Inherited retinal dystrophies and optic neuropathies cause chronic disabling loss of visual function. The development of recombinant adeno-associated viral vectors (rAAV) gene therapies in all disease fields have been promising, but the translation to the clinic has been slow. The safety and efficacy profiles of rAAV are linked to the dose of applied vectors. DNA changes in the rAAV gene cassette affect potency, the expression pattern (cell-specificity), and the production yield. Here, we present a library of rAAV vectors and elements that provide a workflow to design novel vectors. We first performed a meta-analysis on recombinant rAAV elements in clinical trials (2007-2020) for ocular gene therapies. We analyzed 33 unique rAAV gene cassettes used in 57 ocular clinical trials. The rAAV gene therapy vectors used six unique capsid variants, 16 different promoters, and six unique polyadenylation sequences. Further, we compiled a list of promoters, enhancers, and other sequences used in current rAAV gene cassettes in preclinical studies. Then, we give an update on pro-viral plasmid backbones used to produce the gene therapy vectors, inverted terminal repeats, production yield, and rAAV safety considerations. Finally, we assess rAAV transgene and bioactivity assays applied to cells or organoids in vitro, explants ex vivo, and clinical studies.
1. Introduction
Many reviews have been written on recombinant adeno-associated virus vector (rAAV) tropism in ocular tissue, rAAV host cell infection, and potential rAAV-treatable inherited retinal diseases [
1,
2,
3,
4,
5,
6,
7,
8,
9]. Here, we will review the ocular gene therapies developed over the past 20 years focusing on the diversity of elements incorporated in rAAV vectors. Further, we will discuss how the vectors were generated, tested, and further modified to increase the potency and safety of the gene expression vector. After that, we will provide a library of validated elements that allows other researchers to streamline the modification of their vectors. Novel medical therapies, such as gene therapies, need to be carefully optimized to demonstrate their efficacy and safety before going into clinical human trials [
10]. That is why, it is crucial to choose the most relevant biological model(s) (in vitro, in vivo, and ex vivo models) to test an optimized gene therapy vector in a transgene expression assay (TEA) and test the transgene activity in a biological activity assay (BAA). Thus, we will explore the developments in preclinical models, for example, cellular models, human induced pluripotent stem cell (hiPSC) derived retinal organoid disease models, retinal explant models, and tropism studies in non-human primates.
1.1. Why Viral Vector-Based Gene Augmentation Therapy for Ocular Diseases?
The environment of the eye offers a wide range of treatment possibilities due to the blood-retinal barrier of the eye decreasing viral vector diffusion to other organs and systemic immune activation. The retina consists of terminally differentiated cells thus reducing gene integration and chromosomal rearrangements. Many noninvasive techniques are available to monitor the treatment response.
Treatments aim to slow the progression of the inherited retinopathies by reducing retinal cell death, augmenting retinal function, replacing cells, or creating an artificial retina (retinal prosthesis). Preventing retinal cell death may be achieved by gene therapy, cell therapy, other drug treatments, as well as dietary adjustments and even by changes in lifestyle. Such as cigarette smokers giving up smoking or people taking too much high Vitamin E supplements [
11]. Currently, there are only a few scientifically proven preventive or protective actions available to patients with inherited retinal diseases. Some preventative measures may potentially decrease patients’ quality of life, such as the continuous use of eye protection, photochromic lenses, or restriction to light exposure. Successful viral gene therapies can last many years. This could alleviate the disease burden thereby increasing the patients’ quality of life tremendously.
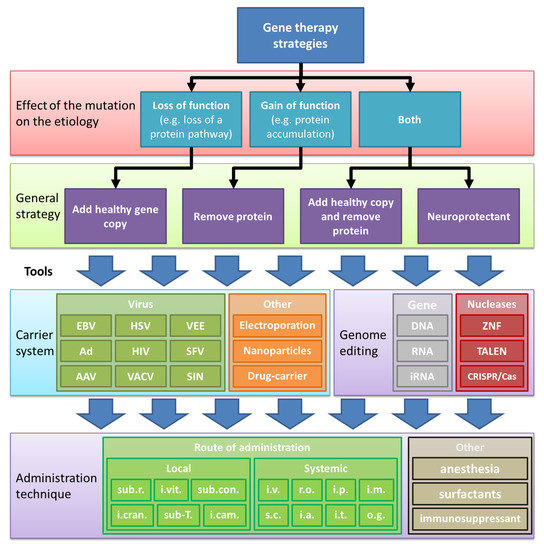
The ocular gene therapy strategy targets the basis of inherited retinopathies: the gene. With this strategy, the variant (disease-causing) gene is silenced, replaced, or repaired in the target cell (see ). The primary gene cassette carrier systems for ocular diseases has been the recombinant viral vector. However, many other potential technologies have been developed alongside, including exosomes/liposomes, antisense oligonucleotides (AONs), electroporation of naked DNA/RNA, or application of nanoparticles [
12,
13]. In most clinical retinal gene therapy studies, the vector of choice is the rAAV gene expression vector [
14]. Two genes that do not fit in a conventional rAAV gene cassette (
MYO7A linked to Usher syndrome Type 1B and
ABCA4 to Stargardt disease) have been delivered to the retina by recombinant lentiviral expression vectors instead [
15,
16,
17,
18]. Moreover, another large gene,
CEP290, can be rescued on the mRNA level by antisense oligonucleotides (AONs; QR-110 for Leber congenital amaurosis (LCA), with
CEP290; QR-421a for LCA with
USH2A) [
19]. An important consideration is how the therapeutic product is administered to the target cells. The most common method is the subretinal or the intravitreal injection. Subretinal injections can target a focal area (e.g., macula), favoring high vector delivery to the retinal pigment epithelium (RPE), photoreceptors, and Müller glial cells. Intravitreal injections efficiently target the ganglion cell layer and spread the rAAV to the whole retina in rodents but not in the primate retina due to the shielding properties of the inner limiting membrane. describes the considerations for choosing a gene therapy strategy.
Figure 1. An overview of retinal gene therapy strategies. The effects of the gene variations determine the gene therapy rescue strategy to be applied. Physical DNA delivery includes electroporation, sonoporation, magnetofection, and bioballistic (gene gun) methods. Viruses: AAV, Adeno-associated virus; Ad, Adenovirus; alpha, alphavirus; Epstein-Barr virus (EBV); FV, Foamy virus; HSV, Herpes simplex virus; HIV, Human immunodeficiency virus; VACV, Vaccinia virus. Nucleases: ZNF, Zinc-finger nuclease; TALEN, transcription activator-like effector nuclease; CRISPR/Cas, clustered regulatory interspaced short palindromic repeat (CRISPR)/Cas-based RNA-guided DNA endonuclease. Route of administration: i.cranial, intracranial; i.a., intraarterial; i.cam., intracameral; i.m., intramuscular; i.v., i.t., intrathecal; intravenous (e.g., tail vein or facial vein); i.vit, intravitreal; o.g., oral gavage; r.o., retro-orbital; sub.r, subretinal; sub-T, sub-Tenon; sub.con., sub conjunctiva; s.c., subcutaneous.
1.2. rAAV Gene Therapy for Ocular Diseases—Advantages and Disadvantages
rAAV DNA carrier systems have been successfully used because they have a plethora of advantages: (1) They express the transgene within days or weeks and can reach full expression levels after 4–6 weeks in vivo [
20,
21]. (2) rAAV DNA carrier systems allow long-term treatment for at least ten years in dogs [
22], (3) and primarily deliver their gene cassette in episomal concatemers into the nucleus [
23]. (4) rAAV DNA carrier systems spread well within tissues to target large retinal patches [
24]. (5) The capsid composition can be adjusted to fit one’s goals [
25]. (6) rAAV injections have been very safe with a low number of serious adverse events (SAE) in clinical trials [
26]. On the other hand, rAAVs similar to other viral strategies, have limitations such as: (1) A small gene cassette capacity (up to 4.6 kb + 2 × 145 bp ITRs). (2) The instability of the inverted terminal repeats (ITRs). (3) The need for high viral load for transgene expression. (4) A humoral immune reaction can be evoked. The immune reactions range from induction of neutralizing antibodies that reduce the number of capsids reaching the target cells to the innate immune pathways silencing the gene cassette within the host cell, as well as the cell-mediated T-cell immune response against foreign protein expression [
27]. It is important to carefully consider the advantages of using rAAVs over other attractive strategies (). In the following, we will describe how successful gene cassettes (vectors) have been designed for rAAV vectors (vectorology) that entered the clinical phase.