
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yutaka Tsutsumi | + 1270 word(s) | 1270 | 2021-06-24 05:02:42 | | | |
| 2 | Enzi Gong | Meta information modification | 1270 | 2021-07-07 03:49:06 | | | | |
| 3 | Enzi Gong | -6 word(s) | 1264 | 2021-07-07 03:49:35 | | | | |
| 4 | Enzi Gong | Meta information modification | 1264 | 2021-07-07 11:12:07 | | |
Video Upload Options
In daily diagnostic pathology services, chromogenic immunostaining using formalin-fixed, paraffin-embedded (FFPE) sections is indispensable to help in the histopathological diagnosis fundamentally based on hematoxylin and eosin (H&E) staining. Differential diagnoses are determined first, and appropriate immunohistochemical markers for the diagnoses are then selected. The expression of lymphocyte surface markers, hormones and tumor markers contributes to the functional classification of tumors. The degree of malignancy can be analyzed by immunostaining for p53 and Ki-67 (MIB-1), a cell proliferation marker, in a variety of malignancies including carcinomas of the oral cavity, colon, and breast.
1. Overview
Chromogenic immunohistochemistry (immunostaining using an enzyme-labeled probe) is an essential histochemical technique for analyzing pathogenesis and making a histopathological diagnosis in routine pathology services. In neoplastic lesions, immunohistochemistry allows the study of specific clinical and biological features such as histogenesis, behavioral characteristics, therapeutic targets, and prognostic biomarkers. The needs for appropriate and reproducible methods of immunostaining are prompted by technical development and refinement, commercial availability of a variety of antibodies, advanced applicability of immunohistochemical markers, accelerated analysis of clinicopathological correlations, progress in molecular targeted therapy, and the expectation of advanced histopathological diagnosis. However, immunostaining does have various pitfalls and caveats. Pathologists should learn from previous mistakes and failures and from results indicating false positivity and false negativity. The present review article describes various devices, technical hints, and trouble-shooting guides to keep in mind when performing immunostaining.
2. Techniques of Immunohistochemistry
Techniques of immunohistochemistry (IHC) or immunostaining have already been established [1][2][3][4][5][6][7][8]. Particularly for diagnostic IHC, chromogenic IHC (immunostaining using enzyme-labeled probes) is commonly utilized. Measures and tools to prevent technical artifacts and appropriate trouble-shooting tips are needed. The present review article provides an overview of technical aspects, knowhow, pitfalls, and trouble-shooting guides for routinely performing chromogenic immunostaining, especially for experienced personnel with immunohistochemical expertise. It should be emphasized that all data presented herein were obtained by the author and/or the author’s colleagues.
3. The Need for Chromogenic Immunostaining in Diagnostic Pathology
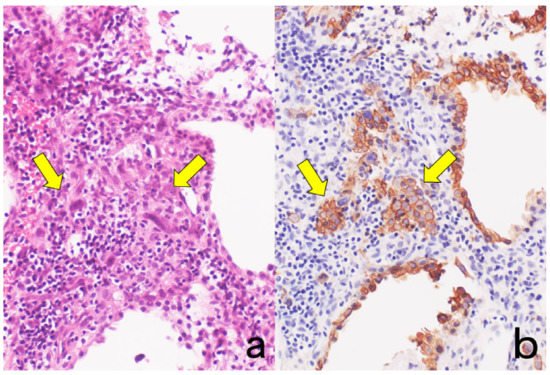
4. Selection of Methodology for Chromogenic Immunostaining
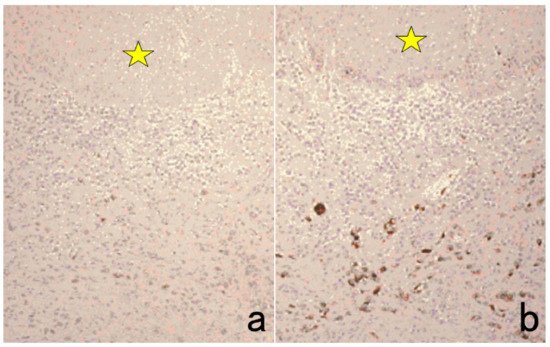
5. Conclusions
References
- Nagura, H.; Osamura, R.Y.; Tsutsumi, Y. Watanabe/Nakane’s Enzyme-Labeled Antibody Method, 4th ed.; Gakusai-Kikaku: Tokyo, Japan, 2002; ISBN 4-906514-43-X. (In Japanese)
- Buchwalow, I.B.; Böcker, W. Immunohistochemistry. Basics Methods 2010, 1, 1–149.
- Shi, S.-R.; Taylor, C.R. Antigen Retrieval Immunohistochemistry Based Research and Diagnostics; John Wiley and Sons, Ltd.: Hoboken, NJ, USA, 2010; ISBN 978-1-118-06030-8.
- Chu, P.; Weiss, L. Modern Immunohistochemistry; Cambridge University Press: Cambridge, UK, 2014; ISBN 13: 9781316183748.
- Lin, F.; Prichard, J. Handbook of Practical Immunohistochemistry: Frequently Asked Questions; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 978-1-4939-1577-4.
- Kim, S.-W.; Roh, J.; Park, C.-S. Immunohistochemistry for pathologists: Protocols, pitfalls, and tips. J. Pathol. Transl. Med. 2016, 50, 411–418.
- Renshaw, S. Immunohistochemistry and Immunocytochemistry: Essential Methods, 2nd ed.; John Wiley and Sons, Ltd.: Hoboken, NJ, USA, 2017; ISBN 9781118717776.
- Dabbs, D.J. Diagnostic Immunohistochemistry, Theranostic and Genomic Applications, 5th ed.; Elsevier: Amsterdam, The Netherlands, 2018; ISBN 9780323477321.
- Motta, R.D.R.; Zettler, C.G.; Cambruzzi, E.; Jotz, G.P.; Berni, R.B. Ki-67 and p53 correlation prognostic value in squamous cell carcinomas of the oral cavity and tongue. Braz. J. Otorhinolaryngol. 2009, 75, 544–549.
- Lumachi, F.; Orlando, R.; Marino, F.; Chiara, G.B.; Basso, S.M.M. Expression of p53 and Ki-67 as prognostic factors for survival of men with colorectal cancer. Anticancer. Res. 2012, 32, 3965–3967.
- Ding, L.; Zhang, Z.; Xu, Y.; Zhang, Y. Comparative study of Her-2, p53, Ki-67 expression and clinicopathological characteristics of breast cancer in a cohort of northern China female patients. Bioengineered 2016, 8, 383–392.
- Kumar, A.; Jagannathan, N. Cytokeratin: A review on current concepts. Int. J. Orofac. Biol. 2018, 2, 6.
- Zaha, D.C. Significance of immunohistochemistry in breast cancer. World J. Clin. Oncol. 2014, 5, 382–392.
- Taylor, C.R. Immunohistochemistry in surgical pathology: Principles and practice. In Histopathology: Methods and Protocols. Methods in Molecular Biology; Springer: Berlin/Heidelberg, Germany, 2014; pp. 81–110.
- Hasui, K.; Murata, F. A new simplified catalyzed signal amplification system for minimizing non-specific staining in tissues with supersensitive immunohistochemistry. Arch. Histol. Cytol. 2005, 68, 1–17.
- Bussolati, G.; Gugliotta, P.; Volante, M.; Pace, M.; Papotti, M. Retrieved endogenous biotin: A novel marker and a potential pitfall in diagnostic immunohistochemistry. Histopathologyc 1997, 31, 400–407.
- Abcam, plc (Cambridge, UK). Antibody Storage Guide. Storage Temperature, Contamination, Freeze/Thaw Damage and Stability. Available online: (accessed on 30 June 2016).





