
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maurizio Nicola D'Alterio | + 833 word(s) | 833 | 2020-03-27 10:14:04 | | | |
| 2 | Maurizio Nicola D'Alterio | + 833 word(s) | 833 | 2020-03-27 13:25:37 | | | | |
| 3 | Rita Xu | Meta information modification | 833 | 2020-04-17 08:38:57 | | | | |
| 4 | Rita Xu | Meta information modification | 833 | 2020-04-17 08:52:30 | | | | |
| 5 | Rita Xu | Meta information modification | 833 | 2020-10-29 10:23:41 | | | | |
| 6 | Rita Xu | + 2060 word(s) | 2893 | 2020-11-06 10:43:04 | | |
Video Upload Options
The purpose of this study was to evaluate the recent literature on the genetic characterization of women affected by endometriosis and to evaluate the influence of polymorphisms of the wingless-type mammalian mouse tumour virus integration site family member 4 (WNT4), vezatin (VEZT), and follicle stimulating hormone beta polypeptide (FSHB) genes, already known to be involved in molecular mechanisms associated with the proliferation and development of endometriotic lesions.
1. Introduction
Endometriosis is a chronic disease of the reproductive age with a prevalence of 5%, reaching its peak between 25 and 35 years of age [1]. The scientific community agrees in recognizing a multifactorial aetiology of endometriosis, with possible genetic, hormonal, immunological and environmental factors as causes. Studies on family aggregation and twins have emphasized the genetic component, demonstrating how predisposition generated by certain susceptibility genes plays an important role in the development, maintenance and recurrence of the disease [2][3][4][5]. Nonetheless, studies on the genetics of endometriosis are complicated by various factors: the phenotypic heterogeneity of the disease; the still unknown prevalence in the population, burdened by the absence of registries and diagnostic underestimation; the invasiveness of diagnostic methods; and various co-morbidities that can generate bias [3]. In the field of research on the genetic basis of endometriosis, Simpson is considered a pioneer. In 1980, he verified in a sample of 123 women with histological diagnosis of endometriosis that 6.9% of first-degree relatives (mother and sisters) were affected, while the disease prevalence in controls (first-degree female relatives of the corresponding “husbands”) was less than 1% [6]. In 1999, Treloar’s work on an Australian population demonstrated a concordance ratio of 2:1 between monozygotic and dizygotic twins and a correspondent genetic risk of 2.34 to affect a sister. The study results show that 51% of genetic influence is responsible for developing endometriosis [7]. There is therefore enough evidence on how endometriosis is clearly heritable, although in what manner is not yet clear. The increased genetic risk in first-degree relatives (5–8%) suggests polygenic and multifactorial inheritance rather than monogenic. However, this recurrence risk is higher than the expected risk for a polygenic pathology (2–5%). The other, more likely, hypothesis is that phenotypic heterogeneity reflects genetic heterogeneity and that therefore not all forms of endometriosis are the same disease; in fact, some forms of endometriosis, due to their characteristics, behave almost like Mendelian pathologies [2]. The aim of this study was to evaluate the recent literature on the genetic characterization of women affected by endometriosis and to evaluate the influence of polymorphisms of the WNT4, VEZT and FSHB genes, known to be involved in molecular mechanisms associated with proliferation and development of endometriotic lesions in a particular Mediterranean population, the Sardinian population. The people of the Mediterranean island of Sardinia are particularly well suited for genetic studies, as is evident from a number of successes in complex trait and disease mapping [8].
The wingless-type mammalian mouse tumour virus integration site family member 4 (WNT4) gene is positioned on chromosome 1p36.23-p35 and codifies a protein which is essential in developing the female reproductive system [9][10][11][12][13][14]. It critically regulates the appropriate postnatal uterine maturation, as well as ovarian antral follicle growth [10]. The WNT class is an extensive group of secreted glycoproteins, codified through 19 different genes implicated in the WNT signalling pathway [9]. WNT-mediated signal transduction pathways address the specific mobilization of groups of genes which are responsible for managing several cellular responses, including cell growth, differentiation, movement, migration, polarity, cell survival and immune response [9]. A study published by Jordan et al. [15] demonstrated that WNT4 is the first signalling molecule causing the chain of events which ends with sex determination, through local secernment of growth factors. Imperfections in WNT4 activity play a role in the development of three important organs deriving from the primordial urogenital ridge—the kidneys, adrenal glands and gonads [9]. This may demonstrate the significant position of WNT4 at an early embryological stage of development. The loss of WNT4 in knockout mice determines the total absence of the Mullerian duct and its derivatives [11]. Apart from being crucial for epithelial–stromal cell communication in the endometrium, WNT signalling is likely important for endometrial maturation and differentiation and embryonic implantation [16]. An association between endometriosis and markers located in or near WNT4 has been highlighted in a number of extensive studies on gene mapping [17][18]. The expression of WNT4 has also been detected at the level of the peritoneum, leading to the consideration of a possible metaplastic hypothesis in promoting the transformation of peritoneal cells into endometriosic cells, through pathways with a role in the development of the female genital tract [12]. Pagliardini et al. demonstrated that a single nucleotide polymorphism (SNP), rs7521902, located 21 kb up/downstream of the WNT4 region, has a susceptibility locus for endometriosis. The functional significance of this SNP in endometriosis remains to be explained [10]. While the SNP rs7521902 was connected to endometriosis susceptibility in British, Australian, Italian and Japanese women [10][18][19], in Belgian [13] and Brazilian women this association was not found [9]. The different genetic backgrounds of the cohorts may be identified as the reason for the lack of association between this polymorphism and endometriosis.
The vezatin (VEZT) gene is located on chromosome 12 locus 12q22; it codifies vezatin, a significant element of the cadherin–catenin complex, which plays a crucial role in the formation and sustenance of adherent joints [20][21][22][23][24]. According to Kussel-Andermann et al., vezatin was found to be a plasma membrane component with a short extracellular domain, a transmembrane domain and an extended intracellular domain. Its intracellular domain connects to myosin VIIA as part of the adherens junctional complex in epithelial cells [21]. Furthermore, several studies on co-immunoprecipitation showed that the system between vezatin and myosin VIIA is able to interact with the system between E-cadherin and catenin, although the specificity of this interaction remains to be determined [21][22]. Also, VEZT is fundamental for implantation; embryos from mice with silenced VEZT cannot develop after the blastocyst stage, because of a loss of adhesion between cells [25]. It has been highlighted that VEZT protein is extensively expressed in human endometrium and myometrium. During the secretory phase of the menstrual cycle, VEZT expression increases in the glandular epithelium in a significant way. The mRNA expression of adherens junction members (E-cadherin and A- and B-catenin) is also enhanced in the secretory phase with respect to the proliferative phase. This indicates that progesterone could be responsible for activating cell-to-cell adhesion [20]. The VEZT promoter does not contain a reaction point for the progesterone receptor (PR), but it contains a nuclear factor kappa B (NF-kB) binding site. As a pro-inflammatory transcription factor, NF-kB is involved in the pathogenesis of endometriosis, showing cycle control in the endometrium and reciprocal management with PR [26]. Therefore, variations in VEZT levels in endometrial glandular cells are likely to occur in response to the dynamic oscillation in progesterone and associated NF-kB changes [20]. Considering the studied physiological roles of VEZT, its potential for a functional role in endometriosis is a likely option, since VEZT has been demonstrated to be upregulated in ectopic endometrium with respect to eutopic endometrium in patients suffering from endometriosis [20][23][24].
The follicle-stimulating hormone beta polypeptide (FSHB) gene, positioned on chromosome 11 locus 11p14.1, codifies subunit b of the hormone-specific follicle-stimulating hormone (FSH), with a crucial role in the growth of ovarian follicles and production of oestrogens [27][28][29]. Recently, some evidence for an association between endometriosis and SNPs of FSHB was reported in independent targets from the UK Biobank, firmly supporting this result [28]. FSH and luteinizing hormone (LH) are related gonadotropin hormones sharing the same alpha subunit. A connection between these SNPs on chromosome 11 with concentrations of both hormones indicates a common mechanism of regulation, with both being key elements in managing follicle development in the ovary, influencing oestradiol release during the proliferative phase of the cycle and contributing to a role for oestradiol in endometriosis risk [27]. Data from the ENCODE project [30] show that the SNP rs11031006 modifies the sequence of 11 protein-binding motifs, including that of oestrogen receptor α, with a possible effect on hormonal feedback inhibition. Recently, allele G of this SNP has been proven to be significantly associated with higher levels of serum FSH [29].
1.1. Review of the Literature
This entry aims to provide a comprehensive and systematic tool for those approaching the genetics of endometriosis. Computerized literature searches were conducted using the Medline/PubMed database and a manual search of relevant and frequently cited publications in the English language from 1999 to 2019. Additional articles were identified by manually searching references from the retrieved eligible articles. Keywords included: endometriosis combined with genes, genetics, SNPs, genome-wide association study (GWAS), WNT4, VEZT, FSHB, next-generation sequencing (NGS) and epigenetics. Review, observational, cohort and case-control studies that evaluated the genetics of endometriosis are herein described.
1.2. Cohort Study
The study was carried out through molecular typing of the following single nucleotide polymorphisms (SNPs): rs7521902, rs10859871 and rs11031006, mapped, respectively, in the WNT4, VEZT and FSHB genes. In this work, we set out to describe the frequency of alleles and genotypes of these SNPs among Sardinian women, and to evaluate their impact on the susceptibility to develop endometriosis. The choice of which polymorphisms were to be analysed fell to WNT4, VEZT and FSHB genes because of their hypothetical correlation with endometriosis, according to recent findings in the literature. Several GWAS meta-analyses have reported a possible pathogenetic role [16][27]. In particular, an association study published in 2017 analysed these SNPs in a Greek population, highlighting a significant connection with the disease [29]. The molecular biological examinations of single-substitution polymorphisms in our study were carried out from whole blood drawn exclusively from Sardinian patients, upon their informed consent. We chose to limit the study to only patients of Sardinian origin, considering the peculiarity of this island, which can be seen as a genetic macro-isolate. The choice of limiting the selection of the target population to patients of Sardinian origin for at least three generations was not intended to exclude the possible influence of genetic features from non-Sardinian distant ancestors. It was, rather, an attempt to make the target more homogeneous.
The genetic analysis of complex traits is simplified in isolated populations such as this one, in which inbreeding and the “founder effect” reduce the genetic diversity of complex and polygenic diseases such as endometriosis. The goal was to identify a genetic characterization of the disease in the Sardinian population, to shed light on the etiopathogenetic mechanisms of endometriosis, and to provide predictive markers for a non-invasive diagnosis of the disease.
Patients and Study Design
The present clinical study represents the initial application, on a small scale, of a study protocol conducted at the Department of Obstetrics and Gynaecology of the Hospital “Duilio Casula” of Monserrato, University of Cagliari, in collaboration with the Laboratory of Genetics and Genomics of the Pediatric Hospital Microcitemico “A. Cao” of Cagliari. Written consent was obtained from the local ethics committee (EndoSNPs, Prot. PG/2019/13157). In line with the Declaration of Helsinki 1975, revised in Hong Kong in 1989, the clinical trial was registered (ClinicalTrials.gov ID: NCT02388854). This study involved a total of 72 women who underwent surgery, 41 representing the cases with clinical and histological diagnosis of endometriosis, and 31 representing the corresponding controls—women without a diagnosis of endometriosis who underwent surgery for benign indications different from endometriosis and chronic pelvic pain. For each patient, a data collection folder was prepared in a database elaborated for the purpose. Apart from personal records, data on geographic origin and anthropometric measurements (weight, height, body mass index (BMI)), the data collection form contained data on habits such as cigarette smoking and alcohol intake, and clinical data on possible co-morbidities or previous surgical interventions. In patients with a diagnosis of endometriosis, data on this pathology were obviously included (familiarity, staging, location of lesions, transvaginal ultrasound data, gynaecological examination, symptomatology, number and type of surgical operations, histological data), type and duration of drug therapy (nonsteroidal anti-inflammatory drugs (NSAIDs), hormone therapy and antibiotics). The diagnosis of endometriosis was clinical, related to ultrasound and histology reports. The staging used was the revised classification proposed by the American Fertility Society/American Society for Reproductive Medicine (AFS/ASRM) [30], which is still the most widespread and widely used, in clinical and academic fields, to describe the severity of endometriosis in a standardized format. Localizations of deep infiltrating endometriosis (DIE) infiltrating the bladder or bowel were classified as severe. For each main symptom (dysmenorrhea, chronic pelvic pain, dyspareunia, intestinal symptoms, urinary symptoms), a score was assigned based on the intensity perceived by the patient (visual analogue scale (VAS) 0–3: mild; 4–7: moderate; 8–10: severe). For the controls, women who had never had a diagnosis or a suspicion of endometriosis were selected. The close and remote pathological medical history of these patients was also negative for dysmenorrhea, dyspareunia, chronic pelvic pain and infertility/sub-fertility, as one of the inclusion criteria was to have given birth to at least 2 children. A common criterion to be included in the study was geographic origin. All participants had an exclusively Sardinian origin for at least 3 generations (maternal and paternal grandparents from Sardinia).
2. Discussion
Our analysis of the recent literature shows evidence of many genes possibly implicated in different pathogenetic mechanisms of endometriosis (Figure 1).
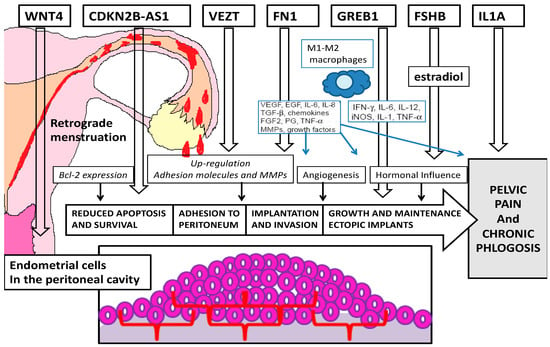
Figure 1. Pathogenetic targets of the most important genes related to endometriosis.
The wingless-type mammalian mouse tumor virus integration site family member 4 (WNT4) gene is positioned on chromosome 1p36.23-p55 and codifies a protein which is essential in developing the female reproductive system [31][32][33][34][35][36]. It critically regulates the appropriate postnatal uterine maturation, as well as ovarian antra follicle growth[32]. The WNT class is an extensive group of secreted glycoproteins, codified through 19 different genes implicated in the WNT signaling pathway[31]. WNT-mediated signal transduction pathways address the specific mobilization of groups of genes which are responsible for managing several cellular responses, including cell growth, differentiation, movement, migration, polarity, cell survival and immune response[31]. Imperfection in WNT4 activity play role in the development of three important organs deriving from the primordial urogenital ridge- the kidneys, adrenal glands and gonads[31]. This may demonstrate the significant position of WNT4 at an early embryological stage of development. The loss of WNT4 in knockout mice determines the total absence of the Mullerian duct and its derivates[33]. Apart from being crucial for epithelial-stromal cell communication in the endometrium, WNT signalling is likely important for endometrial maturation an differentiation and embryonic implantation[37]. An association between endometriosis and markers located in or near WNT4 has been highlighted in a number of extensive studies on gene mapping[38][39]. The expression of WNT4 has also been detected at the level of the peritoneum, leading to the consideration of a possibile metaplastic hypothesis in promoting the transformation of peritoneal cells into endometriosic cells, through pathways with a role in the development of the female genital tract[34]. Pagliardini et al. demonstrated that a single nucleotide polymorphisms (SNP), rs7521902, located 21 kb up/downstream of the WNT4 region, has a susceptibility locus for endometriosis. The functional significance of this SNP in endometriosis remains to be explained[32].
The vezatin (VEZT) gene is located on chromosome 12 locus 12q22; it codifies vexation, a significant element of the cadherin-catenin complex, which plays a crucial role in the formation and sustenance of adherent joints[40][41][42][43]. According to Kussel-Andermann et al., vezatin was found to be a plasma membrane component with a short extracellular domain, a transmembrane domain and an extended intracellular domain. Its intracellular domain connects to myosin VIIA as part of the adherent junctional complex in epithelial cells[41]. Furthermore, several studies on co-immunoprecipitation showed that the system between vezatin and myosin VIIA is able to interact with the system between E-cadherin and catenina, although the specificity of this interaction remains to be determined[41][42]. Also, VEZT is fundamental for implantation; embryos from mice with silenced VEZT cannot develop after the blastocyst stage, because of loss of adhesion between cells[44]. It has been highlighted that VEZT protein is extensively expressed in human endometrium and myometrium. The mRNA expression of adherents junction members is also enhanced in the secretory phase with respect to the proliferative phase. This indicates the progesterone could be responsible for activating cell-to-cell adhesion[40]. The VEZT promote does not contain a reaction point for the progesterone receptor (PR), but it contains a nuclear factor kappa B (NF-kB) binding site. As a pro-inflammatory transcription factor, NF-kB is involved in the pathogenesis of endometriosis, showing cycle control in the endometrium and reciprocal management with PR[45]. Considering the studied physiological roles of VEZT, its potential for a functional role in endometriosis is a likely option, since VEZT has been demonstrated to be upregulated in ectopic endometrium with respect to eutopic endometrium in patients suffering from endometriosis[41][46][43].
The follicle-stimulating hormone beta polypeptide (FSHB) gene, positioned on chromosome 11 locus 11p14.1, codifies subunit b of the hormone-specific-follicle-stimulating hormone (FSH) with a crucial role in the growth of ovarian follicles and production of estrogens[47][48][49]. Recently, some evidence for an association between endometriosis and SNPs of FSHB was reported in independent targets from the UK Biobank, firmly supporting this result[48]. FSH and luteinizing hormone (LH) are related gonodotropin hormones sharing the same alpha subunit. A connection between these SNPs on chromosome 11 with concentrations of both hormones indicates a common mechanism of regulation, with both being key elements in managing follicle development in the ovary, influencing estradiol release during the proliferative phase of the cycle and contributing to a role for estradiol in endometriosis risk[47]. Data from the ENCODE project show that the SNP rs11031006 modifies the sequence of 11 protein-binding motifs, including that of estrogen receptor alfa, with a possibile effect on hormonal feedback inhibition. Recently, allele G of this SNP has been proven to be significantly associated with higher levels of serum FSH[49].
References
- Alio, L.; Angioni, S.; Arena, S.; Bartiromo, L.; Bergamini, N.; Berlanda, N.; Bonanni, V.; Bonin, C., Buggio, L.; Candiani, M.; et al. [ETIC group]. Endometriosis: Seeking optimal management in women approaching menopause. Climacteric 2019, 22, 329–338.
- Deiana, D.; Gessa, S.; Anardu, M.; Danilidis, A.; Nappi, L.; D’Alterio, M.N.; Pontis, A.; Angioni, S. Genetic of endometriosis: A comprehensive review. Gynecol. Endocrinol. 2019, 35, 553–558.
- Melis, I.; Agus, M.; Pluchino, N.; Sardo, A.D.S.; Litta, P.; Melis, G.B.; Angioni, S. Alexithymia in women with deep endometriosis? A pilot. Study. J. Endometr. Pelvic. Pain Dis. 2014, 6, 26–33.
- Angioni, S. New insights on endometriosis. Minerva Ginecol. 2017, 69, 438–439.
- Locci, R.; Nisolle, M.; Angioni, S.; Foidart, J.M.; Munat, C. Expression of the gamma 2 chain of laminin-332 in eutopic and ectopic endometrium of patients with endometriosis. Rep. Biol. Endocrinol. 2013, 11, 94.
- Simpson, J.L.; Elias, S.; Malinak, L.R.; Buttram, V.C. Jr. Heritable aspects of endometriosis. Genetic studies. Am. J. Obstet. Gynecol. 1980, 137, 327–331.
- Treloar, S.A.; O'Connor, D.T.; O'Connor, V.M.; Martin, N.G. Genetic influences on endometriosis in an Australian twin sample. Fertil. Steril. 1999, 71, 701–710.
- Lettre, G.; Hirschhorn J.N. Small island, big genetic discoveries. Nat. Genet. 2015, 47, 1224–1225.
- Mafra, F.; Catto, M.; Bianco, B.; Barbosa, C.P.; Christofolini, D. Association of WNT4 polymorphisms with endometriosis in infertile patients. J. Assist. Reprod. Genet. 2015, 32, 1359–1364.
- Pagliardini, L.; Gentilini, D.; Vigano', P.; Panina-Bordignon, P.; Busacca, M.; Candiani, M.; Di Blasio, A.M. An Italian association study and meta-analysis with previous GWAS confirm WNT4, CDKN2BAS and FN1 as the first identified susceptibility loci for endometriosis. J. Med. Genet. 2013, 50, 43–46.
- Vainio, S.; Heikkilä, M., Kispert, A.; Chin, N.; McMahon, A.P. Female development in mammals is regulated by Wnt-4 signalling. Nature 1999, 397, 405–409.
- Gaetje. R.; Holtrich, U.; Engels, K.; Kissler, S.; Rody, A.; Karn, T.; Kaufmann, M. Endometriosis may be generated by mimicking the ontogenetic development of the female genital tract. Fertil. Steril. 2007, 87, 651–656.
- Sundqvist, J.; Xu, H.; Vodolazkaia, A.; Fassbender, A.; Kyama, C.; Bokor, A.; Gemzell-Danielsson, K.; D'Hooghe, T.M.; Falconer, H. Replication of endometriosis-associated single-nucleotide polymorphisms from genome-wide association studies in a Caucasian population. Hum. Reprod. 2013, 28, 835–839.
- Wu, Z.; Yuan, M.; Li.; Fu, F.; Ma, W.; Li, H.; Wang, W. Wang, S. Analysis of WNT4 polymorphism in Chinese Han women with endometriosis. Reprod Biomed Online 2015, 30, 415–420.
- Jordan, B.K.; Mohammed, M.; Ching, S.T.; Délot, E.; Chen, X.N.; Dewing, P.; Swain, A.; Rao, P.N.; Elejalde, B.R.; Vilain, E. Up-regulation ofWNT-4 signaling and dosage-sensitive sex reversal in humans. Am. J. Hum. Genet. 2001, 68, 1102–1109.
- Rahmioglu, N.; Nyholt, D.R.; Morris, A.P.; Missmer, S.A.; Montgomery, G.W.; Zondervan, K.T. Genetic variants underlying risk of endometriosis: Insights from meta-analysis of eight genome-wide association and replication datasets. Hum. Reprod. Update 2014, 20, 702–716.
- Nyholt, D.R.; Low, S.K.; Anderson, C.A.; Painter, J.N.; Uno, S.; Morris, A.P.; MacGregor, S.; Gordon, S.D.; Henders, A.K.; Martin, N.G.; et al. Genome-wide association meta-analysis identified new endometriosis risk loci. Nat. Genet. 2012, 44, 1355–1359.
- Uno, S.; Zembutsu, H.; Hirasawa, A.; Takahashi, A.; Kubo, M.; Akahane, T.; Aoki, D.; Kamatani, N.; Hirata, K.; Nakamura, Y. A genome-wide association study identifies genetic variants in the CDKN2BAS locus associated with endometriosis in Japanese. Nat. Genet. 2010, 42, 707–710.
- Painter, J.N.; Anderson, C.A.; Nyholt, D.R.; Macgregor, S.; Lin, J.; Lee, S.H.; Lambert, A.; Zhao, Z.Z.; Roseman, F.; Guo, Q.; et al. Genome-wide association study identifies a locus at 7p15.2 associated with the development of endometriosis. Nat. Genet. 2011, 43, 51–54.
- Haldsworth-Carson, S.J.; Fung, J.N.; Luong, H.T.; Sapkota, Y.; Bowdler, L.M.; Wallace, L.; Teh, W.T.; Powell, J.E.; Girling, J.E.; Healey, M.; et al. Endometrial vezatin and it association with endometriosis risk. Hum. Reprod. 2016, 31, 999–1013.
- Küssel-Andermann, P.; El-Amraoui, A.; Safieddine, S.; Nouaille, S.; Perfettini, I.; Lecuit, M.; Cossart, P.; Wolfrum, U. Petit, C. Vezatin, a novel transmembrane protein, bridges myosin VIIA to the cadherin-catenins complex. Embo J. 2000, 19, 6020–6029.
- Blaschuk, O.W.; Rowlands, T.M. Plasma membrane components of adherens junctions. Mol. Membr. Biol. 2002, 19, 75–80.
- Luong, H.T.T.; Painter, J.N.; Sapkota, Y.; Niholt, D.R.; Rogers, P.A.; Montgomery, G.W. Identifying the functional role of VEZT Gene for endometriosis risk. Ann.Transl. Med. 2015, 3, AB028.
- Pagliardini, L.; Gentilini, D.; Sanchez, A.M.; Candiani, M.; Viganò, P.; Di Blasio, A.M. Replication and meta-analysis of previous genome-wide association studies confirm vezatin as the locus with the strongest evidence for association with endometriosis. Hum. Reprod. 2015, 30, 987–993.
- Meola, J.; Rosa, E.; Silva, J.C.; Dentillo, D.B.; Da Silva, W.A. Jr; Veiga-Castelli, L.C.; Bernardes, L.A.; Ferriani, R.A.; De Paz, C.C.; Giuliatti, S.; et al. Differentially expressed genes in eutopic and ectopic endometrium of women with endometriosis. Fertil. Steril. 2010, 93, 1750–1773.
- Guo S-W. Nuclear factor-kappab (NF-κB): An unsuspected major culprit in the pathogenesis of endometriosis that is still at large? Gynecol. Obstet. Investig. 2007, 63, 71–97.
- Sapkota, Y.; Steinthorsdottir, V.; Morris, A.P.; Fassbender, A.; Rahmioglu, N.; De Vivo, I.; Buring, J.E.; Zhang, F.; Edwards, T.L.; Jones, S.; et al. Meta-analysis identifies five novel loci associated with endometriosis highlighting key genes involved in hormone metabolism. Nat. Commun. 2017, 8, 15539.
- Ruth, K.S.; Beaumont, R.N.; Tyrrell, J.; Jones, S.E.; Tuke, M.A.; Yaghootkar, H.; Wood, A.R.; Freathy, R.M.; Weedon, M.N.; Frayling, T.M.; et al.; Genetic evidence that lower circulating FSH levels lengthen menstrual cycle, increase age at menopause and impact female reproductive health. Hum. Reprod. 2016, 31, 473–481.
- Matalliotakis, M.; Zervou, M.I.; Matalliotaki, C.; Ramhioglu, N.; Koumantakis, G.; Kalogiannidis, I.; Prapas, I; Zondervan, K.; Spandidos, D.A.; Matalliotakis, I.; et al. The role of gene polymorphisms in endometriosis. Molec. Med. Rep. 2017, 16, 5881–5886.
- Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil. Steril. 1997, 67, 817–821.
- Fernanda Mafra; Michele Catto; Bianca Bianco; Caio Parente Barbosa; Denise Maria Christofolini; Association of WNT4 polymorphisms with endometriosis in infertile patients. Journal of Assisted Reproduction and Genetics 2015, 32, 1359-1364, 10.1007/s10815-015-0523-1.
- Luca Pagliardini; Davide Gentilini; Paola Viganó; Paola Panina-Bordignon; Mauro Busacca; Massimo Candiani; Anna Maria Di Blasio; An Italian association study and meta-analysis with previous GWAS confirmWNT4,CDKN2BASandFN1as the first identified susceptibility loci for endometriosis. Journal of Medical Genetics 2012, 50, 43-46, 10.1136/jmedgenet-2012-101257.
- Vaino S; Heikkila M; Kispert A; Female Development in Mammals is Regulated by Wnt-4-Signaling. The Endocrinologist 1999, 9, 323, 10.1097/00019616-199907000-00014.
- R Gaetje; Uwe Holtrich; Knut Engels; Stefan Kissler; Achim Rody; Thomas Karn; Manfred Kaufmann; Endometriosis may be generated by mimicking the ontogenetic development of the female genital tract. Fertility and Sterility 2007, 87, 651-656, 10.1016/j.fertnstert.2006.07.1533.
- J. Sundqvist; A. Vodolazkaia; A. Fassbender; C. Kyama; A. Bokor; K. Gemzell-Danielsson; T. M. D'hooghe; Henrik Falconer; H. Xu; Replication of endometriosis-associated single-nucleotide polymorphisms from genome-wide association studies in a Caucasian population. Human Reproduction 2013, 28, 835-839, 10.1093/humrep/des457.
- Zhangying Wu; Ming Yuan; Yan Li; Fangfang Fu; Wenqinq Ma; Haixia Li; Wenwen Wang; Shixuan Wang; Analysis of WNT4 polymorphism in Chinese Han women with endometriosis. Reproductive BioMedicine Online 2015, 30, 415-420, 10.1016/j.rbmo.2014.12.010.
- Nilufer Rahmioglu; Dale R. Nyholt; Andrew P. Morris; Stacey A. Missmer; Grant Montgomery; Krina T. Zondervan; Genetic variants underlying risk of endometriosis: insights from meta-analysis of eight genome-wide association and replication datasets. Human Reproduction Update 2014, 20, 702-16, 10.1093/humupd/dmu015.
- Dale R. Nyholt; Siew-Kee Low; Carl A. Anderson; Jodie N. Painter; Satoko Uno; Andrew P. Morris; Stuart MacGregor; Scott D. Gordon; Anjali Henders; Nicholas G. Martin; et al.John R. AttiaElizabeth G. HollidayMark McEvoyRodney J. ScottStephen H. KennedySusan A. TreloarStacey A. MissmerSosuke AdachiKenichi TanakaYusuke NakamuraKrina T. ZondervanHitoshi ZembutsuGrant Montgomery Genome-wide association meta-analysis identifies new endometriosis risk loci. Nature Genetics 2012, 44, 1355-1359, 10.1038/ng.2445.
- Satoko Uno; Hitoshi Zembutsu; Akira Hirasawa; Atsushi Takahashi; Michiaki Kubo; Tomoko Akahane; Daisuke Aoki; Naoyuki Kamatani; Koichi Hirata; Yusuke Nakamura; et al. A genome-wide association study identifies genetic variants in the CDKN2BAS locus associated with endometriosis in Japanese. Nature Genetics 2010, 42, 707-710, 10.1038/ng.612.
- Sarah Holdsworth-Carson; Jenny Nga Ting Fung; Hien T.T. Luong; Yadav Sapkota; Lisa M. Bowdler; Leanne Wallace; Wan Tinn Teh; Joseph E. Powell; Jane E Girling; Martin Healey; et al.Grant MontgomeryP.A.W. Rogers Endometrial vezatin and its association with endometriosis risk. Human Reproduction 2016, 31, 999-1013, 10.1093/humrep/dew047.
- Polonca Küssel-Andermann; Aziz El-Amraoui; Saaid Safieddine; Sylvie Nouaille; Isabelle Perfettini; Marc Lecuit; Pascale Cossart; Uwe Wolfrum; Christine Petit; Vezatin, a novel transmembrane protein, bridges myosin VIIA to the cadherin–catenins complex. The EMBO Journal 2000, 19, 6020-6029, 10.1093/emboj/19.22.6020.
- Orest William Blaschuk; Tracey M. Rowlands; Plasma membrane components of adherens junctions (Review). Molecular Membrane Biology 2002, 19, 75-80, 10.1080/09687680210132467.
- Luca Pagliardini; Davide Gentilini; Ana Maria Sanchez; Massimo Candiani; Paola Viganó; Anna Maria Di Blasio; Replication and meta-analysis of previous genome-wide association studies confirm vezatin as the locus with the strongest evidence for association with endometriosis. Human Reproduction 2015, 30, 987-993, 10.1093/humrep/dev022.
- Juliana Meola; Júlio César Rosa E Silva; Daniel Dentillo; Wilson Araújo Da Silva; Luciana Caricati Veiga-Castelli; Luciano Ângelo De Souza Bernardes; Rui Alberto Ferriani; Claudia C. P. Paz; Silvana Giuliatti; Lucia Martelli; et al. Differentially expressed genes in eutopic and ectopic endometrium of women with endometriosis. Fertility and Sterility 2010, 93, 1750-1773, 10.1016/j.fertnstert.2008.12.058.
- Sun-Wei Guo; Nuclear Factor-κB (NF-κB): An Unsuspected Major Culprit in the Pathogenesis of Endometriosis That Is Still at Large?. Gynecologic and Obstetric Investigation 2006, 63, 71-97, 10.1159/000096047.
- Hien Tt. Luong; Jodie N. Painter; Yadav Sapkota; Dale R. Nyholt; Peter A. Rogers; Grant W. Montgomery; AB028. Identifying the functional role of VEZT gene for endometriosis risk. Annals of Translational Medicine 1970, 3, 1, 10.3978/j.issn.2305-5839.2015.AB028.
- Yadav Sapkota; Valgerdur Steinthorsdottir; Andrew P. Morris; Amelie Fassbender; Nilufer Rahmioglu; Immaculata De Vivo; Julie E. Buring; Futao Zhang; Todd Edwards; Sarah Jones; et al.Dorien ODaniëlle PeterseKathryn M. RexrodePaul M. RidkerAndrew J. SchorkStuart MacGregorNicholas G. MartinChristian M. BeckerSosuke AdachiKosuke YoshiharaTakayuki EnomotoAtsushi TakahashiYoichiro KamataniKoichi MatsudaMichiaki KuboGudmar ThorleifssonReynir T. GeirssonUnnur ThorsteinsdóttirLeanne WallaceThomas M. WergeJian YangDigna R. Velez EdwardsMette NyegaardSiew-Kee LowKrina T. ZondervanStacey A. MissmerThomas D'hoogheGrant MontgomeryDaniel I. ChasmanKári StefánssonJoyce Y. TungDale R. Nyholt Meta-analysis identifies five novel loci associated with endometriosis highlighting key genes involved in hormone metabolism. Nature Communications 2017, 8, 15539, 10.1038/ncomms15539.
- Katherine S. Ruth; Robin N. Beaumont; Jessica Tyrrell; Samuel E. Jones; Marcus A. Tuke; Hanieh Yaghootkar; Andrew R. Wood; Rachel M. Freathy; Michael Weedon; Timothy M. Frayling; et al.Anna Murray Genetic evidence that lower circulating FSH levels lengthen menstrual cycle, increase age at menopause and impact female reproductive health. Human Reproduction 2016, 31, 473-81, 10.1093/humrep/dev318.
- Michail Matalliotakis; Maria I. Zervou; Charoula Matalliotaki; Nilufer Rahmioglu; George Koumantakis; Ioannis Kalogiannidis; Ioannis Prapas; Krina T. Zondervan; D.A. Spandidos; Ioannis Matalliotakis; et al.G N Goulielmos The role of gene polymorphisms in endometriosis. Molecular Medicine Reports 2017, 16, 5881-5886, 10.3892/mmr.2017.7398.




