
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Claudia Musial | + 3147 word(s) | 3147 | 2021-01-21 10:12:37 | | | |
| 2 | Vivi Li | Meta information modification | 3147 | 2021-01-22 03:28:50 | | |
Video Upload Options
The role of autophagy is to degrade damaged or unnecessary cellular structures. Both in vivo and in vitro studies suggest a dual role of autophagy in cancer—it may promote the development of neoplasms, but it may also play a tumor protective function. The mechanism of autophagy depends on the genetic context, tumor stage and type, tumor microenvironment, or clinical therapy used. Autophagy also plays an important role in cell death as well as in the induction of chemoresistance of cancer cells. The following review describes the extensive autophagic cell death in relation to dietary polyphenols and cancer disease. Polyphenols are organic chemicals that exhibit antioxidant, anti-inflammatory, anti-angiogenic, and immunomodulating properties, and can also initiate the process of apoptosis. In addition, polyphenols reduce oxidative stress and protect against reactive oxygen species. This review presents in vitro and in vivo studies in animal models with the use of polyphenolic compounds such as epigallocatechin-3-gallate (EGCG), oleuropein, punicalgin, apigenin, resveratrol, pterostilbene, or curcumin and their importance in the modulation of autophagy-induced death of cancer cells.
1. The Role of Autophagy in Cancer
Autophagy plays a dual role in cancer [1]. Numerous studies conducted over the past 30 years have demonstrated that the impact of autophagy on the development or limitation of the neoplastic process depends on the genetic context, tumor type, tumor stage, microenvironment, and the applied anticancer treatment [2][3][4].
Under some conditions, autophagy has a cytoprotective function in cancer. Induction of autophagy in cancer cells resulting in their death can prevent the initiation of tumor growth [1]. Autophagy may also exert anticancer effects by increasing cytotoxicity of the chemotherapeutic agents [5][6][7]. Moreover, effects of autophagy on tumor inhibition or growth depend on the type of tumor [8][9][10]. Some types of cancer may undergo autophagic cell death (ACD) due to the use of some antineoplastic drugs [11]. The mechanism of ACD has been described in p53-deficient cancer cells [11]. Available data suggest that the role of autophagy also depends on different stages of tumor development [12]. An example of this is the increased expression of ATG7 as well as down-regulation of ATG5 and beclin-1 during apoptosis of colon cancer cells [12]. It was discovered that chloroquine and oxaliplatin interact synergistically on colon cancer cell lines, which confirmed the silencing of ATG5 through RNA interference, whereas the incubation of cells with beclin-1 resulted in the inhibition of autophagy [12].
It has been hypothesized that autophagy may also promote oncogenesis [5]. The primary stage of neoplastic tumor formation is often associated with hypoxia, resulting in metabolic stress [5]. The upregulation of autophagy under these conditions may provide a suitable environment for cancer cells to enter the dormant state. Thus, autophagy may play a pro-survival role by reducing metabolic stress and enabling proliferation of cancer cells [5].
As mentioned, autophagy may also protect tumors from cell death. Some advanced types of tumors are called “autophagous” tumors due to the increased activity of autophagy compared to normal tissues [12]. These tumors include activated Ras tumors and pancreatic cancer [10][11][12]. Moreover, the results clearly indicate that the use of chloroquine, which inhibits autophagy during treatment with bevacizumab, creates better conditions for tumor control and is associated with inhibition of autophagy [12].
Moreover, it has been suggested that autophagy supports survival of dormant tumor cells [13]. This leads to the theory that inhibition of autophagy may be an appropriate therapeutic strategy in the treatment of particular types of tumors [13]. For this aim, autophagy inhibitors which block autophagy at various stages have been used: Unc-51-like Kinase (ULK) inhibitors, Pan PI3Kinhibitors, VPS34 (PI3K) inhibitors, ATG inhibitors (i.e., NSC185058, tioconazol, UAMC-2526, LV320, FMK-9a), lysosomotropic agents, autophagy formation inhibitors (i.e., verteporfin, chloroquine, hydroxychloroquine), inhibitors of vacuolar H+-ATPase (bafilomycin A1), molecules blocking autophagosome-lysosome fusion, acid protease inhibitors, lysosome inhibitors (ionophores, i.e., tambjamines, monensin, and squaramides) and nanoparticles [13][14][15].
Cancer stem cells (CSCs) are pluripotent cells that were demonstrated in some types of tumors such as colorectal or breast cancer [16]. They are thought to initiate the development of such tumors being responsible for drug resistance, tumor growth, and recurrence due to their ability to renew themselves and differentiate into various types of tumor cells [17]. It was shown that the process of CMA autophagy (the self-eating process) is a leading factor in both resistance and survival of CSCs [17]. For this reason, it has been suggested that inhibition of autophagy in CSCs may help to overcome their resistance to treatment with anti-cancer drugs [17][18].
Importantly, due to the deregulation of the PI3K/Akt/mTOR signaling pathway in neoplastic cells, autophagy is constitutively activated, which allows for adaptation to the microenvironment and the activation of cell proliferation [19]. The data indicate that cancer therapies should primarily target tumor metabolism due to the induction of autophagy determined by metabolic symbiosis [19]. As a result of the regulation of cellular metabolism, the AMP-induced protein kinase (AMPK) is activated by cancer cells [19][20]. The mechanism of AMPK plays a key role in proliferation, maintains energy homeostasis by regulating cellular metabolism, and inhibits cell growth through phosphorylation by inhibiting the TOR pathway [20]. Moreover, the interaction is also directed towards the stabilization of p53 and p27, i.e., cell cycle inhibitors [20]. It is worth noting that AMPK induces autophagy by stimulating the activation of Ulk1 via phosphorylations of Ser 317 and Ser 777 in a glucose starvation setting [21]. Induction of autophagy as well as U1 regulation signaling has been found to occur through nutrient signaling [21].
2. Modulation of Autophagy in Cancer Cells by Polyphenols
Plant-derived polyphenols are divided into two groups, i.e., non-flavonoids and flavonoids (procyanidins) [22]. Both polyphenolic groups exhibit antitumor activity through modulation of non-canonical, i.e., beclin-1-independent, and canonical, beclin-dependent signaling pathways, as well as regulation of tumor suppressors [22]. The strong antioxidant effect of polyphenols has a significant impact on the modulation of autophagic pathways so that the cell can dispose of defective protein aggregates. Moreover, plant polyphenols were shown to induce the death of neoplastic cells in some model systems [22].
2.1. EGCG
Green tea (Camellia sinesis) polyphenols, and more specifically main catechin epigallocatechin-3-gallate (EGCG) (Figure 1), have been subjected to a number of studies suggesting induction of autophagy under cell stress conditions in some cancer cell lines [23][24][25]. EGCG was shown to induce autophagosome induction in human hepatoma (HepG2) cells as well as in bovine endothelial cells [24][25]. The cytoprotective mechanism of EGCG was also confirmed in the endoplasmic reticulum (ER) stress response in human embryonic kidney cell line (HEK293T, ATCC, and CRL-3216) [26][27][28]. The experiment revealed a disturbance of the balance of mTOR-AMPK pathways via GADD34 due to the ER stress [28]. Treatment of cells with EGCG, reduces the negative effect of GADD34 inhibition on the autophagy process [28]. The reduction of the negative impact of GADD34 inhibition in relation to autophagy was achieved by treatment with siGADD34 or with guanabenz [28]. In addition, the thapsigargin (TG) interferes with the accumulation of calcium in the ER [28]. HEK293T cells were treated with rapamycin and EGCG in the experiment. In the study, EGCG induced a delay in apoptotic cell death, also in the absence of GADD34 [28]. This is indicative of a shift in the balance of mTOR-AMPK pathways in the event of ER stress due to EGCG treatment to promote cell survival. The results indicate that EGCG activates autophagy via the mTOR-AMPK pathway, but ULK1 is essential in this process [28]. The research clearly indicates an important therapeutic role of EGCG, also in patients suffering from diseases associated with ER stress [28].
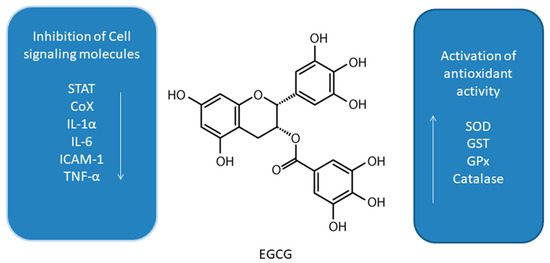
Figure 1. Effect of epigallocatechin-3-gallate (EGCG) on interplay between antioxidant activity and inhibition of cell signaling molecules [23][29][24][25][26][27][28][30][31][32][33][34][35][36][37][38].
EGCG has anti-tumor activity through the regulation of autophagy via effects on reactive oxygen species (ROS) formation in many cancers. In lymphoma, EGCG inhibited the growth of BC-1 and BABL-1 cells, significantly inducing autophagy [30]. Studies in primary exudative lymphoma (PEL), induced by expansion of cells infected with human herpesvirus 8 (HHV8), have shown induction of apoptosis and cell cycle arrest as a result of the treatment with EGCG [30]. The results showed that EGCG increased Bax expression and p53 activation, whereas previous studies showed chemical activation of p53 to induce cell growth inhibition in the PEL model [30]. The available data also indicate an intense induction of autophagy as well as apoptosis in primary glioblastoma cell cultures [30]. The mechanisms were observed after the incubation with 500 μM EGCG. The authors claim that the use of even 100 nM EGCG activates endogenous mechanisms of protection of primary glioblastoma cell cultures and may have a chemopreventive effect [30].
Green tea catechins play a significant role in supporting the treatment [31] of breast [32], pancreatic [33], and lung cancers [34]. For instance, NSCLC A549 cell line was tested to determine the effect of green tea extract on autophagy [34]. The study used colorimetric assay for assessing cell metabolic activity MTT assay, cell death analysis by annexin V/PI assay, transmission electron microscopy (TEM), acridine orange staining (AO), and immunofluorescent labeling of LC3-I [34]. The authors suggested that autophagy was caused by the induction of cytoprotective autophagy by flavonoids present in green tea extracts [34]. Thus, dietary polyphenols alone may not have a significant effect on the growth of neoplastic cells, but will prove useful in combination therapy with cytostatic drugs to increase the effectiveness of oncological therapy [34]. Activating mutations in the EGFR gene in both wild-type and the mutant forms T790M, L858R, and ELREA are often found in NSCLC patients [35]. Experimental studies were carried out with the use of EGCG and EGFR-TKI (erlotinib) [35]. Positive results of the experiment such as in clarifying the differences in the bonds of both wild-type EGFR and mutant types, indicate that the use of polyphenols in the diet may affect the growth of cancer cells, which at the same time is an important aspect as adjunctive therapy [35].
As previously mentioned, EGFR plays a very important role in NSCLC [36]. The main green tea polyphenol, EGCG, was shown to overcome resistance to EGFR tyrosine kinase inhibitors such as gefitinib in wild-type A549 cells and 16HBE cells—the human bronchial epithelial cell line [36]. The results of the study showed the inhibition of ERK phosphorylation by combining gefitinib and EGCG through a reduction of p-ERK and p-MEK [36].
A significant amount of research is required to confirm the significant role of EGCG in autophagy [37]. One of the studies showed that the combination of a proteasome inhibitor bortezomib with EGCG enhances prostate cancer cell death mediated by an increase in ER stress [37]. EGCG caused an increase in autophagy through antagonizing the toxicity of bortezomib [37]. Subsequently, osteosarcoma stem cells were also investigated to develop the basis of anti-cancer drugs based on the results obtained with EGCG and doxyrubicin [38]. The results showed that the use of EGCG significantly reduced the expression of osteosarcoma stem cell markers, and, in particular, SOX2OT V7, by reducing Notch3/DLL3 signaling [38].
2.2. Oleuropein
It was also shown that leaves of European olive (Olea europaea L.), rich in phenolic compounds such as verbascoside, apigenin-7-glucoside, luteolin-7-glucoside, and the main polyphenol—oleuropein (OL, Figure 2), also have antioxidant, anti-inflammatory, and anti-tumor activities [39]. These features are associated with affecting proliferation and apoptosis by modulating expression of many signaling pathways. OL is a phenolic compound, chemically consisting of benzene-1,2-diol (hydroxytyrosol), 4-2-dyroxyethyl (polyphenols), and elenolic acid (secoiroid) [39].
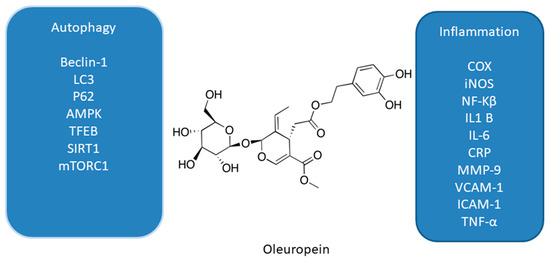
Figure 2. Effect of oleuropein on interplay between autophagy and inflammation [39][40][41][42][43][44][45][46].
The suggested anti-tumor mechanism of OL via the induction of autophagy has been recently confirmed in the estrogen receptor (ER)-positive MCF-7 and T47D breast cancer cell lines [40][41]. In addition to OL, hydroxytyrosol (HT) which is a metabolite of OL was also administered in the study [41]. MCF-7 and T47D cells were treated with OL and HT along with 3-methyladenine, which is an inhibitor of autophagy, with hepatocyte growth factor, or rapamycin, which is an agonist of autophagy [40]. The metastatic capacity and viability of the cells were assessed using the Western blot and transwell test. Depending on the dose, the viability of breast cancer cells decreased. Moreover, HT stronger than OL inhibited migration and invasion of triple-negative breast cancer cells by activating autophagy in the studied breast cancer cell lines [40]. Furthermore, it was showed that by suppressing autophagy, metastases of these cells can be inhibited [40].
OL was also used in a study determining the cellular and molecular activation mechanisms of autophagy [41][42][43][44]. One of the studies was conducted in an animal model and in cultured SH-SY5Y neuroblastoma cells. In the first study, the authors demonstrated a disruption of the autophagy cascade in SH-SY5Y cell line [41]. In a following work, cells were treated with 50 µM OL for 24 h and then assessed for beclin-1 and AMPK phosphorylation. OL increased AMPK phosphorylation, an early marker of autophagy [42]. Moreover, OL use leads to mTOR inhibition through the induction of AMPK in the cortex of TgCRND8 mice [42].
The induction of AMPK-dependent autophagy by OL was noted in an experiment in an animal model of a C57BL/6J mouse [45]. Male and female mice were fed a high-fat and normal diet for eight weeks. Increased expression of Beclin-1, LC3B, and p62/Sqstm1 genes was observed in the study. However, OL had no significant effect on the expression of the apoptotic proteins Bcl-2 and caspase 3 in this animal model [45].
The antiproliferative effect of OL and the induction of autophagy were confirmed in cultures of p-53 negative and androgen insensitive PC-3 human prostate cancer cells [46]. Moreover, the use of OL in the daily diet significantly reduced the dose of the cytotoxic doxorubicin—an anthracycline antibiotic with anti-cancer activity necessary to induce cell death [46], The results indicate a significant intensification of autophagy induction by OL, which was confirmed by the immunoblot analysis for LC3. However, it has to be stressed that the complex processes of autophagy induction due to OL use requires more research [46].
2.3. Pomegranate Extract and Punicalgin
Punicalgin (PUN, Figure 3) is a polyphenol obtained from pomegranate (Punica granatum). Depending on the form of antioxidants, it mainly contains anthocyanins, gallic acid, ellagic acid, ellagic tannins, and polyphenols. [47]. PUN is characterized by antioxidant activity and may inhibit lipoxygenases and cyclooxygenases. In addition, it has potential anti-cancer and chemopreventive effects. Both in vivo and in vitro studies confirm the effectiveness of PUN in the potential prevention of lung, breast, prostate, and colon cancers [45][46][48][49][50][51][52][53][54][55].
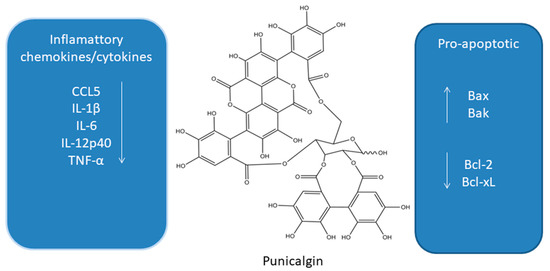
Figure 3. Influence of punicalgin on apoptosis and inflammatory cytokines [45][46][48][49][50][51][52][53][54][55][56][57][58].
The chemopreventive effect of pomegranate polyphenols may be based on the action of a number of proteins and genes, which affect both the growth and progression of cancer [46]. Interestingly, compared with the activity of red wine containing resveratrol and green tea infusion, the antioxidant capacity of pomegranate juice was three times higher [43].
Glioblastoma is one of the most common brain tumors. Unfortunately, the prognosis for this type of cancer remains poor, due to a poor response to medical treatment [57]. Studies indicate that PUN at a concentration of 1–30 μg/ml induced cell death in U87MG glioma cell cultures [57]. It was demonstrated that PUN induced apoptosis by activating the caspase-9/caspase-3 cascade and by cleaving ADP-ribose in the human glioma cell line. In addition, PUN increased autophagosome formation. Since PUN increased the phosphorylation of p27 T198 and of AMP, it was suggested that cell death mediated by PUN occurs via the apoptotic and autophagy pathways [57].
Pomegranate extract (PE) polyphenols show autophagy modulating effects in, inter alia, human syncytiotrophoblast and neuronal human neuroblastoma SY5Y cells [58]. PE activates the upregulation of both lysosomal and autophagosomal compartments, as well as causing autophagosome replacement and induction of autophagosome formation [58]. Importantly, TFEB activation is induced by the use of PE in a cytosolic Ca2+-dependent manner, but has no effect on ERK1/2, AKT and mTOR calcineurin signaling. Moreover, PE stimulated PINK1-Parksi mitophagy during of mitochondrial stress [58].
2.4. Apigenin—An Antioxidant from Acerola Fruit
Acerola (Malpighia glabra) is also known as the Barbados Cherry [59]. Its edible part are small red fruits with juicy flesh. Acerola is a rich source of vitamin C, beneficial to our health [59][51]. Acerola, apart from vitamin C and bioflavonoids, also contains pantothenic acid (vitamin B5), magnesium, potassium, thiamine, niacin, riboflavin, and vitamin A [59][60].
One of the major flavonoids contained in acerola is apigenin (AP, Figure 4) [48]. A number of studies, both in vitro and in vivo, confirmed the chemopreventive effect of AP [61], which occurred due to the inhibition of inflammation and angiogenesis, a significant delay in cell proliferation, a cellular response to oxidative stress, and the induction of apoptosis and autophagy [61].
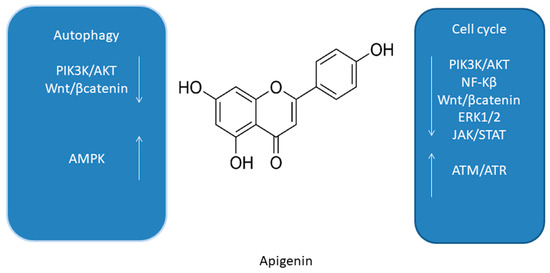
Figure 4. Summary of molecular cell cycle and autophagy signaling pathways of apigenin [48][53][61][62][63].
In one of the studies the influence of AP on the modulation of AMPK in human keratinocytes (HaCaT cell line and primary normal human epidermal keratinocytes) was investigated [62]. AP inhibited the mTOR signaling pathway by inducing autophagy via the activation of AMPK [53]. In addition, results of another study suggest that AP causes autophagy in C6 glioma cells [50]. Treatment of C6 cells resulted in reduced AMPK phosphorylation and, importantly, autophagy was AMPK/mTOR independent [63]. Importantly, it is worth emphasizing that the difference in experimental studies using C6 cells and glioma cells differs in particular in the resistance of HaCaT cells to apoptosis, which can be explained by mutations within the pro-apoptotic p53 protein alleles [50][63].
2.5. Resveratrol
Resveratrol (RSV) (trans-3,4′,5-trihydroxystilbene, Figure 5) is a naturally occurring polyphenol found in berries, peanuts, grapes, and wine [64][65][66]. It has attracted much attention due to the beneficial biological effects related to its anti-oxidant, anti-cancer, anti-inflammatory, anti-diabetic, and cardioprotective activities [67][68][69][70][71]. RSV has been shown to modulate autophagy in many cancer cell lines including leukemia, melanoma, glioma as well as renal, esophageal, liver, colon, prostate, breast, ovarian, oral, and lung cancer cell lines [72][73][74][75][76][77][78][79][80][81][82][83][84][85][86][87][88][89][90][91].
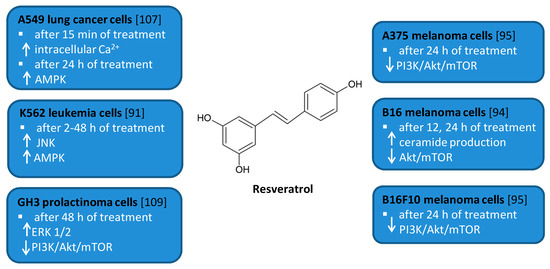
Figure 5. Cell signaling pathways triggered by resveratrol in selected cancer cell lines. ↑ activation/increase; ↓ inhibition/decrease.
Several in vitro studies indicated that RSV is able to induce autophagy that plays a pro-survival role and may serve as a resistance mechanism against apoptotic cell death [77][92]. For example, RSV triggered protective autophagy rough the ceramide accumulation and inhibition of Akt/mTOR pathway in B16 melanoma cells [77]. Inhibition of autophagy in B16 cells markedly increased RSV-induced apoptosis suggesting that RSV-induced autophagy plays a pro-survival role. In addition, the protective role of autophagy was demonstrated in resveratrol-treated U251 human glioma cells [79]. Similar results concerning the pro-survival role of RSV-induced autophagy were observed in GH3 cell line derived from rat pituitary tumor cells [92]. RSV led to the upregulation of ERK1/2 and the downregulation of PI3K/Akt and mTOR phosphorylation in prolactinoma GH3 cells. In some experimental models, RSV-induced autophagy contributed to the elimination of cancer cells [74][78][90]. RSV triggered autophagic cell death in imatinib-sensitive and imatinib-resistant K562 chronic myelogenous leukemia cells [74]. It led to the JNK-mediated p62/SQSTM1 expression and it activated AMPK. Thus, both JNK and AMPK pathways were involved in autophagic elimination in K562 cells. The activation of AMPK and JNK participated independently with the initiation and elongation steps of autophagy, respectively. RSV also suppressed the growth of A375 human melanoma cells and B16 F10 murine melanoma cells by promoting autophagy and inhibiting the PI3K/AKT/mTOR signaling pathway [78]. Moreover, it has also been found to induce autophagic cell death in A549 human lung adenocarcinoma cells via Ca2+/AMPK-mTOR signaling pathway [90].
References
- Dikic, I.; Elazar, Z. Mechanism and medical implications of mammalian autophagy. Nat. Rev. Mol. Cell Biol. 2018, 19, 349–364.
- Lim, J.; Murthy, A. Targeting Autophagy to Treat Cancer: Challenges and Opportunities. Front. Pharmacol. 2020, 11, 590344.
- Kimmelman, A.C. The dynamic nature of autophagy in cancer. Genes Dev. 2011, 25, 1999–2010.
- Janji, B.; Berchem, G.; Chouaib, S. Targeting Autophagy in the Tumor Microenvironment: New Challenges and Opportunities for Regulating Tumor Immunity. Front. Immunol. 2018, 9, 887.
- Liu, E.Y.; Ryan, K.M. Autophagy and cancer—Issues we need to digest. J. Cell Sci. 2012, 12, 2349–2358.
- Karantza-Wadsworth, V.; Patel, S.; Kravchuk, O.; Chen, G.; Mathew, R.; Jin, S.; White, E. Autophagy mitigates metabolic stress and genome damage in mammary tumorigenesis. Genes Dev. 2007, 21, 1621–1635.
- Jin, S.; White, E. Tumor suppression by autophagy through the management of metabolic stress. Autophagy 2008, 4, 563–566.
- Follo, C.; Cheng, Y.; Richards, W.G.; Bueno, R.; Broaddus, V.C. Autophagy facilitates the release of immunogenic signals following chemotherapy in 3D models of mesothelioma. Mol. Carcinog. 2019, 58, 1754–1769.
- Li, X.; He, S.; Ma, B. Autophagy and autophagy-related proteins in cancer. Mol. Cancer 2020, 19, 12.
- Shimgu, T.; Fujiwara, K.; Bogler, O.; Akiyama, Y.; Meritake, K.; Shinojima, N.; Tamacla, Y.; Yokoyama, T.; Kondo, S. Inhibition of autophagy at a late stage enhances imatinib-induced cytotoxicity In human malignant glioma cells. Int. J. Cancer. 2009, 124, 1060–1071.
- Liu, D.; Yang, Y.; Liu, Q.; Wang, J. Inhibition of autophagy by 3-MA potentiates cisplatin-induced apoptosis in esophageal squamous cell carcinoma cells. Med. Oncol. 2011, 28, 105–111.
- Selvakumaran, M.; Amaravadi, R.K.; Vasilevskaya, I.A.; O’Dwyer, P.J. Autophagy inhibition sensitizes colon cancer cells to antiangiogenic and cytotoxic therapy. Clin. Cancer Res. 2013, 19, 2995–3007.
- Pérez-Hernández, M.; Arias, A.; Martínez-García, D.; Pérez-Tomás, R.; Quesada, R.; Soto-Cerrato, V. Targeting Autophagy for Cancer Treatment and Tumor Chemosensitization. Cancers 2019, 11, 1599.
- Boya, P.; Casares, N.; Perfettini, J.; Dessen, P.; Larochette, N.; Métivier, D.; Meley, D.; Souquere, S.; Pierron, G.; Codogno, P.; et al. Inhibition of Macroautophagy Triggers Apoptosis. Mol. Cell. Biol. 2005, 25, 1025–1040.
- Lu, J.; Zhu, L.; Zheng, L.P.; Cui, Q.; Zhu, H.H.; Zhao, H.; Shen, Z.J.; Dong, H.Y.; Chen, S.S.; Wu, W.Z.; et al. Overexpression of ULK1 Represents a Potential Diagnostic Marker for Clear Cell Renal Carcinoma and the Antitumor Effects of SBI-0206965. EBioMedicine 2018, 34, 85–93.
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: the next generation. Cell 2011, 144, 646–674.
- Ojha, R.; Bhattacharyya, S.; Singh, S.K. Autophagy in Cancer Stem Cells: A Potential Link between Chemoresistance, Recurrence, and Metastasis. BioResearch Open Access 2015, 4, 97–108.
- Chavez-Dominguez, R.; Perez-Medina, M.; Lopez-Gonzalez, J.S.; Galicia-Velasco, M.; Aguilar-Cazares, D. The Double-Edge Sword of Autophagy in Cancer: From Tumor Suppression to Pro-tumor Activity. Front. Oncol. 2020, 10, 578418.
- Hardie, D. AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nat. Rev. Mol. Cell Biol. 2007, 8, 774–785.
- Yoshida, G.J. Therapeutic strategies of drug repositioning targeting autophagy to induce cancer cell death: from pathophysiology to treatment. J. Hematol. Oncol. 2017, 9, 10–67.
- Kim, J.; Kundu, M.; Viollet, B. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol. 2011, 13, 132–141.
- Kiruthiga, C.; Devi, K.P.; Nabavi, S.M.; Bishayee, A. Autophagy: A Potential Therapeutic Target of Polyphenols in Hepatocellular Carcinoma. Cancers 2020, 12, 562.
- Musial, C.; Kuban-Jankowska, A.; Gorska-Ponikowska, M. Beneficial Properties of Green Tea Catechins. Int. J. Mol. Sci. 2020, 21, 1744.
- Kapuy, O.; Vinod, P.K.; Bánhegyi, G. mTOR inhibition increases cell viability via autophagy induction during endoplasmic reticulum stress—an experimental and modeling study. FEBS OpenBio 2014, 4, 704–713.
- Karthikeyan, B.; Harini, L.; Krishnakumar, V.; Kannan, V.R.; Sundar, K.; Kathiresan, T. Insights on the involvement of (−)-epigallocatechin gallate in ER stress-mediated apoptosis in age-related macular degeneration. Apoptosis 2017, 22, 72–85.
- Lee, J.H.; Moon, J.H.; Kim, S.W. EGCG-mediated autophagy flux has a neuroprotection effect via a class III histone deacetylase in primary neuron cells. Oncotarget 2015, 6, 9701–9717.
- Holczer, M.; Marton, M.; Kurucz, A.; Banhegyi, G.; Kapuy, O. A comprehensive systems biological study of autophagy-apoptosis crosstalk during endoplasmic reticulum stress. BioMed Res. Int. 2015, 2015, 319589.
- Holczer, M.; Boglárka, B.; Veronika, Z.; Miklós, C.; Gábor, B.; Orsolya, K. Epigallocatechin-3-Gallate (EGCG) Promotes Autophagy-Dependent Survival via Influencing the Balance of mTOR-AMPK Pathways upon Endoplasmic Reticulum Stress. Oxidative Med. Cell. Longev. 2018, 2018, 6721530.
- Arranz, S.; Chiva-Blanch, G.; Valderas-Martínez, P.; Medina-Remón, A.; Lamuela-Raventós, R.M.; Estruch, R. Wine, beer, alcohol and polyphenols on cardiovascular disease and cancer. Nutrients 2012, 4, 759–781.
- Tsai, C.Y.; Chen, C.Y.; Chiou, Y.H. Epigallocatechin-3-gallate suppresses human herpesvirus 8 replication and induces ROS leading to apoptosis and autophagy in primary effusion lymphoma cells. Int. J. Mol. Sci. 2017, 19, 16.
- Grube, S.; Ewald, C.; Kögler, C. Achievable central nervous system concentrations of the green tea catechin EGCG induce stress in glioblastoma cells in vitro. Nutr. Cancer 2018, 70, 1145–1158.
- Abdal, D.A.; Choi, H.Y.; Yang, G.M.; Kim, K.; Saha, S.K.; Cho, S.G. The Anti-Cancer Effect of Polyphenols against Breast Cancer and Cancer Stem Cells: Molecular Mechanisms. Nutrients 2016, 8, 581.
- Zhou, Y.; Zheng, J.; Li, Y.; Xu, D.P.; Li, S.; Chen, Y.M.; Li, H.B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients 2016, 8, 515.
- Izdebska, M.; Klimaszewska-Wiśniewska, A.; Hałas, M.; Gagat, M.; Grzanka, A. Green tea extract induces protective autophagy in A549 non-small lung cancer cell line. Postepy Hig. Med. Dosw. 2015, 31, 1478–1484.
- Minnelli, C.; Laudadio, E.; Mobbili, G.; Galeazzi, R. Conformational Insight on WT- and Mutated-EGFR Receptor Activation and Inhibition by Epigallocatechin-3-Gallate: Over a Rational Basis for the Design of Selective Non-Small-Cell Lung Anticancer Agents. Int. J. Mol. Sci. 2020, 21, 1721.
- Meng, J.; Chang, C.; Chen, Y.; Bi, F.; Ji, C.; Liu, W. EGCG overcomes gefitinib resistance by inhibiting autophagy and augmenting cell death through targeting ERK phosphorylation in NSCLC. Onco Targets Ther. 2019, 12, 6033–6043.
- Modernelli, A.; Naponelli, V.; Giovanna Troglio, M. EGCG antagonizes Bortezomib cytotoxicity in prostate cancer cells by an autophagic mechanism. Sci. Rep. 2015, 5, 15270.
- Wang, W.; Chen, D.; Zhu, K. SOX2OT variant 7 contributes to the synergistic interaction between EGCG and Doxorubicin to kill osteosarcoma via autophagy and stemness inhibition. J. Exp. Clin. Cancer Res. 2018, 37, 37.
- Przychodzen, P.; Wyszkowska, R.; Gorzynik-Debicka, M.; Kostrzewa, T.; Kuban-Jankowska, A.; Gorska-Ponikowska, M. Anticancer Potential of Oleuropein, the Polyphenol of Olive Oil, With 2-Methoxyestradiol, Separately or in Combination, in Human Osteosarcoma Cells. Anticancer Res. 2019, 39, 1243–1251.
- Lu, H.Y.; Zhu, J.S.; Xie, J.; Zhang, Z.; Zhu, J.; Jiang, S.; Shen, W.J.; Wu, B.; Ding, T.; Wang, S.L. Hydroxytyrosol and Oleuropein Inhibit Migration and Invasion via Induction of Autophagy in ER-Positive Breast Cancer Cell Lines (MCF7 and T47D). Nutr. Cancer 2020, 14, 1–11.
- Imran, M.; Nadeem, M.; Gilani, S.A.; Khan, S.; Sajid, M.W.; Amir, R.M. Antitumor Perspectives of Oleuropein and Its Metabolite Hydroxytyrosol: Recent Updates. J. Food Sci. 2018, 83, 1781–1791.
- Grossi, C.; Rigacci, S.; Ambrosini, S.; Ed Dami, T.; Luccarini, I.; Traini, C.; Failli, P.; Berti, A.; Casamenti, F.; Stefani, M. The polyphenol oleuropein aglycone protects TgCRND8 mice against AΔ plaque pathology. PLoS ONE 2013, 8, 71702.
- Rigacci, S.; Miceli, C.; Nediani, C. Oleuropein aglycone induces autophagy via the AMPK/mTOR signalling pathway: A mechanistic insight. Oncotarget 2015, 6, 35344–35357.
- Porcu, C.; Sideri, S.; Martini, M.; Cocomazzi, A.; Galli, A.; Tarantino, G.; Balsano, C. Oleuropein Induces AMPK-Dependent Autophagy in NAFLD Mice, Regardless of the Gender. Int. J. Mol. Sci. 2018, 19, 3948.
- Stacchiotti, A.; Corsetti, G. Natural Compounds and Autophagy: Allies Against Neurodegeneration. Front Cell Dev. Biol. 2020, 22, 555409.
- Turrini, E.; Ferruzzi, L.; Fimognari, C. Potential Effects of Pomegranate Polyphenols in Cancer Prevention and Therapy. Oxidative Med. Cell. Longev. 2015, 2015, 938475.
- Yoshida, G.J. The interplay between apoptosis and ferroptosis mediated by ER stress. Apoptosis 2020, 25, 784–785.
- Mehta, R.; Lansky, E.P. Breast cancer chemopreventive properties of pomegranate (Punica granatum) fruit extracts in a mouse mammary organ culture. Eur. J. Cancer Prev. 2004, 13, 345–348.
- Aslam, M.N.; Lansky, E.P.; Varani, J. Pomegranate as a cosmeceutical source: Pomegranate fractions promote proliferation and procollagen synthesis and inhibit matrix metalloproteinase-1 production in human skin cells. J. Ethnopharmacol. 2006, 103, 311–318.
- Malik, A.; Afaq, F.; Sarfaraz, S.; Adhami, V.M.; Syed, D.N.; Mukhtar, H. Pomegranate fruit juice for chemoprevention and chemotherapy of prostate cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 14813–14818.
- Larrosa, M.; Tomás-Barberán, F.A.; Espín, J.C. The dietary hydrolysable tannin punicalagin releases ellagic acid that induces apoptosis in human colon adenocarcinoma Caco-2 cells by using the mitochondrial pathway. J. Nutr. Biochem. 2006, 17, 611–625.
- Khan, N.; Hadi, N.; Afaq, F.; Syed, D.N.; Kweon, M.H.; Mukhtar, H. Pomegranate fruit extract inhibits prosurvival pathways in human A549 lung carcinoma cells and tumor growth in athymic nude mice. Carcinogenesis 2007, 28, 163–173.
- Syed, D.N.; Malik, A.; Hadi, N.; Sarfaraz, S.; Afaq, F.; Mukhtar, H. Photochemopreventive effect of pomegranate fruil extract on UVA-mediated activation of cellular pathways in normal human epidermal keratinocytes. Photochem. Photobiol. 2006, 82, 398–405.
- Malik, A.; Mukhtar, H. Prostate cancer prevention through pomegranate fruit. Cell Cycle 2006, 5, 371–373.
- Seeram, N.P.; Adams, L.S.; Henning, S.M.; Niu, Y.; Zhang, Y.; Nair, M.G. In vitro antiproliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. J Nutr Biochem. 2005, 16, 360–367.
- Gil, M.I.; Tomas-Barberan, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589.
- Wang, S.G.; Huang, M.H.; Li, J.H.; Lai, F.I.; Lee, H.M.; Hsu, Y.N. Punicalagin induces apoptotic and autophagic cell death in human U87MG glioma cells. Acta Pharmacol. Sin. 2013, 34, 1411–1419.
- Tan, S.; Yu, C.Y.; Sim, Z.W.; Low, Z.S.; Lee, B.; See, F.; Min, N.; Gautam, A.; Chu, J.; Ng, K.W.; et al. Pomegranate activates TFEB to promote autophagy-lysosomal fitness and mitophagy. Sci. Rep. 2019, 9, 727.
- Xu, M.; Shen, C.; Zheng, H. Metabolomic analysis of acerola cherry (Malpighia emarginata) fruit during ripening development via UPLC-Q-TOF and contribution to the antioxidant activity. Food Res. Int. 2020, 130, 108915.
- Mezadri, T.; Villano, D.; Fernández-Pachón, M.S.; García-Parrilla, M.C.; Troncoso, A.M. Antioxidant compounds and antioxidant activity in acerola (Malpighia emarginata DC.) fruits and derivatives. J. Food Compost. Anal. 2008, 21, 282–290.
- Sung, B.; Chung, H.Y.; Kim, N.D. Role of Apigenin in Cancer Prevention via the Induction of Apoptosis and Autophagy. J. Cancer Prev. 2016, 21, 216–226.
- Tong, X.; Smith, K.A.; Pelling, J.C. Apigenin, a chemopreventive bioflavonoid, induces AMP-activated protein kinase activation in human keratinocytes. Mol. Carcinog. 2012, 51, 268–279.
- Jeremic, I.; Tadic, V.; Isakovic, A.; Trajkovic, V.; Markovic, I.; Redzic, Z. The mechanisms of in vitro cytotoxicity of mountain tea, Sideritis scardica, against the C6 glioma cell line. Planta Med. 2013, 79, 516–524.
- Rimando, A.M.; Kalt, W.; Magee, J.B.; Dewey, J.; Ballington, J.R. Resveratrol, pterostilbene, and piceatannol in vaccinium berries. J. Agric. Food Chem. 2004, 52, 4713–4719.
- Liu, C.D.; Wen, Y.Y.; Chiou, J.M.; Wang, K.H.; Chiou, R.Y. Comparative characterization of peanuts grown by aquatic floating cultivation and field cultivation for seed and resveratrol production. J. Agric. Food Chem. 2003, 51, 1582–1585.
- Wang, Y.; Catana, F.; Yang, Y.; Roderick, R.; van Breemen, R.B. An LC-MS method for analyzing total resveratrol in grape juice, cranberry juice, and in wine. J. Agric. Food Chem. 2002, 50, 431–435.
- Stojanovic, S.; Sprinz, H.; Brede, O. Efficiency and mechanism of the antioxidant action of trans-resveratrol and its analogues in the radical liposome oxidation. Arch. Biochem. Biophys. 2001, 391, 79–89.
- Szewczuk, L.M.; Forti, L.; Stivala, L.A.; Penning, T.M. Resveratrol is a peroxidase mediated inactivator of COX-1 but not COX-2: A mechanistic approach to the design of COX-1 selective agents. J. Biol. Chem. 2004, 279, 22727–22737.
- Cao, Z.; Li, Y. Potent induction of cellular antioxidants and phase 2 enzymes by resveratrol in cardiomyocytes: protection against oxidative and electrophilic injury. Eur. J. Pharmacol. 2004, 489, 39–48.
- Rivera, L.; Moron, R.; Zarzuelo, A.; Galisteo, M. Long-term resveratrol administration reduces metabolic disturbances and lowers blood pressure in obese Zucker rats. Biochem. Pharmacol. 2009, 77, 1053–1063.
- Jang, M. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997, 275, 218–220.
- Fan, Y.; Chiu, J.F.; Liu, J.; Deng, Y.; Xu, C.; Zhang, J.; Li, G. Resveratrol induces autophagy-dependent apoptosis in HL-60 cells. BMC Cancer 2018, 18, 581.
- Siedlecka-Kroplewska, K.; Wozniak, M.; Kmiec, Z. The wine polyphenol resveratrol modulates autophagy and induces apoptosis in MOLT-4 and HL-60 human leukemia cells. J. Physiol. Pharmacol. 2019, 70, 1–14.
- Puissant, A.; Robert, G.; Fenouille, N.; Luciano, F.; Cassuto, J.P.; Raynaud, S.; Auberger, P. Resveratrol promotes autophagic cell death in chronic myelogenous leukemia cells via JNK-mediated p62/SQSTM1 expression and AMPK activation. Cancer Res. 2010, 70, 1042–1052.
- Ge, J.; Liu, Y.; Li, Q.; Guo, X.; Gu, L.; Gui Ma, Z.; Zhu, Y.P. Resveratrol induces apoptosis and autophagy in T-cell acute lymphoblastic leukemia cells by inhibiting Akt/mTOR and activating p38-MAPK. Biomed. Environ. Sci. 2013, 26, 902–911.
- Yan, H.W.; Hu, W.X.; Zhang, J.Y.; Wang, Y.; Xia, K.; Peng, M.Y.; Liu, J. Resveratrol induces human K562 cell apoptosis, erythroid differentiation, and autophagy. Tumour Biol. 2014, 35, 5381–5388.
- Wang, M.; Yu, T.; Zhu, C.; Sun, H.; Qiu, Y.; Zhu, X.; Li, J. Resveratrol triggers protective autophagy through the ceramide/Akt/mTOR pathway in melanoma B16 cells. Nutr. Cancer 2014, 66, 435–440.
- Gong, C.; Xia, H. Resveratrol suppresses melanoma growth by promoting autophagy through inhibiting the PI3K/AKT/mTOR signaling pathway. Exp. Ther. Med. 2020, 19, 1878–1886.
- Li, J.; Qin, Z.; Liang, Z. The prosurvival role of autophagy in Resveratrol-induced cytotoxicity in human U251 glioma cells. BMC Cancer 2009, 9, 215.
- Liu, Q.; Fang, Q.; Ji, S.; Han, Z.; Cheng, W.; Zhang, H. Resveratrol-mediated apoptosis in renal cell carcinoma via the p53/AMP activated protein kinase/mammalian target of rapamycin autophagy signaling pathway. Mol. Med. Rep. 2018, 17, 502–508.
- Tang, Q.; Li, G.; Wei, X.; Zhang, J.; Chiu, J.F.; Hasenmayer, D.; Zhang, D.; Zhang, H. Resveratrol-induced apoptosis is enhanced by inhibition of autophagy in esophageal squamous cell carcinoma. Cancer Lett. 2013, 336, 325–337.
- Zhang, B.; Yin, X.; Sui, S. Resveratrol inhibited the progression of human hepatocellular carcinoma by inducing autophagy via regulateng p53 and the phosphoinositide 3 kinase/protein kinase B pathway. Oncol. Rep. 2018, 40, 2758–2765.
- Miki, H.; Uehara, N.; Kimura, A.; Sasaki, T.; Yuri, T.; Yoshizawa, K.; Tsubura, A. Resveratrol induces apoptosis via ROS-triggered autophagy in human colon cancer cells. Int. J. Oncol. 2012, 40, 1020–1028.
- Li, G.; Rivas, P.; Bedolla, R.; Thapa, D.; Reddick, R.L.; Ghosh, R.; Kumar, A.P. Dietary resveratrol prevents development of high-grade prostatic intraepithelial neoplastic lesions: involvement of SIRT1/S6K axis. Cancer Prev. Res. (Phila) 2013, 6, 27–39.
- Selvaraj, S.; Sun, Y.; Sukumaran, P.; Singh, B.B. Resveratrol activates autophagic cell death in prostate cancer cells via downregulation of STIM1 and the mTOR pathway. Mol. Carcinog. 2016, 55, 818–831.
- Scarlatti, F.; Maffei, R.; Beau, I.; Codogno, P.; Ghidoni, R. Role of non-canonical Beclin 1-independent autophagy in cell death induced by resveratrol in human breast cancer cells. Cell Death Differ. 2008, 15, 1318–1329.
- Opipari, A.W., Jr.; Tan, L.; Boitano, A.E.; Sorenson, D.R.; Aurora, A.; Liu, J.R. Resveratrol-induced autophagocytosis in ovarian cancer cells. Cancer Res. 2004, 64, 696–703.
- Lang, F.; Qin, Z.; Li, F.; Zhang, H.; Fang, Z.; Hao, E. Apoptotic Cell Death Induced by Resveratrol Is Partially Mediated by the Autophagy Pathway in Human Ovarian Cancer Cells. PLoS ONE 2015, 10, 0129196.
- Chang, C.H.; Lee, C.Y.; Lu, C.C.; Tsai, F.J.; Hsu, Y.M.; Tsao, J.W.; Juan, Y.N.; Chiu, H.Y.; Yang, J.S.; Wang, C.C. Resveratrol-induced autophagy and apoptosis in cisplatin-resistant human oral cancer CAR cells: A key role of AMPK and Akt/mTOR signaling. Int. J. Oncol. 2017, 50, 873–882.
- Zhang, J.; Chiu, J.; Zhang, H.; Qi, T.; Tang, Q.; Ma, K.; Lu, H.; Li, G. Autophagic cell death induced by resveratrol depends on the Ca(2+)/AMPK/mTOR pathway in A549 cells. Biochem. Pharmacol. 2013, 86, 317–328.
- Wang, J.; Li, J.; Cao, N.; Li, Z.; Han, J.; Li, L. Resveratrol, an activator of SIRT1, induces protective autophagy in non-small-cell lung cancer via inhibiting Akt/mTOR and activating p38-MAPK. Onco Targets Ther. 2018, 11, 7777–7786.
- Zhang, X.; Xu, W.; Su, J.; Chu, M.; Jin, H.; Li, G.; Tan, C.; Wang, X.; Wang, C. The prosurvival role of autophagy in resveratrol-induced cytotoxicity in GH3 cells. Int. J. Mol. Med. 2014, 33, 987–993.




