
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shuvolina Mukherjee | + 2204 word(s) | 2204 | 2021-06-04 10:29:52 | | | |
| 2 | Enzi Gong | + 14 word(s) | 2218 | 2021-06-17 11:53:33 | | |
Video Upload Options
Ovarian cancer (OC) is often used as an umbrella term referring to malignancies caused by ovarian epithelial inclusion cysts that are trapped beneath the surface of the epithelium of the ovary as well as malignancies in the peritoneum and fallopian tube.
1. Overview
Despite recent technological advancements allowing the characterization of cancers at a molecular level along with biomarkers for cancer diagnosis, the management of ovarian cancers (OC) remains challenging. Proteins assume functions encoded by the genome and the complete set of proteins, termed the proteome, reflects the health state. Comprehending the circulatory proteomic profiles for OC subtypes, therefore, has the potential to reveal biomarkers with clinical utility concerning early diagnosis or to predict response to specific therapies. Furthermore, characterization of the proteomic landscape of tumor-derived tissue, cell lines, and PDX models has led to the molecular stratification of patient groups, with implications for personalized therapy and management of drug resistance. Here, we review single and multiple marker panels that have been identified through proteomic investigations of patient sera, effusions, and other biospecimens. We discuss their clinical utility and implementation into clinical practice.
2. Ovarian cancer
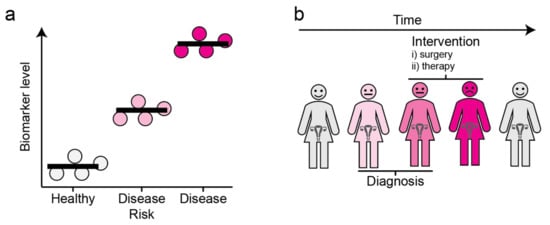
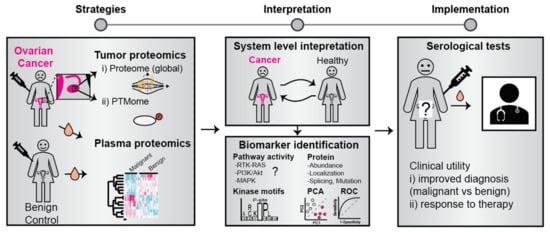
3. Protein Biomarkers Associated with OC
| Marker(s) | Gene ID (If Applicable) |
Source | Type (Circulatory/Tumor-Specific | Utility (Early/Late-Stage Pre/Post-Menopausal) |
Platform & Study Design | Reference |
|---|---|---|---|---|---|---|
| CA-125 | MUC16 | Serum/Plasma | Serum marker-high molecular weight glycoprotein | Monitoring response to chemotherapy and disease activity in clinical trials. | Immunoassays from patient sera using OC125 and M11 antibodies | [18][19][20][21] |
| HE4 | WFDC2 | Serum/Plasma | HE4 is also a secreted glycoprotein that is overexpressed in OCs | FDA approved biomarker for monitoring disease activity | Immunoassays from patient sera | [19][20] |
| MCSF and LPA | CSF1 | Blood/Tumor tissue ascites | Components of the tumor microenvironment | LPA is elevated in the blood, tumor tissue, and ascites. LPA also influences tumor-associated macrophages, which can be used as a therapeutic target | Metanalysis from several studies mostly based on the immunoassay-based determination of markers | [22] |
| CART analysis: CA-125, OVX1, LASA, CA 15-3, CA 72-4) | MUC16, ovx1, MUC1 | Serum | Circulatory markers as well as tumor microenvironment components |
CART analysis (classification and regression tree analysis), uses the sequential analysis of marker concentrations with 5 markers (CA-125, OVX1, LASA, CA 15-3, CA 72-4) to yield a sensitivity of 90.6% and a specificity of 93.2% |
Initial discovery-based studies using radioimmunoassay. Multiple marker analysis performed on ANN based machine learning algorithms |
[23][24][25] |
| A three-panel marker: Apolipoprotein I TransthyretinInter-α-trypsin inhibitor heavy chain H4 (cleavage fragment) |
APOA1, TTR, ITIH4 |
Serum | Components of the circulatory biofluids | Useful for detection of early-stage patients, exhibits higher sensitivity (74%) over CA125 alone (52%) | The study employed SELDI-TOF technology with the ProteinChip Biomarker System (Ciphergen Biosystems) | [25][26] |
| CT45 | CT45A1, CT45A | Tumor tissue (FFPE blocks) | Tumor marker | Reported to be an independent prognostic factor that is associated with a doubling of disease-free survival in advanced-stage HGSCs | Quantitative proteomics on FFPE tumor samples derived from 25 chemotherapy-naive patients with advanced-stage HGSCs | [27] |
| MUCIN-16, SPINT1, TACSTD2, CLEC6A, ICOSLG, MSMB, PROK1, CDH3, WFDC2, KRT19, and FR-alpha | MUCIN-16, SPINT1, TACSTD2, CLEC6A, ICOSLG, MSMB, PROK1, CDH3, WFDC2, KRT19, and FOLR | Plasma | Circulatory markers | Potentially useful for improved diagnosis of adnexal ovarian mass and identification of potential cases for specialized referrals | PEA was implemented utilizing oligonucleotide antibody probes to measure protein abundance | [17] |
4. Conclusions
References
- Ueland, F. A Perspective on Ovarian Cancer Biomarkers: Past, Present and Yet-To-Come. Diagnostics 2017, 7, 14.
- Reid, B.M.; Permuth, J.B.; Sellers, T.A. Epidemiology of ovarian cancer: A review. Cancer Biol. Med. 2017, 14, 9–32.
- Torre, L.A.; Trabert, B.; DeSantis, C.E.; Miller, K.D.; Samimi, G.; Runowicz, C.D.; Gaudet, M.M.; Jemal, A.; Siegel, R.L. Ovarian cancer statistics, 2018. CA. Cancer J. Clin. 2018, 68, 284–296.
- Koshiyama, M.; Matsumura, N.; Konishi, I. Subtypes of Ovarian Cancer and Ovarian Cancer Screening. Diagnostics 2017, 7, 12.
- Cristea, M.; Han, E.; Salmon, L.; Morgan, R.J. Review: Practical considerations in ovarian cancer chemotherapy. Ther. Adv. Med. Oncol. 2010, 2, 175–187.
- Javadi, S.; Ganeshan, D.M.; Qayyum, A.; Iyer, R.B.; Bhosale, P. Ovarian Cancer, the Revised FIGO Staging System, and the Role of Imaging Women’s Imaging Review. AJR 2016, 206.
- King, M.-C.; Marks, J.H.; Mandell, J.B.; New, T.; Breast, Y. Breast and Ovarian Cancer Risks Due to Inherited Mutations in BRCA1 and BRCA2. Science 2003, 302, 643–646.
- Zhang, Z.; Chan, D.W. The road from discovery to clinical diagnostics: Lessons learned from the first FDA-cleared in vitro diagnostic multivariate index assay of proteomic biomarkers. Cancer Epidemiol. Biomark. Prev. 2010, 19, 2995–2999.
- Testa, U.; Petrucci, E.; Pasquini, L.; Castelli, G.; Pelosi, E. Ovarian Cancers: Genetic Abnormalities, Tumor Heterogeneity and Progression, Clonal Evolution and Cancer Stem Cells. Medicines 2018, 5, 16.
- Chen, S.; Parmigiani, G. Meta-analysis of BRCA1 and BRCA2 penetrance. J. Clin. Oncol. 2007, 25, 1329–1333.
- Lawrenson, K.; Gayther, S.A. Ovarian cancer: A clinical challenge that needs some basic answers. PLoS Med. 2009, 6, 126–129.
- Tangjitgamol, S.; Manusirivithaya, S.; Laopaiboon, M.; Lumbiganon, P.; Bryant, A. Interval debulking surgery for advanced epithelial ovarian cancer. Cochrane Database Syst. Rev. 2013, 4, 1–37.
- Van der Burg, M.E.L.; Lammes, F.B.; Verweij, J. CA 125 in ovarian cancer. Neth. J. Med. 1992, 40, 36–51.
- Picotti, P.; Aebersold, R. Selected reaction monitoring-based proteomics: Workflows, potential, pitfalls and future directions. Nat. Methods 2012, 9, 555–566.
- Mor, G.; Visintin, I.; Lai, Y.; Zhao, H.; Schwartz, P.; Rutherford, T.; Yue, L.; Bray-Ward, P.; Ward, D.C. Serum protein markers for early detection of ovarian cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 7677–7682.
- Whitwell, H.J.; Worthington, J.; Blyuss, O.; Gentry-Maharaj, A.; Ryan, A.; Gunu, R.; Kalsi, J.; Menon, U.; Jacobs, I.; Zaikin, A.; et al. Improved early detection of ovarian cancer using longitudinal multimarker models. Br. J. Cancer 2020, 122.
- Enroth, S.; Berggrund, M.; Lycke, M.; Broberg, J.; Lundberg, M.; Assarsson, E.; Olovsson, M.; Stålberg, K.; Sundfeldt, K.; Gyllensten, U. High throughput proteomics identifies a high-accuracy 11 plasma protein biomarker signature for ovarian cancer. Commun. Biol. 2019, 2, 1–12.
- Bast, R.C. Early detection of ovarian cancer: New technologies in pursuit of a disease that is neither common nor rare. Trans. Am. Clin. Climatol. Assoc. 2004, 115, 233.
- Zhang, L.; Chen, Y.; Wang, K. Comparison of CA125, HE4, and ROMA index for ovarian cancer diagnosis. Curr. Probl. Cancer 2019, 43, 135–144.
- Moore, R.G.; McMeekin, D.S.; Brown, A.K.; DiSilvestro, P.; Miller, M.C.; Allard, W.J.; Gajewski, W.; Kurman, R.; Bast, R.C.; Skates, S.J. A novel multiple marker bioassay utilizing HE4 and CA125 for the prediction of ovarian cancer in patients with a pelvic mass. Gynecol. Oncol. 2009, 112, 40–46.
- Sölétormos, G.; Duffy, M.J.; Othman Abu Hassan, S.; Verheijen, R.H.M.; Tholander, B.; Bast, R.C.; Gaarenstroom, K.N.; Sturgeon, C.M.; Bonfrer, J.M.; Petersen, P.H.; et al. Clinical Use of Cancer Biomarkers in Epithelial Ovarian Cancer: Updated Guidelines from the European Group on Tumor Markers. Lippincott Williams and Wilkins. Int. J. Gynecol. Cancer 2016, 26, 43–51.
- Feng, Y.; Xiao, M.; Zhang, Z.; Cui, R.; Jiang, X.; Wang, S.; Bai, H.; Liu, C.; Zhang, Z. Potential interaction between lysophosphatidic acid and tumor-associated macrophages in ovarian carcinoma. J. Inflamm. 2020, 17, 1–5.
- Gadducci, A.; Ferdeghini, M.; Ceccarini, T.; Prontera, C.; Facchini, V.; Bianchi, R.; Fioretti, P. A comparative evaluation of the ability of serum CA 125, CA 19-9, CA 15-3, CA 50, CA 72-4 and TATI assays in reflecting the course of disease in patients with ovarian carcinoma. Eur. J. Gynaecol. Oncol. 1990, 11, 127–133.
- Zhang, Z.; Yu, Y.; Xu, F.; Berchuck, A.; van Haaften-Day, C.; Havrilesky, L.J.; de Bruijn, H.W.A.; van der Zee, A.G.J.; Woolas, R.P.; Jacobs, I.J.; et al. Combining multiple serum tumor markers improves detection of stage I epithelial ovarian cancer. Gynecol. Oncol. 2007, 107, 526–531.
- Woolas, R.P.; Conaway, M.R.; Xu, F.; Jacobs, I.J.; Yu, Y.; Daly, L.; Davies, A.P.; O’Briant, K.; Berchuck, A.; Soper, J.T.; et al. Combinations of multiple serum markers are superior to individual assays for discriminating malignant from benign pelvic masses. Gynecol. Oncol. 1995, 59, 111–116.
- Zhang, Z.; Bast, R.C.; Yu, Y.; Li, J.; Sokoll, L.J.; Rai, A.J.; Rosenzweig, J.M.; Cameron, B.; Wang, Y.Y.; Meng, X.Y.; et al. Three biomarkers identified from serum proteomic analysis for the detection of early stage ovarian cancer. Cancer Res. 2004, 64, 5882–5890.
- Coscia, F.; Lengyel, E.; Duraiswamy, J.; Ashcroft, B.; Bassani-Sternberg, M.; Wierer, M.; Johnson, A.; Wroblewski, K.; Montag, A.; Yamada, S.D.; et al. Multi-level Proteomics Identifies CT45 as a Chemosensitivity Mediator and Immunotherapy Target in Ovarian Cancer. Cell 2018, 175, 159–170.e16.
- Bengtsson, S.; Krogh, M.; Szigyarto, C.A.K.; Uhlen, M.; Schedvins, K.; Silfverswärd, C.; Linder, S.; Auer, G.; Alaiya, A.; James, P. Large-scale proteomics analysis of human ovarian cancer for biomarkers. J. Proteome Res. 2007, 6, 1440–1450.
- Dieters-Castator, D.Z.; Rambau, P.F.; Kelemen, L.E.; Siegers, G.M.; Lajoie, G.A.; Postovit, L.M.; Kobel, M. Proteomics-derived biomarker panel improves diagnostic precision to classify endometrioid and high-grade serous ovarian carcinoma. Clin. Cancer Res. 2019, 25, 4309–4319.
- Kriplani, D.; Patel, M.M. Immunohistochemistry: A diagnostic aid in differentiating primary epithelial ovarian tumors and tumors metastatic to the ovary. South Asian J. Cancer 2013, 2, 254–258.
- Shih, I.M.; Kurman, R.J. Ovarian Tumorigenesis: A Proposed Model Based on Morphological and Molecular Genetic Analysis. Am. J. Pathol. 2004, 164, 1511–1518.
- Dehari, R.; Kurman, R.J.; Logani, S.; Shih, I.M. The development of high-grade serous carcinoma from atypical proliferative (borderline) serous tumors and low-grade micropapillary serous carcinoma: A morphologic and molecular genetic analysis. Am. J. Surg. Pathol. 2007, 31, 1007–1012.
- Wong, K.-K.; Gershenson, D.; Tsai, C.-C. BRAF mutational analysis in ovarian tumors: Recent perspectives. Pathol. Lab. Med. Int. 2015, 7, 75.
- Kurman, R.J.; Shih, I.M. The origin and pathogenesis of epithelial ovarian cancer: A proposed unifying theory. Am. J. Surg. Pathol. 2010, 34, 433–443.
- Davidson, B.; Smith, Y.; Nesland, J.M.; Kærn, J.; Reich, R.; Tropè, C.G. Defining a prognostic marker panel for patients with ovarian serous carcinoma effusion. Hum. Pathol. 2013, 44, 2449–2460.
- Boylan, K.L.M.; Afiuni-Zadeh, S.; Geller, M.A.; Argenta, P.A.; Griffin, T.J.; Skubitz, A.P.N. Evaluation of the potential of Pap test fluid and cervical swabs to serve as clinical diagnostic biospecimens for the detection of ovarian cancer by mass spectrometry-based proteomics. Clin. Proteom. 2021, 18, 4.
- Marcišauskas, S.; Ulfenborg, B.; Kristjansdottir, B.; Waldemarson, S.; Sundfeldt, K. Univariate and classification analysis reveals potential diagnostic biomarkers for early stage ovarian cancer Type 1 and Type 2. J. Proteom. 2019, 196, 57–68.
- Minis, E.; Holcomb, K.; Sisti, G.; Nasioudis, D.; Kanninen, T.T.; Athanasiou, A.; Frey, M.K.; Chapman-Davis, E.; Caputo, T.A.; Witkin, S.S. Evaluation of lysophosphatidic acid in vaginal fluid as a biomarker for ovarian cancer: A pilot study. Eur. J. Obstet. Gynecol. Reprod. Biol. X 2019, 2, 100012.
- Kim, G.; Davidson, B.; Henning, R.; Wang, J.; Yu, M.; Annunziata, C.; Hetland, T.; Kohn, E.C. Adhesion molecule protein signature in ovarian cancer effusions is prognostic of patient outcome. Cancer 2012, 118, 1543–1553.
- Tancioni, I.; Uryu, S.; Sulzmaier, F.J.; Shah, N.R.; Lawson, C.; Miller, N.L.G.; Jean, C.; Chen, X.L.; Ward, K.K.; Schlaepfer, D.D. FAK inhibition disrupts a β5 integrin signaling axis controlling anchorage-independent ovarian carcinoma growth. Mol. Cancer Ther. 2014, 13, 2050–2061.
- Xu, B.; Lefringhouse, J.; Liu, Z.; West, D.; Baldwin, L.A.; Ou, C.; Chen, L.; Napier, D.; Chaiswing, L.; Brewer, L.D.; et al. Inhibition of the integrin/FAK signaling axis and c-Myc synergistically disrupts ovarian cancer malignancy. Oncogenesis 2017, 6, 295.
- He, X.; Lei, S.; Zhang, Q.; Ma, L.; Li, N.; Wang, J. Deregulation of cell adhesion molecules is associated with progression and poor outcomes in endometrial cancer: Analysis of the Cancer Genome Atlas data. Oncol. Lett. 2020, 19, 1906–1914.
- Siravegna, G.; Marsoni, S.; Siena, S.; Bardelli, A. Integrating liquid biopsies into the management of cancer. Nat. Rev. Clin. Oncol. 2017, 14, 531–548.
- Wang, Y.; Sundfeldt, K.; Mateoiu, C.; Shih, I.M.; Kurman, R.J.; Schaefer, J.; Silliman, N.; Kinde, I.; Springer, S.; Foote, M.; et al. Diagnostic potential of tumor DNA from ovarian cyst fluid. Elife 2016, 5, e15175.
- Wang, Y.; Li, L.; Douville, C.; Cohen, J.D.; Yen, T.T.; Kinde, I.; Sundfelt, K.; Kjær, S.K.; Hruban, R.H.; Shih, I.M.; et al. Evaluation of liquid from the Papanicolaou test and other liquid biopsies for the detection of endometrial and ovarian cancers. Sci. Transl. Med. 2018, 10.
- Aktas, B.; Kasimir-Bauer, S.; Heubner, M.; Kimmig, R.; Wimberger, P. Molecular profiling and prognostic relevance of circulating tumor cells in the blood of ovarian cancer patients at primary diagnosis and after platinum-based chemotherapy. Int. J. Gynecol. Cancer 2011, 21, 822–830.
- Mari, R.; Mamessier, E.; Lambaudie, E.; Provansal, M.; Birnbaum, D.; Bertucci, F.; Sabatier, R. Liquid biopsies for ovarian carcinoma: How blood tests may improve the clinical management of a deadly disease. Cancers 2019, 11, 774.
- Zhang, X.; Li, H.; Yu, X.; Li, S.; Lei, Z.; Li, C.; Zhang, Q.; Han, Q.; Li, Y.; Zhang, K.; et al. Cellular Physiology and Biochemistry Cellular Physiology and Biochemistry Analysis of Circulating Tumor Cells in Ovarian Cancer and Their Clinical Value as a Biomarker. Cell Physiol. Biochem. 2018, 48, 1983–1994.
- Van Berckelaer, C.; Brouwers, A.J.; Peeters, D.J.E.; Tjalma, W.; Trinh, X.B.; van Dam, P.A. Current and future role of circulating tumor cells in patients with epithelial ovarian cancer. Eur. J. Surg. Oncol. 2016, 42, 1772–1779.
- Asante, D.B.; Calapre, L.; Ziman, M.; Meniawy, T.M.; Gray, E.S. Liquid biopsy in ovarian cancer using circulating tumor DNA and cells: Ready for prime time? Cancer Lett. 2020, 468, 59–71.
- Bristow, R.E.; Smith, A.; Zhang, Z.; Chan, D.W.; Crutcher, G.; Fung, E.T.; Munroe, D.G. Ovarian malignancy risk stratification of the adnexal mass using a multivariate index assay. Gynecol. Oncol. 2013, 128, 252–259.





