
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Angel J. Matilla | + 1030 word(s) | 1030 | 2020-06-09 08:50:16 | | | |
| 2 | Angel J. Matilla | -8 word(s) | 1022 | 2020-10-27 09:53:11 | | | | |
| 3 | Catherine Yang | Meta information modification | 1022 | 2020-10-29 03:05:29 | | | | |
| 4 | Catherine Yang | -82 word(s) | 940 | 2020-10-30 03:06:27 | | |
Video Upload Options
Phytohormone auxin acts as an outstanding coordinator of plant growth and development. Among other tasks, it has a key role as a signaling molecule that arranges seed life. Recently, auxin has emerged as an essential player that modulates the induction, regulation, and maintenance of primary seed dormancy (PSD). This function was supported by biochemical and genetic evidence. The participation of the transcriptional regulator ABSCISIC ACID (ABA) INSENSITIVE 3 (ABI3) is critical, which demonstrates a cross-talk between auxin and ABA signalings.
1. Introduction
The evolutionary success of higher plants consists of their ability to produce seeds, units responsible for reproduction, dispersal, and survival [1]. Synchronized coordination between hormone signaling networks and environmental cues are being required to control these processes [2]. The viable seed is an entity that originates at the end of the development program progression and it is constituted of three genetically different organs (i.e. endosperm, embryo, and seed-coat) [3][4]. Seed development relies on a strong interdependent control between these three respective organs. Therefore, it is not surprising that all molecular events involved in embryogenesis are tightly coordinated at the genetic and hormonal levels [5][6]. Once all seed tissues are completely differentiated, the embryogenic phase ends and begins the maturation one in which storage compounds accumulate in the endosperm (monocots) or in cotyledons (eudicots). Throughout maturation, desiccation tolerance is acquired, programmed cell death occurs, and PSD is triggered preventing vivipary [7][8][9].
2. Auxin and ABA in seed life
The phytohormone abscisic acid (ABA) is the only hormone known to induce, regulate, and maintain the PSD. Thus, seeds of ABA-deficient mutants germinate faster than the wild-type, and transgenic plants constitutively expressing the ABA biosynthesis gene maintain deep seed dormancy [1][10] . During seed development, ABA is produced in all seed organs, as suggested by the spatiotemporal expression of its biosynthesis genes [11] . ABA synthesized in the endosperm and then transported to the embryo is involved in the induction of seed dormancy [12] . Likewise, ABA shows an accumulation pattern complementary to the gibberellin (GAs), being the main hormone that inhibits all the processes induced by them [13] . On the other hand, auxin represents a key plant hormone that functions as a general coordinator in the execution of multi-functional processes during plant growth and development [14]. A series of evidence clearly relates ABA to the mechanism and mode of action of auxins [15]. Therefore, some evolutionary crosstalk must occur between both plant hormones. However, the study on the participation of auxin in the final part of seed development is not developed enough yet. The auxin is a signaling molecule that is present across all domains of life, including microorganisms [16][17][18] and tryptophan (L-Trp) serves as a common precursor for IAA synthesis in plants and auxin-producing bacteria [14][16]. Regarding the seed, it is now widely accepted that auxin biosynthesis is required for an array of seed developmental processes (e.g., zygotic embryogenesis and endosperm development, among others) [17] . High levels of free-auxins and metabolites found during both early (i.e., cell division and expansion) and last phases of seed development (e.g., endosperm cellularization) suggest that auxin has an essential signaling role [10] . Until now, the role of phytohormones in zygotic embryogenesis mainly refers to the study of eudicots such as Arabidopsis [19][20] . Likewise, the auxin also affects seed germination by altering the ABA/GAs ratio [21] . In order to generate the appropriate response, the auxin polar transport causes its accumulation in specific cellular places, being the embryogenic globular stage the most studied (Figure 1). However, very little is known about auxin biosynthesis and homeostasis, polar auxin transport, and response during early embryogenesis in monocots [22].
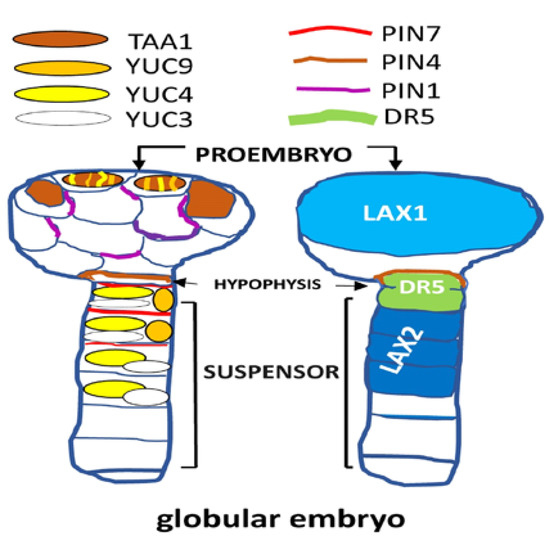
Figure 1. Dynamic of expression and localization (i.e., proembryo, hypophysis, and suspensor) corresponding to several genes for the biosynthesis and transport of auxins in Arabidopsis embryos at the globular stage. LIKE-AUX1/2 (LAX1/2); highly active synthetic auxin response element (AuxRE), is referred to as DR5; auxin efflux carrier PIN-FORMED (PIN); YUCCA (YUC); TRYPTOPHAN AMINOTRANSFERASE OF ARABIDOPSIS (TAA1).
3. Biochemical and genetic evidence supports the auxin key role
Interestingly, some genetic evidence has suggested the involvement of auxin in the maintenance of PSD in Arabidopsis. During the study of auxin involvement in plant immunity, it was evidenced that auxin protects and strictly regulates PSD through enhancing ABA signal transduction, identifying auxin as a seed dormancy promotor [23][24] . These findings were supported, among other consistent experimental verifications, by the dormancy variation among seeds with altered auxin synthesis genes. Thus, the reduced seed dormancy in taa1 and yuc1yuc6 mutants is linked to decreased ABA sensitivity [23]. However, the mechanism by which auxin controls seed dormancy is a question not yet clearly resolved at the molecular level. Parallel, strong genetic evidence supports a model whereby ABA-mediated inhibition of seed germination requires intact auxin biosynthesis, transport, and signaling. The evidence involves ABI3, a transcription factor induced by auxin [25][26] , and implicated in the initiation and maintenance of the maturation phase and considered to be a major downstream component of ABA signaling. Though genetic and biochemical evidence has been shown that ABI3 is required for auxin-activated PSD [23]. At the evolutionary level, it is interesting to note that the auxin regulatory mechanism evidenced by the He’s group [23] was later found and conserved in liverworts [27].
4. Conclusion
A striking aspect of this update lies in the participation of auxin as a key hormone, in conjunction with ABA, in the regulation of specific phases of seed life. That is why this review provides the progress made in recent years on the contribution of auxin in the fertilization process and zygotic and post-zygotic embryogenesis phases. Given the recent demonstration of auxin involvement in the PSD process, this update also considers the events that have led to this outstanding discovery.
References
- Hiroyuki Nonogaki; Seed germination and dormancy: The classic story, new puzzles, and evolution. Journal of Integrative Plant Biology 2019, 61, 541-563, 10.1111/jipb.12762.
- Gerardo Carrera-Castaño; Julián Calleja-Cabrera; Mónica Pernas; Luis Gómez; Luis Oñate-Sánchez; An Updated Overview on the Regulation of Seed Germination. Plants 2020, 9, 703, 10.3390/plants9060703.
- Olivier Coen; Enrico Magnani; Seed coat thickness in the evolution of angiosperms.. Cellular and Molecular Life Sciences 2018, 75, 2509-2518, 10.1007/s00018-018-2816-x.
- Tina Steinbrecher; Gerhard Leubner-Metzger; Tissue and cellular mechanics of seeds. Current Opinion in Genetics & Development 2018, 51, 1-10, 10.1016/j.gde.2018.03.001.
- Kai Shu; Xiao-Dong Liu; Qi Xie; Zuhua He; Two Faces of One Seed: Hormonal Regulation of Dormancy and Germination. Molecular Plant 2016, 9, 34-45, 10.1016/j.molp.2015.08.010.
- Pham A. Tuan; Rohit Kumar; Pawanpuneet K. Rehal; Parneet K. Toora; Belay T Ayele; Molecular Mechanisms Underlying Abscisic Acid/Gibberellin Balance in the Control of Seed Dormancy and Germination in Cereals. Frontiers in Plant Science 2018, 9, 668, 10.3389/fpls.2018.00668.
- Penfield, S. Seed dormancy and Germination. Curr. Biol. 2017, 27, R874–R878.
- Chahtane, H.; Kim, W.; López-Molina, L. Primary seed dormancy: A temporally multilayered riddle waiting to be unlocked. J. Exp. Bot. 2017, 68, 857–869.
- Matilla, A.J. Programmed cell death in seeds: An adaptive mechanism required for life. In Seed Dormancy and Germination; IntechOpen: London, UK, 2019.
- Seo, M.; Marion-Poll, A. Abscisic acid metabolism and transport. In Advances in Botanical Research; Elsevier Inc.: Amsterdam, The Netherlands, 2019; Volume 92, pp. 1–49. ISSN 0065-2296.
- Angel J. Matilla; Nestor Carrillo Barral; María Del Carmen Rodríguez-Gacio; An Update on the Role of NCED and CYP707A ABA Metabolism Genes in Seed Dormancy Induction and the Response to After-Ripening and Nitrate. Journal of Plant Growth Regulation 2014, 34, 274-293, 10.1007/s00344-014-9464-7.
- Hiroyuki Nonogaki; ABA responses during seed development and germination. Advances in Botanical Research 2019b, 92, 171-217, 10.1016/bs.abr.2019.04.005.
- Mine, A. Interactions between abscisic acid and other hormones. In Abscisic Acid in Plants; Seo, M., Marion-Poll, A., Eds.; Academic Press: Cambridge, MA, USA, 2019; ISBN 9780081026205.
- Leyser, O.; Auxin signaling. Plant Physiol. 2018, 176, 465–479.
- Ryan Emenecker; Lucia C Strader; Auxin-Abscisic Acid Interactions in Plant Growth and Development. Biomolecules 2020, 10, 281, 10.3390/biom10020281.
- Matilla, M.A.; Daddaoua, A.; Chini, A.; Morel, B.; Krell, T. An auxin controls bacterial antibiotics production. Nucleic Acids Res. 2018, 46, 11229–11238.
- Zhao, Y. Essential roles of local auxin biosynthesis in plant development and in adaptation to environmental changes. Annu. Rev. Plant Biol. 2018, 69, 417–435.
- Casanova-Sáez, R.; Voß, U. Auxin metabolism controls developmental decisions in land plants. Trends Plant Sci. 2019, 24, 741–754.
- Smit, M.E.; Weijers, D. The role of auxin signaling in early embryo pattern formation. Curr. Opin. Plant Biol. 2015, 28, 99–105.
- Larsson, E.; Vivian-Smith, A.; Offringa, R.; Sundberg, E. Auxin homeostasis in Arabidopsis ovules is anther-dependent at maturation and changes dynamically upon fertilization. Front. Plant Sci. 2017, 8, 1735.
- Haiwei Shuai; Yongjie Meng; Xiaofeng Luo; Feng Chen; Wenguan Zhou; Yujia Dai; Ying Qi; Junbo Du; Feng Yang; Jiang Liu; et al.Wenyu YangKai Shu Exogenous auxin represses soybean seed germination through decreasing the gibberellin/abscisic acid (GA/ABA) ratio. Scientific Reports 2017, 7, 12620, 10.1038/s41598-017-13093-w.
- Michaela Sylvia Matthes; Norman Bradley Best; Janlo M. Robil; Simon Malcomber; Andrea Gallavotti; Paula McSteen; Auxin EvoDevo: Conservation and Diversification of Genes Regulating Auxin Biosynthesis, Transport, and Signaling. Molecular Plant 2019, 12, 298-320, 10.1016/j.molp.2018.12.012.
- Liu, X.; Zhang, H.; Zhaob, Y.; Fenga, Z.; Lia, Q.; Yangc, H.Q.; Luand, S.; Lie, J.; He, Z.H. Auxin controls seed dormancy through stimulation of abscisic acid signaling by inducing ARF-mediated ABI3 activation in Arabidopsis. Proc. Natl. Acad. Sci. USA 2013a, 110, 15485–15490.
- Liu, A.; Gao, F.; Kanno, Y.; Jordan, M.C.; Kamiya, Y.; Seo, M.; Ayele, B.T. Regulation of wheat seed dormancy by after-ripening is mediated by specific transcriptional switches that induce changes in seed hormone metabolism and signaling. PLoS ONE 2013b, 8, e56570.
- Brady, S.M.; Sarkar, S.F.; Bonetta, D.; McCourt, P. The Abscisic acid insensitive 3 (ABI3) gene is modulated by farnesylation and is involved in auxin signaling and lateral root development in Arabidopsis. Plant J. 2003, 34, 67–75.
- Pellizzaro, A.; Neveu, M.; Lalanne, L.; Vu, B.L.; Kanno, Y.; Seo, M.; Leprince, O.; Buitink, J. A role for auxin signaling in the acquisition of longevity during seed maturation. New Phytol. 2020, 225, 284–296.
- D. Magnus Eklund; Kimitsune Ishizaki; Edurado Flores-Sondoval; Saya Kikuchi; Yumiko Takebayashi; Shigeyuki Tsukamoto; Yuki Hirakawa; Maiko Nonomura; Hirotaka Kato; Masaru Kouno; et al.Rishikesh P. BhaleraoUlf LagercrantzHiroyuki KasaharaTakayuki KohchiJohn L. Bowman Auxin Produced by the Indole-3-Pyruvic Acid Pathway Regulates Development and Gemmae Dormancy in the Liverwort Marchantia polymorpha[OPEN]. The Plant Cell 2015, 27, 1650-1669, 10.1105/tpc.15.00065.





