
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Russell Pachynski | + 4105 word(s) | 4105 | 2021-01-25 09:25:16 | | | |
| 2 | Vivi Li | Meta information modification | 4105 | 2021-02-01 04:29:39 | | |
Video Upload Options
Although most prostate cancers are localized, and the majority are curable, recurrences occur in approximately 35% of men. Among patients with prostate-specific antigen (PSA) recurrence and PSA doubling time (PSADT) less than 15 months after radical prostatectomy, prostate cancer accounted for approximately 90% of the deaths by 15 years after recurrence. An immunosuppressive tumor microenvironment (TME) and impaired cellular immunity are likely largely responsible for the limited utility of checkpoint inhibitors (CPIs) in advanced prostate cancer compared with other tumor types. Thus, for immunologically “cold” malignancies such as prostate cancer, clinical trial development has pivoted towards novel approaches to enhance immune responses. Numerous clinical trials are currently evaluating combination immunomodulatory strategies incorporating vaccine-based therapies, checkpoint inhibitors, and chimeric antigen receptor (CAR) T cells. Other trials evaluate the efficacy and safety of these immunomodulatory agents’ combinations with standard approaches such as androgen deprivation therapy (ADT), taxane-based chemotherapy, radiotherapy, and targeted therapies such as tyrosine kinase inhibitors (TKI) and poly ADP ribose polymerase (PARP) inhibitors.
1. Introduction
Per 2019 SEER estimates, prostate cancer comprises approximately 10% of all new cancer diagnoses, with over 98% of the patients alive at 5 years [1]. Recurrence after localized treatment occurs in about 1/3rd of the men, and these patients with recurrent disease eventually develop malignant cells resistant to androgen ablation alone [2][3]. This statistic points out that, while patients with prostate cancer have a low mortality, those with advanced prostate cancer eventually progress to the castrate-resistant disease [4]. Moreover, patients with a shorter prostate-specific antigen doubling time (PSADT) experience increased prostate cancer-specific and all-cause mortality [5].
While androgen deprivation therapy (ADT) is not curative, it does lead to an overall survival (OS) benefit of approximately 30 months in patients with metastatic disease [6]. Androgen deprivation can be achieved by surgical orchiectomy or medical castration using gonadotropin-releasing hormone receptor (GnRH-R) agonists or antagonists [7][8]. Currently, for patients with metastatic hormone-sensitive prostate cancer (mHSPC), additional first-line agents are frequently used in conjunction with an ADT backbone. These include three oral androgen receptor (AR)-targeted drugs—abiraterone acetate, apalutamide, and enzalutamide, as well as docetaxel chemotherapy [9][10][11][12][13][14][15][16].
The addition of these agents for patients with mHSPC has improved patient outcomes [17]. Based on the STAMPEDE clinical trial results, the 3-year failure-free survival (FFS)—defined as radiologic, clinical, or PSA progression or death from prostate cancer, was 75% in patients with mHSPC, treated with a combination of abiraterone and ADT [10]. The CHAARTED clinical trial showed that for patients with mHSPC treated with a combination of ADT and docetaxel, the median time to castrate resistance was 20.2 months [18]. In the phase III ARCHES clinical trial, at a median follow-up of approximately fourteen months, over 70% of the patients had developed castrate resistance while on enzalutamide [15]. Similar outcomes have been observed with apalutamide [14].
2. Vaccines
Bacillus Calmette-Guérin (BCG) vaccine was the first FDA-approved immunotherapy for the treatment of any solid tumor (bladder cancer) in 1990 [19][20]. While the FDA-approval of Sipuleucel-T followed it for mCRPC in 2010, unfortunately, since then, there have not been additional vaccine approvals for prostate cancer [21]. As of July 2020, there are thirteen vaccine clinical trials actively recruiting patients with prostate cancer. Of these, three are being evaluated in patients with mCRPC in combination with CPIs. While vaccine-based therapies have several advantages, one possible drawback is that an effective immune response to a specific TAA might be variable, limited in part by human leukocyte antigen (HLA) expression and haplotype, which affect presentation of the immunogenic epitope(s) [22][23]. High-affinity peptide-MHC (major histocompatibility complex) interactions and increased duration of peptide-MHC interactions may lead to more effective vaccine-induced immunogenicity [24][25]. Vaccine-based therapies can be broadly categorized into four different types of approaches as described below and schematically represented in Figure 1.
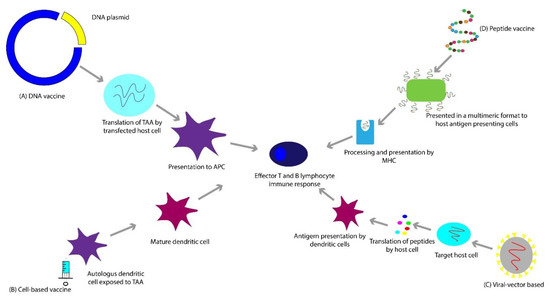
Figure 1. (A) DNA vaccines employ a plasmid, which gets translated to a tumor-associated antigen. (B) Cell-based vaccines utilize whole cells as a source of antigen. (C) Viral-vector based vaccines are based on the process of transfecting the host cell with a viral vector, that contains a gene which encodes a tumor-associated antigen. (D) Peptide-based vaccines consist of subunits of an epitope of a tumor-associated antigen, sometimes presented in a multimeric format (e.g., virus-like particles (VLPs)) to elicit an effective immune response.
2.1. Deoxyribonucleic Acid (DNA)-Based Vaccines
DNA-based vaccines are commonly plasmids taken up by host cells, resulting in host-synthesized TAAs, which elicit an immune response (Figure 1A) [26][27]. As an example, pTVG-HP and pTVG-AR are plasmid DNA vaccines encoding the human PAP and AR antigens, respectively, and are currently being evaluated in clinical trials in mCRPC [28][29][30]. In addition to shared TAAs such as AR and PAP, the DNA vaccine platform can generate personalized cancer vaccines for prostate cancer patients [31], such as the ongoing phase I clinical trial that utilizes a combination of a neoantigen DNA vaccination, nivolumab, ipilimumab, and PROSTVAC for patients with mHSPC (NCT03532217), which takes advantage of both shared and personalized antigen approaches. However, whether or not a personalized approach represents an improvement over the utilization of shared tumor antigens remains to be seen.
2.2. Peptide-Based Vaccines
Peptide-based vaccines are built of subunits containing the specific epitope of an antigen (Figure 1D) [32]. UV1 is a synthetic long peptide vaccine containing a fragment of human telomerase reverse transcriptase (hTERT) administered in combination with granulocyte-macrophage colony-stimulating factor (GM-CSF) in a phase I/II clinical trial for patients with mHSPC (NCT01784913). hTERT plays a pivotal role in cancer development. It is responsible for the immortality and “stemness” of tumor cells and is often overexpressed in cancer cells [33]. Another rapidly evolving approach is developing personalized peptide vaccines that involve identifying an individual patient’s peptide candidates for their ability to induce an immune response in vitro and subsequent administration to the patient [34]. These have the potential to induce clinically meaningful, potent anti-tumor responses.
2.3. Viral Vector-Based Vaccines
This paradigm involves transferring a gene encoding TAA(s) into patients using a vector, in this case, a virus, resulting in stimulation of host immune response against the antigen (Figure 1C) [35][36]. PROSTVAC is a therapeutic vaccine that encodes PSA to generate a T-cell response. It utilizes two different live poxviral-based vectors-PROSTVAC-V, a recombinant vaccinia virus (rilimogene galvacirepvec), and PROSTVAC-F, a recombinant fowlpox virus (rilimogene glafolivec). In addition to induced modified human PSA, they contain three costimulatory domains for T cells, called TRICOM. These are B7.1, leukocyte function-associated antigen-3, and intercellular adhesion molecule-1 [37]. The initial phase I and II trials incorporated the use of granulocyte-macrophage colony-stimulating factor (GM-CSF), which is hypothesized to potentiate T-cell responses [38][39]. The phase III PROSPECT trial failed to show a significant difference in OS [40][41]. It was suggested that the FDA approvals of newer agents such as cabazitaxel, sipuleucel-T, abiraterone acetate, enzalutamide, and radium-223 during the trial period and follow up increased OS in the placebo group, and that the initial expected OS at the time of protocol design was a flawed assumption [42][43][44][45][46][47][48].
2.4. Cell-Based Vaccines
For this vaccine approach, whole cells, autologous or allogeneic, are used as antigen sources (Figure 1B) [49]. The polyvalent source of antigen can theoretically bypass antigen escape [50][51]. These are usually utilized in combination with GM-CSF to induce the growth and differentiation of dendritic cells involved in antigen presentation [50].
One whole tumor cell-based vaccine approach is GVAX-PCa. It consists of two allogeneic prostate cancer cell lines, LNCaP and PC3, modified to secrete GM-CSF. It has been tested in two phase III trials, VITAL-1 and VITAL-2, and failed to show clinical benefit [51][52][53]. There are attempts to improve the efficacy of GVAX-PCa by combining it with CPIs [54].
As mentioned, Sipuleucel-T, an autologous cell vaccine prepared from peripheral blood mononuclear cells (PBMCs), is currently approved for the treatment of patients with mCRPC [55]. The patients undergo leukapheresis, and the cells are exposed in vitro to PA2024—a fusion protein consisting of human GM-CSF and human PAP [55]. Notably, the patients with visceral metastases and symptomatic cancer pain were excluded from the clinical trials [42][56][57]. There were two interesting observations from these trials. Firstly, progression-free survival (PFS) was not prolonged [42]. Secondly, median survival improved significantly in patients with a lower PSA than patients with a higher PSA [58]. The phase III IMPACT trial demonstrated that Sipuleucel-T improved OS by 4.1 months in patients with mCRPC compared to placebo [42].
3. Potential Immune Evasion Mechanisms
The TME comprises immune cells, non-immune cells such as fibroblasts, and endothelial cells embedded in an extracellular matrix (ECM) [59]. The complex interplay between protumorigenic and anti-tumor immune-modulating factors leads to the varying patterns of tumor progression, and our therapeutic decisions influence these opposing factors. The immune evasion mechanism can be categorized into factors that increase or dampen the immune response and are briefly outlined below and depicted schematically in Figure 2.
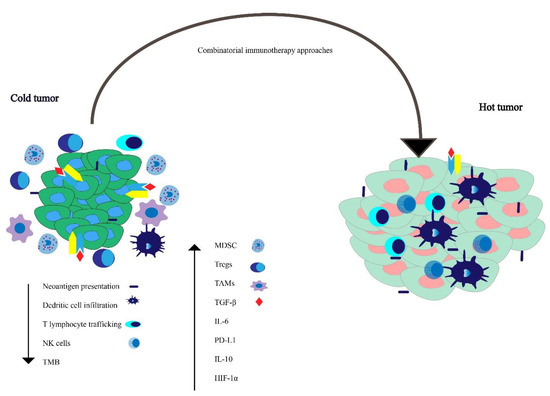
Figure 2. Working model for understanding factors influencing tumor microenvironment in tumors lacking immunological anti-tumor activity—the so-called “cold tumors”—versus tumors with immunological activity against the tumor—“hot tumors.” Combinatorial immunotherapeutic approaches can possibly convert an immunologically cold tumor to a hot tumor.
3.1. Increase Immune Suppressive Factors
There are a variety of immune-suppressive factors in the TME, such as PD-L1, interleukin 10 (IL-10), hypoxia-inducible factor 1-alpha (HIF-1α), and transforming growth factor-β (TGF-β) [60][61][62][63]. There is evidence suggesting an increase in myeloid-derived cells, tumor-associated macrophages (TAMs), and myeloid-derived suppressor cells (MDSCs) in the TME of prostate cancer is associated with tumor progression [64]. The majority of the studies show that pathways such as the loss of phosphatase and tensin homolog (PTEN) increase MDSC infiltration [65]. Chronic inflammation induced by the tumor, inflammatory cytokines such as soluble tumor necrosis factor (sTNF), interleukin 1 beta (IL-1β), TGF-β, and IL-10 cause myeloid cells to differentiate into MDSCs, which have been implicated in worsened prognosis, and resistance to CPI immunotherapy [66][67][68].
TAMs express reduced major histocompatibility complex (MHC) expression and lead to an increase in PD-L1 expression [69]. Increased regulatory T cells (Tregs) in prostate cancer specimens are associated with advanced stage and worsened prognosis [70]. There is an increase in the level of TGF-β in patients with prostate cancer [71], which leads to an increase in tumor growth [72]. Mutations in forkhead box protein A1 (FOXA1), a transcription factor essential for epithelial lineage differentiation, induces TGF-β signaling in CRPC [73].
3.2. Reduced Immune Stimulatory Factors
There is evidence that peripheral natural killer (NK) cell pools may be decreased in patients with mCRPC. One potential mechanism for this decrease in peripheral NK cell pool is an increased NK cell group 2 member D (NKG2D) serum receptor levels derived from the tumor [74]. Membrane-bound and not tumor shed NKG2D ligand is thought to promote CD8 T cell and NK cell response [75]. Circulating NK cell function is reduced in patients with prostate cancer, and this difference is more profound in patients with metastatic disease [64][74]. Moreover, ADT is associated with an increase in CD8 T cells [64]. The incidence of the T-cell receptor zeta (TCR ζ) chain has been correlated with adequate effector cell function, and flow cytometric analysis has revealed a reduction in ζ chain expression in peripheral blood lymphocytes (PBL) of patients with prostate cancer [76].
Tumors with a low TMB, such as prostate cancer, often have reduced expression of neoantigens, which can be recognized by the immune system [77]. Tumors with higher T cell trafficking have been shown to have a higher therapeutic response to immune therapy [78]. Malignant transformation is often associated with changes in HLA expression [79][80][81], and thus these defects in HLA expression and antigen presentation machinery can lead to decreased tumor cell recognition by cognate T cells. This can result in immune evasion by cancer cells and may have therapeutic implications with respect to response to CPIs [82]. This also provides additional rationale for the development of HLA-independent immuno-cell therapies [83].
The impact of therapy on immune responses is complex. ADT can lead to enhanced lymphopoiesis and mitigate immune tolerance to prostate cancer antigens [84]. Androgen receptor (AR) antagonists have been shown to inhibit T cell responses, possibly related to γ-Aminobutyric acid type A (GABA-A) off-target effects [85]. It is postulated that initiating immunotherapy before AR antagonist therapy could improve tumor control by upregulation of the immune response [86]. As our understanding of tumor biology and the effect of treatments evolve, it could influence the choice and sequence of therapies in the future.
4. Ongoing Clinical Trials on Combination with CPIs
Below we discuss some of the ongoing attempts to improve the response to existing immunotherapeutic agents and novel immunotherapeutic agents currently in clinical trials (Figure 3A and Figure 4B). A breakdown of the currently ongoing combination treatments of CPIs with other agents is schematically depicted in Figure 3B,C.
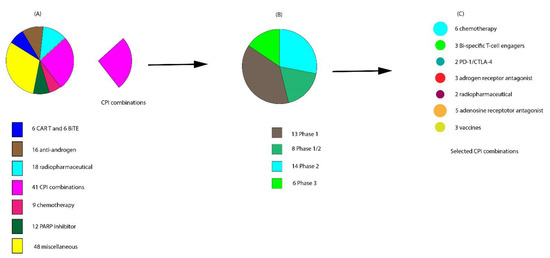
Figure 3. Snapshot of ongoing interventional clinical trials actively recruiting patients with metastatic castration-resistant prostate cancer (mCRPC) in July 2020. (A) Breakdown of the 150 clinical trials ongoing. (B) Of the 41 checkpoint inhibitor (CPI) trials, 13 are in phase I, 8 in phase I/II, 14 in phase II, and 6 in phase III. (C) The agents currently being used for CPI combinatorial therapy.
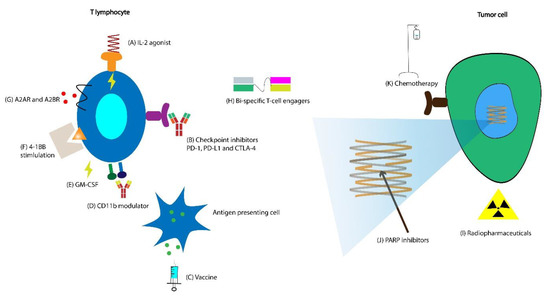
Figure 4. (A) Bempegaldesleukin is an interleukin (IL)-2 agonist currently being tested in combination with avelumab. (B) Multiple new checkpoint inhibitors (CPIs) are in clinical trials in combination with other agents. (C) Currently, there are DNA-, viral vector-, and mRNA-based vaccines in combination with checkpoint inhibitors in clinical trials in mCRPC. (D) GB1275 is a CD11b agonist in clinical trials in combination with CPI. (E) Granulocyte-macrophage colony-stimulating factor (GM-CSF) is used in combination with vaccines to augment immunological response (NCT04090528). (F) 4-1BB stimulation has been employed extensively in chimeric antigen receptor (CAR) T cell therapies, and utomilumab, a monoclonal agonist antibody that binds to the 4-1BB is currently in clinical trials for patients with mCRPC. (G) Currently, there are four clinical trials combining adenosine receptor antagonists with checkpoint inhibitors in patients with mCRPC. (H) After their success in acute lymphoblastic leukemia (ALL) and diffuse large B-cell leukemia (DLBCL), cellular therapies such as CAR T cell and bi-specific T-cell engagers are being studied extensively in clinical trials in numerous solid malignancies, including prostate cancer. (I) There are two clinical trials combining checkpoint inhibitor therapy with radiopharmaceutical Lu 177-PSMA-617, which is a β emitting isotope of lutetium attached to prostate-specific membrane antigen (PSMA). (J) While poly ADP ribose polymerase (PARP) inhibitors have already demonstrated efficacy in patients with homologous recombination deficiency (HRD), clinical trials are currently ongoing in combination with agents such as radiopharmaceuticals, CPIs, and chemotherapy evaluating whether they are effective in patients without HRD. (K) Chemotherapeutic agents have potential synergy with checkpoint inhibitors, and clinical trials to test this hypothesis in patients with mCRPC are currently ongoing.
4.1. Vaccines with CPIs
As of July 2020, three ongoing trials are actively recruiting patients with mCRPC, which combine vaccines with CPIs (Figure 4C). One potential limitation of using self-antigen TAAs as therapeutic vaccine targets is the issue of central T cell tolerance, which must be overcome in order to mount an effective anti-tumor response [87]. CPIs, when combined with vaccines encoding specific TAAs, have the potential to expand antigen-specific T cells [88]. Tumor antigens used in therapeutic vaccines are processed and presented in the context of MHC, and thus predicting these peptide: MHC binding interactions using advanced algorithms to improve T cell responses is of intense clinical interest [89][90]. Thus, there is a sound scientific rationale for combining vaccines with CPIs to potentiate the vaccine’s immune response [91][92][93].
One of the vaccines, ChAdOx1-MVA 5T4, is a combination of two replication-deficient viruses, chimpanzee adenovirus and modified vaccinia ankara, targeting an oncofetal self-antigen 5T4 (NCT03815942) [94]. The 5T4 protein is expressed in numerous malignancies but seldom in normal tissue [95]. It is associated with cells’ movement through the epithelial-mesenchymal transition [96], modulation of chemokine, and Wnt signaling [97][98]. A phase I study revealed 5T4-specific T cell responses [99].
Another vaccine that is currently recruiting patients in a phase II trial is the combination of two DNA vaccines, pTVG-HP and pTVG-AR, which target PAP and the androgen receptor ligand-binding domain (AR LBD), respectively, in combination with Pembrolizumab (NCT04090528) (Figure 4E) [100][101]. pTVG-HP monotherapy, in a phase II trial, revealed a possible effect on micrometastatic bone disease [30]. In a phase I study, patients who received pTVG-AR developed a T helper type 1 (Th1) biased immunity to the AR LBD [29].
The third vaccine in clinical trials is Wpro1 (NCT04382898). It is an mRNA-based vaccine that encodes for five antigens expressed in prostate cancer. Practical applications of mRNA-based vaccines are relatively recent, as they have been limited by instability and inefficient in vivo delivery [102]. Technological advances in delivery platforms have overcome these challenges [103], and several mRNA vaccines are currently in clinical trials. The mRNA is complexed with liposomes to form serum-stable RNA lipoplexes and induce activation of vaccine antigen-specific CD8 and CD4 T cells, in addition to the innate immune system.
The development of genomic approaches that allow the identification of tumor neoantigens has led to the development of clinical trials of personalized neoantigen vaccines [104]. Emerging clinical data reveals that targeting these mutated neoantigens can result in tumor responses [105][106]. This approach will likely lead to the development of clinical trials for patients with mCRPC in the future.
4.2. Combinations of CPIs and TKIs
Due to CPI monotherapy’s limited activity in prostate cancer [107][108], there has been enthusiasm for combining these agents with tyrosine kinase inhibitors (TKIs) [109]. TKIs compete with the adenosine triphosphate (ATP) binding site of the catalytic domain of oncogenic tyrosine kinases [110]. TKIs such as cabozantinib, sunitinib, and axitinib can act through several immunomodulatory mechanisms, such as anti-angiogenesis by inhibition of vascular endothelial growth factor (VEGF) [111]. VEGF overexpression is associated with the inhibition of differentiation of monocytes into dendritic cells [112]. Inhibition of VEGF leads to reduced immune inhibitory stimuli such as Tregs and MDSCs [113][114]. Cabozantinib has also been shown to target tyrosine-protein kinases-c-Met, Tyro3, Axl, and Mer, which have a role in immunosuppression [115][116].
The combination of TKI and CPI has already shown synergy and has been approved in NSCLC and renal cell carcinoma (RCC) [117][118]. Cabozantinib was evaluated in combination with PD-L1 inhibitor atezolizumab, in the phase 1b clinical trial COSMIC-021. One out of the 44 patients enrolled in the mCRPC cohort experienced a grade 5 treatment-related adverse event (TRAE) of dehydration. The ORR was 32%, while 48% of the patients had stable disease, leading to a disease control rate of 80% [119]. The combination of cabozantinib and atezolizumab for patients with mCRPC is also being evaluated in a phase III clinical trial (NCT04446117).
4.3. Combinations of CPIs and Chemotherapy
While chemotherapies are typically immune-suppressive, they can potentiate anti-tumor immunity by enhancing antigen presentation, expressing costimulatory molecules, and downregulating PD-L1 expression [120][121]. This potential synergy has led to the approval of the combination of CPI and chemotherapy in patients with NSCLC and is considered the front line in the metastatic setting [122]. There are currently six ongoing clinical trials that recruit patients with mCRPC, which evaluate a CPI’s efficacy in combination with chemotherapy (Figure 4K). KEYNOTE-365 and KEYNOTE-921 phase I and III trials, respectively, evaluate the combination of PD-1 inhibitor pembrolizumab with docetaxel (NCT02861573, NCT03834506). CheckMate 9KD evaluates the PD-1 inhibitor nivolumab in combination with docetaxel (NCT03338790). PT-112 is a novel molecule comprised of a platinum agent complexed to a pyrophosphate ligand [123]. It has potential synergy with CPIs and is currently being evaluated in combination with the PD-L1 inhibitor avelumab in the phase I/II PAVE-1 clinical trial (NCT03409458).
4.4. Combination of CPIs with Radiopharmaceuticals
There is evidence that targeted radionuclide therapy (TRT) may increase PD-L1 expression on T cells and that the combination of TRT with CPI leads to increased infiltration by CD8 T cells [124]. As of July 2020, there are 20 interventional clinical trials actively recruiting patients that test radiopharmaceuticals’ efficacy in patients with mCRPC. A phase Ib and a phase I/II trial combine pembrolizumab with Lu 177-PSMA-617, a β emitting isotope of lutetium attached to PSMA (NCT03658447, NCT03805594) (Figure 4I). Lu 177-PSMA-617 has shown some efficacy as monotherapy in mCRPC [125]. The combination of radium-223, PD-L1 inhibitor avelumab, and peposertib is being evaluated in a multicenter phase 1/2 clinical trial (NCT04071236). Peposertib is a DNA-dependent protein kinase inhibitor, thereby inhibiting its ability to function in DNA damage response (DDR). By preventing the repair of radiation-induced DNA double-strand breaks (DSBs), peposertib has a potential for synergy with radiation [126].
4.5. Combination of CPIs with Radiation
Stereotactic body radiation therapy (SBRT) offers the benefit of local control for oligometastatic disease with minimal toxicity [127]. There is some evidence that suggests treatment with radiation leads to antigen release and exposure of damage-associated molecular patterns—potentially leading to the “abscopal effect” [128]. In addition to upregulation of MHC I expression, which is potentially immune-stimulating when combined with CPI [129][130], radiation has other potential synergistic effects with CPI, such as increased Fas surface expression leading to interaction with Fas ligand (FAS-L) and enhanced cytotoxic T cell activity [131], dendritic cell activation resulting in enhanced cross-presentation of TAAs [132], and upregulation of PD-L1 [133][134].
Post-hoc analysis of the KEYNOTE-001 trial suggests previous treatment with radiation in patients with NSCLC leads to improved PFS and OS with pembrolizumab therapy [135]. The efficacy of PD-L1 inhibitor durvalumab after concurrent chemoradiotherapy for patients with NSCLC was demonstrated in the phase III PACIFIC clinical trial [136]. The phase I RADVAX clinical trial evaluating the combination of pembrolizumab with hypofractionated radiotherapy (HFRT) in patients with metastatic cancers revealed that in some patients, HFRT could reinvigorate systemic response to pembrolizumab, despite prior progression on PD-1 therapy [137]. One of the PORTER trials’ cohorts evaluates the combination of nivolumab with SBRT in patients with mCRPC (NCT03835533).
4.6. Combination of CPIs with PARP Inhibitors
Poly ADP ribose polymerase (PARP) inhibitors are small molecules that block the repair of single-strand DNA breaks. Because of this, tumors with homologous recombination deficiency (HRD) show particular sensitivity to PARP inhibition [138]. PARP inhibitors have multiple potential synergistic effects when combined with CPIs, such as increased intratumoral CD8 T cell infiltration [139], increased IFNγ production in the TME [140], and PD-L1 upregulation [141].
There are currently two PARP inhibitors approved for administration in patients with mCRPC and HRD. Rucaparib was approved based on the phase II TRITON2 study’s preliminary results, which evaluated men with HRD CRPC [142]. Rucaparib demonstrated an ORR of 44% in patients with BRCA 1/2 mutation and is currently approved for this patient population. Olaparib was approved based on the results of the phase III PROFOUND clinical trial, which enrolled patients with mCRPC who had disease progression while receiving a new hormonal agent (e.g., enzalutamide or abiraterone), and had a qualifying alteration in prespecified genes involved in homologous recombination repair [143]. Olaparib demonstrated a higher ORR in men with previously treated mCRPC and HRD compared to the physician’s choice of enzalutamide or abiraterone (control), and is FDA approved for this patient population.
PARP inhibitors could have efficacy beyond HRD tumors when combined with other agents such as cytotoxic chemotherapy (taxanes, topoisomerase inhibitors, etc.) and radiation due to the accumulation of DNA damage [144]. As of July 2020, there are 14 ongoing trials testing this hypothesis. These ongoing clinical trials evaluate PARP inhibitors’ efficacy in combination with various agents such as chemotherapy, radiopharmaceuticals, and checkpoint inhibitors (Figure 4J). Of these, only five clinical trials are in patients with HRD. PARP inhibition could potentiate immune modulation elicited by CPI by enhanced immune priming and T cell infiltration [145]. KEYNOTE-365 is evaluating the combination of pembrolizumab and olaparib (NCT02861573). Similarly, CheckMate 9KD evaluates the combination of nivolumab and rucaparib [146].
4.7. Combinations of CPIs and Adenosine Receptor Antagonists
A2B adenosine receptor (A2BR) is a G protein-coupled receptor (GPCR), which is expressed at high levels in prostate cancer tissue [147], and its blockade has been associated with inhibition of prostate cancer growth [148]. It has also been observed that PD-1 blockade leads to overexpression of adenosine 2A receptor (A2AR) on tumor-infiltrating CD8 T cells, thereby making them more susceptible to A2A-mediated suppression [149]. Currently, five ongoing trials are evaluating the efficacy of adenosine receptor antagonists in patients with mCRPC. Of these, four combine adenosine receptor antagonists with CPI (NCT04381832, NCT03207867, NCT04089553, NCT03629756) (Figure 4G). NZV930 is a novel anti-CD73 monoclonal antibody that prevents the conversion of extracellular adenosine monophosphate (AMP) to adenosine and is being evaluated in a phase Ib/II study in combination with a CPI (NCT03549000).
4.8. Combination of Interleukin-2 (IL-2) Agonist with CPI
IL-2 is a cytokine that potentiates T cell proliferation and NK cell activity and induces differentiation of Tregs [150]. Based on the interim results of the phase I/II PIVOT-2 clinical trial, IL-2 receptor agonist bempegaldesleukin has demonstrated activity in combination with nivolumab for patients with urothelial carcinoma (Figure 4A). Notably, it converted some PD-L1 negative patients to PD-L1 positive, as assessed by the PharmDx 28-8 assay [151]. It is currently being evaluated in a phase I/II clinical trial in combination with avelumab (NCT04052204).
4.9. Combination of CD11b Agonist with CPI
CD11b is an integrin that promotes pro-inflammatory macrophage polarization and suppresses tumor growth [152]. Preclinical studies have shown it can lead to a reduction in MDSCs and enhance dendritic cell responses, all of which potentially improve anti-tumor T cell immunity and may have synergistic effects with CPIs [153][154]. GB1275 is a CD11b agonist being tested in a phase I/II clinical trial combined with pembrolizumab in patients with solid cancers, including mCRPC (NCT04060342) (Figure 4D).
References
- Howlader, N.A.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; Chen, H.S.; et al. SEER Cancer Statistics Review, 1975–2016. In 2018 Seer Data Submiss; National Cancer Institute: Bethesda, MD, USA, 2019.
- Chandrasekar, T.; Yang, J.C.; Gao, A.C.; Evans, C.P. Mechanisms of resistance in castration-resistant prostate cancer (CRPC). Transl. Urol. 2015, 4, 365–380.
- Pienta, K.J.; Bradley, D. Mechanisms Underlying the Development of Androgen-Independent Prostate Cancer. Clin. Cancer Res. 2006, 12, 1665.
- Shaffer, D.R.; Scher, H.I. Prostate cancer: A dynamic illness with shifting targets. Lancet Oncol. 2003, 4, 407–414.
- Freedland, S.J.; Humphreys, E.B.; Mangold, L.A.; Eisenberger, M.; Dorey, F.J.; Walsh, P.C.; Partin, A.W. Death in patients with recurrent prostate cancer after radical prostatectomy: Prostate-specific antigen doubling time subgroups and their associated contributions to all-cause mortality. J. Clin. Oncol. 2007, 25, 1765–1771.
- Tangen, C.M.; Hussain, M.H.A.; Higano, C.S.; Eisenberger, M.A.; Small, E.J.; Wilding, G.; Donnelly, B.J.; Schelhammer, P.F.; Crawford, E.D.; Vogelzang, N.J.; et al. Improved Overall Survival Trends of Men with Newly Diagnosed M1 Prostate Cancer: A SWOG Phase III Trial Experience (S8494, S8894 and S9346). J. Urol. 2012, 188, 1164–1169.
- Huggins, C.; Hodges, C.V. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. CA A Cancer J. Clin. 1972, 22, 232–240.
- Limonta, P.; Marelli, M.M.; Moretti, R.M. LHRH analogues as anticancer agents: Pituitary and extrapituitary sites of action. Expert Opin. Investig. Drugs 2001, 10, 709–720.
- Fizazi, K.; Tran, N.; Fein, L.; Matsubara, N.; Rodriguez-Antolin, A.; Alekseev, B.Y.; Özgüroğlu, M.; Ye, D.; Feyerabend, S.; Protheroe, A.; et al. Abiraterone plus Prednisone in Metastatic, Castration-Sensitive Prostate Cancer. N. Engl. J. Med. 2017, 377, 352–360.
- James, N.D.; de Bono, J.S.; Spears, M.R.; Clarke, N.W.; Mason, M.D.; Dearnaley, D.P.; Ritchie, A.W.S.; Amos, C.L.; Gilson, C.; Jones, R.J.; et al. Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy. N. Engl. J. Med. 2017, 377, 338–351.
- Gravis, G.; Boher, J.-M.; Chen, Y.-H.; Liu, G.; Fizazi, K.; Carducci, M.A.; Oudard, S.; Joly, F.; Jarrard, D.M.; Soulie, M.; et al. Burden of Metastatic Castrate Naive Prostate Cancer Patients, to Identify Men More Likely to Benefit from Early Docetaxel: Further Analyses of CHAARTED and GETUG-AFU15 Studies. Eur. Urol. 2018, 73, 847–855.
- Sathianathen, N.J.; Philippou, Y.A.; Kuntz, G.M.; Konety, B.R.; Gupta, S.; Lamb, A.D.; Dahm, P. Taxane-based chemohormonal therapy for metastatic hormone-sensitive prostate cancer. Cochrane Database Syst. Rev. 2018.
- Davis, I.D.; Martin, A.J.; Stockler, M.R.; Begbie, S.; Chi, K.N.; Chowdhury, S.; Coskinas, X.; Frydenberg, M.; Hague, W.E.; Horvath, L.G.; et al. Enzalutamide with Standard First-Line Therapy in Metastatic Prostate Cancer. N. Engl. J. Med. 2019, 381, 121–131.
- Chi, K.N.; Agarwal, N.; Bjartell, A.; Chung, B.H.; Pereira de Santana Gomes, A.J.; Given, R.; Juárez Soto, Á.; Merseburger, A.S.; Özgüroğlu, M.; Uemura, H.; et al. Apalutamide for Metastatic, Castration-Sensitive Prostate Cancer. N. Engl. J. Med. 2019, 381, 13–24.
- Armstrong, A.J.; Szmulewitz, R.Z.; Petrylak, D.P.; Holzbeierlein, J.; Villers, A.; Azad, A.; Alcaraz, A.; Alekseev, B.; Iguchi, T.; Shore, N.D.; et al. ARCHES: A Randomized, Phase III Study of Androgen Deprivation Therapy With Enzalutamide or Placebo in Men With Metastatic Hormone-Sensitive Prostate Cancer. J. Clin. Oncol. 2019, 37, 2974–2986.
- Morris, M.J.; Rumble, R.B.; Basch, E.; Hotte, S.J.; Loblaw, A.; Rathkopf, D.; Celano, P.; Bangs, R.; Milowsky, M.I. Optimizing Anticancer Therapy in Metastatic Non-Castrate Prostate Cancer: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2018, 36, 1521–1539.
- Chen, J.; Ni, Y.; Sun, G.; Liao, B.; Zhang, X.; Zhao, J.; Zhu, S.; Wang, Z.; Shen, P.; Zeng, H. Comparison of Current Systemic Combination Therapies for Metastatic Hormone-Sensitive Prostate Cancer and Selection of Candidates for Optimal Treatment: A Systematic Review and Bayesian Network Meta-Analysis. Front. Oncol. 2020, 10, 1806.
- Sweeney, C.J.; Chen, Y.-H.; Carducci, M.; Liu, G.; Jarrard, D.F.; Eisenberger, M.; Wong, Y.-N.; Hahn, N.; Kohli, M.; Cooney, M.M.; et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. N. Engl. J. Med. 2015, 373, 737–746.
- Steinberg, R.L.; Thomas, L.J.; O’Donnell, M.A. Bacillus Calmette-Guérin (BCG) Treatment Failures in Non-Muscle Invasive Bladder Cancer: What Truly Constitutes Unresponsive Disease. Bladder Cancer 2015, 1, 105–116.
- Guallar-Garrido, S.; Julián, E. Bacillus Calmette-Guérin (BCG) Therapy for Bladder Cancer: An Update. Immunotargets 2020, 9, 1–11.
- Guo, C.; Manjili, M.H.; Subjeck, J.R.; Sarkar, D.; Fisher, P.B.; Wang, X.-Y. Therapeutic cancer vaccines: Past, present, and future. Adv. Cancer Res. 2013, 119, 421–475.
- Comber, J.D.; Philip, R. MHC class I antigen presentation and implications for developing a new generation of therapeutic vaccines. Adv. Vaccines 2014, 2, 77–89.
- Khalili, S.; Rahbar, M.R.; Dezfulian, M.H.; Jahangiri, A. In silico analyses of Wilms’ tumor protein to designing a novel multi-epitope DNA vaccine against cancer. J. Theor. Biol. 2015, 379, 66–78.
- Yu, Z.; Theoret, M.R.; Touloukian, C.E.; Surman, D.R.; Garman, S.C.; Feigenbaum, L.; Baxter, T.K.; Baker, B.M.; Restifo, N.P. Poor immunogenicity of a self/tumor antigen derives from peptide–MHC-I instability and is independent of tolerance. J. Clin. Investig. 2004, 114, 551–559.
- Engels, B.; Engelhard, V.H.; Sidney, J.; Sette, A.; Binder, D.C.; Liu, R.B.; Kranz, D.M.; Meredith, S.C.; Rowley, D.A.; Schreiber, H. Relapse or Eradication of Cancer Is Predicted by Peptide-Major Histocompatibility Complex Affinity. Cancer Cell 2013, 23, 516–526.
- Cole, G.; McCaffrey, J.; Ali, A.A.; McCarthy, H.O. DNA vaccination for prostate cancer: Key concepts and considerations. Cancer Nanotechnol. 2015, 6, 2.
- Wolchok, J.D.; Gregor, P.D.; Nordquist, L.T.; Slovin, S.F.; Scher, H.I. DNA vaccines: An active immunization strategy for prostate cancer. Semin. Oncol. 2003, 30, 659–666.
- McNeel, D.G.; Dunphy, E.J.; Davies, J.G.; Frye, T.P.; Johnson, L.E.; Staab, M.J.; Horvath, D.L.; Straus, J.; Alberti, D.; Marnocha, R.; et al. Safety and Immunological Efficacy of a DNA Vaccine Encoding Prostatic Acid Phosphatase in Patients With Stage D0 Prostate Cancer. J. Clin. Oncol. 2009, 27, 4047–4054.
- Kyriakopoulos, C.E.; Eickhoff, J.C.; Ferrari, A.C.; Schweizer, M.T.; Wargowski, E.; Olson, B.M.; McNeel, D.G. Multicenter Phase I Trial of a DNA Vaccine Encoding the Androgen Receptor Ligand-binding Domain (pTVG-AR, MVI-118) in Patients with Metastatic Prostate Cancer. Clin. Cancer Res. 2020, 26, 5162.
- McNeel, D.G.; Eickhoff, J.C.; Johnson, L.E.; Roth, A.R.; Perk, T.G.; Fong, L.; Antonarakis, E.S.; Wargowski, E.; Jeraj, R.; Liu, G. Phase II Trial of a DNA Vaccine Encoding Prostatic Acid Phosphatase (pTVG-HP [MVI-816]) in Patients With Progressive, Nonmetastatic, Castration-Sensitive Prostate Cancer. J. Clin. Oncol. 2019, 37, 3507–3517.
- Peng, M.; Mo, Y.; Wang, Y.; Wu, P.; Zhang, Y.; Xiong, F.; Guo, C.; Wu, X.; Li, Y.; Li, X.; et al. Neoantigen vaccine: An emerging tumor immunotherapy. Mol. Cancer 2019, 18, 128.
- Malonis, R.J.; Lai, J.R.; Vergnolle, O. Peptide-Based Vaccines: Current Progress and Future Challenges. Chem. Rev. 2020, 120, 3210–3229.
- Zanetti, M. A second chance for telomerase reverse transcriptase in anticancer immunotherapy. Nat. Rev. Clin. Oncol. 2017, 14, 115–128.
- Noguchi, M.; Sasada, T.; Itoh, K. Personalized peptide vaccination: A new approach for advanced cancer as therapeutic cancer vaccine. Cancer Immunol. Immunother. 2013, 62, 919–929.
- Rauch, S.; Jasny, E.; Schmidt, K.E.; Petsch, B. New Vaccine Technologies to Combat Outbreak Situations. Front. Immunol. 2018, 9, 1963.
- Bouard, D.; Alazard-Dany, N.; Cosset, F.L. Viral vectors: From virology to transgene expression. Br. J. Pharmacol. 2009, 157, 153–165.
- Madan, R.A.; Arlen, P.M.; Mohebtash, M.; Hodge, J.W.; Gulley, J.L. Prostvac-VF: A vector-based vaccine targeting PSA in prostate cancer. Expert Opin. Investig. Drugs 2009, 18, 1001–1011.
- Arlen, P.M.; Skarupa, L.; Pazdur, M.; Seetharam, M.; Tsang, K.Y.; Grosenbach, D.W.; Feldman, J.; Poole, D.J.; Litzinger, M.; Steinberg, S.M.; et al. Clinical safety of a viral vector based prostate cancer vaccine strategy. J. Urol. 2007, 178, 1515–1520.
- Kantoff, P.W.; Schuetz, T.J.; Blumenstein, B.A.; Glode, L.M.; Bilhartz, D.L.; Wyand, M.; Manson, K.; Panicali, D.L.; Laus, R.; Schlom, J.; et al. Overall survival analysis of a phase II randomized controlled trial of a Poxviral-based PSA-targeted immunotherapy in metastatic castration-resistant prostate cancer. J. Clin. Oncol. 2010, 28, 1099–1105.
- Gulley, J.L.; Borre, M.; Vogelzang, N.J.; Ng, S.; Agarwal, N.; Parker, C.C.; Pook, D.W.; Rathenborg, P.; Flaig, T.W.; Carles, J.; et al. Phase III Trial of PROSTVAC in Asymptomatic or Minimally Symptomatic Metastatic Castration-Resistant Prostate Cancer. J. Clin. Oncol. 2019, 37, 1051–1061.
- Kantoff, P.W.; Gulley, J.L.; Pico-Navarro, C. Revised Overall Survival Analysis of a Phase II, Randomized, Double-Blind, Controlled Study of PROSTVAC in Men With Metastatic Castration-Resistant Prostate Cancer. J. Clin. Oncol. 2017, 35, 124–125.
- Kantoff, P.W.; Higano, C.S.; Shore, N.D.; Berger, E.R.; Small, E.J.; Penson, D.F.; Redfern, C.H.; Ferrari, A.C.; Dreicer, R.; Sims, R.B.; et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N. Engl. J. Med. 2010, 363, 411–422.
- de Bono, J.S.; Oudard, S.; Ozguroglu, M.; Hansen, S.; Machiels, J.-P.; Kocak, I.; Gravis, G.; Bodrogi, I.; Mackenzie, M.J.; Shen, L.; et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: A randomised open-label trial. Lancet 2010, 376, 1147–1154.
- Fizazi, K.; Scher, H.I.; Molina, A.; Logothetis, C.J.; Chi, K.N.; Jones, R.J.; Staffurth, J.N.; North, S.; Vogelzang, N.J.; Saad, F.; et al. Abiraterone acetate for treatment of metastatic castration-resistant prostate cancer: Final overall survival analysis of the COU-AA-301 randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2012, 13, 983–992.
- Ryan, C.J.; Smith, M.R.; Fizazi, K.; Saad, F.; Mulders, P.F.; Sternberg, C.N.; Miller, K.; Logothetis, C.J.; Shore, N.D.; Small, E.J.; et al. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): Final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2015, 16, 152–160.
- Scher, H.I.; Fizazi, K.; Saad, F.; Taplin, M.E.; Sternberg, C.N.; Miller, K.; de Wit, R.; Mulders, P.; Chi, K.N.; Shore, N.D.; et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 2012, 367, 1187–1197.
- Beer, T.M.; Armstrong, A.J.; Rathkopf, D.E.; Loriot, Y.; Sternberg, C.N.; Higano, C.S.; Iversen, P.; Bhattacharya, S.; Carles, J.; Chowdhury, S.; et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N. Engl. J. Med. 2014, 371, 424–433.
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fosså, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 2013, 369, 213–223.
- Sabado, R.L.; Balan, S.; Bhardwaj, N. Dendritic cell-based immunotherapy. Cell Res. 2017, 27, 74–95.
- Warren, T.L.; Weiner, G.J. Uses of granulocyte-macrophage colony-stimulating factor in vaccine development. Curr. Opin. Hematol. 2000, 7, 168–173.
- Small, E.J.; Sacks, N.; Nemunaitis, J.; Urba, W.J.; Dula, E.; Centeno, A.S.; Nelson, W.G.; Ando, D.; Howard, C.; Borellini, F.; et al. Granulocyte Macrophage Colony-Stimulating Factor–Secreting Allogeneic Cellular Immunotherapy for Hormone-Refractory Prostate Cancer. Clin. Cancer Res. 2007, 13, 3883.
- Higano, C.S.; Corman, J.M.; Smith, D.C.; Centeno, A.S.; Steidle, C.P.; Gittleman, M.; Simons, J.W.; Sacks, N.; Aimi, J.; Small, E.J. Phase 1/2 dose-escalation study of a GM-CSF-secreting, allogeneic, cellular immunotherapy for metastatic hormone-refractory prostate cancer. Cancer 2008, 113, 975–984.
- Small, E. A phase III trial of GVAX immunotherapy for prostate cancer in combination with docetaxel versus docetaxel plus prednisone in symptomatic, castration-resistant prostate cancer (CRPC). In Proceedings of the 2009 Genitourinary Cancer Symposium, American Society of Clinical Oncology (ASCO), Orlando, FL, USA, 26–28 February 2009.
- van den Eertwegh, A.J.M.; Versluis, J.; van den Berg, H.P.; Santegoets, S.J.A.M.; van Moorselaar, R.J.A.; van der Sluis, T.M.; Gall, H.E.; Harding, T.C.; Jooss, K.; Lowy, I.; et al. Combined immunotherapy with granulocyte-macrophage colony-stimulating factor-transduced allogeneic prostate cancer cells and ipilimumab in patients with metastatic castration-resistant prostate cancer: A phase 1 dose-escalation trial. Lancet Oncol. 2012, 13, 509–517.
- Burch, P.A.; Breen, J.K.; Buckner, J.C.; Gastineau, D.A.; Kaur, J.A.; Laus, R.L.; Padley, D.J.; Peshwa, M.V.; Pitot, H.C.; Richardson, R.L.; et al. Priming Tissue-specific Cellular Immunity in a Phase I Trial of Autologous Dendritic Cells for Prostate Cancer. Clin. Cancer Res. 2000, 6, 2175.
- Small, E.J.; Schellhammer, P.F.; Higano, C.S.; Redfern, C.H.; Nemunaitis, J.J.; Valone, F.H.; Verjee, S.S.; Jones, L.A.; Hershberg, R.M. Placebo-controlled phase III trial of immunologic therapy with sipuleucel-T (APC8015) in patients with metastatic, asymptomatic hormone refractory prostate cancer. J. Clin. Oncol. 2006, 24, 3089–3094.
- Higano, C.S.; Schellhammer, P.F.; Small, E.J.; Burch, P.A.; Nemunaitis, J.; Yuh, L.; Provost, N.; Frohlich, M.W. Integrated data from 2 randomized, double-blind, placebo-controlled, phase 3 trials of active cellular immunotherapy with sipuleucel-T in advanced prostate cancer. Cancer 2009, 115, 3670–3679.
- Schellhammer, P.F.; Chodak, G.; Whitmore, J.B.; Sims, R.; Frohlich, M.W.; Kantoff, P.W. Lower baseline prostate-specific antigen is associated with a greater overall survival benefit from sipuleucel-T in the Immunotherapy for Prostate Adenocarcinoma Treatment (IMPACT) trial. Urology 2013, 81, 1297–1302.
- Hanahan, D.; Coussens, L.M. Accessories to the Crime: Functions of Cells Recruited to the Tumor Microenvironment. Cancer Cell 2012, 21, 309–322.
- Jiang, X.; Wang, J.; Deng, X.; Xiong, F.; Ge, J.; Xiang, B.; Wu, X.; Ma, J.; Zhou, M.; Li, X.; et al. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol. Cancer 2019, 18, 10.
- Xu, X.; Zheng, L.; Yuan, Q.; Zhen, G.; Crane, J.L.; Zhou, X.; Cao, X. Transforming growth factor-β in stem cells and tissue homeostasis. Bone Res. 2018, 6, 2.
- Masoud, G.N.; Li, W. HIF-1α pathway: Role, regulation and intervention for cancer therapy. Acta Pharm. Sin. B 2015, 5, 378–389.
- Saraiva, M.; O’Garra, A. The regulation of IL-10 production by immune cells. Nat. Rev. Immunol. 2010, 10, 170–181.
- Gannon, P.O.; Poisson, A.O.; Delvoye, N.; Lapointe, R.; Mes-Masson, A.M.; Saad, F. Characterization of the intra-prostatic immune cell infiltration in androgen-deprived prostate cancer patients. J. Immunol. Methods 2009, 348, 9–17.
- Garcia, A.J.; Ruscetti, M.; Arenzana, T.L.; Tran, L.M.; Bianci-Frias, D.; Sybert, E.; Priceman, S.J.; Wu, L.; Nelson, P.S.; Smale, S.T.; et al. Pten null prostate epithelium promotes localized myeloid-derived suppressor cell expansion and immune suppression during tumor initiation and progression. Mol. Cell Biol. 2014, 34, 2017–2028.
- Tesi, R.J. MDSC; the Most Important Cell You Have Never Heard of. Trends Pharmacol. Sci. 2019, 40, 4–7.
- Zhang, S.; Ma, X.; Zhu, C.; Liu, L.; Wang, G.; Yuan, X. The Role of Myeloid-Derived Suppressor Cells in Patients with Solid Tumors: A Meta-Analysis. PLoS ONE 2016, 11, e0164514.
- Gonda, K.; Shibata, M.; Ohtake, T.; Matsumoto, Y.; Tachibana, K.; Abe, N.; Ohto, H.; Sakurai, K.; Takenoshita, S. Myeloid-derived suppressor cells are increased and correlated with type 2 immune responses, malnutrition, inflammation, and poor prognosis in patients with breast cancer. Oncol. Lett. 2017, 14, 1766–1774.
- Lin, Y.; Xu, J.; Lan, H. Tumor-associated macrophages in tumor metastasis: Biological roles and clinical therapeutic applications. J. Hematol. Oncol. 2019, 12, 76.
- Flammiger, A.; Weisbach, L.; Huland, H.; Tennstedt, P.; Simon, R.; Minner, S.; Bokemeyer, C.; Sauter, G.; Schlomm, T.; Trepel, M. High tissue density of FOXP3+ T cells is associated with clinical outcome in prostate cancer. Eur. J. Cancer 2013, 49, 1273–1279.
- Eastham, J.A.; Truong, L.D.; Rogers, E.; Kattan, M.; Flanders, K.C.; Scardino, P.T.; Thompson, T.C. Transforming growth factor-beta 1: Comparative immunohistochemical localization in human primary and metastatic prostate cancer. Lab. Investig. 1995, 73, 628–635.
- Steiner, M.S.; Barrack, E.R. Transforming growth factor-beta 1 overproduction in prostate cancer: Effects on growth in vivo and in vitro. Mol. Endocrinol. 1992, 6, 15–25.
- Song, B.; Park, S.-H.; Zhao, J.C.; Fong, K.-w.; Li, S.; Lee, Y.; Yang, Y.A.; Sridhar, S.; Lu, X.; Abdulkadir, S.A.; et al. Targeting FOXA1-mediated repression of TGF-β signaling suppresses castration-resistant prostate cancer progression. J. Clin. Investig. 2019, 129, 569–582.
- Liu, G.; Lu, S.; Wang, X.; Page, S.T.; Higano, C.S.; Plymate, S.R.; Greenberg, N.M.; Sun, S.; Li, Z.; Wu, J.D. Perturbation of NK cell peripheral homeostasis accelerates prostate carcinoma metastasis. J. Clin. Investig. 2013, 123, 4410–4422.
- Diefenbach, A.; Jensen, E.R.; Jamieson, A.M.; Raulet, D.H. Rae1 and H60 ligands of the NKG2D receptor stimulate tumour immunity. Nature 2001, 413, 165–171.
- Healy, C.G.; Simons, J.W.; Carducci, M.A.; Deweese, T.L.; Bartkowski, M.; Tong, K.P.; Bolton, W.E. Impaired expression and function of signal-transducing zeta chains in peripheral T cells and natural killer cells in patients with prostate cancer. Cytometry 1998, 32, 109–119.
- Maleki Vareki, S. High and low mutational burden tumors versus immunologically hot and cold tumors and response to immune checkpoint inhibitors. J. Immunother. Cancer 2018, 6, 157.
- Gajewski, T.F.; Woo, S.R.; Zha, Y.; Spaapen, R.; Zheng, Y.; Corrales, L.; Spranger, S. Cancer immunotherapy strategies based on overcoming barriers within the tumor microenvironment. Curr. Opin. Immunol. 2013, 25, 268–276.
- Campoli, M.; Ferrone, S. HLA antigen changes in malignant cells: Epigenetic mechanisms and biologic significance. Oncogene 2008, 27, 5869–5885.
- Chang, C.C.; Campoli, M.; Ferrone, S. Classical and nonclassical HLA class I antigen and NK Cell-activating ligand changes in malignant cells: Current challenges and future directions. Adv. Cancer Res. 2005, 93, 189–234.
- Wang, C.; Xiong, C.; Hsu, Y.-C.; Wang, X.; Chen, L. Human leukocyte antigen (HLA) and cancer immunotherapy: HLA-dependent and -independent adoptive immunotherapies. Ann. Blood 2020, 5, 3058–3070.
- Cai, L.; Michelakos, T.; Yamada, T.; Fan, S.; Wang, X.; Schwab, J.H.; Ferrone, C.R.; Ferrone, S. Defective HLA class I antigen processing machinery in cancer. Cancer Immunol. Immunother. 2018, 67, 999–1009.
- Subklewe, M.; von Bergwelt-Baildon, M.; Humpe, A. Chimeric Antigen Receptor T Cells: A Race to Revolutionize Cancer Therapy. Transfus. Med. Hemother. 2019, 46, 15–24.
- Aragon-Ching, J.B.; Williams, K.M.; Gulley, J.L. Impact of androgen-deprivation therapy on the immune system: Implications for combination therapy of prostate cancer. Front. Biosci. 2007, 12, 4957–4971.
- Foster, W.R.; Car, B.D.; Shi, H.; Levesque, P.C.; Obermeier, M.T.; Gan, J.; Arezzo, J.C.; Powlin, S.S.; Dinchuk, J.E.; Balog, A.; et al. Drug safety is a barrier to the discovery and development of new androgen receptor antagonists. Prostate 2011, 71, 480–488.
- Pu, Y.; Xu, M.; Liang, Y.; Yang, K.; Guo, Y.; Yang, X.; Fu, Y.-X. Androgen receptor antagonists compromise T cell response against prostate cancer leading to early tumor relapse. Sci. Transl. Med. 2016, 8, 333ra47.
- Kyewski, B.; Derbinski, J. Self-representation in the thymus: An extended view. Nat. Rev. Immunol. 2004, 4, 688–698.
- Ribas, A.; Wolchok, J.D. Cancer immunotherapy using checkpoint blockade. Science 2018, 359, 1350.
- Leoni, G.; Alise, A.M.; Cotugno, G.; Langone, F.; Garzia, I.; De Lucia, M.; Fichera, I.; Vitale, R.; Bignone, V.; Tucci, F.G.; et al. A Genetic Vaccine Encoding Shared Cancer Neoantigens to Treat Tumors with Microsatellite Instability. Cancer Res. 2020, 80, 3972.
- Hundal, J.; Kiwala, S.; McMichael, J.; Miller, C.A.; Xia, H.; Wollam, A.T.; Liu, C.J.; Zhao, S.; Feng, Y.-Y.; Graubert, A.P.; et al. pVACtools: A Computational Toolkit to Identify and Visualize Cancer Neoantigens. Cancer Immunol. Res. 2020, 8, 409.
- Gubin, M.M.; Zhang, X.; Schuster, H.; Caron, E.; Ward, J.P.; Noguchi, T.; Ivanova, Y.; Hundal, J.; Arthur, C.D.; Krebber, W.J.; et al. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature 2014, 515, 577–581.
- Cimen Bozkus, C.; Roudko, V.; Finnigan, J.P.; Mascarenhas, J.; Hoffman, R.; Iancu-Rubin, C.; Bhardwaj, N. Immune Checkpoint Blockade Enhances Shared Neoantigen-Induced T-cell Immunity Directed against Mutated Calreticulin in Myeloproliferative Neoplasms. Cancer Discov 2019, 9, 1192–1207.
- Sahin, U.; Oehm, P.; Derhovanessian, E.; Jabulowsky, R.A.; Vormehr, M.; Gold, M.; Maurus, D.; Schwarck-Kokarakis, D.; Kuhn, A.N.; Omokoko, T.; et al. An RNA vaccine drives immunity in checkpoint-inhibitor-treated melanoma. Nature 2020, 585, 107–112.
- Cappuccini, F.; Bryant, R.; Pollock, E.; Carter, L.; Verrill, C.; Hollidge, J.; Poulton, I.; Baker, M.; Mitton, C.; Baines, A.; et al. Safety and immunogenicity of novel 5T4 viral vectored vaccination regimens in early stage prostate cancer: A phase I clinical trial. J. Immunother. Cancer 2020, 8, e000928.
- Stern, P.L.; Harrop, R. 5T4 oncofoetal antigen: An attractive target for immune intervention in cancer. Cancer Immunol. Immunother. 2017, 66, 415–426.
- Damelin, M.; Geles, K.G.; Follettie, M.T.; Yuan, P.; Baxter, M.; Golas, J.; DiJoseph, J.F.; Karnoub, M.; Huang, S.; Diesl, V.; et al. Delineation of a cellular hierarchy in lung cancer reveals an oncofetal antigen expressed on tumor-initiating cells. Cancer Res. 2011, 71, 4236–4246.
- Southgate, T.D.; McGinn, O.J.; Castro, F.V.; Rutkowski, A.J.; Al-Muftah, M.; Marinov, G.; Smethurst, G.J.; Shaw, D.; Ward, C.M.; Miller, C.J.; et al. CXCR4 mediated chemotaxis is regulated by 5T4 oncofetal glycoprotein in mouse embryonic cells. PLoS ONE 2010, 5, e9982.
- Kagermeier-Schenk, B.; Wehner, D.; Ozhan-Kizil, G.; Yamamoto, H.; Li, J.; Kirchner, K.; Hoffmann, C.; Stern, P.; Kikuchi, A.; Schambony, A.; et al. Waif1/5T4 inhibits Wnt/β-catenin signaling and activates noncanonical Wnt pathways by modifying LRP6 subcellular localization. Dev. Cell 2011, 21, 1129–1143.
- Redchenko, I.; Cappuccini, F.; Pollock, E.; Bryant, R.J.; Carter, L.; Verrill, C.; Hollidge, J.; Goodwin, L.; Harrop, R.; Romero, P.J.; et al. VANCE: First-in-human phase I study of a novel ChAdOx1-MVA 5T4 vaccine in low and intermediate risk prostate cancer. J. Clin. Oncol. 2018, 36, 3018.
- Johnson, L.E.; Frye, T.P.; Chinnasamy, N.; Chinnasamy, D.; McNeel, D.G. Plasmid DNA vaccine encoding prostatic acid phosphatase is effective in eliciting autologous antigen-specific CD8+ T cells. Cancer Immunol. Immunother. 2007, 56, 885–895.
- Olson, B.M.; Johnson, L.E.; McNeel, D.G. The androgen receptor: A biologically relevant vaccine target for the treatment of prostate cancer. Cancer Immunol. Immunother. 2013, 62, 585–596.
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. mRNA vaccines—A new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279.
- Kranz, L.M.; Diken, M.; Haas, H.; Kreiter, S.; Loquai, C.; Reuter, K.C.; Meng, M.; Fritz, D.; Vascotto, F.; Hefesha, H.; et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature 2016, 534, 396–401.
- Schumacher, T.N.; Schreiber, R.D. Neoantigens in cancer immunotherapy. Science 2015, 348, 69–74.
- Keskin, D.B.; Anandappa, A.J.; Sun, J.; Tirosh, I.; Mathewson, N.D.; Li, S.; Oliveira, G.; Giobbie-Hurder, A.; Felt, K.; Gjini, E.; et al. Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial. Nature 2019, 565, 234–239.
- Robbins, P.F.; Lu, Y.C.; El-Gamil, M.; Li, Y.F.; Gross, C.; Gartner, J.; Lin, J.C.; Teer, J.K.; Cliften, P.; Tycksen, E.; et al. Mining exomic sequencing data to identify mutated antigens recognized by adoptively transferred tumor-reactive T cells. Nat. Med. 2013, 19, 747–752.
- Thompson, R.H.; Allison, J.P.; Kwon, E.D. Anti-cytotoxic T lymphocyte antigen-4 (CTLA-4) immunotherapy for the treatment of prostate cancer. Urol. Oncol. 2006, 24, 442–447.
- Antonarakis, E.S.; Piulats, J.M.; Gross-Goupil, M.; Goh, J.; Ojamaa, K.; Hoimes, C.J.; Vaishampayan, U.; Berger, R.; Sezer, A.; Alanko, T.; et al. Pembrolizumab for Treatment-Refractory Metastatic Castration-Resistant Prostate Cancer: Multicohort, Open-Label Phase II KEYNOTE-199 Study. J. Clin. Oncol. 2020, 38, 395–405.
- García-Aranda, M.; Redondo, M. Targeting Protein Kinases to Enhance the Response to anti-PD-1/PD-L1 Immunotherapy. Int. J. Mol. Sci. 2019, 20, 2296.
- Arora, A.; Scholar, E.M. Role of Tyrosine Kinase Inhibitors in Cancer Therapy. J. Pharmacol. Exp. Ther. 2005, 315, 971.
- Qin, S.; Li, A.; Yi, M.; Yu, S.; Zhang, M.; Wu, K. Recent advances on anti-angiogenesis receptor tyrosine kinase inhibitors in cancer therapy. J. Hematol. Oncol. 2019, 12, 27.
- Ott, P.A.; Hodi, F.S.; Buchbinder, E.I. Inhibition of Immune Checkpoints and Vascular Endothelial Growth Factor as Combination Therapy for Metastatic Melanoma: An Overview of Rationale, Preclinical Evidence, and Initial Clinical Data. Front. Oncol. 2015, 5, 202.
- Adotevi, O.; Pere, H.; Ravel, P.; Haicheur, N.; Badoual, C.; Merillon, N.; Medioni, J.; Peyrard, S.; Roncelin, S.; Verkarre, V.; et al. A decrease of regulatory T cells correlates with overall survival after sunitinib-based antiangiogenic therapy in metastatic renal cancer patients. J. Immunother. 2010, 33, 991–998.
- Ko, J.S.; Zea, A.H.; Rini, B.I.; Ireland, J.L.; Elson, P.; Cohen, P.; Golshayan, A.; Rayman, P.A.; Wood, L.; Garcia, J.; et al. Sunitinib mediates reversal of myeloid-derived suppressor cell accumulation in renal cell carcinoma patients. Clin. Cancer Res. 2009, 15, 2148–2157.
- Balan, M.; Mier y Teran, E.; Waaga-Gasser, A.M.; Gasser, M.; Choueiri, T.K.; Freeman, G.; Pal, S. Novel roles of c-Met in the survival of renal cancer cells through the regulation of HO-1 and PD-L1 expression. J. Biol. Chem. 2015, 290, 8110–8120.
- Paolino, M.; Penninger, J.M. The Role of TAM Family Receptors in Immune Cell Function: Implications for Cancer Therapy. Cancers (Basel) 2016, 8, 97.
- Molife, C.; Hess, L.M.; Cui, Z.L.; Li, X.I.; Beyrer, J.; Mahoui, M.; Oton, A.B. Sequential therapy with ramucirumab and/or checkpoint inhibitors for non-small-cell lung cancer in routine practice. Future Oncol. 2019, 15, 2915–2931.
- Rini, B.I.; Plimack, E.R.; Stus, V.; Gafanov, R.; Hawkins, R.; Nosov, D.; Pouliot, F.; Alekseev, B.; Soulières, D.; Melichar, B.; et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2019, 380, 1116–1127.
- Agarwal, N.; Loriot, Y.; McGregor, B.A.; Dreicer, R.; Dorff, T.B.; Maughan, B.L.; Kelly, W.K.; Pagliaro, L.C.; Srinivas, S.; Squillante, C.M.; et al. Cabozantinib in combination with atezolizumab in patients with metastatic castration-resistant prostate cancer: Results of cohort 6 of the COSMIC-021 study. J. Clin. Oncol. 2020, 38, 5564.
- Emens, L.A.; Middleton, G. The interplay of immunotherapy and chemotherapy: Harnessing potential synergies. Cancer Immunol. Res. 2015, 3, 436–443.
- Lake, R.A.; Robinson, B.W.S. Immunotherapy and chemotherapy—a practical partnership. Nat. Rev. Cancer 2005, 5, 397–405.
- Leonetti, A.; Wever, B.; Mazzaschi, G.; Assaraf, Y.G.; Rolfo, C.; Quaini, F.; Tiseo, M.; Giovannetti, E. Molecular basis and rationale for combining immune checkpoint inhibitors with chemotherapy in non-small cell lung cancer. Drug Resist. Updates 2019, 46, 100644.
- Yamazaki, T.; Buqué, A.; Ames, T.D.; Galluzzi, L. PT-112 induces immunogenic cell death and synergizes with immune checkpoint blockers in mouse tumor models. Oncoimmunology 2020, 9, 1721810.
- Chen, H.; Zhao, L.; Fu, K.; Lin, Q.; Wen, X.; Jacobson, O.; Sun, L.; Wu, H.; Zhang, X.; Guo, Z.; et al. Integrin α(v)β(3)-targeted radionuclide therapy combined with immune checkpoint blockade immunotherapy synergistically enhances anti-tumor efficacy. Theranostics 2019, 9, 7948–7960.
- Kim, Y.J.; Kim, Y.I. Therapeutic Responses and Survival Effects of 177Lu-PSMA-617 Radioligand Therapy in Metastatic Castrate-Resistant Prostate Cancer: A Meta-analysis. Clin. Nucl. Med. 2018, 43, 728–734.
- van Bussel, M.T.J.; Awada, A.; de Jonge, M.J.A.; Mau-Sørensen, M.; Nielsen, D.; Schöffski, P.; Verheul, H.M.W.; Sarholz, B.; Berghoff, K.; El Bawab, S.; et al. A first-in-man phase 1 study of the DNA-dependent protein kinase inhibitor peposertib (formerly M3814) in patients with advanced solid tumours. Br. J. Cancer 2020.
- Palacios-Eito, A.; Béjar-Luque, A.; Rodríguez-Liñán, M.; García-Cabezas, S. Oligometastases in prostate cancer: Ablative treatment. World J. Clin. Oncol. 2019, 10, 38–51.
- Suek, N.; Campesato, L.F.; Merghoub, T.; Khalil, D.N. Targeted APC Activation in Cancer Immunotherapy to Enhance the Abscopal Effect. Front. Immunol. 2019, 10, 604.
- Sharabi, A.B.; Nirschl, C.J.; Kochel, C.M.; Nirschl, T.R.; Francica, B.J.; Velarde, E.; Deweese, T.L.; Drake, C.G. Stereotactic Radiation Therapy Augments Antigen-Specific PD-1–Mediated Antitumor Immune Responses via Cross-Presentation of Tumor Antigen. Cancer Immunol. Res. 2015, 3, 345.
- Reits, E.A.; Hodge, J.W.; Herberts, C.A.; Groothuis, T.A.; Chakraborty, M.; Wansley, E.K.; Camphausen, K.; Luiten, R.M.; de Ru, A.H.; Neijssen, J.; et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J. Exp. Med. 2006, 203, 1259–1271.
- Chakraborty, M.; Abrams, S.I.; Camphausen, K.; Liu, K.; Scott, T.; Coleman, C.N.; Hodge, J.W. Irradiation of Tumor Cells Up-Regulates Fas and Enhances CTL Lytic Activity and CTL Adoptive Immunotherapy. J. Immunol. 2003, 170, 6338.
- Gupta, A.; Probst, H.C.; Vuong, V.; Landshammer, A.; Muth, S.; Yagita, H.; Schwendener, R.; Pruschy, M.; Knuth, A.; van den Broek, M. Radiotherapy Promotes Tumor-Specific Effector CD8+ T Cells via Dendritic Cell Activation. J. Immunol. 2012, 189, 558.
- Dovedi, S.J.; Adlard, A.L.; Lipowska-Bhalla, G.; McKenna, C.; Jones, S.; Cheadle, E.J.; Stratford, I.J.; Poon, E.; Morrow, M.; Stewart, R.; et al. Acquired Resistance to Fractionated Radiotherapy Can Be Overcome by Concurrent PD-L1 Blockade. Cancer Res. 2014, 74, 5458.
- Sharabi, A.B.; Lim, M.; DeWeese, T.L.; Drake, C.G. Radiation and checkpoint blockade immunotherapy: Radiosensitisation and potential mechanisms of synergy. Lancet Oncol. 2015, 16, e498–e509.
- Shaverdian, N.; Lisberg, A.E.; Bornazyan, K.; Veruttipong, D.; Goldman, J.W.; Formenti, S.C.; Garon, E.B.; Lee, P. Previous radiotherapy and the clinical activity and toxicity of pembrolizumab in the treatment of non-small-cell lung cancer: A secondary analysis of the KEYNOTE-001 phase 1 trial. Lancet Oncol. 2017, 18, 895–903.
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929.
- Maity, A.; Mick, R.; Huang, A.C.; George, S.M.; Farwell, M.D.; Lukens, J.N.; Berman, A.T.; Mitchell, T.C.; Bauml, J.; Schuchter, L.M.; et al. A phase I trial of pembrolizumab with hypofractionated radiotherapy in patients with metastatic solid tumours. Br. J. Cancer 2018, 119, 1200–1207.
- McCabe, N.; Turner, N.C.; Lord, C.J.; Kluzek, K.; Białkowska, A.; Swift, S.; Giavara, S.; Connor, M.J.; Tutt, A.N.; Zdzienicka, M.Z.; et al. Deficiency in the Repair of DNA Damage by Homologous Recombination and Sensitivity to Poly(ADP-Ribose) Polymerase Inhibition. Cancer Res. 2006, 66, 8109.
- Huang, J.; Wang, L.; Cong, Z.; Amoozgar, Z.; Kiner, E.; Xing, D.; Orsulic, S.; Matulonis, U.; Goldberg, M.S. The PARP1 inhibitor BMN 673 exhibits immunoregulatory effects in a Brca1(-/-) murine model of ovarian cancer. Biochem. Biophys. Res. Commun. 2015, 463, 551–556.
- Higuchi, T.; Flies, D.B.; Marjon, N.A.; Mantia-Smaldone, G.; Ronner, L.; Gimotty, P.A.; Adams, S.F. CTLA-4 Blockade Synergizes Therapeutically with PARP Inhibition in BRCA1-Deficient Ovarian Cancer. Cancer Immunol. Res. 2015, 3, 1257.
- Jiao, S.; Xia, W.; Yamaguchi, H.; Wei, Y.; Chen, M.-K.; Hsu, J.-M.; Hsu, J.L.; Yu, W.-H.; Du, Y.; Lee, H.-H.; et al. PARP Inhibitor Upregulates PD-L1 Expression and Enhances Cancer-Associated Immunosuppression. Clin. Cancer Res. 2017, 23, 3711.
- Abida, W.; Campbell, D.; Patnaik, A.; Shapiro, J.D.; Sautois, B.; Vogelzang, N.J.; Voog, E.G.; Bryce, A.H.; McDermott, R.; Ricci, F.; et al. Non-BRCA DNA Damage Repair Gene Alterations and Response to the PARP Inhibitor Rucaparib in Metastatic Castration-Resistant Prostate Cancer: Analysis from the phase 2 TRITON2 study. Clin. Cancer Res. 2020.
- de Bono, J.; Mateo, J.; Fizazi, K.; Saad, F.; Shore, N.; Sandhu, S.; Chi, K.N.; Sartor, O.; Agarwal, N.; Olmos, D.; et al. Olaparib for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 2020, 382, 2091–2102.
- Dréan, A.; Lord, C.J.; Ashworth, A. PARP inhibitor combination therapy. Crit. Rev. Oncol. Hematol. 2016, 108, 73–85.
- Li, A.; Yi, M.; Qin, S.; Chu, Q.; Luo, S.; Wu, K. Prospects for combining immune checkpoint blockade with PARP inhibition. J. Hematol. Oncol. 2019, 12, 98.
- Fizazi, K.; Drake, C.G.; Shaffer, D.R.; Pachynski, R.; Saad, F.; Ciprotti, M.; Kong, G.; Ryan, C.J.; Petrylak, D.P. An open-label, phase 2 study of nivolumab in combination with either rucaparib, docetaxel, or enzalutamide in men with castration-resistant metastatic prostate cancer (mCRPC.; CheckMate 9KD). J. Clin. Oncol. 2018, 36, TPS3126.
- Li, S.; Huang, S.; Peng, S.B. Overexpression of G protein-coupled receptors in cancer cells: Involvement in tumor progression. Int. J. Oncol. 2005, 27, 1329–1339.
- Wei, Q.; Costanzi, S.; Balasubramanian, R.; Gao, Z.-G.; Jacobson, K.A. A2B adenosine receptor blockade inhibits growth of prostate cancer cells. Purinergic Signal. 2013, 9, 271–280.
- Beavis, P.A.; Milenkovski, N.; Henderson, M.A.; John, L.B.; Allard, B.; Loi, S.; Kershaw, M.H.; Stagg, J.; Darcy, P.K. Adenosine Receptor 2A Blockade Increases the Efficacy of Anti-PD-1 through Enhanced Antitumor T-cell Responses. Cancer Immunol. Res. 2015, 3, 506–517.
- Liao, W.; Lin, J.-X.; Leonard, W.J. IL-2 family cytokines: New insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Curr. Opin. Immunol. 2011, 23, 598–604.
- Diab, A.; Tannir, N.M.; Bentebibel, S.-E.; Hwu, P.; Papadimitrakopoulou, V.; Haymaker, C.; Kluger, H.M.; Gettinger, S.N.; Sznol, M.; Tykodi, S.S.; et al. Bempegaldesleukin (NKTR-214) plus Nivolumab in Patients with Advanced Solid Tumors: Phase I Dose-Escalation Study of Safety, Efficacy, and Immune Activation (PIVOT-02). Cancer Discov. 2020.
- Schmid, M.C.; Khan, S.Q.; Kaneda, M.M.; Pathria, P.; Shepard, R.; Louis, T.L.; Anand, S.; Woo, G.; Leem, C.; Faridi, M.H.; et al. Integrin CD11b activation drives anti-tumor innate immunity. Nat. Commun. 2018, 9, 5379.
- Panni, R.Z.; Herndon, J.M.; Zuo, C.; Hegde, S.; Hogg, G.D.; Knolhoff, B.L.; Breden, M.A.; Li, X.; Krisnawan, V.E.; Khan, S.Q.; et al. Agonism of CD11b reprograms innate immunity to sensitize pancreatic cancer to immunotherapies. Sci. Transl. Med. 2019, 11, eaau9240.
- Rasco, D.W.; Bendell, J.C.; Wang-Gillam, A.; Park, W.; O’Reilly, E.M.; Zhou, L.; Galkin, A.; Carter, L.L.; Nickle, D.; Li, J.; et al. A phase I/II study of GB1275, a first-in-class oral CD11b modulator, alone, and combined with pembrolizumab in specified advanced solid tumors or with chemotherapy in metastatic pancreatic cancer (KEYNOTE-A36). J. Clin. Oncol. 2020, 38, 3085.




