| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | ANTONELLA PATTI | + 3210 word(s) | 3210 | 2021-01-20 09:59:32 | | | |
| 2 | Catherine Yang | -506 word(s) | 2704 | 2021-02-09 02:32:02 | | |
Video Upload Options
The textile production is constantly increasing by offering products to fashion, style and marketing needs, or even to progressively competitive technical challenges. The perspective of innovation and development is in contrast with the highly polluting impact on the surroundings of this industrial activity.
1. Introduction
Since the Prehistoric Age, one of the human needs has been to cover and protect the body against the cold and/or bad weather. Many anthropological and archeological studies have demonstrated the practical and symbolic importance of fabrics in human history, especially in rituals, events, and ceremonies.[1]. A first finding of woven fibers date back to the Neolithic era in the Egyptian land: it was made of hemp, i.e. linen, of very fine quality in a complex pattern (“brocaded”) with fringes at the edges [2]. In Iran, evidence of the trade of the wool, and in Babylon garments made of wool. Around 3000 BC, spinning and weaving of linen and cotton were well developed in Egypt and India, respectively. In the Bronze Age (2000 BC) discoveries mainly of animal fibers, for burial were found in Northern Europe. In the Middle Age, Turkey became skilled in carpet manufacturing; Palermo (Sicily) became famous for the production of elaborate silk and gold fabrics, and Lyon (France) became the most important in Europe for the silk manufacturing. Before the industrial revolution, the textile industry expanded more as a trade in fine ornamental yarns and beautiful textiles, than in terms of technological progress. However, already at the end of 1500, in England, W. Lee of Woodborough invented the knitting machine; then, in the 1793 the invention of the cotton gin has followed, and continuing the flying shuttle for weaving by John Kay, up to the mechanized loom by Edmund Cartwright [3]. These technological advances have led to the expansion of the British textile industry at exponential speed. For example the cotton consumption increased from four in 1764 to 300 million pounds in 1833. In the early 1900s, the manager of the Chemical Department of du Pont understood that the progress would be accelerated by the entry of the university and research in the industrial world. Research activities developed in the field of high polymers have conducted to the synthesis of long molecules with similar properties compared to the natural cellulose, rubber, and silk. Hence the introduction of synthetic fibers as nylon 6, nylon 66, acrylic and polyester fibers in the textile market. [4]
From this moment, the textile production, based both on natural fibers and synthetic ones, has undergone considerable development linked to the population growth, and economic and commercial aspects of the fashion and style.[5]
Statistical studies by Garside M. (2019) [6] report that the production of chemical and textile fibers from 1975 to 2018 has increased approximately from 23.94 million metric tons to 105.6 million metric tons: this means an increment of more than four times in about 40 years. As the demand for fabrics has grown, aspects linked to the environmental impact of fiber production, and the following disposal operations, have also become increasingly present. Numerous and recent studies show the danger of the textile industry for the freshwater and atmosphere micro-system, due to the usage of significant amounts of industrial harmful and toxic chemicals during the manufacturing process and the release of pollutants during the lifecycle of a textile product [7]. Other forms of contamination arise from high energy consumption, heavy transportation, and excessive packing material. Finally, given the huge waste generation, the problem of disposing of large solid volumes has arisen. Twenty percent of earth pollution has been attributed to the textile industry, which for these reasons has been considered among the most harmful and detrimental manufacturing processes for our ecosystem [8].
In the light of these complex ecological issues, over the years until 2020, European directives have promoted and encouraged the recovery of textiles in a board plan for new circular economic action for a cleaner and more competitive country [9].
2. Textile Fibrous Structures and Their Involvement in the Common and Technical Uses
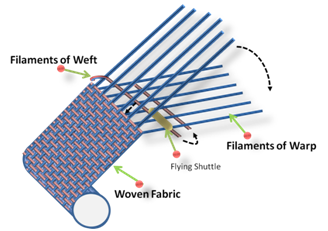
The term textile is derived from the Latin “textilis” and the French “texere”, meaning “to weave”, and it originally referred only to woven fabrics; then, it was extended to fabrics produced by other methods, such as threads, cords, ropes, braids, lace, embroidery, nets, and fabrics made by weaving that are woven, knitted, braided, or non-woven [10].Technically, the basic principle of weaving involves two series of filaments: the first one, called the warp, is pre-established and fixed, while the second, namely the weft, is in turn inserted within the first structure. The weaving process (Figure 1) consists of three steps: (i) shedding, during which the warp sheet is divided into two patterns, one spaced, separated, and raised from the other so as to create an opening between the warp series; (ii) filling insertion, which provides for the insertion of a yarn inside the created opening; and (iii) beat-up, in which process the filling yarn is pushed inside the weave of the fabric [11].Through this method, 2D planar weaving structures, i.e., with in-plane oriented fibers, can be produced.
Figure 1. Basic scheme of the production process for woven textiles: the filaments of the warp are opened, while the ones constituting the weft are driven orthogonally in the two opposite directions.
Depending on the interlocking types, other conventional techniques exist, by which another kind of weaving, such as knitting, braiding, and non-woven fabrics, can be obtained. In detail, four main categories of textile constructions can be recognized (as seen in Figure 2): (i) woven, made by weaving two sets of filaments (warp and weft) by the orthogonal interlacement, continuously arranged at 0° and 90°; (ii) knitted fabrics, created by intermeshing loops and segments of fibers with different orientation; (iii) braided fabrics, in which the fibers are placed in the bias direction; and (iv) non-woven, in which the fibers are randomly oriented in the structure in a discontinuous way. In the nineteenth century, three dimensional (3D) structures from a set of fibers in the multiaxial orientation were developed, essentially made from fibers oriented in the three dimensions. These structures were born to meet the requirements imposed by aerospace’s industry of materials able to resist under multi-axial loads and severe thermal conditions; indeed, compared to 2D, the 3D textiles possess a higher mechanical resistance in x, y, and z directions [12].
Figure 2. 2D planar and 3D fibrous structures: (a) woven, (b) braid, (c) knit, (d) non woven, (e) tridimensional.
The applications of each of the described types of fabric are the most varied, classified into products for clothing, household, and technical textiles [13]. In the case of woven fabrics, in the book of the Kim Ghandi [14], different technical applications for filtration in industry activities have been reported, as in the mechanical shaker cleaning method, together with air filtration in automobile engineers, or sound control and speak quality in the electronic devices (acoustic, loud-speakers, and microphones), liquid filtration techniques for chemicals in paper and pulp industry, and precipitation of metal oxides from solvent slurries. Typical structural components are made by 2D or 3D, woven, braided, knitted, or non-woven fabrics, such as knot elements (beams, shells, exhaust, seats, and chassis) or geotextiles (asphalt overlay, soil stabilization, drainage, sedimentation, and erosion control), to fulfill general requirements of easy manufacturability, low cost, sound and energy absorption, and corrosion resistance. In addition, all of them are utilized for ballistic applications as protective clothing or products, endowed with extremely high strength and modulus, ability in absorbing energy and sound, and in combating against the chemical and biological warfare agents. In aerospace applications, woven and braided fabrics are applied in soft space suits for astronauts, space shuttle components, and aircraft seat cushions, as well as in automobile interior for airbags or car seat parts [15].
3. Environmental Impact of the Textile Production
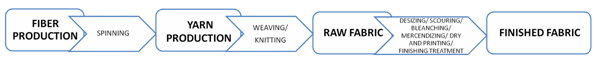
The different steps starting from the fibers’ production that lead to the fabric realization can be described in the following operations: fiber production, yarn production, fabric production, pre-treatment, dyeing and printing, and finishing treatment. The common flow chart of the textile production starting from the fiber up to the finished fabric is reported in Figure 3.
Figure 3. Typical progressive phases of the textile production.
As just predicted in the introduction, the environmental impact of these operations concern different aspects all connected to eachother that take into account the usage of harmful chemicals, the water and energy consumption, the air emission, the transportation, and the packaging.
The raw materials involved in the textile manufacturing are divided into two main categories: (i) natural fibers, deriving from vegetables and plants (cotton, flax, sisal, hemp, ramie, jute, banana, pineapple, coir, and oil palm), animals (wool, angora, cashmere, and silk), and minerals (asbestos); and (ii) man-made fibers, synthetic or regenerated, coming from petroleum-based resources, such as polyester, nylon, spandex, acrylic, and polypropylene. These primary materials, depending on their nature, require being treated with different chemical agents [16].
For instance, in the fiber production, pesticides, insecticides, and fertilizers are applied in the case of cellulose or natural fibers, to allow and facilitate the growth and development of plants, whereas for protein fibers, parasites are used for the animal, and after the fibers shredding, the utilization of chemicals for the cleaning operations [5]. In the case of the synthetic fibers, the preparation of the agents, polymerization, polymer recovery and extrusion, and spinning for arriving to the filaments required the use of monomers and catalyst that generate a series of byproducts [7].
Then, during the yarn production, particularly in the spinning, oil is required for reducing the friction among the parts, and again, in the fabric production, the sizing species together with the lubricants should be used for avoiding the breakage of the fibers during the process. Once obtained, and before the dyeing step, the fabric should be suitably prepared with a multiple pre-treatments through the following processes: (i) de-sizing, consisted in the removal of the starch (sizing chemical) and the improvement of the absorbent capability, since the starch hampers the diffusion of the dye molecule into the yarn/fabric; through enzymatic, or dilute mineral acid hydrolysis, or oxidation, the starch is transformed in water soluble constituents; (ii) scouring, during which wax, fats, pectin, and lubrication oil are removed by using aqueous sodium hydroxide together with the surfactant; (iii) bleaching, to get white fibers by decolorizing their natural creamy appearance, during which oxidant agents, such as sodium hypochlorite, sodium chlorite, and hydrogen peroxide, are always applied; and (iv) mercerizing, realized by immersion in a high concentration of sodium hydroxide solution to improve fabric features, in terms of tensile strength, hygroscopicity, and dye absorbency, brightness, and dimensional stability, occurred through the swelling, the untwisting, and the fiber re-orientation.
In fact, during the mercerizing, the wetted fibers go through a longitudinal shrinkage that can be avoided by elongation and holding the fibers under an applied uniaxial stress. The excess of caustic soda is removed by water washing, (v) dying, and printing to impart the color to the fabric or yarns. All the chromophore agents, such as azo (-N=N-), carbonyl (-C=O), nitro (-N=O), and also amine, carboxyl, sulfonate, and hydroxyl groups are considered water contaminants, because they confer unacceptable color to the wash water [5][17].
Several other chemicals are involved in the dyeing and printing depending on the chemical nature of the fibers: reactive dyes, direct dyes, naphthol dyes, and indigo dyes in the case of cellulose fibers, acid dyes and Lanaset dyes in the case of protein fibers, and finally dispersed dyes, basic dyes, and direct dyes for the synthetic fibers. In order to promote the link between fibers and pigments, binder and polymeric resins should be applied, while the pigments in excess should be removed by washing with detergents, such as alkyl aryl-sulfonates, sulfated alkyl phenol polyglycol, alkylphenol ethoxylates, sodium palmitate, and sodium stearate [5].
Finally, specific finishing treatments should be designed for imparting particular features to the latest products: in the case of water and oil repellency, paraffin (waxes), silicones, fluorocarbon, and stearic-acid melamine may be suggested [18]; for the antibacterial activity metallic salts (Ag+ and Cu2+), triclorosan (2,4,4-hydrophenyl trichloro (II) ether), quaternary ammonium compounds, chitosan, and cyclodextrin are the most common antiseptic products [19]; again, for the flame retardancy halogen based formulations, phosphor, or nitrogen based coating systems, silicone based species are usually implied [20].
In general, all the listed components implied in the fabric production can have different life paths: they can remain attached to the fabric or can evaporate, end up in the wastewater, and be poured into the environment. Although the dilution in large volume of air and water, many of these species may not degrade rapidly, and therefore can be transported at very high distances and accumulate in sediments or organisms (“persistence”); additionally, they can enter the body absorbed through food and skin and accumulate in it (“bioaccumulation”).
Many of these species can be considered toxic (“toxicity”) due to the potential or established carcinogenic and/or mutagenic effects, the risk of physical malformation for the fetuses, and the unleashing of allergies of various kinds [8].
A summary scheme of pollutants involved in each operation of the textile industry is reported in the work of Holkar et al. [17]. For example, in the desizing operation, enzymes and waxes are commonly adopted, in scouring fat, soap, surfactants, pectin, and oils are usually applied, for the bleaching hydrogen peroxide, sodium silicate, organic stabilizer, and alkaline pH conditions are necessary; again, metals, salts, alkaline/acid species color, and metals are used; the printing required formaldehyde, urea, solvent, and metals; finally, solvents, waxes, resins, and softeners are needed.
By increasing the complexity of the processes, the machinery technologies, and the automation of the implants, gradually increments of energy consumption correlated to the textile manufacturing have been recorded. In the work of Dhayaneswaran and Ashokkumar [21]: it is highlighted that the energy cost is around 15–20% over the production cost, and it stands next to raw material cost, and the power distribution in textile mills including productive and non-productive machines is shown. On the basis of this research, it appears that the highest power is absorbed by ring frame, about 588 KWh, following by speed frame, carding, and compressor.
It can be easily understood that large volumes of water are required for all the described operations that for this reason are referred to the wet process category. In the recent review of Holkar et al. on the textile wastewater treatments, it has been reported that the employment of water for an average sized textiles is about 200 L per kg of fabric processed per day, and finishing treatment generates around 17 to 20 percent of industrial waste water [17].
The implants for the textile production occupy large areas and need huge available spaces. In general, for this reason, the factories are located in low-cost countries, so as to require the transport of goods from the producing countries to the consuming countries. In this way, not only the fuel of non-renewable sources is consumed, but photochemical oxidizing products are also emitted into the air by generating smog. The final products to be transported and sold are protected, preserved, and stored in packaging, generally consisting of non-biodegradable plastic materials, derived from non-renewable sources, paper, aluminum, and cotton, which ends up representing yet another outline of waste and refusal of the textile industry. In the last period, for promoting eco-friendly activities, companies implement the use of recycled or bio-degradable materials for the textile packaging [8][22].
The European Environmental Agency has estimated that clothing, footwear, and household production represented the fifth highest greenhouse production with an amount of released CO equal to 15–35 ton per ton of produced textiles [23].
Depending on the material nature, the environmental impact of the fiber production is correlated to a complexity of the foresaid multiple factors. Shen et al. [24] performed a comparative life cycle assessment (LCA) of three types of man-made cellulose fibers (Viscose, Modal, and Tencel), against the conventional textile fibers based on cotton, polyester (PET), and polypropylene (PP). They concluded that the Viscose and Modal could be considered advantageous from an environmental point of view, given the low fossil energy requirements in the pulp and fiber production, while the Tencel resulted in an overall reduction of the energy consumption, chemical use, CO and SO emissions, and water consumptions. In the case of cotton, even if it can be considered not an energy-intensive product, the major environmental issues were regarded as the highest fresh water ecotoxicity and terrestrial ecotoxicity, mainly due to pesticide use.
References
- Nosch, M.-L.; C_cile, M.; Harlow, M. Prehistoric, Ancient Near Eastern & Aegean Textiles and Dress; Oxbow Books, 2014;
- Barber, E.J.W. Prehistoric Textiles: The Development of Cloth in the Neolithic and Bronze; Princeton University Press, 1991;
- Fisher, C.H. History of Natural Fibers. J. Macromol. Sci. Part A - Chem. 1981, 15, 1345–1375, doi:10.1080/00222338108056788.
- Heckert, W.W. Synthetic fibers. J. Chem. Educ. 1953, 30, 166–178.
- Pensupa, N.; Leu, S.-Y.; Hu, Y.; Du, C.; Liu, H.; Jing, H.; Wang, H.; Carol Sze Ki Lin Recent Trends in Sustainable Textile Waste Recycling Methods: Current Situation and Future Prospects. In; Springer, Cham, 2017; pp. 189–228.
- Garside, M. Chemical and textile fibers production worldwide 1975-2018 | Statista Available online: https://www.statista.com/statistics/263154/worldwide-production-volume-of-textile-fibers-since-1975/#statisticContainer (accessed on May 15, 2020).
- Stone, C.; Windsor, F.M.; Munday, M.; Durance, I. Natural or synthetic – how global trends in textile usage threaten freshwater environments. Sci. Total Environ. 2020, 718, 134689, doi:10.1016/j.scitotenv.2019.134689.
- Roy Choudhury, A.K. Environmental Impacts of the Textile Industry and Its Assessment Through Life Cycle Assessment. In Roadmap to Sustainable Textiles and Clothing. Textile Science and Clothing; Muthu, S., Ed.; Singapore, 2014; pp. 1–39.
- EUR-Lex - 52020DC0098 - EN - EUR-Lex Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1583933814386&uri=COM:2020:98:FIN (accessed on Jun 4, 2020).
- Nawab, Y.; Hamdani, S.T.A.; Shaker, K. Structural Textile Design: Interlacing and Interlooping - Google Libri; CRC Press, Ed.; 2017;
- Mohamed, M.H.; Bogdanovich, A.E. COMPARATIVE ANALYSIS OF DIFFERENT 3D WEAVING PROCESSES, MACHINES AND PRODUCTS. In Proceedings of the Proceeding of the 16th international conference on composites materials ; Kyoto, Japan., 2007.
- Fangueiro, R.; Soutinho, F. Textile structures. In Fibrous and Composite Materials for Civil Engineering Applications; Elsevier, 2011; pp. 62–91.
- Buhu, A.; Buhu, L. Woven Fabrics for Technical and Industrial Products. In Textiles for Advanced Applications; InTech, 2017.
- Gandhi, K. Woven Textiles: Principles, Technologies and Applications; 2nd ed.; Woodhead Publishing, 2019;
- Bilisik, K.; Karaduman, N.S.; Bilisik, N.E. 3D Fabrics for Technical Textile Applications. In Non-woven Fabrics; InTech, 2016.
- Jabbar, M.; Shaker, K. Textile raw materials in: Physical Sciences Reviews Volume 1 Issue 7 (2016). Phys. Sci. Rev. 2016, 1, 20160022–20160033.
- Holkar, C.R.; Jadhav, A.J.; Pinjari, D. V.; Mahamuni, N.M.; Pandit, A.B. A critical review on textile wastewater treatments: Possible approaches. J. Environ. Manage. 2016, 182, 351–366.
- Schindler, W.D. (Wolfgang D.; Hauser, P.J. (Peter J.; Textile Institute (Manchester, E. Chemical finishing of textiles; Elsevier, Ed.; CRC: Boca Raton, Fl, 2004; ISBN 1845690370.
- Shahidi, S.; Wiener, J. Antibacterial Agents in Textile Industry. In Antimicrobial Agents; InTech, 2012.
- Neisius, M.; Stelzig, T.; Liang, S.; Gaan, S. Flame retardant finishes for textiles. In Functional Finishes for Textiles: Improving Comfort, Performance and Protection; Elsevier Inc., 2015; pp. 429–461 ISBN 9780857098450.
- Dhayaneswaran, Y.; Ashokkumar, L. A Study on Energy Conservation in Textile Industry. J. Inst. Eng. Ser. B 2013, 94, 53–60, doi:10.1007/s40031-013-0040-5.
- Choudhury, A.K.R. Sustainable chemical technologies for textile production. In Sustainable Fibres and Textiles; Elsevier Inc., 2017; pp. 267–322 ISBN 9780081020425.
- Textile waste — European Environment Agency Available online: https://www.eea.europa.eu/media/infographics/textile-waste/view (accessed on Jun 9, 2020).
- Shen, L.; Worrell, E.; Patel, M.K. Environmental impact assessment of man-made cellulose fibres. Resour. Conserv. Recycl. 2010, 55, 260–274, doi:10.1016/j.resconrec.2010.10.001.