In recent years, the interaction between light and metallic films have been proven to be a highly powerful tool for optical sensing applications. There has been continuous effort to integrate this plasmonic sensing technology using micro and nanofabrication techniques with the optical fiber sensor world, trying to get better, smaller and cost-effective high performance sensing solutions. In this work, we present a review of the latest and more relevant scientific contributions to the optical fiber sensors field using plasmonic materials over the last decade. The combination of optical fiber technology with metallic micro and nanostructures that allow plasmonic interactions have opened a complete new and promising field of study. We review the main advances in the integration of such metallic micro/nanostructures onto the optical fibers, discuss the most promising fabrication techniques and show the new trends in physical, chemical and biological sensing applications.
1. Introduction
The plasmonic resonance of light with metallic thin films, especially gold, has become very important in the sensor field. One of the most used phenomenon is Surface Plasmon Resonance (SPR). Such SPR sensor devices take advantage of the coupling of certain incident electromagnetic radiation to a surface wave polariton along a dielectric-metallic interface. This resonant coupling occurs under certain strict optical conditions including the permittivity of the dielectric and the metal, the incident wavelength, angle and polarization, etc. As it is a surface resonant phenomenon that is extremely sensitive to slight variations of the optical properties of the media, if there is any change on the external medium in the range of several tens of nanometers from the surface, the resonance condition gets affected, yielding a sensing signal. Due to the surface nature of this optical phenomenon that is ideal for the detection of adsorption processes, it has been successfully used in biological sensing applications, where traditional immunoassays such as ELISA, DNA detection, etc. are based on the selective adsorption of biomolecules (antibody-antigen, DNA hybridization, etc.)
[1]. Currently, there are several commercial SPR sensor platforms available from companies such as Cytiva (former Biacore), Xantec, IBIS Technologies, etc.
When this dielectric-metal resonance occurs in a metallic nanoparticle (NP) instead of a metal continuous thin film, the plasmonic resonance is confined within the nanoparticle, resulting in a collective oscillation of the electrons. Such phenomenon is known as Localized Surface Plasmon Resonance (LSPR), and has the same basis as SPR. Since the resonant condition occurs in the metallic nanoparticles typically dispersed in a dielectric medium, the incident angle and the polarization of the light is less important. This LSPR phenomenon is extremely concentrated into the surrounding of the nanoparticle, giving an intense optical absorption band that is very dependent on changes in the optical properties of the dielectric medium in the range of a few nanometers away from the NP surface. This phenomenon has been exploited in many research works where metallic nanoparticles are used as plasmonic nanodetectors.
When plasmonic resonances are induced under certain conditions, it is possible to observe significant increments on the local electrical field intensity around the metallic surface. Such high concentrations of electric field can induce other phenomena that can be used also for sensing applications such as Surface-Enhanced Raman Scattering (SERS).
Optical fibers are very interesting for optical sensing applications because they show some significant advantages compared to conventional volume optics and planar optics solutions. Beyond the traditional advantages of optical fibers (small, biocompatible, multiplexing, etc.) the concept of having an optrode (optical fiber tip sensor) that can perform highly sensitive measurements in almost any media is very interesting. Even more so that there is now a huge effort to combine traditional fabrication techniques with micro and nanofabrication tools in order to create structured optical fiber tip sensors, walking towards the Lab on Fiber and Lab on Tip concepts
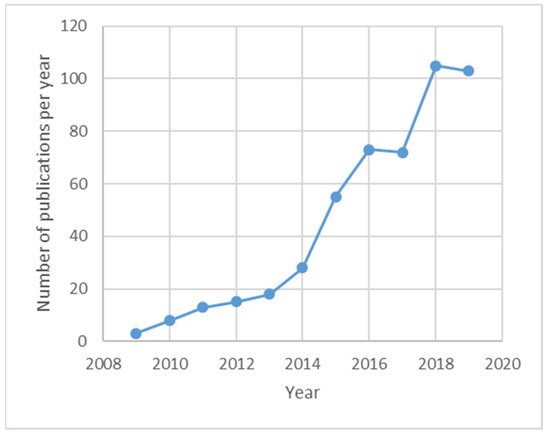
[2][3]. There is no doubt that the use of plasmonic interactions is a very promising trend in the optical fiber sensor field, as shown by the evolution of the number of publications per year with the word “plasmonic” in their title, abstract or keywords shown in
Figure 1.
Figure 1. Evolution of the number of publications per year in the Optical Fiber Sensors field with the word “plasmonic” in their abstract, title or keywords. Source: Scopus.
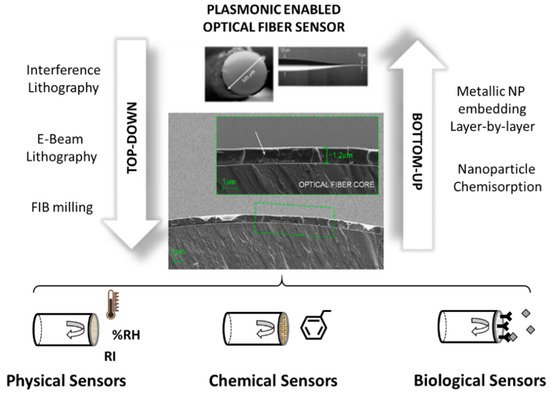
There are two different strategies to incorporate metallic micro and nanostructures onto an optical fiber so that plasmonic interactions can be used for sensing applications. On one hand, there is the bottom-up approach, where metallic micro and nanostructures are previously created and further immobilized over the optical fiber surface. For this purpose, researchers have used different techniques such as nanoparticle-chemisorption strategies or the immobilization of the nanoparticles within thin film coatings. On the other hand, several techniques fit with the top-down approach, where metallic micro and nanostructures are created using lithographic techniques and some other microfabrication tools to sculpt the metallic microstructures out of gold or silver thin films. In the last decade, there has been an extraordinary boost in adapting microfabrication tools to nonconventional substrates, such as optical fibers. This opens the door to a complete revolution in the optical fiber sensor field, turning them into advanced “multifunctional plug and play optrodes.”
Figure 2 shows a schematic description of both approaches.
Figure 2. Schematic view of the two main different approaches used to create plasmonic optical fiber sensors. The Bottom-Up approach starts from metallic nanoparticles (NPs) that are assembled to the optical fiber sensor, and the Top-Bottom approach starts with metallic thin films further patterned using lithographic techniques or Focused-Ion-Beam milling.
In the following sections, there is a review of the most relevant contributions published in the last decade 2010–2020, so it is possible to figure out the current state of the art in the plasmonic optical fiber sensor field, and picture a roadmap with the most promising trends in this interesting field.
2. LSPR Optical Fiber Sensors Functionalized with Metallic Nanoparticles
In this section, the main approaches for creating bottom-up plasmonic sensing structures in the end-face of an optical fiber are summarized. This bottom-up approach starts from metallic nanoparticles that can inherently show plasmonic absorption bands, immobilized onto the optical fiber sensor. In most of the cases, a non-organized distribution of the metallic nanoparticles can be achieved by using different wet-chemistry methodologies. In the first subsection, we summarize the main optical fiber-functionalization approaches focused on chemically activation of the surface of fiber with specific functional groups that can effectively bind metallic nanoparticles of a wide variety of morphologies (i.e., spherical, nanoflowers, nanostars and nanorods, among others). In the second subsection, the Layer-by-Layer nanoassembly technique is presented as a promising and interesting technique to immobilize metallic nanoparticles into a multilayer polyelectrolyte structure. The main sensing applications of these LSPR-based optical fiber devices is summarized in a wide variety of research fields of interest in the scientific industry for the detection of physical, chemical and biological parameters.
2.1. Chemisorpted Nanoparticle-Monolayers
One of the most straightforward techniques for the optical fiber functionalization to immobilize nanoparticles is the direct chemisorption of the nanoparticles onto the optical fiber surface. In this sense, one of the most used techniques is the silanization of the optical fiber substrate with bifunctional silanes. For this purpose, firstly the unclad core is immersed in a piranha solution at a ratio of 3:1 (concentrated H
2SO
4 and H
2O
2) with the aim to hydrolyze the surface of the optical fiber core, making possible the generation of additional SiOH sites in the outer surface of the fiber to allow a further chemical bonding. Later, the hydrolyzed optical fiber surface is exposed to specific Organically Modified Silicates (ORMOSILs) such as aminosilanes or mercaptosilanes. These ORMOSILS are used to create self-assembled monolayers onto the optical fiber surface, which is used to attach the metal nanoparticles with a desired shape by the chemical interaction of specific functional groups such mercapto (-SH) or amine (-NH
2) of the corresponding organosilanes. Consequently, this leads to obtain a very robust metallic nanoparticles integration over the optical fiber. More details about the fabrication of monolayers based on the Chemisorption of nanoparticles can be found in
[4][5].
2.1.1. LSPR in NP Monolayers for Physical Detection
One of the most representative physical parameters is the refractive index, whose detection is of great interest in the industry. According to this, refractometers are a very powerful tool in the field of sensing devices because they can be used for a direct measurement of the surrounding medium refractive index (SMRI) or even they can be also used combined with a sensitive coating whose refractive index depends on a specific parameter. The use of resonance-based optical fiber refractometers can take advantage of the intrinsic properties associated with optical fiber as well as the advantage of wavelength detection technique associated with the resonance phenomenon of the LSPR of the metallic nanoparticles. A clear example can be found in
[6] where the development of an LSPR-based U-bent plastic optical fiber based on the immobilization of gold nanoflowers (denoted as AuNFs) is presented for measuring refractive index changes. The experimental results have corroborated that the U-bent LSPR sensor has shown an 8-fold improvement in refractive index sensitivity in comparison with the bare sensor. Other interesting approach for the detection of refractive index changes is also presented in
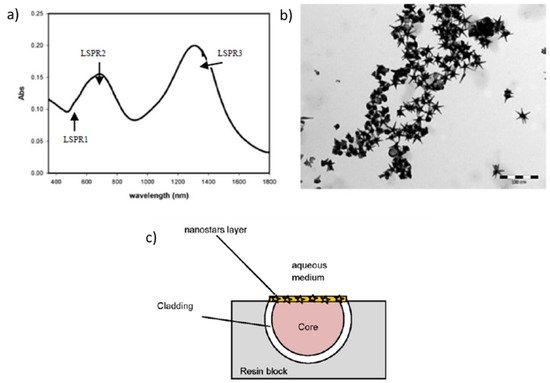
[7]. In this work, the immobilization of five-branched gold nanostars (denoted as GNS) onto the part of the core is performed (see
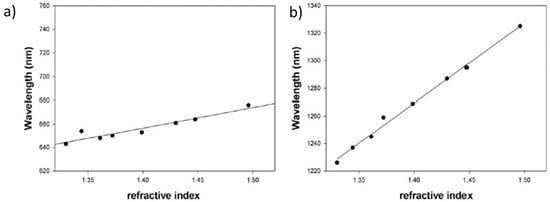
Figure 3), which exhibits three localized surface resonances (LSPR1, LSPR2 and LSPR3). Two of these LSPR bands have presented a wavelength dependence as a function of variations of the refractive index of the surrounding medium, being the sensitivity for LSPR3 (580 nm/RIU) greater than LSPR2 (175 nm/RIU), as shown in the linear fitting in
Figure 4. Finally, a relevant aspect to remark is that GNS can be also used as a powerful tool for nanomedicine, exploiting the 700–1000 nm transparent window of biological matter for treatments against tumors or even multidrug resistant bacterial infections, among others.
Figure 3. (
a) UV-Vis-NIR absorption spectrum of the synthesized gold nanostars (GNS) with the three characteristic Localized Surface Plasmon Resonance (LSPR) absorption bands; (
b) TEM image of the five-branched GNS; (
c) Aspect in detail of the LSPR optical fiber. Reprinted with permission of
[7].
Figure 4. Wavelength shift of LSPR2 (
a) and LSPR3 (
b) when the GNS are suspended in different solvents with refractive index values from 1.3300 up to 1.4476. Reprinted with permission of
[7].
A similar wavelength-based LSPR optical fiber sensor can be found in
[8], although in this work two different types of gold morphologies such as gold nanospheres (GNSs) and gold nanorods (GNRs) have been immobilized, being the sensitivity to refractive index variation of 914 nm/RIU for GNSs and 601 nm/RIU for GNRs, respectively. A very promoting result is presented in
[9] because the sensitivity of the LSPR optical fiber sensor has been increased up to 1933 nm/RIU by using hollow gold nanostructures (denoted as gold nanocages, AuNCs).
Other works are devoted to the use of silver nanoparticles for sensing applications. In this sense, presented in
[10] is a comparative study based on the immobilization of silver nanoparticles with different shapes such as triangular and spherical onto U-shaped fiber sensors. The results have indicated that triangle silver nanoparticles have shown the highest sensitivity (1116.8 nm/RIU) in comparison with the spherical silver nanoparticles (342.7 nm/RIU). In other works, it is simulated by using theoretical studies the influence in the resultant refractive index of two input parameters such as the thickness layer and particle size of different types of metal nanoparticles (gold, silver, copper, platinum)
[11][12].
The combination of different fiber structures or different coupling effects can also be employed for the detection of refractive index changes. According to this, an optical fiber hetero-core sensor consisting of a piece of single mode fiber longitudinally placed between two multimode fibers is used for refractive index sensing
[13]. Finally, other interesting approach based on the electric field coupling effect between both Au film surface plasmon resonance (SPR) and gold nanoparticle localized surface plasmon resonance (LSPR) is presented in
[14]. In this work, the SPR-LSPR coupling effect has considerably improved the resultant refractive index sensitivity with a value of 3074 nm/RIU, showing a considerable improvement in the sensitivity in comparison with only Au film SPR optical fiber.
2.1.2. LSPR in NP Monolayers for Chemical Detection
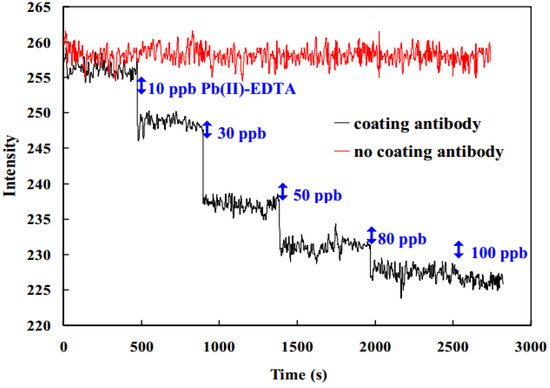
Other interesting research field based on the design of LSPR optical fibers is for the detection and quantization of heavy metals (i.e., lead, cadmium, mercury) because they are considered as harmful to the human body. A clear example is presented in
[15] where a monoclonal antibody-functionalized fiber-based biosensor using the LSPR effect has been developed to evaluate the concentration of lead ions (Pb
2+). As observed in
Figure 5, a change of 12.2% in absorbability has been observed for detecting 10 to 100 ppb Pb(II)-EDTA complex, showing a limit of detection of 0.27 ppb. In addition, an important aspect to remark is that the biosensor retains 92.7% of its original activity, giving reproducible results after storage in Trehalose dehydrate solution at 4 °C for 35 days. This same solution is used in
[16] in order to retain the corresponding activity for a long period of time, although in this work is presented a fiber-optic biosensor for the detection of cadmium. In this work, the resultant LSPR biosensor has been fabricated by using phytochelatines (PCs) onto AuNPs, and the experimental results have shown a good limit of detection (0.16 ppb) and sensitivity (1.24 ppb
−1), respectively.
Figure 5. Difference in the response signal in the range of 10~100 ppb by two optical fiber sensors with antibody coating (black line) and without antibody coating (red line). Reprinted with permission of
[15].
Other interesting work based on LSPR optical fiber biosensor for the detection of heavy metals is presented in
[17]. In this work, AuNPs have been succesfully functionalized with mercapto-undecanoic acid (MUA), showing a sensitivity of 0.28 nm/mM and a limit of detection of 65 ppm for lead ions. In addition, a similar response has been also observed for the detection of cadmium ions. Finally, one aspect to remark is that among all the known heavy metals, mercury ions can easily enter the human body through skin, respiratory or even gastrointestinal tissues, and due to this, their detection is of great interest because an excess can damage brain or nervous system. According to this, an interesting approach is presented in
[18] where a U-shaped optical fiber sensor is fabricated for mercury ions detection by using glucose capped silver nanoparticles. In this work, a good limit of detection (2 ppb) has been obtained, although this limit has been improved in
[19] by using chitosan capped AuNPs, obtaining 0.1 ppb in tap water and 0.2 ppb in sea fish and vegetable samples with a negligible cross sensitivity towards other metal ions.
More recently, some authors have focused their research on the development of optical fiber sensors involving novel materials that allow plasmonic interactions (beyond Au and Ag nanoparticles
[20]). In this sense, the 2-D plasmonic materials are very promising due to their intense light-matter interaction due to the plasmon confinement, leading to very intense plasmonic bands. Nevertheless, currently most of the 2D materials locate intrinsically at terahertz or mid-infrared range, which is a limitation for their practical application nowadays, especially for optical fiber sensing applications. Nevertheless, there are transition metal oxide 2-D crystals whose properties can be adjustable in the VIS-NIR range, and their plasmonic interactions are far more intense than in traditional materials. For example, in
[21] the authors report the use of 2-D MoS
2 crystals to modify and enhance the plasmonic behavior of a Ti thin film over a partially uncladded SMF fiber. The authors have proven that the plasmonic bands of the sensor are sensitive to the external refractive index, opening the door to highly sensitive chemical sensors, if such structures are properly combined with further sensitive layers. Another interesting work was published in
[22] where it is reported how 2-D WOx flat nanocrystals can show plasmonic bands matched with conventional telecommunications wavelength in the 1550 nm range. In this particular case, the authors report that the integration of such 2-D nanocrystals over a D-shape optical fiber can lead to highly sensitive gas sensors, in this case showing a LOD as low as 8 ppb of NO
2.
Finally, Surface-enhanced Raman Scattering (SERS)-based molecular sensing can be also used as an emerging tool for remote test in trace chemicals or even in the detection of food additives. According to this, a very representative work can be found in
[23], where the development of a fiber-optrode is presented by using silver-coated gold nanostars (Au@Ag NS) as highly enhancing SERS active substrate. In this work, a chemical colorant such as Rhodamine B has been chosen as an analytical target due to its illegal use as food additive and rinsing concerns on the food safety
[24]. In addition, the optimization of the optrode configuration has been performed by variating the density of the gold nanostars and the resultant roughness of the fiber end-face with the aim to maximize the SERS signal. Finally, the experimental results have demonstrated that the optrode can perfectly detect different types of analytes with a LOD for Rhodamine B between 10
−7 and 10
−8 M, respectively.
2.1.3. LSPR in NP Monolayers for Biological Detection
Other aspect to remark is that the use of plasmonic fiber-optic biosensors can be considered as a very promising alternative in comparison with traditional methods for biomolecule detection, showing important advances in clinical diagnostics
[25][26][27]. This is possible because the metallic nanoparticles can be further functionalized in order to immobilize specific enzymes or antibodies for the detection of specific targets.
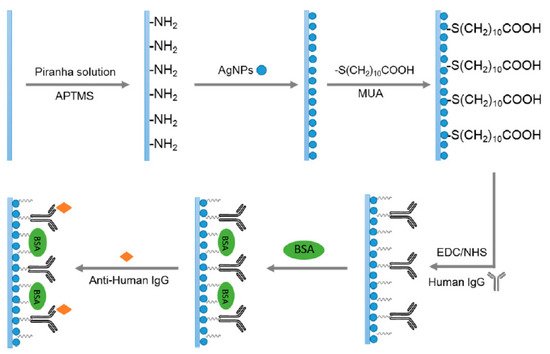
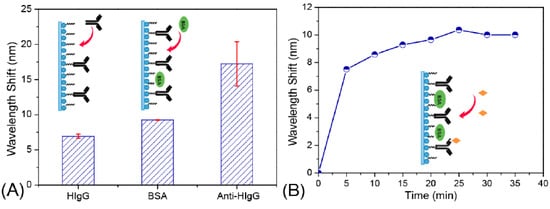
Figure 6 shows the immobilization of AgNPs with a further functionalization in order to obtain a biosensing approach for monitoring antigen-antibody interactions
[26]. The optical setup as well as the morphology and optical properties of the AgNPs are observed in
Figure 7, whereas the biosensing results for anti-human IgG are presented in
Figure 8.
Figure 6. A summary scheme for the functionalization process of the AgNPs for biosensing applications. Reprinted with permission of
[26].
Figure 7. (
A) A summary scheme of the reflection configuration setup; (
B) SEM image of the synthesized AgNPs used for LSPR biosensing; (
C) UV-Vis spectra of AgNPs solutions; (
D) Histogram with the particle size distribution of AgNPs. Reprinted with permission of
[26].
Figure 8. (
A) LSPR shift spectra after different stages of surface modification; (
B) LSPR peak wavelength changes during the process of anti-human IgG immobilization on the Ag NP-based sensor surface. Reprinted with permission of
[26].
A very interesting work based on a functionalized Long Period Grating plasmonic fiber sensor for the detection of glyphosate in water is presented in
[27]. The sensor operation is based on the reaction between glyphosate molecules and cysteamine-functionalized gold nanoparticles that modifies the effective refractive index of the long-period grating cladding modes, showing a limit of detection about 0.02 µM. Other representative study can be found in
[28] where an optical tapered fiber-based biosensor is developed in order to detect uric acid concentration in human serum. In this work, a comparative study in sensitivity by immobilizing gold nanoparticles of two different sizes (10 and 30 nm) is presented. In
[29], by using the same tapered optical fiber configuration, an LSPR biosensor is presented for variable monoclonal mouse IgE-antiDNP antibody detection, showing an LSPR transmission intensity change as a function of the analyte concentration with a limit of detection of 4.8 pM. A cholesterol biosensor is presented in
[30] by using a single-mode (SMF) and a hollow core fiber (HCF) with the aim to detect cholesterol concentration in the human body. In this work, AuNPs have been functionalized with a specific enzyme such as cholesterol oxidase (ChOx), showing a detection limit of 25.5 nM. The use of aptamers can be another novel alternative route for functionalizing the surface of the nanoparticles. A clear example is observed in
[31] where gold nanorods (GNRs) were modified by an aptamer for the aim to detect ochratoxin A (OTA) which is a very harmful mycotoxin produced by several fungi. A spectral red-shift in the concentration range from 10 pM to 100 nM has been obtained with a limit of detection of 12 pM.
An approach based on co-immobilization of both enzyme glutamate dehydrogenase and coenzyme nicotamide adenine nucleotide onto AuNPs is presented in
[32] for the detection of a specific amino acid such as the glutamate which plays important roles in the formation of synapses, learning and memory. The experimental results indicate that the resultant biosensor has presented a limit of detection of 0.36 mM with a sensitivity of 0.0048 AU/mM, showing a good selectivity for the analyte of interest. A similar approach can be found in
[33] where a highly sensitive biosensor for the detection of taurine is presented by immobilizing taurine dioxygenase enzyme over the AuNPs. In this case, the LSPR biosensor has presented a higher limit of detection of 53 µM with a sensitivity of 0.0190 AU/mM, respectively. In addition, the LSPR biosensor is highly selective to taurine because a minimum change in the absorbance related to the LSPR with other interfering agents (creatinine, lactate or glutamate) has been observed. Presented in
[34] is an LSPR-based fiber-optic sensor for the detection of triacylglycerides by immobilizing lipase enzyme on silver nanoparticles (AgNPs), showing a sensitivity of 28.5 nm/mM with a good selectivity, stability and reproducibility in the entire physiological range. Presented in
[35] is a U-shaped fiber optic biosensor for the detection of blood glucose, showing the maximum sensitivity for a bending radius around 0.982 mm, respectively. Other interesting work can be found in
[36] where an LSPR biosensor has been fabricated for the detection antibody-antigen reaction of interferon-gamma (IFN-γ) by the immobilization of AuNPs at the end-face of an optical fiber, showing a limit of detection of 2 pg/mL. This same biosensor has also been used for the detection of a prostate-specific antigen (PSA), showing a limit of detection for PSA 1 pg/mL below, respectively.
Alternatively there are some works that report optical fiber biosensors based on 2D plasmonic materials such as MoO
3 [37]. Such Molybdenum trioxide nanoflakes can be chemically modified to heavily dope them with free electrons MoO
3−x so their plasmonic properties can be tuned, in this case, placing their resonance band at 735 nm. The authors functionalized a D-shaped optical fiber with these nanoflakes, showing a good affinity and optical sensitivity to negatively charged biomolecules. As a proof of concept, the authors report Bovine Serum Albumin (BSA) detection with a LOD of 1 pg/mL.
Finally, evanescent wave-based excitation of noble metal structures on fiber core surface are also highly suitable for the design and development of SERS-based biosensor applications. A versatile approach toward a shape-controlled noble metal nanostructure-sensitized tapered fiber probe for SERS-based detection is presented in
[38]. According to this, charged plasmonic structures with various morphologies (Au nanospheres, Ag nanocubes, Au nanorods and Au@Ag core-shell nanorod) can be perfectly assembled onto tapered silica fiber probes and the in situ Raman measurements indicate a detection concentration of methyl parathion down to 10
−8 M can be performed. Presented in
[39] is a comparative study based on plamonically active U-bent plastic optical fibers (POF) obtained by different techniques such as electroless deposition, sputtering and chemisorption of AuNPs. The experimental results indicate that the AuNPs immobilized by chemisorption process has shown a higher enhancement factor (1.24 × 10
9) in comparison with gold sputtered films (3.29 × 10
8) or electroless (5.42 × 10
7), respectively. In a later work
[40], it is demonstrated a biosensor application of the POF-based SERS sensor based on the chemisorption of AuNPs by realizing a sandwich immunoassay with 4-mercapto benzoic acid (4-MBA) as Raman label.
A summary of the different optical fiber sensors based on Chemisorpted nanoparticles monolayers is presented in
Table 1.
| Type of NPs |
Optical Structure |
Target |
Sensing Mechanism |
Wavelength
Range |
Sensitivity/LOD |
Ref |
| Gold nanoflowers |
Cladding removed U-bent plastic optical fiber |
Refractive index |
LSPR intensity |
Visible region (400–700 nm) |
[5.57ΔA560 nm
/ΔRIU]/- |
[6] |
| Gold nanostars |
Cladding removed plastic optical fiber |
Refractive index |
LSPR wavelength shift |
UV-Vis-nIR (380–1800 nm) |
[175 nm/RIU (LSPR2) and 580 nm/RIU (LSPR3)]/- |
[7] |
| Gold nanospheres and gold nanorods |
Reflection configuration |
Refractive index and Human IgG |
LSPR wavelength shift |
400–1100 nm |
[914 nm/RIU (GNSs) and 601 nm/RIU (GNRs)]/- |
[8] |
| Hollow gold nanocages |
Reflection configuration |
Refractive index |
LSPR wavelength shift |
300–1100 nm |
[1933 nm/RIU]/- |
[9] |
| Triangle and spherical silver nanoparticles |
Cladding removed U-bent plastic optical fiber |
Refractive index |
LSPR wavelength shift |
350–900 nm |
[1116.8 nm/RIU (triangle NPs) and 342.7 nm/RIU (spherical NPs)]/- |
[10] |
| Gold nanoparticles |
Hetero-core structured fiber |
Refractive index |
LSPR wavelength shift |
400–800 nm |
[765 nm/RIU]/- |
[13] |
| Gold nanoparticles |
Cladding removed D-type optical fiber |
Refractive index |
LSPR wavelength shift |
500–900 nm |
[3074.34 nm/RIU]/- |
[14] |
| Gold nanoparticles |
Reflection configuration |
Lead ions |
LSPR absorbance |
Visible region |
-/0.27 ppb |
[15] |
| Gold nanoparticles |
Reflection configuration |
Cadmium ions |
LSPR absorbance |
Visible region |
0.16 ppb/
1.24 ppb-1 |
[16] |
| Gold nanoparticles |
Reflection configuration |
Lead and Cadmium ions |
LSPR wavelength shift |
400–900 nm |
[0.28 nm/mM ]/
65 ppm |
[17] |
| Glucose capped silver nanoparticles |
Cladding removed U-shaped optical fiber |
Mercury ions |
LSPR absorbance |
Visible region |
-/2 ppb |
[18] |
| Chitosan capped gold nanoparticles |
Cladding removed U-bent optical fiber |
Mercury ions |
LSPR absorbance |
Visible region |
-/0.1–0.2 ppb |
[19] |
| Silver-coated gold nanostars |
Reflection configuration |
Rhodamine B |
SERS |
350–850 nm |
-/10−8 M |
[23] |
| Gold nanoparticles |
Hollow core fiber structure |
Cholesterol |
LSPR intensity |
Visible region |
[16.149 nm/µM]/
25.5 nM |
[30] |
| Gold nanorods |
Reflection configuration |
Ochratoxin A |
LSPR wavelength shift |
Visible region |
-/12 pM |
[31] |
| Gold nanoparticles |
Cladding removed multimode fiber |
Glutamate |
LSPR absorbance |
450–800 nm |
[0.0048AU/mM]/
0.36 mM |
[32] |
| Gold nanoparticles |
Cladding removed multimode fiber |
Taurine |
LSPR absorbance |
500–800 nm |
[0.0190AU/mM]/
53 µM |
[33] |
| Silver nanoparticles |
Cladding removed plastic optical fiber |
Triacylglycerides |
LSPR wavelength shift |
Visible region |
[28.5 nm/mM]/
0.016 mM |
[34] |
| Gold nanoparticles |
Cladding removed U-bent plastic optical fiber |
Blood glucose |
LSPR absorbance |
400–700 nm |
Not reported |
[35] |
| Gold nanoparticles |
Reflection configuration |
Interferon gamma (IFN-γ) and prostate specific antigen (PSA) |
LSPR intensity |
400–700 nm |
IFN- γ: 2 pg/mL
PSA: 1 pg/mL below |
[36] |
| Au nanosphere, Ag nanocube, Au nanorod and Au@Ag core-shell nanorod |
|
Metyl parathion |
SERS |
800–2000 cm−1 |
Not reported |
[38] |
| Gold nanoparticles |
|
Mercapto benzoic acid |
SERS |
400–1000 nm |
EF: 1.77 − 109 |
[40] |
2.2. Layer-By-Layer Nanoparticles Assembled Thin Films
In this section, the most relevant strategies by using the Layer-by-Layer assembly onto optical fiber are presented due to its easiness for scaling-up and a great precision on the resultant thickness
[41]. The basis of this nanodeposition technique is related to the electrostatic attraction between aqueous polyelectrolytes of opposite charge (denoted as polycations and polyanions), which can overlap at the molecular level
[42][43][44]. These polyelectrolytes can be employed as a polyelectrolyte template structure for a deposition of the metal nanoparticles or even can be used as encapsulating agents during the fabrication of the thin-films
[45][46]. According to this criteria, several works can be found in the bibliography related to the incorporation of metallic Ag or AuNPs with a well-defined morphology by using different polyelectrolytes
[47][48][49]. A representative example is shown in
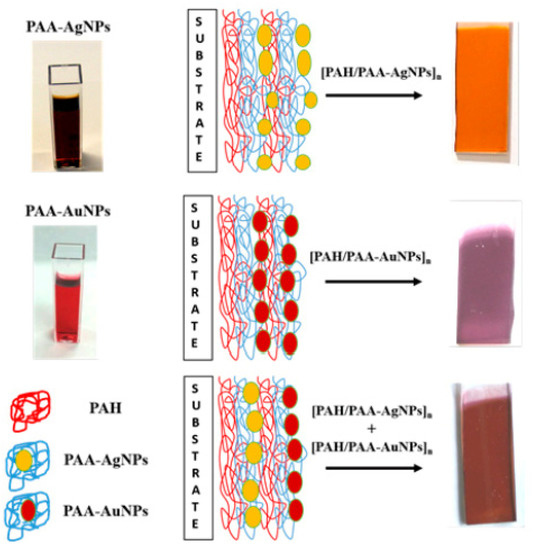
Figure 9 where poly(acrylic acid) (PAA) is used as protective agent of the silver (PAA-AgNPs) and gold (PAA-AuNPs) nanoparticles, and the presence of these nanoparticles into the LbL films can be perfectly visible to the naked eye
[50].
Figure 9. Aspect of the silver nanoparticles (PAA-AgNPs) and gold nanoparticles (PAA-AuNPs) with a schematic summary for their incorporation into LbL films. Reprinted with permission of
[50].
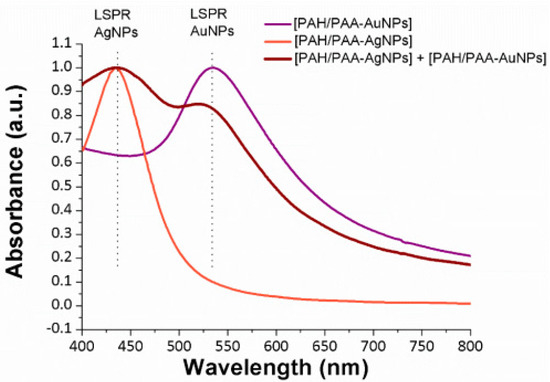
In addition, these LbL films have been also characterized in terms of optical properties (see
Figure 10), and the UV-Vis spectra corroborate the presence of the LSPR absorption bands located at 430 nm (LSPR AgNPs) and 530 nm (LSPR AuNPs), indicating the successful incorporation of metallic nanoparticles with a spherical morphology.
Figure 10. UV-Vis spectra of the LbL films for [PAH/PAA-AuNPs] (violet line), [PAH/PAA-AgNPs] (orange line) and [PAH/PAA-AgNPs] + [PAH/PAA-AuNPs] (red line), respectively. Reprinted with permission of
[50].
2.2.1. LSPR in LbL Overlays for Physical Detection
The use of the optical fiber technology for monitoring the Relative Humidity (RH %) has been continuously increasing in industrial and environmental control processes. Rivero et al. have reported the first works based on the immobilization of AgNPs by using the Layer-by-Layer assembly for this purpose
[51][52][53]. As the metal nanoparticles are immersed in a polyelectrolyte multilayer (PEM) structure, a change in the optical response is associated with a swelling/deswelling phenomenon derived from this PEM structure, showing a change in the their aggregation state and the refractive index of the surrounding medium. In addition, using AgNPs has shown an additional advantage because the growth of microorganisms or even bacteria can be prevented due to the intrinsic properties of AgNPs when the sensitive regions is exposed in high humidity ambient
[54]. Furthermore, the integration of both metal nanoparticles and fiber technology makes it possible to combine the plasmonic interactions associated with the metal structures with other optical resonant phenomena denoted as Lossy-Mode Resonances (LMR). LMRs consist of the optical coupling of certain guided modes to an external optical fiber overlay, when certain optical conditions are fulfilled
[55]. This approach can be found in
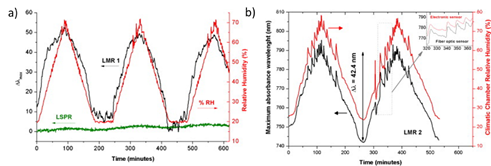
[52] where it is presented for the first time the fabrication and characterization of an optical fiber humidity sensor based on the simultaneous observation of LSPR and LMR absorption bands. As can be appreciated in
Figure 11, a considerable difference in sensitivity to RH changes is presented. In this sense, LSPR related to the immobilization of AgNPs has shown a very slight wavelength variation in comparison with the LMRs, which present a strong wavelength response to RH changes. In a later work
[53], also evaluated is this high wavelength response associated with the LMR band for high relative humidity performance such as human breathing.
Figure 11. Evolution of the dynamic response of the optical fiber sensors composed of 25 bilayers (
a) and 40 bilayers (
b), respectively. Reprinted with permission of
[52].
An interesting aspect associated with the LbL assembly is its versatility, making possible the incorporation of different types of metal nanoparticles into the polyelectrolyte multilayer structure. According to this, presented in
[56] is a self-assembled monolayer of gold nanospheres coated LSPR fiber sensor for the measurement of the refractive index. An increase in the size and density of the nanoparticles enables a better sensitivity, being around 2016.224 nm/RIU. Another aspect to remark is the possibility of encapsulating nanoparticles of different shapes into the LbL films. Rivero et al. have also incorporated AuNPs with a spherical shape by using the Layer-by-Layer Embedding (LbL-E), and as a result, by controlling the resultant thickness coating, an optical fiber device based on LSPR and LMR has been successfully designed for the detection of refractive index changes
[57]. Furthermore, in a later work, other type shapes of metal structures such as gold nanorods (GNRs) have been also deposited onto the optical fiber core by using the same nanodeposition technology (LbL-E). In this work, an optical fiber sensor is presented for the simultaneous detection of refractive index and relative humidity changes
[58]. These GNRs present two representative LSPR peaks (corroborated by UV-Vis spectra) which are denoted as LSPR-T (transversal plasmon resonance) and LSPR-L (longitudinal plasmon resonance), showing different sensitivities to refractive index changes (see
Figure 12). When the thickness coating of the LbL-E films is increased, it makes possible the appearance of a new LMR band. The experimental results showed an excellent sensitivity of 11.2 nm/%RH for the LMR, confirming the potential of this type of optical fiber sensor based on the combination of LSPRs and LMR bands.
Finally, in other works, the Layer-by-Layer assembly has been used as an effective supporting layer for a further immobilization of the metallic nanoparticles onto the polyelectrolyte structure. In
[59], gold nanorod colloids with the aspect ratio of 3 were synthesized and fixed onto a polyelectrolyte structure on the sidewall of an optical fiber. The resultant LSPR sensor shows a sensitivity of 468 nm/RIU, when the sensitive coating was immersed in sucrose solutions with variable refractive indices from 1.33 up to 1.3749, respectively. In addition, a red shift of the LSPR band is observed when the concentration of sucrose solutions is increased, whereas a blue shift is observed when the concentration of sucrose solution is decreased.
2.2.2. LSPR in LbL Overlays for Chemical Detection
As previously commented, the presence of heavy ions such mercury ions (Hg
2+) in the environment is a real concern because it is considered as a highly toxic element that can cause DNA damage or central nervous system disorders due to its toxicity and carcinogenicity. The immobilization of the metal nanoparticles into LbL thin films deposited onto optical fiber is also used for its detection thanks to the sensing signal derived from LSPR. According to this, presented in
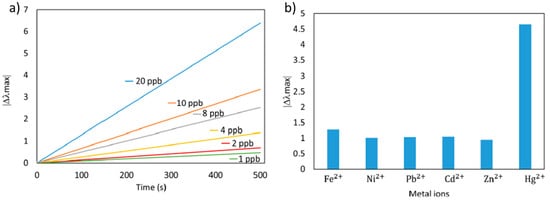
[60] is a highly sensitive optical fiber sensor using the LbL nano-assembly technique with AuNPs for mercury (Hg
2+) detection. The resultant sensor has presented a limit of detection of 0.7 ppb and low cross-sensitivity towards other heavy metal ions (see
Figure 13). Other novel work to detect Hg
2+ ions can be found in
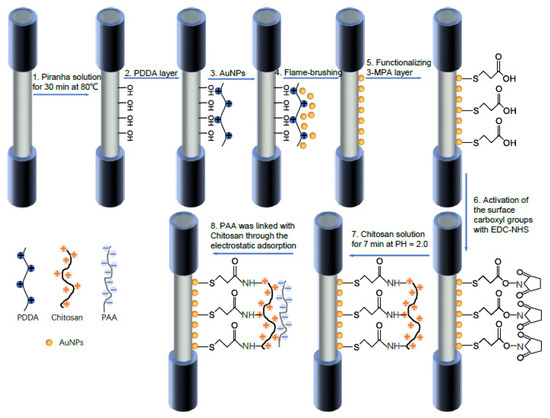
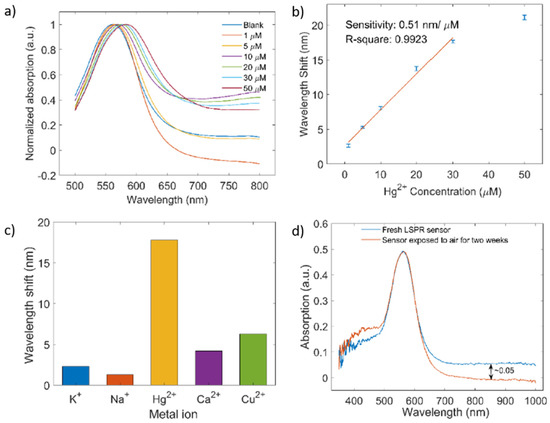
[61]. In this work, the optical fiber has been functionalized with gold nanoparticles (AuNPs) by using a flame-brushing technology and a polyelectrolyte structure composed of chitosan (CS) and polyacrylic acid (PAA) bilayers, which facilitates Hg
2+ ions adsorption on the sensor for a further chemical detection. A summary of the synthetic route for the functionalization of the optical fiber is presented in
Figure 14. The experimental results have demonstrated a linear shift with concentrations from 1 to 30 µM (see
Figure 15) and a sensitivity of around 0.51 nm/µM, showing a good specificity and longtime stability, respectively.
Figure 13. (
a) Response of the LSPR optical fiber sensor for variable Hg concentration from 1 to 20 ppb; (
b) Selectivity of the LSPR sensor against different heavy ions (Fe
2+, Ni
2+, Pb
2+, Cd
2+, Zn
2+and Hg
2+). Reprinted with permission of
[60].
Figure 14. A summary scheme for the functionalization of the optical fiber in order to obtain the LSPR sensor. Reprinted with permission of
[61].
Figure 15. (
a) Hg
2+ detection of the LSPR sensor; (
b) Sensitivity of the LSPR sensor as a function of the wavelength shift for variable concentrations of the analyte; (
c) Specificity of the LSPR sensor against different heavy metal ions; (
d) Stability of the proposed LSPR sensor. Reprinted with permission of
[61].
This LSPR sensing signal can be employed for the detection of volatile organic compounds (VOCs) or chemical substances, which can be dangerous for the human health. In this sense, an interesting approach based on a fiber optic LSPR sensor anchored with metal organic framework film is presented for acetone sensing
[62]. In this work, it has been observed a redshift of the resonance wavelength with a total reversibility to acetone which is directly associated with an increase in the local refractive index induced by the acetone adsorption into the sensitive coating.
The analytical detection of hydrogen peroxide (H
2O
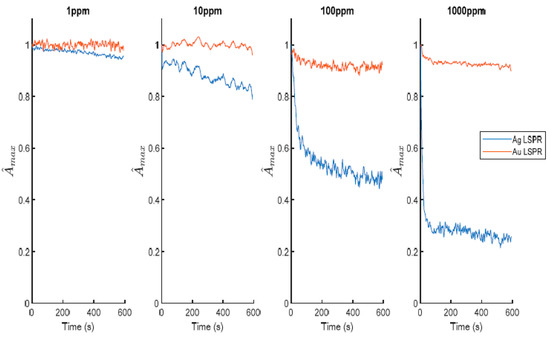
2) is a key factor in the biosensing field because it is considered as an important indicator of several diseases such as Parkinson, Alzheimer, asthma or breast cancer, among others. According to this, the use of sensitive coatings onto optical fiber can be used as an efficient technology for its detection. A representative work is presented in
[50] where a simple and robust hydrogen peroxide (H
2O
2) optical fiber sensor is proposed based on LSPR sensitivities of AgNPs and AuNPs, respectively. In
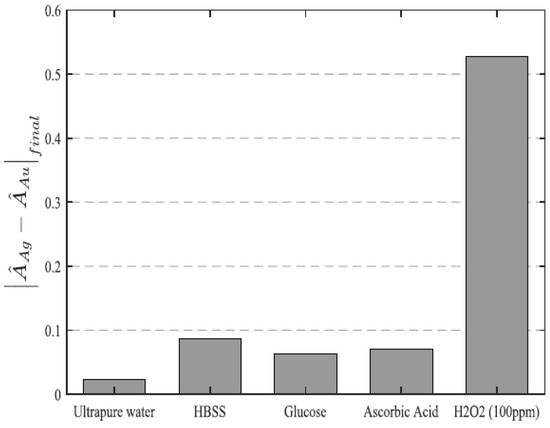
Figure 16, it can be appreciated the difference in sensitivity of both LSPR bands, where it is practically maintained at the same maximum absorbance for AuNPs, whereas the maximum absorbance for AgNPs is decreased by a high magnitude when the analyte concentration is continuously increased (from 1 to 1000 ppm). The novelty of this work is that the LSPR signal associated with the AuNPs can be used as a stable reference to get a differential measurement estimator that is noticeably more robust than the simple typical intensity-based measurements. The experimental results (see
Figure 17) have shown a good selectivity towards this specific analyte in comparison with other interfering agents (glucose, ascorbic acid) or even chemical solutions (ultrapure water, Hank’s balanced salt solution).
Figure 16. Evolution of the normalized LSPR spectra for different concentrations of the analyte (H
2O
2) from 1 up to 100 ppm. Reprinted with permission of
[50].
Figure 17. Evolution of the normalized LSPR sensor against different solutions (ultrapure water, Hanks’ Balanced Salt Solution-HBSS) or interfering agents (glucose, ascorbic acid). Reprinted with permission of
[50].
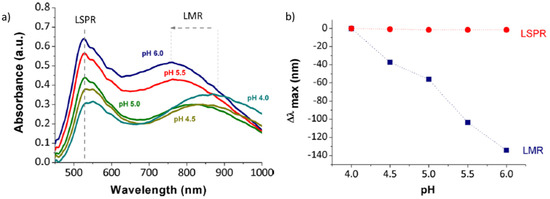
Finally, LbL-E of metal nanoparticles technique deposited onto optical fiber can be used for the chemical detection of pH variations. According to this, a representative work based on the immobilization of AuNPs into LbL films can be found in
[63] for the detection of pH changes by using both LSPR and LMR phenomena. In concordance with previous works, there is a similarity in the optical behavior of these absorption bands because the dynamic range for LMR band (67.35 nm/pH unit) is greater than LSPR band (0.75 nm/pH unit) in the pH range from 4.0 to 6.0, as shown in
Figure 18.
Figure 18. (
a) Response of the optical fiber sensor for variable pH values from 4.0 to 6.0; (
b) Shift of the LSPR and LMR bands as a function of the pH changes. Reprinted with permission of
[63].
2.2.3. LSPR in LbL Overlays for Biological Detection
The use of the sensing signal of the LSPR into LbL films can be also extrapolated as a powerful tool in protein sensing applications by using different optical fiber configurations. An interesting approach is found in
[64] where a highly sensitive protein sensor based on tapered optical fiber modified with AuNPs is presented. The basis of the sensing methodology is related to changes in the refractive index of the polyelectrolyte structure by the streptavidin (SV) binding to the biotin, showing a limit of detection (LOD) of 271 pM, respectively. However, in
[65] it is coated the end-face of optical fiber and the reflection mode has been performed to the detection of the biotin-streptavidin bioconjugate pair, showing a sensitivity of around 800 pg/mm
2, respectively. Evaluated in
[66] is the effective plasmon penetration depth by using AuNPs of variable size with the aim to optimize and improve the sensitivity of the LSPR biosensor. Another representative application of the LSPR is the detection of different types of antigens such as Immunoglobulins (Ig). Presented in
[67] are gold nanoparticle multilayers which are highly sensitive and selective to surface modifications, detecting stable binding of antigen (Immunoglobulin G, IgG), whereas other novel works are focused on the immobilization of silica core gold shell nanoparticles (denoted as SiO
2@AuNPs) onto optical fiber long period gratings (LPGs) for the detection of human IgM
[68]. In this work, the dynamic binding of IgM on the LPG can be observed at a concentration of 0.3 µM, showing a limit of detection of 0.0218 ng/mm
2. In addition, this same sensitive coating has been employed in
[69] for streptavidin detection, showing a high sensitivity with a limit of detection of 0.86 pg/mm
2, respectively. The effective plasmon penetration depth of hollow gold nanostructures (HGNS) immobilized onto U-bent optical fiber has been studied in
[70] for the detection of E. Coli B40 strain using bacteriophage T4. The results corroborate that the response of the sensor has been better for the HGNS in comparison with spherical gold nanoparticles.
As it was previously commented, the Layer-by-Layer polyelectrolyte multilayer structure can be also employed as an efficient matrix for a further assembly of the metallic nanoparticles
[71]. The experimental results indicate that the LSPR sensor shows good refractive index sensitivity and is also used to conduct real-time and label free monitoring of Ribonuclease and Concavidine A (Con A) biomolecular interaction. Other interesting work for conducting real-time and label free monitoring of both IgG/anti-IgG and Con A/RNase B biomolecular interaction is presented in
[72]. Finally, in
[73] gold nanoparticles with spherical shape and variable size (48 ± 6 nm and 23 ± 2 nm, respectively) are successfully self-assembled onto a previously fabricated trilayer polyelectrolyte structure, as observed in the SEM images in
Figure 19. These assembled AuNPs are uniformly dispersed onto the LbL film without showing any aggregates. In addition, the response of LSPR optical fiber sensors to different concentrations of sucrose solutions is presented in
Figure 20. In both cases, a red shift of the LSPR band has been observed and the transmission peak intensities have been reduced when the sucrose concentration has been increased, showing a good and linear correlation. This optical fiber LSPR sensor has been also used for the immunoassay of goat anti-rabbit IgG and the lowest detection concentration for this biosensing analyte has been 11.1 ng/mL.
A summary of the different optical fiber sensors based on LbL nanoparticles assembled thin films is presented in
Table 2.
| Type of NPs |
Optical Structure |
Target |
Sensing Mechanism |
Wavelength
Range |
Sensitivity/LOD |
Ref |
Spherical
AgNPs |
Cladding removed multimode fiber |
Relative humidity |
LSPR intensity |
400–700 nm |
Not reported |
[51] |
Spherical
AgNPs |
Cladding removed multimode fiber |
Relative humidity |
LSPR intensity and LMR wavelength shift |
400–1100 nm |
0.943 nm/RH% (LMR2) 0.126 nm/RH% (LMR3)/- |
[52] |
Spherical
AgNPs |
Cladding removed multimode fiber |
Human breathing |
LMR wavelength shift |
900–1600 nm |
[0.455 nm/RH%]/- |
[53] |
| Gold nanospheres |
Reflection configuration |
Refractive index |
LSPR wavelength shift |
400–900 nm |
[2016.224 nm/RIU]/- |
[56] |
Spherical
AuNPs |
Cladding removed multimode fiber |
Refractive index |
LSPR intensity and LMR wavelength shift |
400–1100 nm |
8922 nm/RIU (LMR1), 4037 nm/RIU (LMR2), 1906 nm/RIU (LMR3)/- |
[57] |
| Gold nanorods (GNRs) |
Cladding removed multimode fiber |
Refractive index and relative humidity |
LSPR-L intensity, LSPR-T intensity and LMR wavelength shift |
400–1600 nm |
75.69 dB/RIU (LSPR-T), 50.46 dB/RIU (LSPR-L)
11.2 nm/%RH (LMR)/- |
[58] |
| Gold nanorods |
Cladding removed multimode fiber |
Refractive index |
LSPR wavelength shift |
Visible region |
[468 nm/RIU]/- |
[59] |
Spherical
AuNPs |
Cladding removed multimode fiber |
Hg2+ |
LSPR wavelength shift |
420–670 |
-/0.7 ppb |
[60] |
| AuNPs |
Cladding removed fiber by flame-brushing technology |
Hg2+ |
LSPR wavelength shift |
400–950 nm |
[0.51 nm/µM]/- |
[61] |
| AuNPs |
Reflection |
Acetone |
LSPR wavelength shift |
400–850 nm |
Not reported |
[62] |
| Spherical AgNPs+AuNPs |
Cladding removed multimode fiber |
Hydrogen peroxide |
LSPR intensity |
400–900 nm |
-/2.7 ppm |
[50] |
Spherical
AuNPs |
Cladding removed multimode fiber |
pH changes |
LSPR and LMR wavelength shift |
450–1000 nm |
0.75 nm/pH unit (LSPR), 67.35 nm/pH unit (LMR)/- |
[63] |
| AuNPs |
Taper |
Streptavidin |
Wavelength shift |
380–1000 nm |
-/271 pM |
[64] |
| AuNPs |
Reflection |
Streptavidin |
LSPR wavelength shift |
350–1000 nm |
465.5 nm/(mg/mL)/- |
[65] |
| SiO2@AuNPs |
Long period grating |
HIgM |
Wavelength shift |
650–1000 nm |
11 nm/(ng/mm2)/
0.0218 ng/mm2 |
[68] |
| SiO2@AuNPs |
Long period grating |
Streptavidin |
Wavelength shift |
650–1000 nm |
3.88 (ng/mm2)−1/0.86pg/mm2 |
[69] |
| Hollow gold nanostructures |
Cladding removed U-bent plastic optical fiber |
E. Coli |
LSPR absorbance |
450–800 nm |
Not reported |
[70] |
| AuNPs |
Cladding removed multimode fiber |
Concavidine A |
LSPR intensity |
400–900 nm |
Not reported |
[71] |
Spherical
AuNPs |
Cladding removed multimode fiber |
Goat anti-rabbit IgG |
LSPR intensity |
400–800 nm |
-/11.1 ng/mL |
[73] |