
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hideo Kimura | + 1980 word(s) | 1980 | 2021-06-22 05:47:00 | | | |
| 2 | Peter Tang | Meta information modification | 1980 | 2021-06-23 04:44:23 | | |
Video Upload Options
We discovered H2S as a signaling molecule which is produced by enzymes to modulate the synaptic transmission and relax vasculature. The cytoprotective effect, anti-inflammatory activity, energy formation, and oxygen sensing by H2S have been subsequently demonstrated. Two additional pathways for the production of H2S with 3-mercaptopyruvate sulfurtransferase (3MST) from l- and d-cysteine have been identified. We also discovered that hydrogen polysulfides (H2Sn, n ≥ 2) are potential signaling molecules produced by 3MST. H2Sn regulate the activity of ion channels and enzymes, as well as even the growth of tumors. S-Sulfuration (S-sulfhydration) proposed by Snyder is the main mechanism for H2S/H2Sn underlying regulation of the activity of target proteins.
1. Identification of H2S as a Signaling Molecule
2. Identification of H2Sn as Signaling Molecules
3. Synergy and Crosstalk between H2S and NO
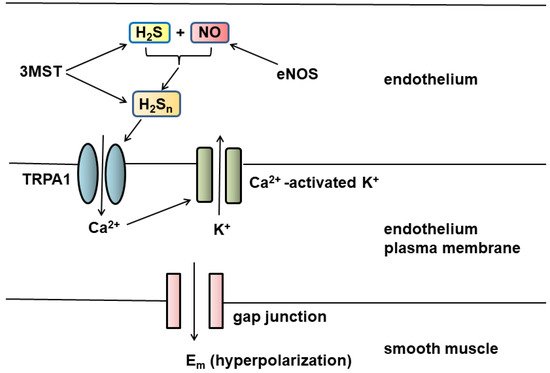
4. Vascular Tone Regulation by H2S and H2Sn
5. Cytoprotective Effect of H2S, H2Sn, and H2SO3
6. Signaling by H2S, H2Sn through S-Sulfuration and Bound Sulfane Sulfur
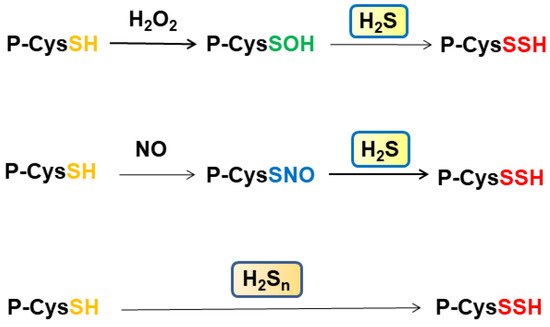
Some cysteine residues are oxidized by H2O2 to generate S-nitrosylated cysteine residues, and some others are S-nitrosylated by NO. These oxidized cysteine residues are S-sulfurated by H2S rather than H2Sn (Figure 2). Cys150 and Cys156 of GAPDH may be in the different oxidation state as described previously [26][64]. Zivanovic et al. demonstrated that the activity of manganese superoxide dismutase is suppressed through S-sulfenylation by H2O2, while the activity is recovered by H2S, which S-sulfurates the S-sulfenylated cysteine residues [65]. The same group showed that epidermal growth factor (EGF) activates its receptor in which the levels of S-sulfenylated cysteine residues are increased at the early phase, and those of S-sulfurated residues are increased at late phase when the expression of H2S producing enzymes is enhanced. H2S S-sulfurates those S-sulfenylated cysteine residues to regulate their activity (Figure 2).
Another role of S-sulfuration is that it enables proteins to recover their functions from over-oxidization. Sulfinic (Protein-CysSO2H) and sulfonic acids (Protein-CysSO3H) are not reduced back to Protein-CysSH by thioredoxin and deteriorate the protein function. In contrast, S-sulfurated proteins P-CysSSO2H and P-CysSSO3H can be reduced by thioredoxin to P-CysSH [65][66].
References
- Reiffenstein, R.J.; Hulbert, W.C.; Roth, S.H. Toxicology of hydrogen sulfide. Annu. Rev. Pharmacol. Toxic 1992, 32, 109–134.
- Warenycia, M.W.; Goodwin, L.R.; Benishin, C.G.; Reiffenstein, R.J.; Grancom, D.M.; Taylor, J.D.; Dieken, F.P. Acute hydrogen sulfide poisoning. Demonstration of selective uptake of sulfide by the brainstem by measurement of brain sulfide levels. Biochem. Pharmacol. 1989, 38, 973–981.
- Stipanuk, M.H.; Beck, P.W. Characterization of the enzymic capacity for cysteine desulphhydration in liver and kidney of the rat. Biochem. J. 1982, 206, 267–277.
- Griffith, O.W. Mammalian Sulfur Amino Acid Metabolism: An Overview. In Methods in Enzymology; Academic Press: New York, NY, USA, 1987; Volume 143, pp. 366–376.
- Abe, K.; Kimura, H. The possible role of hydrogen sulfide as an endogenous neuromodulator. J. Neurosci. 1996, 16, 1066–1071.
- O’Dell, T.J.; Hawkins, R.D.; Kandel, E.R.; Arancio, O. Tests of the roles of two diffusible substances in long-term potentiation: Evidence for nitric oxide as a possible early retrograde messenger. Proc. Natl. Acad. Sci. USA 1991, 88, 11285–11289.
- Schuman, E.M.; Madison, D.V. A requirement for the intercellular messenger nitric oxide in long-term potentiation. Science 1991, 254, 1503–1506.
- Haley, J.E.; Wilcox, G.L.; Chapman, P.F. The role of nitric oxide in hippocampal long-term potentiation. Neuron 1992, 8, 211–216.
- Stevens, C.F.; Wang, Y. Reversal of long-term potentiation by inhibitors of haem oxygenase. Nature 1993, 364, 147–149.
- Zhuo, M.; Small, S.A.; Kandel, E.R.; Hawkins, R.D. Nitric oxide and carbon monoxide produce activity-dependent long-term synaptic enhancement in hippocampus. Science 1993, 260, 1946–1950.
- Aizenman, E.; Lipton, D.A.; Loring, R.H. Selective modulation of NMDA responses by reduction and oxidation. Neuron 1989, 2, 1257–1263.
- Travis, J. The rotten smell of memory: It’s a gas. Sci. News 1996, 149, 116.
- Searcy, D.G.; Lee, S.H. Sulfur reduction by human erythrocytes. J. Exp. Zool. 1998, 282, 310–322.
- Nagai, Y.; Tsugane, M.; Oka, J.-I.; Kimura, H. Polysulfides induce calcium waves in rat hippocampal astrocytes. J. Pharmacol. Sci. 2006, 100, 200.
- Oosumi, K.; Tsugane, M.; Ishigami, M.; Nagai, Y.; Iwai, T.; Oka, J.-I.; Kimura, H. Polysulfide activates TRP channels and increases intracellular Ca2+ in astrocytes. Bull. Jpn. Soc. Neurochem. 2010, 49, 517.
- Kimura, Y.; Mikami, Y.; Osumi, K.; Tsugane, M.; Oka, J.; Kimura, H. Polysulfides are possible H2S-derived signaling molecules in rat brain. FASEB J. 2013, 27, 2451–2457.
- Kimura, Y.; Toyofuku, Y.; Koike, S.; Shibuya, N.; Nagahara, N.; Lefer, D.; Ogasawara, Y.; Kimura, H. Identification of H2S3 and H2S produced by 3-mercaptopyruvate sulfurtransferase in the brain. Sci. Rep. 2015, 5, 14774.
- Kimura, Y.; Koike, S.; Shibuya, N.; Lefer, D.; Ogasawara, Y.; Kimura, H. 3-Mercaptopyruvate sulfurtransferase produces potential redox regularots cysteine- and glutathione-persulfide (Cys-SSH and GSSH) together with signaling molecules H2S2, H2S3 and H2S. Sci. Rep. 2017, 7, 10459.
- Nagahara, N.; Koike, S.; Nirasawa, T.; Kimura, H.; Ogasawara, Y. Alternative pathway of H2S and polysulfides production from sulfurated catalytic-cysteine of reaction intermediates of 3-mercaptopyruvate sulfurtransferase. Biochem. Biophys. Res. Commun. 2018, 496, 648–653.
- Miyamoto, R.; Koike, S.; Takano, Y.; Shibuya, N.; Kimura, Y.; Hanaoka, K.; Urano, Y.; Ogasawara, Y.; Kimura, H. Polysulfides (H2Sn) produced from the interaction of hydrogen sulfide (H2S) and nitric oxide (NO) activate TRPA1 channels. Sci. Rep. 2017, 7, 45995.
- Kharma, A.; Grman, M.; Misak, A.; Domínguez-Álvarez, E.; Nasim, M.J.; Ondrias, K.; Chovanec, M.; Jacob, C. Inorganic Polysulfides and Related Reactive Sulfur–Selenium Species from the Perspective of Chemistry. Molecules 2019, 24, 1359.
- Koike, S.; Kawamura, K.; Kimura, Y.; Shibuya, N.; Kimura, H.; Ogasawara, Y. Analysis of endogenous H2S and H2Sn in mouse brain by high-performance liquid chromatography with fluorescence and tandem mass spectrometric detection. Free Radic. Biol. Med. 2017, 113, 355–362.
- Nagai, Y.; Tsugane, M.; Oka, J.; Kimura, H. Hydrogen sulfide induces calcium waves in astrocytes. FASEB J. 2004, 18, 557–559.
- Wang, L.; Cvetkov, T.L.; Chance, M.R.; Moiseenkova-Bell, V.Y. Identification of in Vivo Disulfide Conformation of TRPA1 Ion Channel. J. Biol. Chem. 2012, 287, 6169–6176.
- Hatakeyama, Y.; Takahashi, K.; Tominaga, M.; Kimura, H.; Ohta, T. Polysulfide Evokes Acute Pain through the Activation of Nociceptive TRPA1 in Mouse Sensory Neurons. Mol. Pain 2015, 11, 24.
- Mustafa, A.K.; Gadalla, M.M.; Sen, N.; Kim, S.; Mu, W.; Gazi, S.K.; Barrow, R.K.; Yang, G.; Wang, R.; Snyder, S.H. H2S Signals Through Protein S-Sulfhydration. Sci. Signal. 2009, 2, ra72.
- Mishanina, T.V.; Libiad, M.; Banerjee, R. Biogenesis of reactive sulfur species for signaling by hydrogen sulfide oxidation pathways. Nat. Chem. Biol. 2015, 11, 457–464.
- Hosoki, R.; Matsuki, N.; Kimura, H. The Possible Role of Hydrogen Sulfide as an Endogenous Smooth Muscle Relaxant in Synergy with Nitric Oxide. Biochem. Biophys. Res. Commun. 1997, 237, 527–531.
- Teague, B.; Asiedu, S.; Moore, P.K. The smooth muscle relaxant effect of hydrogen sulphide in vitro: Evidence for a physiological role to control intestinal contractility. Br. J. Pharmacol. 2002, 137, 139–145.
- Whiteman, M.; Li, L.; Kostetski, I.; Chu, S.H.; Siau, J.L.; Bhatia, M.; Moore, P.K. Evidence for the formation of a novel nitrosothiol from the gaseous mediators nitric oxide and hydrogen sulphide. Biochem. Biophys. Res. Commun. 2006, 343, 303–310.
- Filipovic, M.R.; Miljkovic, J.; Allgauer, A.; Chaurio, R.; Shubina, T.; Herrmann, M.; Ivanovic-Burmazovic, I. Biochemical insight into physiological effects of H2S: Reaction with peroxynitrite and formation of a new nitric oxide donor, sulfinyl nitrite. Biochem. J. 2012, 441, 609–621.
- Eberhardt, M.; Dux, M.; Namer, B.; Miljkovic, J.; Cordasic, N.; Will, C.; Kichko, T.I.; De La Roche, J.; Fischer, M.J.; Suárez, S.A.; et al. H2S and NO cooperatively regulate vascular tone by activating a neuroendocrine HNO–TRPA1–CGRP signaling pathway. Nat. Commun. 2014, 5, 4381.
- Cortese-Krott, M.M.; Kuhnle, G.G.C.; Dyson, A.; Fernandez, B.O.; Grman, M.; Dumond, J.F.; Barrow, M.P.; McLeod, G.; Nakagawa, H.; Ondrias, K.; et al. Key bioactive reaction products of the NO/H2S interaction are S/N-hybrid species, polysulfides, and nitroxyl. Proc. Natl. Acad. Sci. USA 2015, 112, E4651–E4660.
- Bogdándi, V.; Ditrói, T.; Bátai, I.Z.; Sándor, Z.; Minnion, M.; Vasas, A.; Galambos, K.; Buglyó, P.; Pintér, E.; Feelisch, M.; et al. Nitrosopersulfide (SSNO−) Is a Unique Cysteine Polysulfidating Agent with Reduction-Resistant Bioactivity. Antioxid. Redox Signal. 2020, 33, 1277–1294.
- Zhao, W.; Zhang, J.; Lu, Y.; Wang, R. The vasorelaxant effect of H2S as a novel endogenous gaseous KATP channel opener. EMBO J. 2001, 20, 6008–6016.
- Mustafa, A.K.; Sikka, G.; Gazi, S.K.; Steppan, J.; Jung, S.M.; Bhunia, A.K.; Barodka, V.M.; Gazi, F.K.; Barrow, R.K.; Wang, R.; et al. Hydrogen Sulfide as Endothelium-Derived Hyperpolarizing Factor Sulfhydrates Potassium Channels. Circ. Res. 2011, 109, 1259–1268.
- Chen, G.F.; Cheung, D.W. Characterization of acetylcholine-induced membrane hyperpolarization in endothelial cells. Circ. Res. 1992, 70, 257–263.
- Eckman, D.M.; Frankovich, J.D.; Keef, K.D. Comparison of the actions of acetylcholine and BRL 38227 in the guinea-pig coronary artery. Br. J. Pharmacol. 1992, 106, 9–16.
- Garland, C.; Plane, F.; Kemp, B.K.; Cocks, T.M. Endothelium-dependent hyperpolarization: A role in the control of vascular tone. Trends Pharmacol. Sci. 1995, 16, 23–30.
- Shibuya, N.; Mikami, Y.; Kimura, Y.; Nagahara, N.; Kimura, H. Vascular Endothelium Expresses 3-Mercaptopyruvate Sulfurtransferase and Produces Hydrogen Sulfide. J. Biochem. 2009, 146, 623–626.
- Shibuya, N.; Tanaka, M.; Yoshida, M.; Ogasawara, Y.; Togawa, T.; Ishii, K.; Kimura, H. 3-Mercaptopyruvate Sulfurtransferase Produces Hydrogen Sulfide and Bound Sulfane Sulfur in the Brain. Antioxid. Redox Signal. 2009, 11, 703–714.
- Earley, S. TRPA1 channels in the vasculature. Br. J. Pharmacol. 2012, 167, 13–22.
- Kimura, Y.; Kimura, H. Hydrogen sulfide protects neurons from oxidative stress. FASEB J. 2004, 18, 1165–1167.
- Kimura, Y.; Goto, Y.-I.; Kimura, H. Hydrogen Sulfide Increases Glutathione Production and Suppresses Oxidative Stress in Mitochondria. Antioxid. Redox Signal. 2010, 12, 1–13.
- Kimura, Y.; Dargusch, R.; Schubert, D.; Kimura, H. Hydrogen Sulfide Protects HT22 Neuronal Cells from Oxidative Stress. Antioxid. Redox Signal. 2006, 8, 661–670.
- Elrod, J.W.; Calvert, J.W.; Morrison, J.; Doeller, J.E.; Kraus, D.W.; Tao, L.; Jiao, X.; Scalia, R.; Kiss, L.; Szabo, C.; et al. Hydrogen sulfide attenuates myocardial ischemia-reperfusion injury by preservation of mitochondrial function. Proc. Natl. Acad. Sci. USA 2007, 104, 15560–15565.
- Cooper, A.J.L. Biochemistry of Sulfur-Containing Amino Acids. Annu. Rev. Biochem. 1983, 52, 187–222.
- Shibuya, N.; Koike, S.; Tanaka, M.; Ishigami-Yuasa, M.; Kimura, Y.; Ogasawara, Y.; Fukui, K.; Nagahara, N.; Kimura, H. A novel pathway for the production of hydrogen sulfide from D-cysteine in mammalian cells. Nat. Commun. 2013, 4, 1366.
- Hylin, J.W.; Wood, J.L. Enzymatic Formation of Polysulfides from Mercaptopyruvate. J. Biol. Chem. 1959, 234, 2141–2144.
- Landry, A.P.; Ballou, D.P.; Banerjee, R. H2S oxidation by nanodisc-embedded human sulfide quinone oxidoreductase. J. Biol. Chem. 2017, 292, 11641–11649.
- Vitvitsky, V.; Kabil, O.; Banerjee, R. High Turnover Rates for Hydrogen Sulfide Allow for Rapid Regulation of Its Tissue Concentrations. Antioxid. Redox Signal. 2012, 17, 22–31.
- Searcy, D.; Whitehead, J.; Maroney, M. Interaction of Cu, Zn Superoxide Dismutase with Hydrogen Sulfide. Arch. Biochem. Biophys. 1995, 318, 251–263.
- Searcy, D.G. HS−: O2 oxidoreductase activity of Cu, Zn superoxide dismutase. Arch. Biochem. Biophys. 1996, 334, 50–58.
- Olson, K.R.; Gao, Y.; Arif, F.; Arora, K.; Patel, S.; DeLeon, E.R.; Sutton, T.R.; Feelisch, M.; Cortese-Krott, M.; Straub, K.D. Metabolism of hydrogen sulfide (H2S) and Production of Reactive Sulfur Species (RSS) by superoxide dismutase. Redox Biol. 2018, 15, 74–85.
- Olson, K.R.; Gao, Y.; DeLeon, E.R.; Arif, M.; Arif, F.; Arora, N.; Straub, K.D. Catalase as a sulfide-sulfur oxido-reductase: An ancient (and modern?) regulator of reactive sulfur species (RSS). Redox Biol. 2017, 12, 325–339.
- Akaike, T.; Ida, T.; Wei, F.-Y.; Nishida, M.; Kumagai, Y.; Alam, M.; Ihara, H.; Sawa, T.; Matsunaga, T.; Kasamatsu, S.; et al. Cysteinyl-tRNA synthetase governs cysteine polysulfidation and mitochondrial bioenergetics. Nat. Commun. 2017, 8, 1177.
- Nakamura, S.; Nakamura, M.; Yamazaki, I.; Morrison, M. Reactions of ferryl lactoperoxidase (compound II) with sulfide and sulfhydryl compounds. J. Biol. Chem. 1984, 259, 7080–7085.
- Garai, D.; Ríos-González, B.B.; Furtmüller, P.G.; Fukuto, J.M.; Xian, M.; López-Garriga, J.; Obinger, C.; Nagy, P. Mechanisms of myeloperoxidase catalyzed oxidation of H2S by H2O2 or O2 to produce potent protein Cys–polysulfide-inducing species. Free Radic. Biol. Med. 2017, 113, 551–563.
- Warenycia, M.W.; Goodwin, L.R.; Francom, D.M.; Dieken, F.P.; Kombian, S.B.; Reiffenstein, R.J. Dithiothreitol liberates non-acid labile sulfide from brain tissue of H2S-poisoned animals. Arch. Toxicol. 1990, 64, 650–655.
- Ogasawara, Y.; Ishii, K.; Togawa, T.; Tanabe, S. Determination of Bound Sulfur in Serum by Gas Dialysis/High-Performance Liquid Chromatography. Anal. Biochem. 1993, 215, 73–81.
- Ogasawara, Y.; Isoda, S.; Tanabe, S. Tissue and Subcellular Distribution of Bound and Acid-Labile Sulfur, and the Enzymic Capacity for Sulfide Production in the Rat. Biol. Pharm. Bull. 1994, 17, 1535–1542.
- Ishigami, M.; Hiraki, K.; Umemura, K.; Ogasawara, Y.; Ishii, K.; Kimura, H. A Source of Hydrogen Sulfide and a Mechanism of Its Release in the Brain. Antioxid. Redox Signal. 2009, 11, 205–214.
- Go, Y.-M.; Jones, D.P. The Redox Proteome. J. Biol. Chem. 2013, 288, 26512–26520.
- Jarosz, A.P.; Wei, W.; Gauld, J.W.; Auld, J.; Özcan, F.; Aslan, M.; Mutus, B. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) is inactivated by S-sulfuration in vitro. Free Radic. Biol. Med. 2015, 89, 512–521.
- Zivanovic, J.; Kouroussis, E.; Kohl, J.B.; Adhikari, B.; Bursac, B.; Schott-Roux, S.; Petrovic, D.; Miljkovic, J.L.; Thomas-Lopez, D.; Jung, Y.; et al. Selective Persulfide Detection Reveals Evolutionarily Conserved Antiaging Effects of S-Sulfhydration. Cell Metab. 2019, 30, 1152–1170.e13.
- Nagahara, N.; Nirasawa, T.; Yoshii, T.; Niimura, Y. Is Novel Signal Transducer Sulfur Oxide Involved in the Redox Cycle of Persulfide at the Catalytic Site Cysteine in a Stable Reaction Intermediate of Mercaptopyruvate Sulfurtransferase? Antioxid. Redox Signal. 2012, 16, 747–753.





