targeting the P2X7 Receptor to Reduce Neoangiogenesis
2.3. P2X7 Receptor and Neuronal Damage
2.4. Other Potential Roles of the P2X7 Receptor as a Therapeutic Target in Diabetic Retinopathy
References
- Fletcher, E.L.; Wang, A.Y.; Jobling, A.I.; Rutar, M.V.; Greferath, U.; Gu, B.; Vessey, K.A. Targeting P2X7 receptors as a means for treating retinal disease. Drug Discov. Today 2019, 24, 1598–1605.
- Solini, A.; Novak, I. Role of the P2X7 receptor in the pathogenesis of type 2 diabetes and its microvascular complications. Curr. Opin. Pharmacol. 2019, 47, 75–81.
- Argaw, A.T.; Gurfein, B.T.; Zhang, Y.; Zameer, A.; John, G.R. VEGF-mediated disruption of endothelial CLN-5 promotes blood-brain barrier breakdown. Proc. Natl. Acad. Sci. USA 2009, 106, 1977–1982.
- Platania, C.B.M.; Giurdanella, G.; Di Paola, L.; Leggio, G.M.; Drago, F.; Salomone, S.; Bucolo, C. P2X7 receptor antagonism: Implications in diabetic retinopathy. Biochem. Pharmacol. 2017, 138, 130–139.
- Portillo, J.-A.C.; Greene, J.A.; Okenka, G.; Miao, Y.; Sheibani, N.; Kern, T.S.; Subauste, C.S. CD40 promotes the development of early diabetic retinopathy in mice. Diabetologia 2014, 57, 2222–2231.
- Portillo, J.-A.C.; Corcino, Y.L.; Miao, Y.; Tang, J.; Sheibani, N.; Kern, T.S.; Dubyak, G.; Subauste, C.S. CD40 in Retinal Müller Cells Induces P2X7-Dependent Cytokine Expression in Macrophages/Microglia in Diabetic Mice and Development of Early Experimental Diabetic Retinopathy. Diabetes 2017, 66, 483–493.
- Subauste, C.S. The CD40-ATP-P2X7 Receptor Pathway: Cell to Cell Cross-Talk to Promote Inflammation and Programmed Cell Death of Endothelial Cells. Front. Immunol. 2019, 10.
- Sugiyama, T.; Kobayashi, M.; Kawamura, H.; Li, Q.; Puro, D.G. Enhancement of P2X7-Induced Pore Formation and Apoptosis: An Early Effect of Diabetes on the Retinal Microvasculature. Investig. Opthalmol. Vis. Sci. 2004, 45, 1026–1032.
- Klaassen, I.; Van Noorden, C.J.; Schlingemann, R.O. Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions. Prog. Retin. Eye Res. 2013, 34, 19–48.
- Platania, C.B.M.; Lazzara, F.; Fidilio, A.; Fresta, C.G.; Conti, F.; Giurdanella, G.; Leggio, G.M.; Salomone, S.; Drago, F.; Bucolo, C. Blood-retinal barrier protection against high glucose damage: The role of P2X7 receptor. Biochem. Pharmacol. 2019, 168, 249–258.
- Fresta, C.G.; Caruso, G.; Fidilio, A.; Platania, C.B.M.; Musso, N.; Caraci, F.; Drago, F.; Bucolo, C. Dihydrotanshinone, a Natural Diterpenoid, Preserves Blood-Retinal Barrier Integrity via P2X7 Receptor. Int. J. Mol. Sci. 2020, 21, 9305.
- Amoroso, F.S.; Capece, M.; Rotondo, A.; Cangelosi, D.; Ferracin, M.; Franceschini, A.; Raffaghello, L.; Pistoia, V.; Varesio, L.; Adinolfi, E. The P2X7 receptor is a key modulator of the PI3K/GSK3β/VEGF signaling network: Evidence in experimental neuroblastoma. Oncogene 2015, 34, 5240–5251.
- Amoroso, F.S.; Falzoni, S.; Adinolfi, E.; Ferrari, D.; Di Virgilio, F. The P2X7 receptor is a key modulator of aerobic glycolysis. Cell Death Dis. 2012, 3, e370.
- Adinolfi, E.; Raffaghello, L.; Giuliani, A.L.; Cavazzini, L.; Capece, M.; Chiozzi, P.; Bianchi, G.; Kroemer, G.; Pistoia, V.; Di Virgilio, F. Expression of P2X7 Receptor Increases In Vivo Tumor Growth. Cancer Res. 2012, 72, 2957–2969.
- Clapp, C.; Diaz-Lezama, N.; Adan-Castro, E.; Ramirez-Hernandez, G.; Moreno-Carranza, B.; Sarti, A.C.; Falzoni, S.; Solini, A.; Di Virgilio, F. Pharmacological blockade of the P2X7 receptor reverses retinal damage in a rat model of type 1 diabetes. Acta Diabetol. 2019, 56, 1031–1036.
- Resta, V.; Novelli, E.; Vozzi, G.; Scarpa, C.; Caleo, M.; Ahluwalia, A.; Solini, A.; Santini, E.; Parisi, V.; Di Virgilio, F.; et al. Acute retinal ganglion cell injury caused by intraocular pressure spikes is mediated by endogenous extracellular ATP. Eur. J. Neurosci. 2007, 25, 2741–2754.
- Sugiyama, T. Role of P2X7 receptors in neuronal death in the retina. Neural Regen. Res. 2014, 9, 579–581.
- Dvoriantchikova, G.; Ivanov, D.; Barakat, D.; Grinberg, A.; Wen, R.; Slepak, V.Z.; Shestopalov, V.I. Genetic Ablation of Pannexin1 Protects Retinal Neurons from Ischemic Injury. PLoS ONE 2012, 7, e31991.
- Fowler, B.J.; Gelfand, B.D.; Kim, Y.; Kerur, N.; Tarallo, V.; Hirano, Y.; Amarnath, S.; Fowler, D.H.; Radwan, M.; Young, M.T.; et al. Nucleoside reverse transcriptase inhibitors possess intrinsic anti-inflammatory activity. Science 2014, 346, 1000–1003.
- Pavlou, S.; Augustine, J.; Cunning, R.; Harkin, K.; Stitt, A.W.; Xu, H.; Chen, M. Attenuating Diabetic Vascular and Neuronal Defects by Targeting P2rx7. Int. J. Mol. Sci. 2019, 20, 2101.
- Yang, K.; Liu, J.; Zhang, X.; Ren, Z.; Gao, L.; Wang, Y.; Lin, W.; Ma, X.; Hao, M.; Kuang, H. H3 Relaxin Alleviates Migration, Apoptosis and Pyroptosis Through P2X7R-Mediated Nucleotide Binding Oligomerization Domain-Like Receptor Protein 3 Inflammasome Activation in Retinopathy Induced by Hyperglycemia. Front. Pharmacol. 2020, 11.
- Marques-Da-Silva, C.; Chaves, M.M.; Castro, N.; Coutinho-Silva, R.; Guimaraes, M.Z. Colchicine inhibits cationic dye uptake induced by ATP in P2X2 and P2X7 receptor-expressing cells: Implications for its therapeutic action. Br. J. Pharmacol. 2011, 163, 912–926.





