
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Daniel Grande | + 1733 word(s) | 1733 | 2021-05-13 11:53:47 | | | |
| 2 | Lindsay Dong | Meta information modification | 1733 | 2021-06-18 03:33:27 | | |
Video Upload Options
Osteoarthritis of the knee is one of the most common chronic, debilitating musculoskeletal conditions. Exosomes derived from non-classical sources (not bone marrow or adipose derived MSCs derived MSCs) for osteoarthritis of the knee have potential clinical use.
1. Introduction
Osteoarthritis (OA) is a leading cause of chronic disability and pain with an estimated nearly 9.6% of men and 18% of women over the age of 60 across the world suffering from OA [1][2]. Current conservative management consisting of non-steroid anti-inflammatory drugs (NSAIDs), intra-articular glucocorticoid injections, physical therapy, and more are often unsatisfactory. Currently there are no drugs that directly treat the disease to prevent the pathologic progression to its end stage, to which the only treatment is total knee arthroplasty (TKA). Due to the lack of curative therapy, TKA had its utilization rates more than double from 1999 to 2008 [3]. Most importantly, an estimated nearly more than $27 billion in healthcare costs annually can be attributed to knee OA [4][5][6]. Given such large healthcare expenditures and current inadequate multimodal treatment regimens, developing novel non-surgical or minimally invasive therapeutics is critical.
One growing area of research regarding alternative methods for treating OA has been focused on the use of mesenchymal stem cells (MSCs) due to their ability to proliferate and differentiate into other cell lineages, thereby potentially regenerating cartilage, bone, and other structures [7][8]. MSCs have been shown both in vivo and in vitro to play an important role in protecting against the development of OA or articular cartilage destruction via anti-apoptotic activity, anti-fibrotic activity, promoting chondrogenesis, immunosuppression, regulating metabolic activity, and more [9]. Contrary to conventional thoughts, further investigations with MSCs showed that many of its beneficial effects in OA likely involve paracrine signaling, of which one is via the release of extracellular vesicles (EVs) [10][11]. A subtype of these EVs are exosomes, 30-150 nm diameter particles secreted from cells for intercellular communication that carry proteins, lipids, nucleic acids (including mRNAs and microRNAs (miRNA)), and more [12]. As such, many investigations are currently in progress to better understand the roles and mechanisms by which exosomes function and the potential for them to be used clinically in OA and other pathologies.
There are multiple advantages and properties of exosomes compared to MSCs that improve the likelihood of them being translated clinically and thus the recent rapid rise in the amount of literature about them. For example, given the small size and hypoimmunogenic nature of exosomes, there is a lower likelihood of rejection [13][14]. Additionally, as exosomes are non-viable, lower costs are involved in their maintenance and storage relative to MSCs, as cells need to be maintained in a viable state [13][14]. Furthermore, exosomes have the ability to cross the blood-brain barrier, opening the possibility for any therapies targeting the central nervous system or for future use as drug-delivery vehicles [15].
2. Exosomes Derived from Non-Classic Sources for Treatment of Osteoarthritis
2.1. Synovial Derived Exosomes
Given that the knee joint is lined by a thin layer of synovial membrane, the function and downstream effects of EVs produced by the synovial membrane on nearby structures have been of keen interest. Several recent in vivo studies have shown promising results, with all having a relatively similar methodology, as listed in Table 1. OA was induced in all mice models via transection of the medial collateral ligament (MCL), medial meniscus, and anterior collateral ligament (ACL). All three studies exhibited the beneficial effects of SMSCs-Exos in OA models.
Table 1. In vivo studies with exosomes derived from non-classic sources for knee osteoarthritis and cartilage injuries.
| Source | Dose/Volume | Animal | Animal Model | Results | Reference |
|---|---|---|---|---|---|
| Platelet-Rich Plasma | 100 μg/mL | Rabbits | Transection of MCL, medial meniscus, ACL | Reversed the decrease in collagen II and RUNX2 protein expression, promoted cartilage repair, inhibited OA | Liu et al., 2019 [16] |
| Infrapatellar fat pad | 10 μL 1010 particles/mL |
Mice | Destabilization of the medial meniscus (DMM) surgery | Alleviate articular cartilage damage and improve gait, likely via miR-100-5p downregulation of mTOR | Wu et al., 2019 [17] |
| Synovial Membrane | 100 μL 1011 particles/mL |
Rats | Transection of MCL, medial meniscus, ACL | Enhance cartilage tissue regeneration and prevent OA | Tao et al., 2017 [18] |
| Synovial Membrane | 5 μL particles/mL | Mice | Transection of MCL, medial meniscus, ACL | Reduced cartilage damage and restored structure of cartilage surface | Wang et al., 2020 [19] |
| Synovial Membrane | 30 μL 1011 particles/mL |
Mice | Transection of MCL, medial meniscus, ACL | Prevent OA, promote cartilage regeneration and improve articular cartilage damage | Wang et al., 2020 [20] |
| Umbilical cord | 500 μL 1010 particles/mL |
Rabbits | rabbit cartilage defect model via surgery at the trochlear grooves of the distal femur | Repair cartilage defects via promoting migration and proliferation of chondrocytes | Yan and Wu, 2019 [21] |
| Umbilical Cord | 100 μL 1 mg/mL |
Rats | Distal femur cartilage defect | Improved cartilage defects via increased collagen II secretion and matrix synthesis, possibly involving lncRNA H19 | Yan et al., 2020 [22] |
| Umbilical Cord | 200 μl injection 1 mg/mL |
Rats | Unilateral cartilage defect on the femoral trochelear groove | Improve cartilage repair via lncRNA H19/miR-29b-3p/FoxO3 axis | Yan et al., 2021 [23] |
| Human Embryonic Stem Cell | 100 µg exosomes per 100 μL injection | Rats | Osteochondral defects surgically created at trochlear grove of distal femur | Better hyaline cartilage formation and subchondral bone regeneration | Zhang et al., 2016 [11] |
| Human Embryonic Stem Cell | 100 µg exosomes per 100 μL injection | Rats | Osteochondral defects surgically created at trochlear grove of distal femur | Regulate apoptotic and cellular proliferation genes | Zhang et al., 2018 [24] |
| Human Embryonic Stem Cell | 5 µL exosomes | Mice | Destabilization of the medial meniscus (DMM) surgery | Modulate cartilage extracellular matrix synthesis and degradation | Wang et al., 2017 [25] |
| Amniotic Fluid | 100 µg exosomes in 50 µL | Rats | Monoiodoacetate (MIA) injections | Protection from cartilage damage | Zavatti et al., 2019 [26] |
| Tendon, Ligament, Subchondral | - | - | - | - | - |
Abbreviations: MCL, medial collateral ligament; ACL, anterior cruciate ligament.
2.2. Infra-patellar Fat Pad Derived Exosomes
IPFP could serve as useful source of regenerative cells for cartilae repair as evidence shows it is responsible for some spontaneous joint repair in OA [27], and MSCs from the IPFP possess significnt chondrogenic potential [28][29]. Both MSCs and EVs including exosomes that are derived from IPFPs have been of interest for OA patients. Early success with IPFP-derived MSCs, as reviewed by Huri et al., spurred interest in IPFP-MSC derived exosomes (IPFP-Exos) [30]. Thus far, there has been only one in vivo study with IPFP-Exos for studying knee OA by Wu et al. [20].
2.3. PRP Derived Exosomes
Platelet-rich plasma (PRP) is human plasma that has an increased concentration of platelets. PRP has been of high interest due to platelets carrying large amounts of growth factors and anti-inflammatory cytokines that promote cartilage extracellular matrix sythesis and directly inhibit the expression of MMPs and other known mediators of OA [31]. However, due to the large variation in bioactive components, PRP efficacy as an OA treatment is heterogenous. For example, PRP was found in recent meta-analyses to be overall effective in releaving knee OA related pain, but in several studies the results are not statistically significant or clinically meaningful [32][33][34]. The concentrations of bioactive proteins vary largely depending on numerous factors, such as platelet, volume of PRP, use of activating factors, and residual red and white blood cell count. Thus, this variation can be eliminated by isolating and applying only the bioactive proteins, which are released via the secretion of exosomess. Prior research suggests that exosomes at least partially mediate the effects of PRP in certain processes such as for reepithelialization of chronic cutaneous wounds and preventing GC-induced apoptosis in a rat model of osteonecrosis of the femoral head, but the exact mechanisms still remain unclear, especially in knee OA [35][36][37].
2.4. Amniotic Fluid Stem Cell (AFSC)-Derived Exosomes
Zavatti et al. created a rat OA model using monoiodoacetate (MIA) injections and then treated these mice 3 weeks after with 100 ug exosomes (on the basis that this is the amount of exosomes produced by 500,000 AFSC cells) [17]. OA mice treated with AFSC-Exos showed a significantly lower OARSI score, greater percentage of cartilage covering the joint surface on histology, less fibrous-connective tissue covering the joint surface, and greater pain tolerance compared to MIA-induced OA mice that were not treated with AFSC-Exos [17].
2.5. Umbilical cord MSCs-Derived Exosomes
Umbilical cord MSC-derived exosomes (UMSC-Exos) have also been of high interest, especially due to a recent study showing that UMSCs are able to generate four times more exosomes per cell than BMSCs or AMSCs [38].To our knowledge, three in vivo studies with UMSC-Exos for knee cartilage defects has been conducted to date [21][24][26].
2.6. Embryonic Stem Cell-induced MSCs Derived Exosomes
Many advancements in understanding the role of exosomes derived from human embryonic mesenchymal stem cells (human eMSCs-Exos) in OA of the knee have been made, as several in vivo studies have been completed successfully with comparisons between their methodology listed in Table 1. One of the first in vivo studies using human eMSCs-Exos for OA was by Zhang et al., where osteochondral defects were created surgically in the trochlear groove of the distal femur in a rat model, following the surgical procedure, 100 ug exosomes were administered weekly for 12 weeks [11]. At the end of 12 weeks, human eMSCs-Exos treated rats had better hyaline cartilage formation and subchondral bone regeneration with good surface regularity compared to control, defect knees treated with PBS only, which primarily continued to show fibrous and non-cartilaginous tissue [11].
3. Future Directions
Moving forward, many aspects of exosome research still need to be thoroughly explored, but in particular is the contents of exosomes, including any proteins, RNAs, small RNAs, miRNAs, and more. As mentioned in several studies earlier, many of the downstream therapeutic effects of exosomes are likely due to these contents targeting the expression of certain genes at the target cell.
In moving towards clinical use, many challenges in using exosomes still exist, including but not limited to dosage, frequency, safety, kinetics, and control batch differences. For exosomes derived from different sources, finding the ideal concentrations and optimal frequency of exosome administration in animal models and humans needed for therapeutic effects is critical. Likewise, standardization of the isolation, extraction, and storage processes is needed as well. In regards to safety, an early study has shown transplantation of human UMSC-Exos to be safe in animal models [39], and others have reported minimal immunogenicity of MSC-derived exosomes [40][41]. Nevertheless, study of immunogenicity of exosomes from an allogenic source might be necessary. More similar studies using the aforementioned exosomal sources is needed. As much of our current research and understanding is in the early stage, there remains much time until regulatory guidelines for exosomes can be properly established by the Food and Drug Administration (FDA). Some of the key considerations regarding the future directions of exosomes as therapies are summarized in Figure 1.
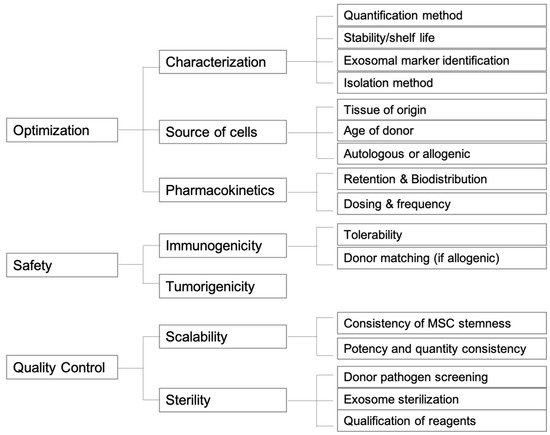
Figure 1. Graphical scheme of future directions for exosome therapeutic products.
References
- Maiese, K. Picking a bone with WISP1 (CCN4): New strategies against degenerative joint disease. J. Transl. Sci. 2016, 1, 83–85.
- Grazio, S.C.; Balen, D. Obesity: Risk factor and predictor of osteoarthritis [Debljina: imbenik rizika i prediktor razvoja osteoartritisa]. Lijec. Vjesn. 2009, 131, 22–26.
- Losina, E.; Thornhill, T.S.; Rome, B.N.; Wright, J.; Katz, J.N. The dramatic increase in total knee replacement utilization rates in the United States cannot be fully explained by growth in population size and the obesity epidemic. J. Bone Jt. Surg. Ser. A 2012, 94, 201–207.
- Losina, E.; Paltiel, A.D.; Weinstein, A.M.; Yelin, E.; Hunter, D.J.; Chen, S.P.; Klara, K.; Suter, L.G.; Solomon, D.H.; Burbine, S.A.; et al. Lifetime medical costs of knee osteoarthritis management in the United States: Impact of extending indications for total knee arthroplasty. Arthritis Care Res. 2015, 67, 203–215.
- Murphy, L.; Helmick, C.G. The Impact of osteoarthritis in the United States: A population-health perspective: A population-based review of the fourth most common cause of hospitalization in U.S. adults. Orthop. Nurs. 2012, 31, 85–91.
- Ong, K.L.; Runa, M.; Lau, E.; Altman, R.D. Cost-of-illness of knee osteoarthritis: Potential cost savings by not undergoing arthroplasty within the first 2 years. Clin. Outcomes Res. 2019, 11, 245–255.
- Smirnov, S.V.; Harbacheuski, R.; Lewis-Antes, A.; Zhu, H.; Rameshwar, P.; Kotenko, S.V. Bone-marrow-derived mesenchymal stem cells as a target for cytomegalovirus infection: Implications for hematopoiesis, self-renewal and differentiation potential. Virology 2007, 360, 6–16.
- Crapnell, K.; Blaesius, R.; Hastings, A.; Lennon, D.P.; Caplan, A.I.; Bruder, S.P. Growth, differentiation capacity, and function of mesenchymal stem cells expanded in serum-free medium developed via combinatorial screening. Exp. Cell Res. 2013, 319, 1409–1418.
- Mancuso, P.; Raman, S.; Glynn, A.; Barry, F.; Murphy, J.M. Mesenchymal stem cell therapy for osteoarthritis: The critical role of the cell secretome. Front. Bioeng. Biotechnol. 2019, 7, 9.
- Qi, X.; Zhang, J.; Yuan, H.; Xu, Z.; Li, Q.; Niu, X.; Hu, B.; Wang, Y.; Li, X. Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells repair critical-sized bone defects through enhanced angiogenesis and osteogenesis in osteoporotic rats. Int. J. Biol. Sci. 2016, 12, 836.
- Zhang, S.; Chu, W.C.; Lai, R.C.; Lim, S.K.; Hui, J.H.P.; Toh, W.S. Exosomes derived from human embryonic mesenchymal stem cells promote osteochondral regeneration. Osteoarthr. Cartil. 2016, 24, 2135–2140.
- Nikfarjam, S.; Rezaie, J.; Zolbanin, N.M.; Jafari, R. Mesenchymal stem cell derived-exosomes: A modern approach in translational medicine. J. Transl. Med. 2020, 18.
- Elahi, F.M.; Farwell, D.G.; Nolta, J.A.; Anderson, J.D. Preclinical translation of exosomes derived from mesenchymal stem/stromal cells. Stem Cells 2020, 38, 15–21.
- Chen, T.S.; Arslan, F.; Yin, Y.; Tan, S.S.; Lai, R.C.; Choo, A.B.H.; Padmanabhan, J.; Lee, C.N.; de Kleijn, D.P.V.; Lim, S.K. Enabling a robust scalable manufacturing process for therapeutic exosomes through oncogenic immortalization of human ESC-derived MSCs. J. Transl. Med. 2011, 9.
- Chen, C.C.; Liu, L.; Ma, F.; Wong, C.W.; Guo, X.E.; Chacko, J.V.; Farhoodi, H.P.; Zhang, S.X.; Zimak, J.; Ségaliny, A.; et al. Elucidation of Exosome Migration Across the Blood–Brain Barrier Model In Vitro. Cell. Mol. Bioeng. 2016, 9, 509–529.
- Liu, X.; Wang, L.; Ma, C.; Wang, G.; Zhang, Y.; Sun, S. Exosomes derived from platelet-rich plasma present a novel potential in alleviating knee osteoarthritis by promoting proliferation and inhibiting apoptosis of chondrocyte via Wnt/β-catenin signaling pathway. J. Orthop. Surg. Res. 2019, 14, 470.
- Zavatti, M.; Beretti, F.; Casciaro, F.; Bertucci, E.; Maraldi, T. Comparison of the therapeutic effect of amniotic fluid stem cells and their exosomes on monoiodoacetate-induced animal model of osteoarthritis. BioFactors 2020, 46, 106–117.
- Wang, Y.; Yu, D.; Liu, Z.; Zhou, F.; Dai, J.; Wu, B.; Zhou, J.; Heng, B.C.; Zou, X.H.; Ouyang, H.; et al. Exosomes from embryonic mesenchymal stem cells alleviate osteoarthritis through balancing synthesis and degradation of cartilage extracellular matrix. Stem Cell Res. Ther. 2017, 8, 189.
- Tao, S.C.; Yuan, T.; Zhang, Y.L.; Yin, W.J.; Guo, S.C.; Zhang, C.Q. Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics 2017, 7, 180–195.
- Wu, J.; Kuang, L.; Chen, C.; Yang, J.; Zeng, W.N.; Li, T.; Chen, H.; Huang, S.; Fu, Z.; Li, J.; et al. miR-100-5p-abundant exosomes derived from infrapatellar fat pad MSCs protect articular cartilage and ameliorate gait abnormalities via inhibition of mTOR in osteoarthritis. Biomaterials 2019, 206, 87–100.
- Yan, L.; Liu, G.; Wu, X. Exosomes derived from umbilical cord mesenchymal stem cells in mechanical environment show improved osteochondral activity via upregulation of LncRNA H19. J. Orthop. Transl. 2020, 26, 111–120.
- Wang, Z.; Yan, K.; Ge, G.; Zhang, D.; Bai, J.; Guo, X.; Zhou, J.; Xu, T.; Xu, M.; Long, X.; et al. Exosomes derived from miR-155-5p–overexpressing synovial mesenchymal stem cells prevent osteoarthritis via enhancing proliferation and migration, attenuating apoptosis, and modulating extracellular matrix secretion in chondrocytes. Cell Biol. Toxicol. 2020.
- Wang, K.; Li, F.; Yuan, Y.; Shan, L.; Cui, Y.; Qu, J.; Lian, F. Synovial Mesenchymal Stem Cell-Derived EV-Packaged miR-31 Downregulates Histone Demethylase KDM2A to Prevent Knee Osteoarthritis. Mol. Ther. Nucleic Acids 2020, 22, 1078–1091.
- Yan, L.; Liu, G.; Wu, X. The umbilical cord mesenchymal stem cell-derived exosomal lncRNA H19 improves osteochondral activity through miR-29b-3p/FoxO3 axis. Clin. Transl. Med. 2021, 11, e255.
- Zhang, S.; Chuah, S.J.; Lai, R.C.; Hui, J.H.P.; Lim, S.K.; Toh, W.S. MSC exosomes mediate cartilage repair by enhancing proliferation, attenuating apoptosis and modulating immune reactivity. Biomaterials 2018, 156, 16–27.
- Yan, L.; Wu, X. Exosomes produced from 3D cultures of umbilical cord mesenchymal stem cells in a hollow-fiber bioreactor show improved osteochondral regeneration activity. Cell Biol. Toxicol. 2020, 36, 165–178.
- English, A.; Jones, E.A.; Corscadden, D.; Henshaw, K.; Chapman, T.; Emery, P.; McGonagle, D. A comparative assessment of cartilage and joint fat pad as a potential source of cells for autologous therapy development in knee osteoarthritis. Rheumatology 2007, 46, 1676–1683.
- Buckley, C.T.; Vinardell, T.; Kelly, D.J. Oxygen tension differentially regulates the functional properties of cartilaginous tissues engineered from infrapatellar fat pad derived MSCs and articular chondrocytes. Osteoarthr. Cartil. 2010, 18, 1345–1354.
- Luo, L.; Thorpe, S.D.; Buckley, C.T.; Kelly, D.J. The effects of dynamic compression on the development of cartilage grafts engineered using bone marrow and infrapatellar fat pad derived stem cells. Biomed. Mater. 2015, 10, 055011.
- Huri, P.Y.; Hamsici, S.; Ergene, E.; Huri, G.; Doral, M.N. Infrapatellar Fat Pad-Derived Stem Cell-Based Regenerative Strategies in Orthopedic Surgery. Knee Surg. Relat. Res. 2018, 30, 179–186.
- Sundman, E.A.; Cole, B.J.; Karas, V.; Della Valle, C.; Tetreault, M.W.; Mohammed, H.O.; Fortier, L.A. The anti-inflammatory and matrix restorative mechanisms of platelet-rich plasma in osteoarthritis. Am. J. Sports Med. 2014, 42, 35–41.
- Khoshbin, A.; Leroux, T.; Wasserstein, D.; Marks, P.; Theodoropoulos, J.; Ogilvie-Harris, D.; Gandhi, R.; Takhar, K.; Lum, G.; Chahal, J. The efficacy of platelet-rich plasma in the treatment of symptomatic knee osteoarthritis: A systematic review with quantitative synthesis. Arthrosc. J. Arthrosc. Relat. Surg. Off. Publ. Arthrosc. Assoc. N. Am. Int. Arthrosc. Assoc. 2013, 29, 2037–2048.
- Belk, J.W.; Kraeutler, M.J.; Houck, D.A.; Goodrich, J.A.; Dragoo, J.L.; McCarty, E.C. Platelet-Rich Plasma Versus Hyaluronic Acid for Knee Osteoarthritis: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Am. J. Sports Med. 2021, 49, 249–260.
- Phillips, M.; Vannabouathong, C.; Devji, T.; Patel, R.; Gomes, Z.; Patel, A.; Dixon, M.; Bhandari, M. Differentiating factors of intra-articular injectables have a meaningful impact on knee osteoarthritis outcomes: A network meta-analysis. Knee Surg. Sports Traumatol. Arthrosc. 2020, 28, 3031–3039.
- Guo, S.C.; Tao, S.C.; Yin, W.J.; Qi, X.; Yuan, T.; Zhang, C.Q. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics 2017, 7, 81–96.
- Tao, S.C.; Yuan, T.; Rui, B.Y.; Zhu, Z.Z.; Guo, S.C.; Zhang, C.Q. Exosomes derived from human platelet-rich plasma prevent apoptosis induced by glucocorticoid-associated endoplasmic reticulum stress in rat osteonecrosis of the femoral head via the Akt/Bad/Bcl-2 signal pathway. Theranostics 2017, 7, 733–750.
- Torreggiani, E.; Perut, F.; Roncuzzi, L.; Zini, N.; Baglìo, S.R.; Baldini, N. Exosomes: Novel effectors of human platelet lysate activity. Eur. Cells Mater. 2014, 28, 137–151.
- Haraszti, R.A.; Miller, R.; Stoppato, M.; Sere, Y.Y.; Coles, A.; Didiot, M.C.; Wollacott, R.; Sapp, E.; Dubuke, M.L.; Li, X.; et al. Exosomes Produced from 3D Cultures of MSCs by Tangential Flow Filtration Show Higher Yield and Improved Activity. Mol. Ther. 2018, 26, 2838–2847.
- Sun, L.; Xu, R.; Sun, X.; Duan, Y.; Han, Y.; Zhao, Y.; Qian, H.; Zhu, W.; Xu, W. Safety evaluation of exosomes derived from human umbilical cord mesenchymal stromal cell. Cytotherapy 2016, 18, 413–422.
- Burrello, J.; Monticone, S.; Gai, C.; Gomez, Y.; Kholia, S.; Camussi, G. Stem Cell-Derived Extracellular Vesicles and Immune-Modulation. Front. Cell Dev. Biol. 2016, 4, 83.
- Guo, M.; Yin, Z.; Chen, F.; Lei, P. Mesenchymal stem cell-derived exosome: A promising alternative in the therapy of Alzheimer’s disease. Alzheimers. Res. Ther. 2020, 12, 109.





