
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tina Maver | + 3148 word(s) | 3148 | 2021-07-09 09:58:30 | | | |
| 2 | Conner Chen | Meta information modification | 3148 | 2021-07-13 04:13:28 | | |
Video Upload Options
Astrocytes are key cells in the central nervous system. They are involved in many functions under physiological and pathological conditions. Primary cultures of astrocytes represent an important object for basic and translational neuroscience research, especially for in vitro cell models. Astrocyte cultures for functional cell models are most commonly isolated from rodent brains, because they are easily accessible and grow rapidly. Tissue engineering and biomaterial development represent a promising alternative to animal testing and provide an ideal opportunity to develop and test various biomaterials as scaffolds for purposes such as cell ingrowth and tissue repair.
1. The Sources of Human Astrocytes and Their Importance for Cell Models in Biomaterial Research
Astrocytes are critical cells in the central nervous system [1][2]. They are involved in many vital functions under physiological and pathological conditions. Primary cultures of astrocytes represent an essential target for basic and translational neuroscience research, especially for in vitro cell models [3].
Primary cell cultures of astrocytes have been isolated from a variety of sources, usually rodent brains. Despite the abundance of experimental plants, there are significant differences between human and rodent astrocytes [4][5]. Human astrocytes differ from rodent cells in many ways. They are larger and more structurally complex and exhibit differences in calcium signaling. They also contact many more synapses than their rodent counterparts. In addition, humans and primates have astrocyte types that are not found in rodents [6][7]. These differences are the main reason for advancing and improving isolation methods and pushing forward studies on primary adult human astrocytes. On the other hand, human cells have the advantage of more accurately representing the environment of the central nervous system. Therefore, these cells are often used to study human central nervous system physiology and metabolic processes that would not otherwise be possible in vivo [8][9][10].
There have not been many reports on the isolation of human astrocytes [11][12]. The significant advantages of the in vitro culture of human astrocytes include the ability to perform biochemical analyses of individual identified cell types, reduced cell complexity (compared with whole-brain), the ability to fully control the cellular environment, the imaging and electrophysiology of individual cells, co-culturing, and manipulation of gene expression [13]. Mature astrocytes contain well-established connections and are more organized than newborn tissue, which is plastic and unstable in response to stimuli. Consequently, human astrocyte cultures respond more reliably and may help clarify the role of astrocytes in in vivo situations [6][14][15]. Therefore, it is more beneficial to study these cells separately under in vitro conditions [13][16][17].
Astrocytes can be isolated from different parts of neonatal or adult brains [6][18]. However, human tissue samples are usually obtained from neonatal brains. In rare cases, adult patients have also been designated as donors, mainly those who have undergone craniotomy for trauma, tumor, or epilepsy surgery, or have undergone surgery for various hemorrhages, such as arteriovenous malformations, intracerebral hematomas, and aneurysms. Postmortem specimens have also been collected [11][19]. When establishing cultures of human astrocytes, it is necessary to obtain a healthy part of the brain. If tumor cells are involved, tumor tissue can be used, as in various gliomas. When harvesting tissue, it is important to consider the pathology and the conditions under which the cells were harvested. For example, the brain substance surrounding the hematoma (i.e., the penumbra) is often not suitable for culture and isolation because it is necrotic or sub-vital, resulting in lower cell yield and quality [11][20].
Compared to adult astrocytes, neonatal astrocytes begin to show signs of ageing relatively late, after three to six months in culture. Initially, they grow and proliferate at a fast rate. Adult astrocytes have very limited proliferative activity in vitro and therefore do not remain in culture for long. They cannot be readily subcultured. These cultures are therefore of limited use. In addition, the differentiation of neonatal cells may be incomplete because they lack normal cell partners or differentiation signals [21]. They are also considered to be more activated than adult brain cells, which are mature [22]. This is particularly important when cell culture is used to study neurodegenerative diseases. The experimental results obtained from neonatal cells cannot be directly transferred to adult cells [23][24]. Therefore, adult brain-derived neuroglial cells form a useful and convenient model for experiments, as their pathophysiological mechanisms cannot be equally studied in neonatal culture. In recent years, isolation and culturing techniques have enabled better isolation capabilities for adult astrocytes [22][23]. In adult brains, tissue for isolation is much more readily available, both in quantity and frequency of collection, compared to neonatal brains. Neonatal brains can be obtained from fetuses, usually at 9 to 22 weeks of age, from elective abortions [25]. Furthermore, the timing of tissue collection is problematic and strict collaboration between the clinical department and the laboratory is necessary. Not all fetuses are suitable for isolation. Only brain-shaped fetuses collected after the surgical procedure of vacuum aspiration can be used. Tissue from fetuses that have undergone abortion after a medical procedure is not suitable, because the pharmaceutical agents used to kill the fetus can alter the viability of the cells and thus hinder the development of the primary culture [25][26]. On the other hand, adult tissue is readily available, as there are many more surgical procedures that can make tissue available for experimentation.
Transport to the cell laboratory is significant and the time and mode may vary. It is usually longer for neonatal brain samples collected during abortions. The transport time is typically less than two hours [27]. In adults, on the other hand, the tissue is usually more stable, as it is collected during resections and biopsies and reaches the laboratory much more quickly [28][29].
In the isolation and purification of astrocytes, one of the major limitations is that culture methods for mature astrocytes are not yet fully developed [30]. The technique developed by McCarthy and de Vellis in 1980, in which astrocytes were prepared from a neonatal rodent brain, has long served as a prototype for astrocyte isolation [31]. Much of our knowledge of astrocytes, synaptogenesis, and their role in neuronal survival comes from studies of these cells [32][33]. Although cultures isolated using this technique have increased our understanding of astrocytic function, they have a number of drawbacks. One is that these cultures select for populations of cells that express astrocytic markers but appear to have an immature or reactive phenotype. During the isolation process, only a small percentage of cells survive and proliferate, and the population is not prospective. The cells eventually stratify into two populations, the astrocytes and the oligodendrocytes. The latter grow on top of the astrocytes in culture and can be separated, leaving astrocytes in culture [30][34]. This technique is sophisticated and is used to select cells in neonatal animals that can survive and proliferate in vitro. It was originally developed for the isolation of rodent astrocytes and, with some modifications, has been applied to their human counterparts.
Immunopanning, on the other hand, is a new technique that allows the prospective isolation of astrocytes (Figure 1) [35]. This involves direct cell selection without multiple steps, allowing a representative population of astrocytes to be selected from an entire cell suspension. Immunopanning is a very gentle procedure and yields viable cells that can be cultured in serum-free medium containing heparin-binding EGF-like growth factor (HBEGF) at the end of the preparation. This factor is critical for the survival of astrocytes in culture, and in which they can be maintained for longer than two weeks. In contrast, astrocytes isolated by other techniques have a shorter lifespan in vitro [35][36][37].
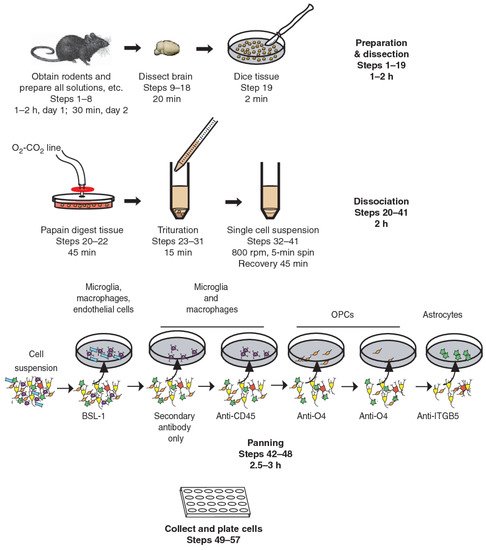
Figure 1. Immunopanning of astrocytes with anti-ITGB5 [38].
Astrocytes isolated by immunopanning preserve their gene profiles and phenotypic characteristics. They promote neuronal survival and synapse formation and function in vitro [39]. Studies on immunopanned astrocytes revealed some of their important properties, such as findings on their phagocytic functions and the need for trophic support for their survival. These results indicate the importance of immunopanning-based purification of astrocytes for the study of their biology and function, and this isolation technique has been used for other species, not only rodents [30][34][35].
The progress made in recent decades in the isolation of astrocytes is also due to new technological achievements, laboratory techniques, and new surgical capabilities, including neuroendoscopic and neuronavigational methods and instruments. These allow tissue harvesting from different sites of the central nervous system, with minimal possible morbidity, and that is less invasive, more frequent, and with less tissue damage, which contributes to a higher cell yield when isolated in the laboratory [40][41]. There are numerous neurosurgical approaches used in clinical practice that provide a welcome source of both healthy and diseased brain tissue [42][43]. Over the past few decades, clinicians, researchers and patients have benefited from surgical techniques that optimize surgical outcomes, help limit the potential for neurologic morbidity, and increase the ability to obtain an ideal tissue sample, which forms the basis for successful cell isolation [38][42][44].
2. The Need for Tissue Engineering for Future Astrocyte Implantation
Spinal cord and brain injuries remain an important clinical problem for treatment, and functionally successful neuronal regeneration has not yet been achieved. Traumatic brain and spinal cord injury, stroke, and neurodegenerative diseases are a major case of morbidity and mortality worldwide and present a treatment challenge for clinicians and rehabilitation specialists [45][46]. Traumatic brain injury induces functional deficits due to axonal destruction and formation of cystic cavitations, scar tissue, and physical lacunae. In addition, reactive oxygen species are produced, leading to massive neuronal death, which worsens the course of secondary injury and may lead to disability and death [47][48]. In spinal cord injury, the mechanisms of neuronal damage are similar. In stroke, primary ischemic changes are followed by edema and altered vascular permeability, resulting in secondary brain injury, which further worsens the clinical condition and prognosis of patients. In such insults, not only neurons are affected, but also many other cells in the brain and spinal cord, such as astrocytes, microglia, oligodendrocytes, endothelial cells, and pericytes, which enter these pathological circuits [49][50]. As the central nervous system has a limited capacity to counteract the damage and dysfunction of axonal pathways and replace the lost neurons, these diseases often result in permanent neurological deficits [45][51][52]. Many attempts have been made to limit the extent of neuronal damage and to promote the recovery of the damaged brain and spinal cord areas, including limiting penumbra and promoting the regeneration of central nervous system cells. In in vitro research using biomaterials, cell-based approaches have been widely used in attempts to overcome the effects of glial scarring and replenish the lost cells, mainly neurons. The idea of bioengineering is production of cell carriers for implantation of axon growth promoting glia, and for supportive integration with host cells [47][50][53].
During the development of the central nervous system, neuronal migration and axonal expansion occur along corridors formed by other cells, particularly astrocytes [54]. These pathways or corridors are called living scaffolds. In regenerative medicine of the central nervous system, the goal is to simulate such scaffolds with artificial implants made of tissue-engineered biomaterials populated by one or more specific cell types, to promote neuron regeneration, enable targeted reconstruction, replace neural circuitry, and limit glial scar formation [55]. Such living scaffolds are constructed in vitro and can be implanted in vivo (Figure 2) to present cell adhesion molecules and neurotrophic and chemotactic signals that actively regulate neural migration and axonal growth during regenerative processes [54][56].
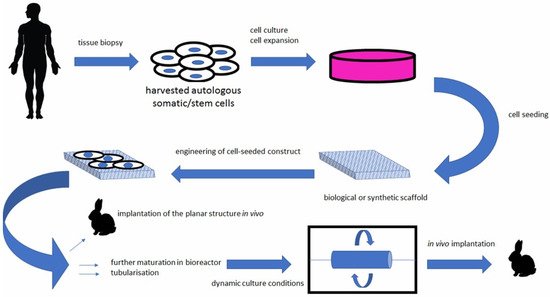
Figure 2. Bioengineered scaffolds as substitutes for grafts [57].
A variety of bioengineered scaffolds have been developed to promote axonal regeneration in damaged neural tissue. An ideal scaffold for such purposes does not yet exist. Among the most popular materials in bioengineering are collagens [46][53]. Biomaterials made from collagen have numerous advantages over many synthetic polymers, including having stability, non-toxic degradation products, biocompatibility, and the induction of minimal foreign body reaction [58].
Glial cells contained within engineered living scaffolds modulate numerous developmental mechanisms in the brain. Such tissue-engineered scaffolds often contain astrocytes, being the most numerous cells in the central nervous system. For example, hydrogel scaffolds may be coated with an extracellular collagen matrix and populated with astrocytes. Such scaffolds induce astrocytes to grow and orient into dense three-dimensional bundles of bipolar, longitudinally aligned projections. These aligned astrocyte networks provide a favorable substrate for neuronal attachment and neurite outgrowth. Moreover, these bio-manufactured scaffolds maintain their integrity and orientation even when detached from hydrogels, making them suitable for implantation into the central nervous system [51][54][59][60][61]. The neuroanatomical properties and potential of regenerative mechanisms may lead to a new class of engineered glial-based living scaffolds that can guide and promote the growth and expansion of immature neurons during migration and aid in axonal pathfinding through otherwise non-permissive environments. This can potentially mitigate the effects of neuronal degeneration that are so common in central nervous system injury and disease. So far, living scaffolds are only experimental and limited to use in animal research. A biomimetic, self-assembling peptide hydrogel has been tested in rats as stabilizing scaffolds and a vehicle for grafted cells following brain and spinal cord injury. They have been shown to be a suitable cell and drug delivery system in the injured central nervous system [54][56][62][63].
Biotechnological bridging materials include animal collagen, not just hydrogel. Experiments with glial cells and neurons have been performed on microstructured porcine collagen scaffolds. These contain densely packed and highly oriented channels that form a tri-structure that facilitates cell attachment, proliferation, and migration, making them suitable for tissue culture. Such biocompatible scaffolds that promote glial cell attachment and migration will be essential for future repair strategies for injured neural tissue [47][49][60].
Astrocytes are also known to be an essential component of the blood–brain barrier. They play an important role in its maintenance and repair, regulate amino acid, ion, and water homeostasis, and produce proteins to reinforce the blood–brain barrier [51][59]. Many in vitro models of the human blood–brain barrier integrate astrocytes and combine them with other cells, such as endothelial cells. Tight junctions of endothelial cells are essential in the blood–brain barrier models, indicating that these in vitro conditions are suitable for establishing important features of astrocyte and endothelial cell functions in the brain. The in vitro models include hydrogels that serve as platforms for the study of the blood–brain barrier and new methods for tumor treatment, limiting the damage of stroke and promoting the uptake of therapeutic agents into the central nervous system. To date, such experimental blood–brain barrier models have not been implanted or integrated in humans [52].
3. Astrocytes Derived from Stem Cells and Their Potential in Tissue Engineering
Astrocytes have important functions in normal and pathological states. Activated astrocytes are present in almost all neurological diseases. Most studies to date have been performed on animal experimental systems, mostly because of ease of access, maintenance in culture, and because of difficulty in obtaining primary human astrocytes. Due to interspecies differences, human astrocytes are preferred in experiments. In addition to the isolation of human astrocytes from various sources, as described above, induced pluripotent stem cells (iPSCs) have also been a focus of research. Almost all types of neural cells, including neurons, astrocytes, oligodendrocytes, neural stem cells, pericytes, and microglia, can be derived from iPSCs by considering developmental principles [64][65]. The iPSC-derived neural cells are a valuable tool for research, such as developing new therapeutic strategies, elucidation neurological disease mechanisms, and studying the physiology of the nervous system in health and disease [65][66].
The source of iPSCs are somatic cells that can be reprogrammed with transcription factors, such as SOX2, OCT4, KLF4, and MYC. This transformation technique has allowed the study of a variety of diseases and generated the concept of “disease in a dish”, which allows the modeling of disease phenotypes in a tissue culture dish [66][67]. The iPSCs are pluripotent like embryonic stem cells. They can be efficiently expanded and induced into all cell types in the human body under appropriate culture conditions. The iPSCs provide an unlimited source for subsequent differentiation into cell types of interest. Since they are reprogrammed from human somatic cells, concerns about species differences associated with animal models can be avoided. They also retain their original genomic features, such as chromosomal abnormalities and gene mutations. They remain intact after differentiation and can be used to study the effects of genomic defects on cellular functions, which is particularly valuable in drug development research (Figure 3) [66][68].
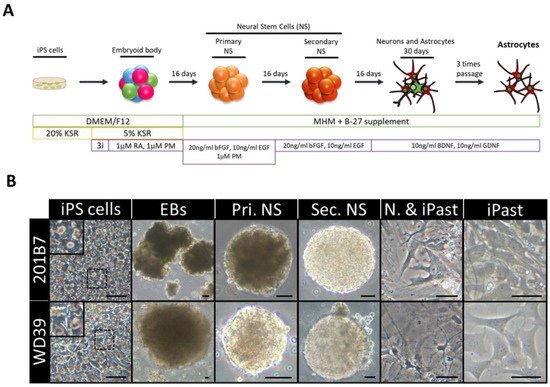
Figure 3. Induction of Human Astrocytes (iPasts). (A) Design of the human iPast induction protocol under serum free condition. Abbreviations: DMEM/F12: Dulbecco’s Modified Eagle Medium: Nutrient Mixture F-12; MHM: Media Hormone mix; B-27™: optimized serum-free supplement; KSR: KnockOut™ Serum Replacement; 3i: 3 µM CHIR99021, 3 µM 431542, 3 µM Dorsomorphine; RA: Retinoic acid; PM: Purmorphamine; bFGF (FGF-2): Basic fibroblast growth factor; EGF: Epidermal Growth Factor; BDNF: Brain-Derived Neurotrophic Factor; GDNF: Glial cell line-Derived Neurotrophic Factor. (B) Representative images of cells at each step of iPast induction process from the two iPSC control lines 201B7 and WD39. Abbreviations: EBs: Embryoid Bodies; Pri. NS: primary neurospheres; Sec. NS: secondary neurospheres; N. & iPasts: Neurons and iPasts; iPasts: human iPSC-derived astrocytes. The cells in the insets at iPSC stage are higher magnifications of cells in dashed boxes and indicative of good quality iPSCs with clear perinuclear halos. Scale bars: 50 µm (20 µm for iPast stage). [69].
Human astrocytes derived from human iPSCs exhibit typical characteristics of physiological astrocytes and respond to various stimuli. Therefore, they are a suitable experimental model for studying astrocyte functions and reactivation under healthy and pathological conditions of the human nervous system [70]. Astrocyte differentiation techniques using human iPSCs are far from simple. They are lengthy and complex and the cells require the use of serum containing factors known to promote glial differentiation from neural precursor cells [71][72]. This is the reason why the isolation of astrocytes from human brain is still attractive and preferred by many laboratories over the iPSC technique. Compared to neurons, astrocytes are formed at a much later stage of embryonic development. This means that the differentiation process of astrocytes from iPSCs takes longer than that of neurons. Reports in the literature on the differentiation time of astrocytes from human iPSC vary and can last between 80 and 180 days [66][73][74].
The use of human iPSC-derived astrocytes in neurological disease modeling dates back to 2012. Early studies on neurodegenerative diseases were based on two-dimensional cell cultures. Although they provided important insights into brain dysfunction at a cellular level, their major limitation was that they did not allow for the proper spatial organization and developmental progression of cells in the brain. In contrast, neural cells derived from human iPSCs allow growth in cerebral organoids, which are self-organized, three-dimensional aggregates with cellular diversity and cytoarchitectures more similar to the human brain. They provide more sophisticated tissue architecture and microenvironmental signals than a traditional two-dimensional system [75][76][77]. Pathological changes in various neurodegenerative diseases are also reflected in astrocytes derived from human iPSCs. For example, iPSC-derived astrocytes in Alzheimer’s disease show a different morphology, with lower complexity and aberrant marker localization, compared to normal astrocytes [78]. The iPSC-derived astrocytes from patients with frontotemporal dementia affect neurons by inducing increased oxidative stress and transcriptional profile changes in previously healthy neurons [79]. Three-dimensional culture systems with hydrogel and iPSC-derived neurons and astrocytes are used to study Rett syndrome [66][80][81].
References
- Bedner, P.; Jabs, R.; Steinhäuser, C. Properties of human astrocytes and NG2 glia. Glia 2020, 68, 756–767.
- Sajin, B.; Steindler, D. Cells on the edge: Boundary astrocytes and neurons. Perspect. Dev. Neurobiol. 1994, 2, 275–289.
- Nimmerjahn, A.; Bergles, D.E. Large-scale recording of astrocyte activity. Curr. Opin. Neurobiol. 2015, 32, 95–106.
- Lange, S.C.; Bak, L.K.; Waagepetersen, H.S.; Schousboe, A.; Norenberg, M.D. Primary Cultures of Astrocytes: Their Value in Understanding Astrocytes in Health and Disease. Neurochem. Res. 2012, 37, 2569–2588.
- Allen, N.J.; Barres, B.A. Glia—More than just brain glue. Nature 2009, 457, 675–677.
- Oberheim, N.A.; Goldman, S.A.; Nedergaard, M. Heterogeneity of astrocytic form and function. In Astrocytes; Springer: Berlin/Heidelberg, Germany, 2012; pp. 23–45.
- Herculano-Houzel, S. The glia/neuron ratio: How it varies uniformly across brain structures and species and what that means for brain physiology and evolution. Glia 2014, 62, 1377–1391.
- Rustenhoven, J.; Park, T.I.; Schweder, P.; Scotter, J.; Correia, J.; Smith, A.M.; Gibbons, H.M.; Oldfield, R.L.; Bergin, P.S.; Mee, E.W. Isolation of highly enriched primary human microglia for functional studies. Sci. Rep. 2016, 6, 1–11.
- Giffard, R.; Ouyang, Y.-B. Cell Culture: Primary Neural Cells; Elsevier: Amsterdam, The Netherlands, 2009.
- Allen, N.J. Astrocyte regulation of synaptic behavior. Annu. Rev. Cell Dev. Biol. 2014, 30, 439–463.
- Kimelberg, H.K.; Nedergaard, M. Functions of astrocytes and their potential as therapeutic targets. Neurotherapeutics 2010, 7, 338–353.
- Gradisnik, L.; Maver, U.; Bosnjak, R.; Velnar, T. Optimised isolation and characterisation of adult human astrocytes from neurotrauma patients. J. Neurosci. Meth. 2020, 341, 108796.
- John, G.R. Investigation of astrocyte–oligodendrocyte interactions in human cultures. In Astrocytes; Springer: Berlin/Heidelberg, Germany, 2012; pp. 401–414.
- Tanti, G.K.; Srivastava, R.; Kalluri, S.R.; Nowak, C.; Hemmer, B. Isolation, culture and functional characterization of glia and endothelial cells from adult pig brain. Front. Cell. Neurosci. 2019, 13, 333.
- Wolf, F.; Kirchhoff, F. Neuroscience. Imaging astrocyte activity. Science 2008, 320, 1597–1599.
- Denis-Donini, S.; Glowinski, J.; Prochiantz, A. Glial heterogeneity may define the three-dimensional shape of mouse mesencephalic dopaminergic neurones. Nature 1984, 307, 641–643.
- Zhang, Y.; Barres, B.A. Astrocyte heterogeneity: An underappreciated topic in neurobiology. Curr. Opin. Neurobiol. 2010, 20, 588–594.
- Bellaver, B.; Souza, D.G.; Souza, D.O.; Quincozes-Santos, A. Hippocampal Astrocyte Cultures from Adult and Aged Rats Reproduce Changes in Glial Functionality Observed in the Aging Brain. Mol. Neurobiol. 2017, 54, 2969–2985.
- Mizee, M.R.; Miedema, S.S.; van der Poel, M.; Schuurman, K.G.; van Strien, M.E.; Melief, J.; Smolders, J.; Hendrickx, D.A.; Heutinck, K.M.; Hamann, J.; et al. Isolation of primary microglia from the human post-mortem brain: Effects of ante- and post-mortem variables. Acta Neuropathol. Commun. 2017, 5, 16.
- Kimelberg, H.K.; Schools, G.P.; Cai, Z.; Zhou, M. Freshly isolated astrocyte (FIA) preparations: A useful single cell system for studying astrocyte properties. J. Neurosci. Res. 2000, 61, 577–587.
- Uliasz, T.F.; Hamby, M.E.; Jackman, N.A.; Hewett, J.A.; Hewett, S.J. Generation of primary astrocyte cultures devoid of contaminating microglia. In Astrocytes; Springer: Berlin/Heidelberg, Germany, 2012; pp. 61–79.
- Welser, J.V.; Milner, R. Derivation of Microglia-Free Astrocyte Cultures from Neural Stem Cells. In Astrocytes; Springer: Berlin/Heidelberg, Germany, 2012; pp. 81–91.
- Eng, L.F.; Ghirnikar, R.S.; Lee, Y.L. Glial fibrillary acidic protein: GFAP-thirty-one years (1969–2000). Neurochem. Res. 2000, 25, 1439–1451.
- Wang, D.D.; Bordey, A. The astrocyte odyssey. Prog. Neurobiol. 2008, 86, 342–367.
- Kim, H.J.; Magrané, J. Isolation and culture of neurons and astrocytes from the mouse brain cortex. In Neurodegeneration; Springer: Berlin/Heidelberg, Germany, 2011; pp. 63–75.
- Mizee, M.R.; van der Poel, M.; Huitinga, I. Purification of cells from fresh human brain tissue: Primary human glial cells. Handb. Clin. Neurol. 2018, 150, 273–283.
- Jakovcevski, I.; Filipovic, R.; Mo, Z.; Rakic, S.; Zecevic, N. Oligodendrocyte development and the onset of myelination in the human fetal brain. Front. Neuroanat. 2009, 3, 5.
- Chaboub, L.S.; Deneen, B. Developmental origins of astrocyte heterogeneity: The final frontier of CNS development. Dev. Neurosci. 2012, 34, 379–388.
- Goldman, S.A.; Kuypers, N.J. How to make an oligodendrocyte. Development 2015, 142, 3983–3995.
- Foo, L.C. Purification and culture of astrocytes. Cold Spring Harb. Protoc. 2013, 2013, top070912.
- Mccarthy, K.D.; De Vellis, J. Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J. Cell Biol. 1980, 85, 890–902.
- Banker, G.A. Trophic interactions between astroglial cells and hippocampal neurons in culture. Science 1980, 209, 809–810.
- Eroglu, C.; Barres, B.A. Regulation of synaptic connectivity by glia. Nature 2010, 468, 223–231.
- Cahoy, J.D.; Emery, B.; Kaushal, A.; Foo, L.C.; Zamanian, J.L.; Christopherson, K.S.; Xing, Y.; Lubischer, J.L.; Krieg, P.A.; Krupenko, S.A. A transcriptome database for astrocytes, neurons, and oligodendrocytes: A new resource for understanding brain development and function. J. Neurosci. 2008, 28, 264–278.
- Foo, L.C. Purification of rat and mouse astrocytes by immunopanning. Cold Spring Harb. Protoc. 2013, 2013, prot074211.
- Barres, B.A.; Silverstein, B.E.; Corey, D.P.; Chun, L.L. Immunological, morphological, and electrophysiological variation among retinal ganglion cells purified by panning. Neuron 1988, 1, 791–803.
- Yang, P.; Hernandez, M.R. Purification of astrocytes from adult human optic nerve heads by immunopanning. Brain Res. Protoc. 2003, 12, 67–76.
- Asthagiri, A.R.; Pouratian, N.; Sherman, J.; Ahmed, G.; Shaffrey, M.E. Advances in brain tumor surgery. Neurol. Clin. 2007, 25, 975–1003.
- Foo, L.C.; Allen, N.J.; Bushong, E.A.; Ventura, P.B.; Chung, W.-S.; Zhou, L.; Cahoy, J.D.; Daneman, R.; Zong, H.; Ellisman, M.H. Development of a method for the purification and culture of rodent astrocytes. Neuron 2011, 71, 799–811.
- Sudhakar, V.; Richardson, R.M. Gene therapy for neurodegenerative diseases. Neurotherapeutics 2019, 16, 166–175.
- Savu, R.; Mohan, D.; Mohan, A.; Moisa, H.; Ciurea, A. A modern minimally invasive transcallosal approach to the third ventricle: A personal experience of 30 patients. Chirurgia 2014, 109, 80–89.
- Tatter, S.B. Neurosurgical management of brain tumors. Neuroimaging Clin. N. Am. 1999, 9, 779–799.
- Teo, C. The concept of minimally invasive neurosurgery. Neurosurg. Clin. N. Am. 2010, 21, 583–584.
- Save, A.V.; Gill, B.J.; D’amico, R.S.; Canoll, P.; Bruce, J.N. Fluorescein-guided resection of gliomas. J. Neurosurg. Sci. 2019, 63, 648–655.
- Montani, L.; Petrinovic, M.M. Targeting axonal regeneration: The growth cone takes the lead. J. Neurosci. 2014, 34, 4443–4444.
- Horner, P.J.; Gage, F.H. Regenerating the damaged central nervous system. Nature 2000, 407, 963–970.
- Möllers, S.; Heschel, I.; Damink, L.H.O.; Schügner, F.; Deumens, R.; Müller, B.; Bozkurt, A.; Nava, J.G.; Noth, J.; Brook, G.A. Cytocompatibility of a novel, longitudinally microstructured collagen scaffold intended for nerve tissue repair. Tissue Eng. Part A 2009, 15, 461–472.
- Qian, F.; Han, Y.; Han, Z.; Zhang, D.; Zhang, L.; Zhao, G.; Li, S.; Jin, G.; Yu, R.; Liu, H. In Situ implantable, post-trauma microenvironment-responsive, ROS Depletion Hydrogels for the treatment of Traumatic brain injury. Biomaterials 2021, 270, 120675.
- Gao, Y.; Yang, Z.; Li, X. Regeneration strategies after the adult mammalian central nervous system injury—Biomaterials. Regen. Biomater. 2016, 3, 115–122.
- Cregg, J.M.; DePaul, M.A.; Filous, A.R.; Lang, B.T.; Tran, A.; Silver, J. Functional regeneration beyond the glial scar. Exp. Neurol. 2014, 253, 197–207.
- Struzyna, L.A.; Harris, J.P.; Katiyar, K.S.; Chen, H.I.; Cullen, D.K. Restoring nervous system structure and function using tissue engineered living scaffolds. Neural Regen. Res. 2015, 10, 679.
- Placone, A.L.; McGuiggan, P.M.; Bergles, D.E.; Guerrero-Cazares, H.; Quiñones-Hinojosa, A.; Searson, P.C. Human astrocytes develop physiological morphology and remain quiescent in a novel 3D matrix. Biomaterials 2015, 42, 134–143.
- Lie, D.C.; Song, H.; Colamarino, S.A.; Ming, G.-l.; Gage, F.H. Neurogenesis in the adult brain: New strategies for central nervous system diseases. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 399–421.
- Katiyar, K.S.; Winter, C.C.; Gordián-Vélez, W.J.; O’Donnell, J.C.; Song, Y.J.; Hernandez, N.S.; Struzyna, L.A.; Cullen, D.K. Three-dimensional tissue engineered aligned astrocyte networks to recapitulate developmental mechanisms and facilitate nervous system regeneration. JoVE J. Vis. Exp. 2018, 131, e55848.
- Silver, J.; Miller, J.H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 2004, 5, 146–156.
- Struzyna, L.A.; Katiyar, K.; Cullen, D.K. Living scaffolds for neuroregeneration. Curr. Opin. Solid State Mater. Sci. 2014, 18, 308–318.
- Culenova, M.; Bakos, D.; Ziaran, S.; Bodnarova, S.; Varga, I.; Danisovic, L. Bioengineered Scaffolds as Substitutes for Grafts for Urethra Reconstruction. Materials 2019, 12, 3449.
- Huebner, E.A.; Strittmatter, S.M. Axon regeneration in the peripheral and central nervous systems. Cell Biol. Axon 2009, 305–360.
- Buffo, A.; Rolando, C.; Ceruti, S. Astrocytes in the damaged brain: Molecular and cellular insights into their reactive response and healing potential. Biochem. Pharmacol. 2010, 79, 77–89.
- Toy, D.; Namgung, U. Role of glial cells in axonal regeneration. Exp. Neurobiol. 2013, 22, 68.
- Wiseman, T.M.; Baron-Heeris, D.; Houwers, I.G.; Keenan, R.; Williams, R.J.; Nisbet, D.R.; Harvey, A.R.; Hodgetts, S.I. Peptide hydrogel scaffold for mesenchymal precursor cells implanted to injured adult rat spinal cord. Tissue Eng. Part A 2020.
- Kaneko, N.; Marín, O.; Koike, M.; Hirota, Y.; Uchiyama, Y.; Wu, J.Y.; Lu, Q.; Tessier-Lavigne, M.; Alvarez-Buylla, A.; Okano, H. New neurons clear the path of astrocytic processes for their rapid migration in the adult brain. Neuron 2010, 67, 213–223.
- David, S.; Aguayo, A.J. Axonal elongation into peripheral nervous system” bridges” after central nervous system injury in adult rats. Science 1981, 214, 931–933.
- Okita, K.; Ichisaka, T.; Yamanaka, S. Generation of germline-competent induced pluripotent stem cells. Nature 2007, 448, 313–317.
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872.
- Li, L.; Chao, J.; Shi, Y. Modeling neurological diseases using iPSC-derived neural cells. Cell Tissue Res. 2018, 371, 143–151.
- Shi, Y.; Inoue, H.; Wu, J.C.; Yamanaka, S. Induced pluripotent stem cell technology: A decade of progress. Nat. Rev. Drug Discov. 2017, 16, 115–130.
- Liu, J.; Nie, H.; Xu, Z.; Niu, X.; Guo, S.; Yin, J.; Guo, F.; Li, G.; Wang, Y.; Zhang, C. The effect of 3D nanofibrous scaffolds on the chondrogenesis of induced pluripotent stem cells and their application in restoration of cartilage defects. PLoS ONE 2014, 9, e111566.
- Leventoux, N.; Morimoto, S.; Imaizumi, K.; Sato, Y.; Takahashi, S.; Mashima, K.; Ishikawa, M.; Sonn, I.; Kondo, T.; Watanabe, H.; et al. Human Astrocytes Model Derived from Induced Pluripotent Stem Cells. Cells 2020, 9, 2680.
- Soubannier, V.; Maussion, G.; Chaineau, M.; Sigutova, V.; Rouleau, G.; Durcan, T.M.; Stifani, S. Characterization of human iPSC-derived astrocytes with potential for disease modeling and drug discovery. Neurosci. Lett. 2020, 731, 135028.
- Suga, M.; Kondo, T.; Inoue, H. Modeling neurological disorders with human pluripotent stem cell-derived astrocytes. Int. J. Mol. Sci. 2019, 20, 3862.
- Roybon, L.; Lamas, N.J.; Garcia-Diaz, A.; Yang, E.J.; Sattler, R.; Jackson-Lewis, V.; Kim, Y.A.; Kachel, C.A.; Rothstein, J.D.; Przedborski, S. Human stem cell-derived spinal cord astrocytes with defined mature or reactive phenotypes. Cell Rep. 2013, 4, 1035–1048.
- Yuan, S.H.; Martin, J.; Elia, J.; Flippin, J.; Paramban, R.I.; Hefferan, M.P.; Vidal, J.G.; Mu, Y.; Killian, R.L.; Israel, M.A. Cell-surface marker signatures for the isolation of neural stem cells, glia and neurons derived from human pluripotent stem cells. PLoS ONE 2011, 6, e17540.
- Ren, B.; Dunaevsky, A. Modeling Neurodevelopmental and Neuropsychiatric Diseases with Astrocytes Derived from Human-Induced Pluripotent Stem Cells. Int. J. Mol. Sci. 2021, 22, 1692.
- Lancaster, M.A.; Renner, M.; Martin, C.-A.; Wenzel, D.; Bicknell, L.S.; Hurles, M.E.; Homfray, T.; Penninger, J.M.; Jackson, A.P.; Knoblich, J.A. Cerebral organoids model human brain development and microcephaly. Nature 2013, 501, 373–379.
- Qian, X.; Song, H.; Ming, G.-L. Brain organoids: Advances, applications and challenges. Development 2019, 146, dev166074.
- Jones, V.C.; Atkinson-Dell, R.; Verkhratsky, A.; Mohamet, L. Aberrant iPSC-derived human astrocytes in Alzheimer’s disease. Cell Death Dis. 2017, 8, e2696.
- Hallmann, A.-L.; Araúzo-Bravo, M.J.; Mavrommatis, L.; Ehrlich, M.; Röpke, A.; Brockhaus, J.; Missler, M.; Sterneckert, J.; Schöler, H.R.; Kuhlmann, T. Astrocyte pathology in a human neural stem cell model of frontotemporal dementia caused by mutant TAU protein. Sci. Rep. 2017, 7, 1–10.
- Zhang, Z.-N.; Freitas, B.C.; Qian, H.; Lux, J.; Acab, A.; Trujillo, C.A.; Herai, R.H.; Huu, V.A.N.; Wen, J.H.; Joshi-Barr, S. Layered hydrogels accelerate iPSC-derived neuronal maturation and reveal migration defects caused by MeCP2 dysfunction. Proc. Natl. Acad. Sci. USA 2016, 113, 3185–3190.
- Julia, T.; Wang, M.; Pimenova, A.A.; Bowles, K.R.; Hartley, B.J.; Lacin, E.; Machlovi, S.I.; Abdelaal, R.; Karch, C.M.; Phatnani, H. An efficient platform for astrocyte differentiation from human induced pluripotent stem cells. Stem Cell Rep. 2017, 9, 600–614.
- Hughes, C.S.; Postovit, L.M.; Lajoie, G.A. Matrigel: A complex protein mixture required for optimal growth of cell culture. Proteomics 2010, 10, 1886–1890.





