
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Zhonglei Wang | + 2243 word(s) | 2243 | 2021-06-21 10:48:52 | | | |
| 2 | Conner Chen | Meta information modification | 2243 | 2021-06-28 09:41:16 | | |
Video Upload Options
As a traditional source for modern pharmaceutical discovery and potential drug leads, natural products have played an integral role in treating patients due to their unique structural, chemical, and biological diversity. A wide range of natural products can be considered promising anti-COVID-19 or anti-lung cancer agents have gained widespread attention, including natural products as monotherapy for the treatment of SARS-CoV-2 (ginkgolic acid, shiraiachrome A, resveratrol, and baicalein) or lung cancer (daurisoline, graveospene A, deguelin, and erianin) or in combination with FDA-approved anti-SARS-CoV-2 agents (cepharanthine plus nelfinavir, linoleic acid plus remdesivir) and anti-lung cancer agents (curcumin and cisplatin, celastrol and gefitinib).
1. Natural Products as Monotherapy for the Treatment of SARS-CoV-2
Natural products have demonstrated potential value, which supports this strategy as an indispensable research focus in the fight against the COVID-19 epidemic [1][2]. The chemical structures of the components described in this section are shown in Figure 1. The SARS-CoV-2 main protease (Mpro), also called the 3C-like protease (3CLpro), has a vital function in viral replication and is, therefore, a preferred drug target [3]. The papain-like protease (PLpro), another prime therapeutic target, plays an essential role in maturing viral RNA polyproteins and dysregulation of host inflammation [4]. Ginkgolic acid, a phenolic acid, is an essential component of the traditional herbal medicine Ginkgo biloba (EGb) [5]. A study has demonstrated that ginkgolic acid is characterized by half-maximal inhibitory concentration (IC50) values of 1.79 μM and 16.3 μM against SARS-CoV-2 Mpro and SARS-CoV-2 PLpro, respectively [6]. The study unambiguously showed that ginkgolic acid exerts good dual-inhibitory effects through its irreversible binding to SARS-CoV-2 cysteine proteases [6].
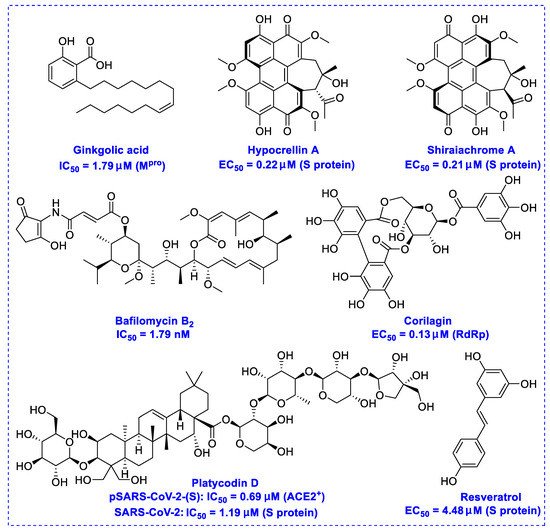
Figure 1. Promising natural products for treating SARS-CoV-2.
Angiotensin-converting enzyme 2 (ACE2), an essential ingredient of the renin–angiotensin–aldosterone system (RAAS), is a critical host cell surface receptor for viral infection [7]. The glycosylated spike protein (S protein) plays an essential role in mediating viral entry via interactions with the ACE2 cell surface receptor [8]. Hypocrellin A and shiraiachrome A, two-axial chiral perylenequinones, have been reported to exhibit potent effects on the infected monkey Vero E6 cell line by inhibiting the activity of the SARS-CoV-2 S protein at EC50 values of 0.22 μM and 0.21 μM, respectively, while at doses of up to 10 μM, these presented no observable cytotoxicity against these cells [9].
Transmembrane protease serine 2 (TMPRSS2), a critical factor enabling SARS-CoV-2 infection, can interact with ACE2 [10]. It has been reported that platycodin D, a triterpenoid saponin isolated from Platycodon grandiflorum, prevents TMPRSS2-driven infection in vitro by impairing membrane fusion [11]. Platycodin D has IC50 values of 0.69 μM and 0.72 μM for SARS-CoV-2 pseudovirus (pSARS-CoV-2) overexpression of ACE2 (ACE2+) and ACE2/TMPRSS2+, respectively, and IC50 values of 1.19 μM and 4.76 μM for SARS-CoV-2 in TMPRSS2-negative Vero cells and TMPRSS2-positive Calu-3 cells, respectively [11]. Resveratrol, a remarkable phytoalexin, may effectively inhibit the replication of SARS-CoV-2 S protein in Vero E6 cells at an EC50 of 4.48 μM [12], and has an excellent safety tracking record, with no cytotoxicity even up to a concentration of 150 µM [13].
The RNA-dependent RNA polymerase (RdRp) of SARS-CoV-2 is another promising target that regulates the replication of the viral genome [14]. Corilagin, a non-nucleoside inhibitor, is a gallotannin isolated from the medicinal plant Phmllanthi Fructus [15]. Corilagin has been reported to inhibit SARS-CoV-2 infection with an EC50 value of 0.13 μM in a concentration-dependent manner by preventing the conformational change of RdRp and inhibits SARS-CoV-2 replication [16]. Furthermore, corilagin, as identified via molecular dynamics simulation-guided studies, could also be used as an endogenous Mpro candidate, with an 88% anti-SARS-CoV-2 Mpro activity at concentrations of 20 μM in vitro [17].
Bafilomycin B2, which can be isolated from Streptomyces sp. HTL16, indicates enhanced inhibitory potency against SARS-CoV-2 at IC50 values of 5.11 nM (in the full-time approach) and 8.32 nM (in the pretreatment-of-virus approach) in Vero E6 cells, respectively [18]. While bafilomycin B2 has demonstrated potential effectiveness in inhibiting the viral entry process, evidence of its utility as anti-SARS-CoV-2 agents in vivo is currently insufficient.
The above evidence supports the potential value of the above natural products as therapeutic agents for the treatment of the novel SARS-CoV-2 infection, suggesting more validation studies (both in vitro and in animal models as well as on humans) could be encouraged to perform. Besides the above-mentioned molecules, several other natural products have also been shown to exhibit potent anti-SARS-CoV-2 activities in vitro. Table 1 summarizes a range of studies investigating the in vitro effects of anti-SARS-CoV-2 agents since 2020.
Table 1. Other natural products with anti-SARS-CoV-2 activities in vitro.
| No. | Name | Structure | EC50 or IC50 (μM) | Strain | Refs |
|---|---|---|---|---|---|
| 1 | Acetoside | 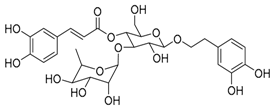 |
0.043 | Vero E6 cells | [19] |
| 2 | Anacardic acid | 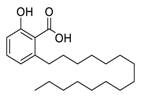 |
2.07 | USA-WA1/2020 | [6] |
| 3 | Andrographolide |  |
0.034 | Calu-3 cells | [20][21] |
| 4 | Apigenin-7-O-glucoside | 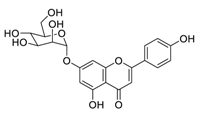 |
0.074 | Vero E6 cells | [19] |
| 5 | Artemisinin | 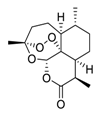 |
64.45 | Vero E6 cells | [21][22] |
| 6 | Azithromycin | 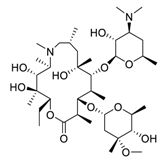 |
2.12 | Caco-2 cells | [23] |
| 7 | Baicalin | 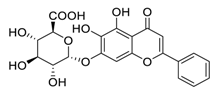 |
7.98 | Vero E6 cells | [24] |
| 8 | Cannabidiol | 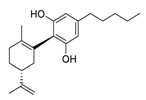 |
7.91 | Vero E6 cells | [25][26] |
| 9 | Catechin-3-O-gallate | 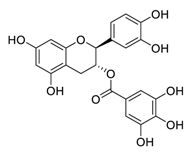 |
2.98 | Vero E6 cells | [27] |
| 10 | Chebulagic acid | 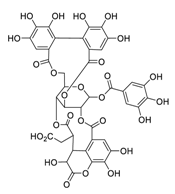 |
9.76 | Vero E6 cells | [28] |
| 11 | Daurisoline | 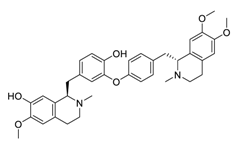 |
3.66 | Vero E6 cells | [29] |
| 12 | EGCG | 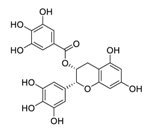 |
0.874 | Vero E6 cells | [24][30] |
| 13 | Emetine | 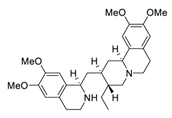 |
0.46 | Vero E6 cells | [31][32] |
| 14 | Epicatechin-3-O-gallate | 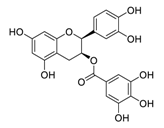 |
5.21 | Vero E6 cells | [27] |
| 15 | Gallinamide A | 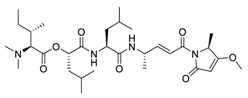 |
0.028 | Vero E6 cells | [33] |
| 16 | Gallocatechin-3-O-gallate | 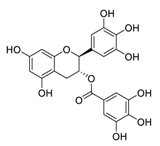 |
6.38 | Vero E6 cells | [27] |
| 17 | Hopeaphenol | 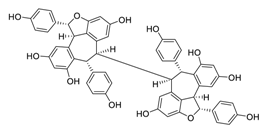 |
2.3 | B.1.351 | [34] |
| 18 | Ipomoeassin F | 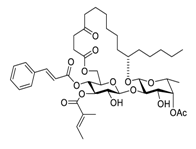 |
semi-permeabilized mammalian cells | [35] | |
| 19 | Kobophenol A | 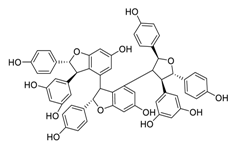 |
1.81 | Vero E6 cells | [36] |
| 20 | Myricetin | 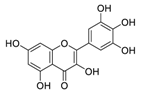 |
0.22 | Vero E6 cells | [37][38] |
| 21 | Naringenin | 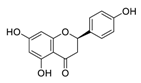 |
0.092 | Vero E6 cells | [19][39] |
| 22 | Osajin | 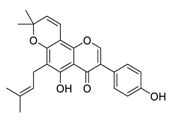 |
3.87 | Vero E6 cells | [40] |
| 23 | 2,3′,4,5′,6-Pentahydroxybenzophenone | 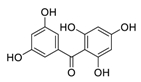 |
0.102 | Vero E6 cells | [19] |
| 24 | Procyanidin B2 | 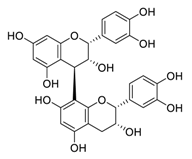 |
75.3 | Vero E6 cells | [27] |
| 25 | Punicalagin | 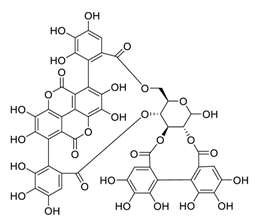 |
7.20 | Vero E6 cells | [28] |
| 26 | Sennoside B | 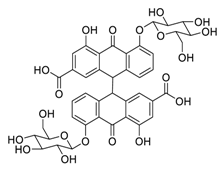 |
0.104 | Vero E6 cells | [19] |
| 27 | Shikonin | 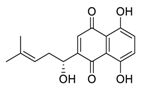 |
15.75 | Vero E6 cells | [41] |
| 28 | Δ9-Tetrahydrocannabinol | 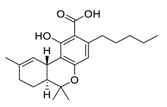 |
10.25 | Vero E6 cells | [25] |
| 29 | Tetrandrine | 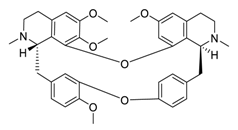 |
3.00 | Vero E6 cells | [40] |
| 30 | Theaflavin | 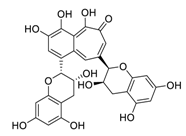 |
8.44 | HEK293T human embryonic kidney cells |
[42] |
Traditional Chinese medicines have attracted considerable attention due to their ability to effectively inhibit SARS-CoV-2 [43][44][45]. For example, the Qingfei Paidu decoction (QFPD) has shown an ability to treat COVID-19 patients at all stages with excellent clinical efficacy (cure rate >90%) [46][47]. Shuanghuanglian oral liquid or injection (SHL), another well-known traditional Chinese medicine, dose-dependently inhibits SARS-CoV-2 Mpro replication [48]. In addition to the above-mentioned QFPD and SHL, several other traditional Chinese medicines (such as Kegan Liyan oral liquid and Toujie Quwen granule) listed in Table 2 contain Scutellaria baicalensis Georgi (Chinese name: Huangqin), whose major component is baicalein, exerts a marked anti-SARS-CoV-2 effect (IC50 of 0.94 μM, and SI > 212) [49]. Furthermore, it is crucial to investigate how herbal medicine affects SARS-CoV-2 infection by studying its active ingredients. To elucidate the underlying molecular mechanisms, a crystal structure of SARS-CoV-2 Mpro complexed with baicalein was constructed at a resolution of 2.2 Å (the Protein Data Bank (PDB) ID: 6M2N) [48]. Analysis of the core of the substrate-binding pocket revealed multiple interactions (such as hydrogen bonding with Leu141/Gly143 and Ser144/His163, π–π interactions with Cys145 and His4, and hydrophobic interactions with Met49 and His41), which effectively blocked SARS-CoV-2 replication via noncovalent incorporation [48]. The relevant studies [50][51][52] provided direct data for a better understanding of the molecular mechanisms of Chinese herbal medicine by studying its active ingredients.
Table 2. Registered clinical trials relating to traditional Chinese medicine prescriptions containing baicalein (active ingredient of Huangqin) for treatment of COVID-19 patients (Chinese Clinical Trial Registry, www.chictr.org/cn/ (accessed on 31 January 2021).
| Baicalein (The Active Ingredient of Huangqin) | Molecular Mechanisms of Baicalein | Herbal Formula Containing Huangqin | Registration Number | Sample Size of the Control Group |
|---|---|---|---|---|
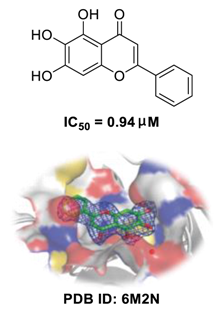 |
RdRp inhibitor via noncovalent incorporation [53], potent antagonists against TMPRSS2 [50], improving respiratory function, decreasing IL-1β and TNF-α levels, and inhibiting cell infiltration [51][52]. | Qingfei Paidu decoction | ChiCTR2000029433 | 120 |
| ChiCTR2000030883 | 100 | |||
| ChiCTR2000032767 | 782 | |||
| Xinguan I decoction | ChiCTR2000029637 | 50 | ||
| Tanreqing capsules | ChiCTR2000029813 | 36 | ||
| Tanreqing injection | ChiCTR2000029432 | 72 | ||
| Kegan Liyan oral liquid | ChiCTR2000033720 | 240 | ||
| ChiCTR2000033745 | 240 | |||
| ChiCTR2000031982 | 240 | |||
| Shuanghuanglian oral liquid | ChiCTR2000033133 | 30 | ||
| ChiCTR2000029605 | 100 | |||
| Toujie Quwen granule | ChiCTR2000031888 | 150 |
2. Natural Products as Monotherapy for the Treatment of Lung Cancer
There is no doubt that natural products have always been recognized as promising anti-lung cancer agents. Daurisoline, an autophagy blocker, is a bisbenzylisoquinoline alkaloid extracted from the herbal medicine Nelumbo nucifera Gaertn [54]. The chemical structures of the molecules discussed in this section are shown in Figure 2. Daurisoline increases the degradation of β-catenin by targeting heat shock protein 90 (HSP90) directly and decreases the expression of MYC proto-oncogene (c-MYC) and cyclin D1, which resulted in cell cycle arrest at the G1 phase in human lung cancer A549 cells and Hop62 cells lines to exert its anti-lung cancer activity [55]. More importantly, in animals, daurisoline has been reported to be a promising anti-lung cancer agent (by inhibiting tumor growth in lung cancer xenografts) with no observable side effects, thus highlighting a potential role for daurisoline in the treatment of lung cancer [55]. Another recent study has shown that daurisoline can effectively inhibit SARS-CoV-2 replication at IC50 values of 3.664 μM and 0.875 μM in Vero E6 cells and in human pulmonary alveolar epithelial cells (HPAEpiC), respectively [29].
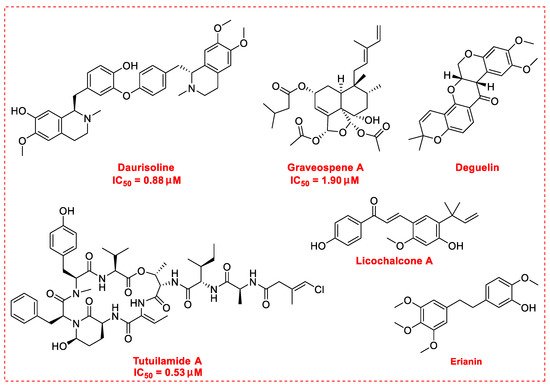
Figure 2. Promising natural products for treating lung cancer.
Graveospene A, isolated from the leaves of Casearia graveolens, is a new clerodane diterpenoid that has been reported to induce apoptosis in A549 cells with an IC50 value of 1.9 μM by inducing cell cycle arrest in phase G0/G1 [56]. Deguelin, a protein kinase B (AKT) kinase inhibitor, is isolated from the African plant Mundulea sericea (Leguminosae) and is commonly used to inhibit the growth of several types of human cancer cell lines [57]. Deguelin promoted the phosphorylation of myeloid cell leukemia sequence-1 (Mcl-1) protein and induced the inhibition of the wildtype and mutated epidermal growth factor receptor (EGFR)-Akt signaling pathway, which resulted in activation of downstream GSK3β/FBW7 and profound anti-NSCLC activity with no obvious side effects in vivo [58].
Licochalcone A is a natural flavonoid derived from Xinjiang licorice and Glycyrrhiza inflata. Licochalcone A is known to possess a broad spectrum of activities with important pharmacological effects in various cancer cell lines [59]. Licochalcone A can significantly increase autophagic cytotoxicity (in both A549 and H460 cell lines) and downregulated the expression of c-IAP1, c-IAP2, XIAP, survivin, c-FLIPL, and RIP1, apoptosis-related proteins via inhibiting the activity of phosphorylated extracellular signal-regulated kinase (ERK) and autophagy [60]. In addition, licochalcone A has been reported to abolish the expression of programmed death ligand-1 (PD-L1) by increasing reactive oxygen species (ROS) levels in a time-dependent manner and interfering with protein translation in cancer cells [61]. Further, licochalcone A can inhibit PD-L1 translation likely through the inhibition of the phosphorylation of 4EBP1 and activation of the PERK-eIF2α signaling pathway [61]. Licochalcone A plays a vital role in reversing the ectopic expression of key microRNA (miR-144-3p, miR-20a-5p, miR-29c-3p, let-7d-3p, and miR-328-3p) to elicit lung cancer chemopreventive activities both in vivo and in vitro [62]. In addition, licochalcone A has been reported to inhibit EGFR signaling and reduced the expression of Survivin protein in a cap-dependent translation manner to exhibit profound activity in mutated NSCLC cells [63].
Erianin, a novel dibenzyl compound, can be isolated from the traditional herbal medicine Dendrobium chrysotoxum Lindl and has been proposed as an apoptosis-inducing agent in human lung cancer cells [64]. The main mechanisms of its anti-lung cancer activity involve the induction of ferroptosis by activating Ca2+/calmodulin signaling, inhibition of cell proliferation and metastasis, and induction of cell cycle arrest in phase G2/M [65].
Tutuilamide A, isolated from marine cyanobacteria Schizothrix sp., is a novel cyclic peptide reported to exhibit moderate cytotoxicity activity in the H-460 human lung cancer cell line with an IC50 value of 0.53 μM [66]. Tutuilamide A, with the help of the vinyl chloride side chain, showed enhanced inhibitory potency with high selectivity (IC50 0.73 nM) for human neutrophil elastase, which is associated mainly with the migration and metastasis of lung cancer cells [67]. Besides the above-mentioned molecules, Table 3 also exhibits other natural products (including their underlying molecular mechanisms) with notable anti-lung cancer activities reported since 2020.
Table 3. The mechanism involved in anticancer activities of other natural products (reported since 2020).
| No. | Name | Structure | Mechanism of Anti-Lung Cancer | Refs |
|---|---|---|---|---|
| 1 | Acovenoside A | 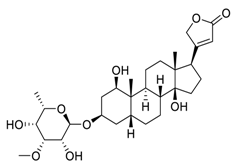 |
Inhibit the adenosine triphosphate (ATP)-dependent Na+/K+ exchange through the Na+/K+-ATPase | [68] |
| 2 | Asiatic acid | 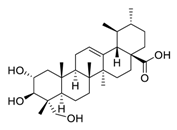 |
Inhibited the ionizing radiation-induced migration and invasion | [69] |
| 3 | Baicalein | 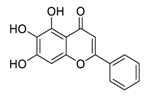 |
Restrained ezrin tension by decreasing inducible nitric oxide synthase expression levels, suppress invasion, reduced vasculogenic mimicry formation | [70][71][72] |
| 4 | Baicalin | 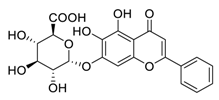 |
Inhibited the invasion, migration, angiogenesis, and Akt/mTOR pathway | [73][74] |
| 5 | Casticin | 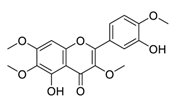 |
Induced the expressions and nuclear translocation of phosphorylation of H2AX | [75] |
| 6 | Dioscin | 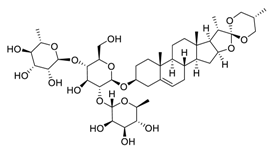 |
Down-regulated signal transducer and activator of transcription 3 and c-Jun N-terminal kinase signaling pathways | [76] |
| 7 | EGCG | 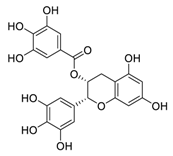 |
Regulated CTR1 expression through the ERK1/2/NEAT1 signaling pathway | [77][78] |
| 8 | Ellagic acid | 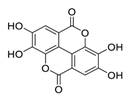 |
Inhibited tumor growth, increased p-AMPK, and suppressed hypoxia-inducible factor 1α levels | [79] |
| 9 | Erianthridin | 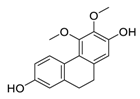 |
Attenuated extracellular signal-regulated kinase activity and mediated apoptosis, matrix-degrading metalloproteinases (MMPs) expression | [80][81] |
| 10 | Eugenol | 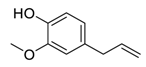 |
Restriction of β-catenin nuclear transportation | [82] |
| 11 | Formononetin | 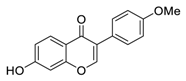 |
Inhibited EGFR-Akt signaling, which in turn activates GSK3β and promotes Mcl-1 phosphorylation in NSCLC cells | [83][84] |
| 12 | Gallic Acid | 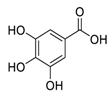 |
Inhibited of EGFR activation and impairment, inhibition of phosphoinositide 3-kinase (PI3K) and AKT phosphorylation | [85][86] |
| 13 | Glochidiol | 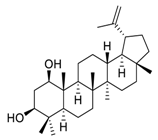 |
Inhibited tubulin polymerization | [87] |
| 14 | Gracillin | 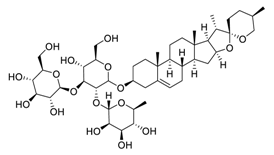 |
Inhibited both glycolysis and mitochondria-mediated bioenergetics, induced apoptosis through the mitochondrial pathway | [88][89] |
| 15 | Hispidulin | 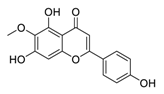 |
Promoted apoptosis by hispidulin via increased generation of ROS | [90] |
| 16 | Icaritin | 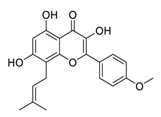 |
Downregulated the immunosuppressive cytokine (TNF-α, IL10, IL6) and upregulated chemotaxis (CXCL9 and CXCL10) | [91] |
| 17 | Isoharringtonine | 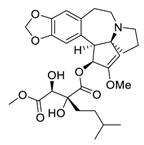 |
Induced death tumor spheroids by activating the intrinsic apoptosis pathway | [92] |
| 18 | Kaempferol | 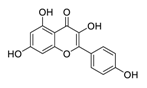 |
Inhibitor of nuclear factor erythroid 2-related factor 2 | [93] |
| 19 | Liriopesides B | 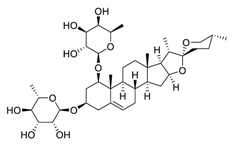 |
Reduced proliferation, and induced apoptosis and cell cycle arrest, inhibited the progression of the cell cycle from the G1 to the S phase | [94] |
| 20 | Nagilactone E | 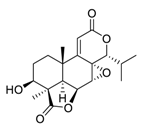 |
Activated the c-Jun N-terminal kinases, increased the phosphorylation, and promoted the localization of c-Jun in the nucleus | [95][96] |
| 21 | 8-Oxo-epiberberine | 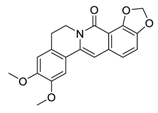 |
Inhibited TGF-β1-induced epithelial-mesenchymal transition (EMT) possibly by interfering with Smad3 | [97] |
| 22 | Parthenolide | 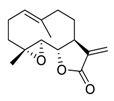 |
Reduced the phosphorylation of EGFR and downstream signaling pathways mitogen-activated protein kinase (MAPK)/ERK, inhibited PI3K/Akt/FoxO3α signaling | [98][99][100] |
| 23 | PDB-1 | 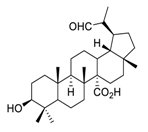 |
Suppressed lung cancer cell migration and invasion via FAK/Src and MAPK signaling pathways | [101] |
| 24 | Polyphyllin I | 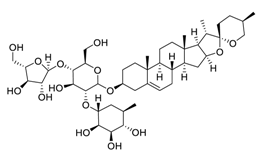 |
Induced autophagy by activating AMPK and then inhibited mTOR signaling, promoted apoptosis, modulated the PI3K/Akt signaling | [102][103] |
| 25 | Quercetin | 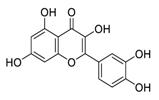 |
Inhibited proliferation and induced apoptosis | [104] |
| 26 | Silibinin | 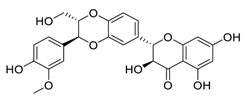 |
Inhibited cell proliferation, migration, invasion, and EMT expression | [105] |
| 27 | Sinomenine | 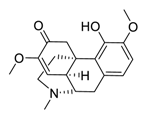 |
Downregulated expression of MMPs and miR-21, suppressed α7 nicotinic acetylcholine receptors expression | [106][107][108] |
| 28 | Toxicarioside O | 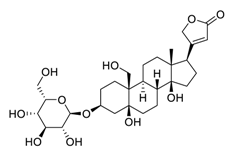 |
Decreased the expression of trophoblast cell surface antigen 2, resulting in inhibition of the PI3K/Akt pathway and EMT program | [109] |
| 29 | Vincamine | 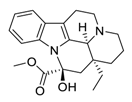 |
Interaction with the apoptotic protein caspase-3 | [110] |
| 30 | Xanthohumol | 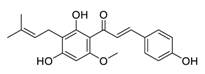 |
Suppressed ERK1/2 signaling and reduced the protein levels of FOS-related antigen 1, decreased the mRNA level of cyclin D1 | [111] |
References
- Verma, S.; Twilley, D.; Esmear, T.; Oosthuizen, C.B.; Reid, A.M.; Nel, M.; Lall, N. Anti-SARS-CoV natural products with the potential to inhibit SARS-CoV-2 (COVID-19). Front. Pharmacol. 2020, 11, 561334.
- Wang, Z.; Yang, L. Turning the tide: Natural products and natural-product-inspired chemicals as potential counters to SARS-CoV-2 infection. Front. Pharmacol. 2020, 11, 1013.
- Qiao, J.; Li, Y.S.; Zeng, R.; Liu, F.L.; Luo, R.H.; Huang, C.; Wang, Y.F.; Zhang, J.; Quan, B.; Shen, C.; et al. SARS-CoV-2 Mpro inhibitors with antiviral activity in a transgenic mouse model. Science 2021, 371, 1374–1378.
- Shin, D.; Mukherjee, R.; Grewe, D.; Bojkova, D.; Baek, K.; Bhattacharya, A.; Schulz, L.; Widera, M.; Mehdipour, A.R.; Tascher, G.; et al. Papain-like protease regulates SARS-CoV-2 viral spread and innate immunity. Nature 2020, 587, 657–662.
- Yang, J.; Feng, Z.; Liu, W.; Wang, Y.; Wang, G.; Yu, W.; Yang, G.; Yang, T.; Wang, Y.; Li, M. Exogenous hormone on episperm development and ginkgolic acid accumulation in Ginkgo biloba L. Ind. Crop. Prod. 2021, 160, 113140.
- Chen, Z.; Cui, Q.; Cooper, L.; Zhang, P.; Lee, H.; Chen, Z.; Wang, Y.; Liu, X.; Rong, L.; Du, R. Ginkgolic acid and anacardic acid are specific covalent inhibitors of SARS-CoV-2 cysteine proteases. Cell Biosci. 2021, 11, 45.
- Medina-Enríquez, M.M.; Lopez-León, S.; Carlos-Escalante, J.A.; Aponte-Torres, Z.; Cuapio, A.; Wegman-Ostrosky, T. ACE2: The molecular doorway to SARS-CoV-2. Cell Biosci. 2020, 10, 148.
- Benton, D.J.; Wrobel, A.G.; Xu, P.; Roustan, C.; Martin, S.R.; Rosenthal, P.B.; Skehel, J.J.; Gamblin, S.J. Receptor binding and priming of the spike protein of SARS-CoV-2 for membrane fusion. Nature 2020, 588, 327–330.
- Li, Y.T.; Yang, C.; Wu, Y.; Lv, J.J.; Feng, X.; Tian, X.; Zhou, Z.; Pan, X.; Liu, S.; Tian, L.W. Axial chiral binaphthoquinone and perylenequinones from the stromata of hypocrella bambusae are SARS-CoV-2 entry inhibitors. J. Nat. Prod. 2021, 84, 436–443.
- Stopsack, K.H.; Mucci, L.A.; Antonarakis, E.S.; Nelson, P.S.; Kantoff, P.W. TMPRSS2 and COVID-19: Serendipity or opportunity for intervention? Cancer Discov. 2020, 10, 779–782.
- Kim, T.Y.; Jeon, S.; Jang, Y.; Gotina, L.; Won, J.; Ju, Y.H.; Kim, S.; Jang, M.W.; Won, W.; Park, M.G.; et al. Platycodin D prevents both lysosome- and TMPRSS2-driven SARS-CoV-2 infection in vitro by hindering membrane fusion. Exp. Mol. Med. 2021.
- Yang, M.; Wei, J.; Huang, T.; Lei, L.; Shen, C.; Lai, J.; Yang, M.; Liu, L.; Yang, Y.; Liu, G.; et al. Resveratrol inhibits the replication of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in cultured Vero cells. Phytother. Res. 2021, 35, 1127–1129.
- Ter Ellen, B.M.; Dinesh Kumar, N.; Bouma, E.M.; Troost, B.; van de Pol, D.P.I.; van der Ende-Metselaar, H.H.; Apperloo, L.; van Gosliga, D.; van den Berge, M.; Nawijn, M.C.; et al. Resveratrol and pterostilbene potently inhibit SARS-CoV-2 replication in vitro. bioRxiv 2020.
- Yin, W.; Mao, C.; Luan, X.; Shen, D.D.; Shen, Q.; Su, H.; Wang, X.; Zhou, F.; Zhao, W.; Gao, M.; et al. Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir. Science 2020, 368, 1499–1504.
- Wu, C.; Huang, H.; Choi, H.Y.; Ma, Y.; Zhou, T.; Peng, Y.; Pang, K.; Shu, G.; Yang, X. Anti-esophageal cancer effect of corilagin extracted from Phmllanthi fructus via the mitochondrial and endoplasmic reticulum stress pathways. J. Ethnopharmacol. 2021, 269, 113700.
- Li, Q.; Yi, D.; Lei, X.; Zhao, J.; Zhang, Y.; Cui, X.; Xiao, X.; Jiao, T.; Dong, X.; Zhao, X.; et al. Corilagin inhibits SARS-CoV-2 replication by targeting viral RNA-dependent RNA polymerase. Acta Pharm. Sin. B 2021.
- Loschwitz, J.; Jäckering, A.; Keutmann, M.; Olagunju, M.; Eberle, R.J.; Coronado, M.A.; Olubiyi, O.O.; Strodel, B. Novel inhibitors of the main protease enzyme of SARS-CoV-2 identified via molecular dynamics simulation-guided in vitro assay. Bioorg. Chem. 2021, 111, 104862.
- Xie, X.; Lu, S.; Pan, X.; Zou, M.; Li, F.; Lin, H.; Hu, J.; Fan, S.; He, J. Antiviral bafilomycins from a feces-inhabiting Streptomyces sp. J. Nat. Prod. 2021, 84, 537–543.
- Abdallah, H.; El-Halawany, A.; Sirwi, A.; El-Araby, A.; Mohamed, G.; Ibrahim, S.; Koshak, A.; Asfour, H.; Awan, Z.; Elfaky, M.A. Repurposing of Some Natural Product Isolates as SARS-COV-2 Main Protease Inhibitors via In Vitro Cell Free and Cell-Based Antiviral Assessments and Molecular Modeling Approaches. Pharmaceuticals 2021, 14, 213.
- Sa-ngiamsuntorn, K.; Suksatu, A.; Pewkliang, Y.; Thongsri, P.; Kanjanasirirat, P.; Manopwisedjaroen, S.; Charoensutthivarakul, S.; Wongtrakoongate, P.; Pitiporn, S.; Chaopreecha, J.; et al. Anti-SARS-CoV-2 Aactivity of Andrographis paniculata extract and its major component andrographolide in human lung epithelial cells and cytotoxicity evaluation in major organ cell representatives. J. Nat. Prod. 2021, 84, 1261–1270.
- Hu, Y.; Liu, M.; Qin, H.; Lin, H.; An, X.; Shi, Z.; Song, L.; Yang, X.; Fan, H.; Tong, Y. Artemether, artesunate, arteannuin B, echinatin, licochalcone B and andrographolide effectively inhibit SARS-CoV-2 and related viruses in vitro. Front. Cell. Infect. Microbiol. 2021.
- Cao, R.; Hu, H.; Li, Y.; Wang, X.; Xu, M.; Liu, J.; Zhang, H.; Yan, Y.; Zhao, L.; Li, W.; et al. Anti-SARS-CoV-2 potential of artemisinins in vitro. ACS Infect. Dis. 2020, 6, 2524–2531.
- Touret, F.; Gilles, M.; Barral, K.; Nougairède, A.; Decroly, E.; de Lamballerie, X.; Coutard, B. In vitro screening of a FDA approved chemical library reveals potential inhibitors of SARS-CoV-2 replication. Sci. Rep. 2020, 10, 13093.
- Hong, S.; Seo, S.H.; Woo, S.J.; Kwon, Y.; Song, M.; Ha, N.C. Epigallocatechin gallate inhibits the uridylate-specific endoribonuclease Nsp15 and efficiently neutralizes the SARS-CoV-2 strain. J. Agric. Food Chem. 2021.
- Raj, V.; Park, J.G.; Cho, K.H.; Choi, P.; Kim, T.; Ham, J.; Lee, J. Assessment of antiviral potencies of cannabinoids against SARSCoV-2 using computational and in vitro approaches. Int. J. Biol. Macromol. 2021, 168, 474–485.
- Nguyen, L.C.; Yang, D.; Nicolaescu, V.; Best, T.; Chen, S.; Friesen, J.B.; Drayman, N.; Mohamed, A.; Dann, C.; Silva, D.; et al. Cannabidiol inhibits SARS-CoV-2 replication and promotes the host innate immune response. bioRxiv 2021.
- Zhu, Y.; Xie, D.Y. Docking characterization and in vitro inhibitory activity of flavan-3-ols and dimeric proanthocyanidins against the main protease activity of SARS-CoV-2. Front. Plant Sci. 2020, 11, 601316.
- Du, R.; Cooper, L.; Chen, Z.; Lee, H.; Rong, L.; Cui, Q. Discovery of chebulagic acid and punicalagin as novel allosteric inhibitors of SARS-CoV-2 3CLpro. Antivir. Res. 2021, 190, 105075.
- Wang, P.; Luo, R.; Zhang, M.; Wang, Y.; Song, T.; Tao, T.; Li, Z.; Jin, L.; Zheng, H.; Chen, W.; et al. A cross-talk between epithelium and endothelium mediates human alveolar-capillary injury during SARS-CoV-2 infection. Cell Death Dis. 2020, 11, 1042.
- Du, A.; Zheng, R.; Disoma, C.; Li, S.; Chen, Z.; Li, S.; Liu, P.; Zhou, Y.; Shen, Y.; Liu, S.; et al. Epigallocatechin-3-gallate, an active ingredient of Traditional Chinese Medicines, inhibits the 3CLpro activity of SARS-CoV-2. Int. J. Biol. Macromol. 2021, 176, 1–12.
- Choy, K.T.; Wong, A.Y.L.; Kaewpreedee, P.; Sia, S.F.; Chen, D.D.; Hui, K.P.Y.; Chu, D.K.W.; Chan, M.C.W.; Cheung, P.P.H.; Huang, X.; et al. Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro. Antivir. Res. 2020, 178, 104786.
- Kumar, R.; Afsar, M.; Khandelwal, N.; Chander, Y.; Riyesh, T.; Dedar, R.K.; Gulati, B.R.; Pal, Y.; Barua, S.; Tripathi, B.N.; et al. Emetine suppresses SARS-CoV-2 replication by inhibiting interaction of viral mRNA with eIF4E. Antivir. Res. 2021, 189, 105056.
- Ashhurst, A.; Tang, A.; Fajtova, P.; Yoon, M.; Aggarwal, A.; Stoye, A.; Larance, M.; Beretta, L.; Drelich, A.; Skinner, D.; et al. Potent in vitro anti-SARS-CoV-2 activity by gallinamide A and analogues via inhibition of cathepsin L. bioRxiv 2020.
- Tietjen, I.; Cassel, J.; Register, E.T.; Zhou, X.Y.; Messick, T.E.; Keeney, F.; Lu, L.D.; Beattie, K.D.; Rali, T.; Ertl, H.C.J.; et al. The natural stilbenoid (-)-hopeaphenol inhibits cellular entry of SARS-CoV-2 USA-WA1/2020, B.1.1.7 and B.1.351 variants. bioRxiv 2021.
- O’Keefe, S.; Roboti, P.; Duah, K.B.; Zong, G.; Schneider, H.; Shi, W.Q.; High, S. Ipomoeassin-F inhibits the in vitro biogenesis of the SARS-CoV-2 spike protein and its host cell membrane receptor. J. Cell Sci. 2021, 134, jcs257758.
- Gangadevi, S.; Badavath, V.N.; Thakur, A.; Yin, N.; Jonghe, S.D.; Acevedo, O.; Jochmans, D.; Leyssen, P.; Wang, K.; Neyts, J.; et al. Kobophenol A inhibits binding of host ACE2 receptor with Spike RBD domain of SARS-CoV-2, a lead compound for blocking COVID-19. J. Phys. Chem. Lett. 2021, 12, 1793–1802.
- Kuzikov, M.; Costanzi, E.; Reinshagen, J.; Esposito, F.; Vangeel, L.; Wolf, M.; Ellinger, B.; Claussen, C.; Geisslinger, G.; Corona, A.; et al. Identification of inhibitors of SARS-CoV-2 3CL-Pro enzymatic activity using a small molecule in-vitro repurposing screen. ACS Pharmacol. Transl. Sci. 2021.
- Xiao, T.; Cui, M.; Zheng, C.; Wang, M.; Sun, R.; Gao, D.; Bao, J.; Ren, S.; Yang, B.; Lin, J.; et al. Myricetin inhibit SARS-CoV-2 viral replication by targeting Mpro and ameliorates pulmonary inflammation. Front. Pharmacol. 2021.
- Clementi, N.; Scagnolari, C.; D’Amore, A.; Palombi, F.; Criscuolo, E.; Frasca, F.; Pierangeli, A.; Mancini, N.; Antonelli, G.; Clementi, M.; et al. Naringenin is a powerful inhibitor of SARS-CoV-2 infection in vitro. Pharmacol. Res. 2021, 163, 105255.
- Jeon, S.; Ko, M.; Lee, J.; Choi, I.; Byun, S.Y.; Park, S.; Shum, D.; Kim, S. Identification of antiviral drug candidates against SARS-CoV-2 from FDA-approved drugs. Antimicrob. Agents Chemother. 2020, 64, e00819-20.
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature 2020, 582, 289–293.
- Jang, M.; Park, Y.I.; Cha, Y.E.; Park, R.; Namkoong, S.; Lee, J.I.; Park, J. Tea polyphenols EGCG and theaflavin inhibit the activity of SARS-CoV-2 3CL-protease in vitro. Evid. Based Compl. Alt. Med. 2020, 2020, 5630838.
- Choudhry, N.; Zhao, X.; Xu, D.; Zanin, M.; Chen, W.; Yang, Z.; Chen, J. Chinese Therapeutic Strategy for Fighting COVID-19 and Potential Small-Molecule Inhibitors against Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). J. Med. Chem. 2020, 63, 13205–13227.
- Wang, Z.; Yang, L. Chinese herbal medicine: Fighting SARS-CoV-2 infection on all fronts. J. Ethnopharmacol. 2021, 270, 113869.
- Huang, K.; Zhang, P.; Zhang, Z.; Youn, J.Y.; Zhang, H.; Cai, H.L. Traditional Chinese Medicine (TCM) in the treatment of viral infections: Efficacies and mechanisms. Pharm. Ther. 2021, 225, 107843.
- National Health Commission of the People’s Republic of China. Notice on the Issunance of Guidelines of Diagnosis and Treatment for 2019-nCoV Infected Pneumonia (Version 7). 2020. Available online: (accessed on 3 March 2020).
- Xinhua Net. Academician Xiaolin Tong: The Total Effective Rate of Qingfeipaidu Formula was 97%, none Transfer from Mild to Severe Cases. 2020. Available online: (accessed on 18 March 2020).
- Su, H.X.; Yao, S.; Zhao, W.F.; Li, M.J.; Liu, J.; Shang, W.J.; Xie, H.; Ke, C.Q.; Hu, H.C.; Gao, M.N.; et al. Anti-SARS-CoV-2 activities in vitro of Shuanghuanglian preparations and bioactive ingredients. Acta Pharmacol. Sin. 2020, 41, 1167–1177.
- Liu, H.; Ye, F.; Sun, Q.; Liang, H.; Li, C.; Li, S.; Lu, R.; Huang, B.; Tan, W.; Lai, L. Scutellaria baicalensis extract and baicalein inhibit replication of SARS-CoV-2 and its 3C-like protease in vitro. J. Enzym. Inhib. Med. Chemother. 2021, 36, 497–503.
- Pooja, M.; Reddy, G.J.; Hema, K.; Dodoala, S.; Koganti, B. Unravelling high-affinity binding compounds towards transmembrane protease serine 2 enzyme in treating SARS-CoV-2 infection using molecular modelling and docking studies. Eur. J. Pharmacol. 2021, 890, 173688.
- Song, J.; Zhang, L.; Xu, Y.; Yang, D.; Zhang, L.; Yang, S.; Zhang, W.; Wang, J.; Tian, S.; Yang, S.; et al. The comprehensive study on the therapeutic effects of baicalein for the treatment of COVID-19 in vivo and in vitro. Biochem. Pharmacol. 2021, 183, 114302.
- Ibrahim, M.A.A.; Mohamed, E.A.R.; Abdelrahman, A.H.M.; Allemailem, K.S.; Moustafa, M.F.; Shawky, A.M.; Mahzari, A.; Hakami, A.R.; Abdeljawaad, K.A.A.; Atia, M.A.M. Rutin and flavone analogs as prospective SARS-CoV-2 main protease inhibitors: In silico drug discovery study. J. Mol. Graph. Model. 2021, 105, 107904.
- Zandi, K.; Musall, K.; Oo, A.; Cao, D.; Liang, B.; Hassandarvish, P.; Lan, S.; Slack, R.L.; Kirby, K.A.; Bassit, L.; et al. Baicalein and baicalin inhibit SARS-CoV-2 RNA-dependent-RNA polymerase. Microorganisms 2021, 9, 893.
- Qian, J.Q. Cardiovascular pharmacological effects of bisbenzylisoquinoline alkaloid derivatives. Acta Pharm. Sin. 2002, 23, 1086–1092.
- Huang, X.H.; Yan, X.; Zhang, Q.H.; Hong, P.; Zhang, W.X.; Liu, Y.P.; Xu, W.W.; Li, B.; He, Q.Y. Direct targeting of HSP90 with daurisoline destabilizes β-catenin to suppress lung cancer tumorigenesis. Cancer Lett. 2020, 489, 66–78.
- Liu, F.; Ma, J.; Shi, Z.; Zhang, Q.; Wang, H.; Li, D.; Song, Z.; Wang, C.; Jin, J.; Xu, J.; et al. Clerodane diterpenoids isolated from the leaves of Casearia graveolens. J. Nat. Prod. 2020, 83, 36–44.
- Tuli, H.S.; Mittal, S.; Loka, M.; Aggarwal, V.; Aggarwal, D.; Masurkar, A.; Kaur, G.; Varol, M.; Sak, K.; Kumar, M.; et al. Deguelin targets multiple oncogenic signaling pathways to combat human malignancies. Pharmacol. Res. 2021, 166, 105487.
- Gao, F.; Yu, X.; Li, M.; Zhou, L.; Liu, W.; Li, W.; Liu, H. Deguelin suppresses non-small cell lung cancer by inhibiting EGFR signaling and promoting GSK3β/FBW7-mediated Mcl-1 destabilization. Cell Death Dis. 2020, 11, 143.
- Wu, C.P.; Lusvarghi, S.; Hsiao, S.H.; Liu, T.C.; Li, Y.Q.; Huang, Y.H.; Hung, T.H.; Ambudka, S.V. Licochalcone A selectively resensitizes ABCG2-overexpressing multidrug-resistant cancer cells to chemotherapeutic drugs. J. Nat. Prod. 2020, 83, 1461–1472.
- Luo, W.; Sun, R.; Chen, X.; Li, J.; Jiang, J.; He, Y.; Shi, S.; Wen, H. ERK activation-mediated autophagy induction resists licochalcone A-induced anticancer activities in lung cancer cells in vitro. OncoTargets Ther. 2020, 13, 13437–13450.
- Yuan, L.W.; Jiang, X.M.; Xu, Y.L.; Huang, M.Y.; Chen, Y.C.; Yu, W.B.; Su, M.X.; Ye, Z.H.; Chen, X.; Wang, Y.; et al. Licochalcone A inhibits interferon-gamma-induced programmed death-ligand 1 in lung cancer cells. Phytomedicine 2021, 80, 153394.
- Li, B.; Zhou, D.; Li, S.; Feng, Y.; Li, X.; Chang, W.; Zhang, J.; Sun, Y.; Qing, D.; Chen, G.; et al. Licochalcone A reverses NNK-induced ectopic miRNA expression to elicit in vitro and in vivo chemopreventive effects. Phytomedicine 2020, 76, 153245.
- Gao, F.; Li, M.; Yu, X.; Liu, W.; Zhou, L.; Li, W. Licochalcone A inhibits EGFR signalling and translationally suppresses survivin expression in human cancer cells. J. Cell Mol. Med. 2021, 25, 813–826.
- Yang, A.; Li, M.Y.; Zhang, Z.H.; Wang, J.Y.; Xing, Y.; Ri, M.; Jin, C.H.; Xu, G.H.; Piao, L.X.; Jin, H.L.; et al. Erianin regulates programmed cell death ligand 1 expression and enhances cytotoxic T lymphocyte activity. J. Ethnopharmacol. 2021, 273, 113598.
- Chen, P.; Wu, Q.; Feng, J.; Yan, L.; Sun, Y.; Liu, S.; Xiang, Y.; Zhang, M.; Pan, T.; Chen, X.; et al. Erianin, a novel dibenzyl compound in Dendrobium extract, inhibits lung cancer cell growth and migration via calcium/calmodulin-dependent ferroptosis. Signal Transduct. Tar. 2020, 5, 51.
- Keller, L.; Canuto, K.M.; Liu, C.; Suzuki, B.M.; Almaliti, J.; Sikandar, A.; Naman, C.B.; Glukhov, E.; Luo, D.; Duggan, B.M.; et al. Tutuilamides A-C: Vinyl-chloride-containing cyclodepsipeptides from marine cyanobacteria with potent elastase inhibitory properties. ACS Chem. Biol. 2020, 15, 751–757.
- Chen, Q.Y.; Luo, D.; Seabra, G.M.; Luesch, H. Ahp-Cyclodepsipeptides as tunable inhibitors of human neutrophil elastase and kallikrein 7: Total synthesis of tutuilamide A, serine protease selectivity profile and comparison with lyngbyastatin 7. Bioorg. Med. Chem. 2020, 28, 115756.
- Hafner, S.; Lang, S.J.; Gaafary, M.E.; Schmiech, M.; Simmet, T.; Syrovets, T. The cardenolide glycoside acovenoside A interferes with epidermal growth factor receptor (EGFR) trafficking in non-small cell lung cancer cells. Front. Pharmacol. 2021, 11, 611657.
- Han, A.R.; Lee, S.; Han, S.; Lee, Y.J.; Kim, J.B.; Seo, E.K.; Jung, C.H. Triterpenoids from the leaves of Centella asiatica inhibit ionizing radiation-induced migration and invasion of human lung cancer cells. Evid. Based Compl. Alt. 2020, 2020, 3683460.
- Zhang, X.; Ruan, Q.; Zhai, Y.; Lu, D.; Li, C.; Fu, Y.; Zheng, Z.; Song, Y.; Guo, J. Baicalein inhibits non-small-cell lung cancer invasion and metastasis by reducing ezrin tension in inflammation microenvironment. Cancer Sci. 2020, 111, 3802–3812.
- Zhang, Z.; Nong, L.; Chen, M.; Gu, X.; Zhao, W.; Liu, M.; Cheng, W. Baicalein suppresses vasculogenic mimicry through inhibiting RhoA/ROCK expression in lung cancer A549 cell line. Acta Bioch. Bioph. Sin. 2020, 52, 1007–1015.
- Li, J.; Yan, L.; Luo, J.; Tong, L.; Gao, Y.; Feng, W.; Wang, F.; Cui, W.; Li, S.; Sun, Z. Baicalein suppresses growth of non-small cell lung carcinoma by targeting MAP4K3. Biomed. Pharmacother. 2021, 133, 110965.
- Sui, X.; Han, X.; Chen, P.; Wu, Q.; Feng, J.; Duan, T.; Chen, X.; Pan, T.; Yan, L.; Jin, T.; et al. Baicalin induces apoptosis and suppresses the cell cycle progression of lung cancer cells through downregulating Akt/mTOR signaling pathway. Front. Mol. Biosci. 2021, 7, 602282.
- Yan, Y.; Yao, L.; Sun, H.; Pang, S.; Kong, X.; Zhao, S.; Xu, S. Effects of wogonoside on invasion and migration of lung cancer A549 cells and angiogenesis in xenograft tumors of nude mice. J. Thorac. Dis. 2020, 12, 1552–1560.
- Cheng, Z.Y.; Hsiao, Y.T.; Huang, Y.P.; Peng, S.F.; Huang, W.W.; Liu, K.C.; Hsia, T.C.; Way, T.D.; Chung, J.G. Casticin induces DNA damage and affects DNA repair associated protein expression in human lung cancer A549 cells. Molecules 2020, 25, 341.
- Cui, L.; Yang, G.; Ye, J.; Yao, Y.; Lu, G.; Chen, J.; Fang, L.; Lu, S.; Zhou, J. Dioscin elicits anti-tumour immunity by inhibiting macrophage M2 polarization via JNK and STAT3 pathways in lung cancer. J. Cell. Mol. Med. 2020, 24, 9217–9230.
- Chen, A.; Jiang, P.; Zeb, F.; Wu, X.; Xu, C.; Chen, L.; Feng, Q. EGCG regulates CTR1 expression through its pro-oxidative property in non-small-cell lung cancer cells. J. Cell. Physiol. 2020, 235, 7970–7981.
- Wei, R.; Wirkus, J.; Yang, Z.; Machuca, J.; Esparza, Y.; Mackenzie, G.G. EGCG sensitizes chemotherapeutic-induced cytotoxicity by targeting the ERK pathway in multiple cancer cell lines. Arch. Biochem. Biophys. 2020, 692, 108546.
- Duan, J.; Li, Y.; Gao, H.; Yang, D.; He, X.; Fang, Y.; Zhou, G. Phenolic compound ellagic acid inhibits mitochondrial respiration and tumor growth in lung cancer. Food Funct. 2020, 11, 6332–6339.
- Boonjing, S.; Pothongsrisit, S.; Wattanathamsan, O.; Sritularak, B.; Pongrakhananon, V. Erianthridin induces non-small cell lung cancer cell apoptosis through the suppression of extracellular signal-regulated kinase activity. Planta Med. 2021, 87, 283–293.
- Pothongsrisit, S.; Arunrungvichian, K.; Hayakawa, Y.; Sritularak, B.; Mangmool, S.; Pongrakhananon, V. Erianthridin suppresses non-small-cell lung cancer cell metastasis through inhibition of Akt/mTOR/p70 S6K signaling pathway. Sci. Rep. 2021, 11, 6618.
- Choudhury, P.; Barua, A.; Roy, A.; Pattanayak, R.; Bhattacharyya, M.; Saha, P. Eugenol emerges as an elixir by targeting β-catenin, the central cancer stem cell regulator in lung carcinogenesis: An in vivo and in vitro rationale. Food Funct. 2021, 12, 1063–1078.
- Yu, X.; Gao, F.; Li, W.; Zhou, L.; Liu, W.; Li, M. Formononetin inhibits tumor growth by suppression of EGFR-Akt-Mcl-1 axis in non-small cell lung cancer. J. Exp. Clin. Canc. Res. 2020, 39, 62.
- Yang, B.; Yang, N.; Chen, Y.; Maomao, Z.; Lian, Y.; Xiong, Z.; Wang, B.; Feng, L.; Jia, X. An integrated strategy for effective-component discovery of Astragali Radix in the treatment of lung cancer. Front. Pharmacol. 2021, 12, 580978.
- Kang, D.Y.; Sp, N.; Jo, E.S.; Rugamba, A.; Hong, D.Y.; Lee, H.G.; Yoo, J.S.; Liu, Q.; Jang, K.J.; Yang, Y.M. The inhibitory mechanisms of tumor PD-L1 expression by natural bioactive gallic acid in non-small-cell lung cancer (NSCLC) cells. Cancers 2020, 12, 727.
- Wang, D.; Bao, B. Gallic acid impedes non-small cell lung cancer progression via suppression of EGFR-dependent CARM1-PELP1 complex. Drug Des. Dev. Ther. 2020, 14, 1583–1592.
- Chen, H.; Miao, L.; Huang, F.; Yu, Y.; Peng, Q.; Liu, Y.; Li, X.; Liu, H. Glochidiol, a natural triterpenoid, exerts its anti-cancer effects by targeting the colchicine binding site of tubulin. Invest. N. Drug. 2021, 39, 578–586.
- Min, H.Y.; Pei, H.; Hyun, S.Y.; Boo, H.J.; Jang, H.J.; Cho, J.; Kim, J.H.; Son, J.; Lee, H.Y. Potent anticancer effect of the natural steroidal saponin gracillin is produced by inhibiting glycolysis and oxidative phosphorylation-mediated bioenergetics. Cancers 2020, 12, 913.
- Yang, J.; Cao, L.; Li, Y.; Liu, H.; Zhang, M.; Ma, H.; Wang, B.; Yuan, X.; Liu, Q. Gracillin isolated from Reineckia carnea induces apoptosis of A549 Cells via the mitochondrial pathway. Drug Des. Dev. Ther. 2021, 2021, 233–243.
- Lv, L.; Zhang, W.; Li, T.; Jiang, L.; Lu, X.; Lin, J. Hispidulin exhibits potent anticancer activity in vitro and in vivo through activating ER stress in non-small-cell lung cancer cells. Oncol. Rep. 2020, 43, 1995–2003.
- Zhao, X.; Lin, Y.; Jiang, B.; Yin, J.; Lu, C.; Wang, J.; Zeng, J. Icaritin inhibits lung cancer-induced osteoclastogenesis by suppressing the expression of IL-6 and TNF-a and through AMPK/mTOR signaling pathway. Anti-Cancer Drug. 2020, 31, 1004–1011.
- Lee, J.H.; Park, S.Y.; Hwang, W.; Sung, J.Y.; Shim, M.L.C.J.; Kim, Y.N.; Yoon, K. Isoharringtonine induces apoptosis of non-small cell lung cancer cells in tumorspheroids via the intrinsic pathway. Biomolecules 2020, 10, 1521.
- Fouzder, C.; Mukhuty, A.; Kundu, R. Kaempferol inhibits Nrf2 signalling pathway via downregulation of Nrf2 mRNA and induces apoptosis in NSCLC cells. Arch. Biochem. Biophys. 2021, 697, 108700.
- Sheng, H.; Lv, W.; Zhu, L.; Wang, L.; Wang, Z.; Han, J.; Hu, J. Liriopesides B induces apoptosis and cell cycle arrest in human non-small cell lung cancer cells. Int. J. Mol. Med. 2020, 46, 1039–1050.
- Chen, Y.C.; Huang, M.Y.; Zhang, L.L.; Feng, Z.L.; Jiang, X.M.; Yuan, L.W.; Huang, R.Y.; Liu, B.; Yu, H.; Wang, Y.T.; et al. Nagilactone E increases PD-L1 expression through activation of c-Jun in lung cancer cells. Chin. J. Nat. Med. 2020, 18, 517–525.
- Zhang, L.L.; Guo, J.; Jiang, X.M.; Chen, X.P.; Wang, Y.T.; Li, A.; Lin, L.G.; Li, H.; Lu, J.J. Identification of nagilactone E as a protein synthesis inhibitor with anticancer activity. Acta Pharmacol. Sin. 2020, 41, 698–705.
- Liu, X.; Zhang, Y.; Zhou, G.J.; Hou, Y.; Kong, Q.; Lu, J.J.; Zhang, Q.; Chen, X. Natural alkaloid 8-oxo-epiberberine inhibited TGF-β1-triggred epithelial-mesenchymal transition by interfering Smad3. Toxicol. Appl. Pharm. 2020, 404, 115179.
- Li, X.; Huang, R.; Li, M.; Zhu, Z.; Chen, Z.; Cui, L.; Luo, H.; Luo, L. Parthenolide inhibits the growth of non-small cell lung cancer by targeting epidermal growth factor receptor. Cancer Cell Int. 2020, 20, 561.
- Sun, L.; Yuan, W.; Wen, G.; Yu, B.; Xu, F.; Gan, X.; Tang, J.; Zeng, Q.; Zhu, L.; Chen, C.; et al. Parthenolide inhibits human lung cancer cell growth by modulating the IGF-1R/PI3K/Akt signaling pathway. Oncol. Rep. 2020, 44, 1184–1193.
- Wu, L.M.; Liao, X.Z.; Zhang, Y.; He, Z.R.; Nie, S.Q.; Ke, B.; Shi, L.; Zhao, J.F.; Chen, W.H. Parthenolide augments the chemosensitivity of non-small-cell lung cancer to cisplatin via the PI3K/AKT signaling pathway. Front. Cell Dev. Biol. 2021, 8, 610097.
- Meng, N.; Zhang, R.; Liu, C.; Wang, Q.; Wang, X.; Guo, X.; Wang, P.; Sun, J. PDB-1 from potentilla discolor bunge suppresses lung cancer cell migration and invasion via FAK/Src and MAPK signaling pathways. Med. Chem. Res. 2020, 29, 887–896.
- Wu, Y.; Si, Y.; Xiang, Y.; Zhou, T.; Liu, X.; Wu, M.; Li, W.; Zhang, T.; Xiang, K.; Zhang, L.; et al. Polyphyllin I activates AMPK to suppress the growth of non-small-cell lung cancer via induction of autophagy. Arch. Biochem. Biophys. 2020, 687, 108285.
- Lai, L.; Shen, Q.; Wang, Y.; Chen, L.; Lai, J.; Wu, Z.; Jiang, H. Polyphyllin I reverses the resistance of osimertinib in non-small cell lung cancer cell through regulation of PI3K/Akt signaling. Toxicol. Appl. Pharm. 2021, 419, 115518.
- Guo, H.; Ding, H.; Tang, X.; Liang, M.; Li, S.; Zhang, J.; Cao, J. Quercetin induces pro-apoptotic autophagy via SIRT1/AMPK signaling pathway in human lung cancer cell lines A549 and H1299 in vitro. Thoracic Cancer 2021, 12, 1415–1422.
- Xu, S.; Zhang, H.; Wang, A.; Ma, Y.; Gan, Y.; Li, G. Silibinin suppresses epithelial-mesenchymal transition in human non-small cell lung cancer cells by restraining RHBDD1. Cell. Mol. Biol. Lett. 2020, 25, 36.
- Shen, K.H.; Hung, J.H.; Liao, Y.C.; Tsai, S.T.; Wu, M.J.; Chen, P.S. Sinomenine inhibits migration and invasion of human lung cancer cell through downregulating expression of miR-21 and MMPs. Int. J. Mol. Sci. 2020, 21, 3080.
- Bai, S.; Wen, W.; Hou, X.; Wu, J.; Yi, L.; Zhi, Y.; Lv, Y.; Tan, X.; Liu, L.; Wang, P.; et al. Inhibitory effect of sinomenine on lung cancer cells via negative regulation of α7 nicotinic acetylcholine receptor. J. Leukocyte Biol. 2021, 109, 843–852.
- Liu, W.; Yu, X.; Zhou, L.; Li, J.; Li, M.; Li, W.; Gao, F. Sinomenine inhibits non-small cell lung cancer via downregulation of hexokinases II-mediated aerobic glycolysis. OncoTargets Ther. 2020, 13, 3209–3221.
- Zheng, W.; Huang, F.Y.; Dai, S.Z.; Wang, J.Y.; Lin, Y.Y.; Sun, Y.; Tan, G.H.; Huang, Y.H. Toxicarioside O inhibits cell proliferation and epithelial-mesenchymal transition by downregulation of Trop2 in lung cancer cells. Front. Oncol. 2020, 10, 609275.
- Al-Rashed, S.; Baker, A.; Ahmad, S.S.; Syed, A.; Bahkali, A.H.; Elgorban, A.M.; Khan, M.S. Vincamine, a safe natural alkaloid, represents a novel anticancer agent. Bioorg. Chem. 2021, 107, 104626.
- Gao, F.; Li, M.; Zhou, L.; Liu, W.; Zuo, H.; Li, W. Xanthohumol targets the ERK1/2-Fra1 signaling axis to reduce cyclin D1 expression and inhibit non-small cell lung cancer. Oncol. Rep. 2020, 44, 1365–1374.





