| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Garry Kerch | + 3932 word(s) | 3932 | 2021-02-13 04:31:03 | | | |
| 2 | Vicky Zhou | Meta information modification | 3932 | 2021-02-14 13:51:29 | | | | |
| 3 | Vicky Zhou | -3120 word(s) | 812 | 2022-04-13 10:27:22 | | |
Video Upload Options
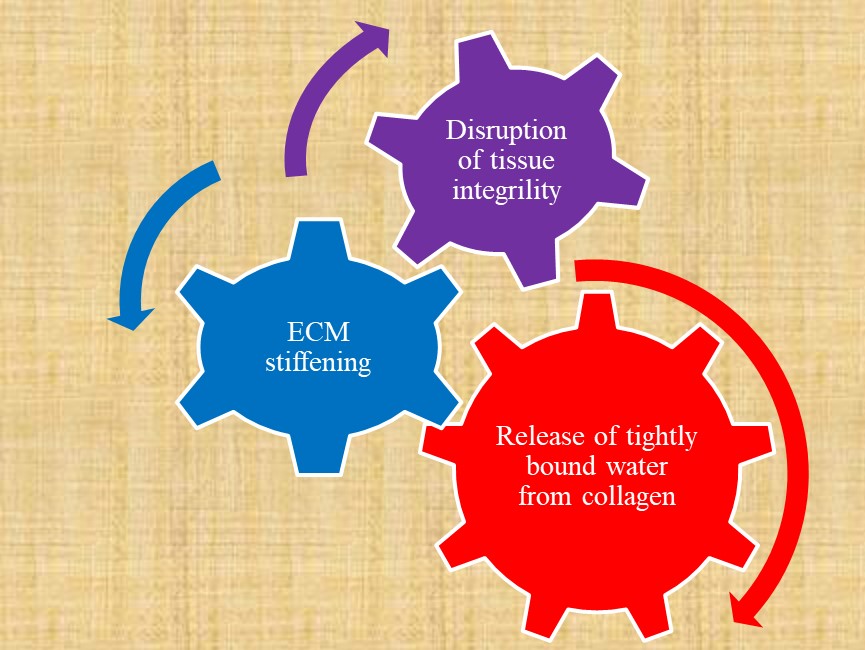
Tissue integrity depends on biological tissue stiffness. Tissue integrity can protect both against age-related diseases and against severity of COVID-19. The disruption of tight junctions and increase of tissue permeability with advancing age can be related with age-related diseases as well as with age-dependent COVID-19. Release of tightly bound water from collagen fibrils leads to the increase of extracellular matrix stiffness and to the associated with matrix stiffness increased tissue permeability. The link between arterial stiffness and oxidative stress has been reported and is expected to be studied in more detail in the future. Trehalose can be suggested for retardation of tightly bound water release and subsequent extracellular matrix crosslinking by advanced glycation end products. Increase in tissue permeability can be blocked by polyphenols that inhibit ICAM-1 expression and mitigate cytoskeleton reorganization. NF-κB activation as a result of increased stiffness and cytoskeleton reorganization can cause both cardiovascular pathologies and COVID-19. Increased cholesterol content in cell membrane leads to increased virus entry into cell and increase of cholesterol is linked with cardiovascular diseases. Statins and chitosan are known as cholesterol-lowering substances. Nrf2 inhibits NF-κB activation and NF-κB inhibits Nrf2 pathway.
References
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72314 Cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242.
- Kerch, G. Role of changes in state of bound water and tissue stiffness in development of age-related diseases. Polymers (Basel) 2020, 12, 1362.
- Bonanad, C.; García-Blas, S.; Tarazona-Santabalbina, F.; Sanchis, J.; Bertomeu-González, V.; Fácila, L.; Ariza, A.; Núñez, J.; Cordero, A. The Effect of Age on Mortality in Patients With COVID-19: A Meta-Analysis With 611,583 Subjects. J. Am. Med. Dir. Assoc. 2020, 21, 915–918.
- Baumann, K. Environment dictates behaviour. Nat. Rev. Mol. Cell Biol. 2010, 11, 679.
- Janmey, P.A.; Fletcher, D.A.; Reinhart-King, C.A. Stiffness sensing by cells. Physiol. Rev. 2020, 100, 695–724.
- Lacolley, P.; Regnault, V.; Avolio, A.P. Smooth muscle cell and arterial aging: Basic and clinical aspects. Cardiovasc. Res. 2018, 114, 513–528.
- Mitchell, J.B.; Dugas, J.P.; McFarlin, B.K.; Nelson, M.J. Effect of exercise, heat stress, and hydration on immune cell number and function. Med. Sci. Sports Exerc. 2002, 34, 1941–1950.
- Jacobs, P.J.; Oosthuizen, M.K.; Mitchell, C.; Blount, J.D.; Bennett, N.C. Heat and dehydration induced oxidative damage and antioxidant defenses following incubator heat stress and a simulated heat wave in wild caught four-striped field mice Rhabdomys dilectus. PLoS ONE 2020, 15, e0242279.
- Kerch, G.; Zicans, J.; Merijs Meri, R.; Stunda-Ramava, A.; Jakobsons, E. The use of thermal analysis in assessing the effect of bound water content and substrate rigidity on prevention of platelet adhesion. J. Therm. Anal. Calorim. 2015, 120, 533–539.