Video Upload Options
Perylene imide (PI) molecules and materials have been extensively studied for optical chemical sensors, particularly those based on fluorescence and colorimetric mode, taking advantage of the unique features of PIs such as structure tunability, good thermal, optical and chemical stability, strong electron affinity, strong visible light absorption and high fluorescence quantum yield. PIs-based optical chemosensors have now found broad applications in gas phase detection of chemicals, including explosives, biomarkers of some food and diseases (such as organic amines (alkylamines and aromatic amines)), benzene homologs, organic peroxides, phenols and nitroaromatics, etc.
1. Introduction
Developing chemosensor techniques for trace-level detection of vapor analytes, especially volatile organic compounds (VOCs), and nitro explosives, which are of various health and security concern, has drawn increasing research interest and efforts in the past decades [1][2][3]. As we know, conventional analytical instrumentations such as gas chromatography (GC), mass spectrometry (MS), infrared spectroscopy (IR), high-performance liquid chromatography (HPLC) [4][5][6][7][8], as well as some sensor methods such as surface acoustic wave (SAW) sensors [9], quartz crystal microbalances [10] and electrochemical resistors [11] are normally managed in lab detection by professional and technical staff due to their expensive cost, bulk size or complicated and time-consuming operation. In contrast, chemosensors such as electrochemical sensors, field-effect transistor sensors, chemiresistive sensors, fluorometric and colorimetric sensors exhibit superiority regarding system simplicity for facile operation, and non-destructive detection with high sensitivity and selectivity [12][13][14]. Among these chemosensing methods, optical sensors (including fluorometric and colorimetric types) are highly attractive for chemical vapor detection not only for real-time operation, compact size, immunity to electromagnetic interferences and remote sensing capabilities, but also for simple readout of output signals (sometimes just by naked eyes). These vapor chemosensors are usually fabricated from fluorophores or chromophores as active sensing materials (also known as probes) in the form of solid films, relying on the rational design of the molecular structures. Compared to those molecule or particle-based sensors in solution-phase detection, these sensing films are usually prepared by immobilization of fluorescent or colorimetric sensors (in molecule, particle or fibril form) on suitable solid substrates, providing advantages in terms of their reusability and reproducibility, which are two crucial factors for practical applications and actual device development of chemosensors [14][15][16].
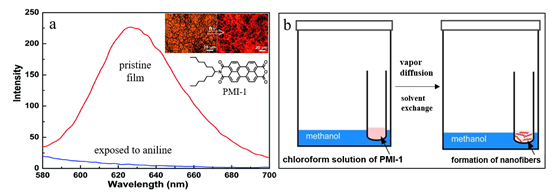
Among the widely diverse optical sensor materials studied thus far, perylene imides (PIs) have attained significant attention in the past decades, because PIs are exceptional n-type organic materials with excellent electron affinity, strong fluorescence (high quantum yield), strong visible light absorption, high thermal stability and photostability under ambient conditions. Indeed, the combination of these unique features makes PIs ideal materials for development in various electronic and optoelectronic devices including solar cells, light-emitting diodes, photoelectric sensors and chemosensors [17][18]. For example, PIs-based nanofiber films have been extensively studied by our and others’ groups for sensing a wide range of reducing gaseous species (i.e., electron-donating), such as ammonia (NH3) and volatile organic amines, by measuring the change of fluorescence intensity upon exposure to the analyte vapor [17][19]. The key of PIs-based chemosensors for successful vapor detection is introducing suitable functional groups on the imide or bay position to enable strong specific binding towards target analytes, while still maintaining the active fluorescence response and certain solubility for solution processing [17][20]. The two kinds of PIs widely used in the field of chemosensors are perylene diimides (PDIs) and perylene monoimides (PMIs), with most research focused on PDIs mainly due to their facile synthesis and good solution processability. However, PMIs usually retain the strong fluorescence upon assembly (aggregation) into solid state, while the solid state of PDIs is normally very weak in fluorescence mainly due to the p-p stacking quenching. In view of the primary requirement of sufficient fluorescence for sensor application (particularly those based on fluorescence quenching mode), PMIs provide some unique opportunities for sensor development, complementary to PDIs. Specifically, the anhydride group at one side of PMIs is intrinsically a strong and specific binder to amines through hydrogen bonding in concert with donor-acceptor interaction [17]. In 2008, our group reported on a porous fluorescent film chemosensor for the vapor detection of aniline; the film was composed of intertwined nanofibers assembled from a PMI molecule (PMI-1 shown in Figure 1) [21]. The successful fabrication of shape-defined, highly crystalline nanofibers was mainly driven by the one-dimensional (1D) intermolecular arrangement, while the tilted p-p stacking still allows for sufficient fluorescence in the solid state. Upon surface adsorption of amines (electron donor) like aniline, the fluorescence of PMIs gets efferently quenched through photoinduced electron transfer (PET). Such fluorescence quenching based sensors have proven highly sensitive for vapor detection of amines with limit of detection down to ppb level for aniline, taking advantage mostly of the unique nano-porosity of the nanofiber film, wherein the intertwined nanofibers form a porous “super net” structure with a large, open surface area that is conducive to both molecular diffusion and adsorption. The innovative materials design, along with specific nanoscale engineering, opened a new, promising direction for the development of solid-state fluorescent chemosensors [21], as evidenced from many follow-up studies in developing various PIs-based fluorescence sensors for chemical vapor detection [22][23][24][25]. Table 1 lists some representative PDIs and PMIs that have been developed and proven effective chemosensors for vapor detection in recent years.
Figure 1. (a) Fluorescence spectra of PMI-1 nanofibril film before (red) and after (blue) exposure to the saturated vapor of aniline (880 ppm) for 10 s. Inset shows bright field and fluorescence optical microscopy image of the film; (b) A diagram showing the procedure of fabrication of the nanofibers via slow solvent vapor transfer.
So far, some reviews have been published by our group and others on the solid phase PDI-based chemosensors, which are capable of both gas and solution phase detection. For example, Fu, Y reported on a review mainly focused on the detection of organic amines, and the associated sensing mechanism and the design principles of probes. Fang’s group [26] summarized the sensory materials and device fabrication. Our group recently published a review on the recent advance in the development of PDI-based optical sensors for environmental detection of heavy metal ions, inorganic anions, amines and other organic pollutants in aqueous and other liquid phase solutions. The PDI sensors covered in the previous review are mostly based on molecular state and the sensing response is caused by molecular binding or reactions in liquid solutions, such as ion coordination or complexation, protonation/deprotonation or other chemical reactions. In sharp contrast, the sensors covered in this review are based on solid aggregate of PDIs and work for gas phase detection of chemicals. We also published another review specifically focused on the nanoscale structural control of PDIs nanofibril interfacial heterojunctions via molecular self-assembly, with the aim to optimize the chemiresistive sensing performance so as to achieve trace level chemical vapor detection [27]. Meanwhile, Kumar, K. and co-workers [28] published a specific review on the aesthetic design strategies and sensing mechanism of PDI-based sensors. However, the field still lacks a comprehensive review on optical chemosensors covering both PDIs and PMIs-based sensor materials, which combined would enable much broader detection of chemical vapors beyond the common species like amines. Moreover, multimodal sensors and array systems involving multiple sensor components have not been adequately reviewed for PDIs or PMIs sensors, though the multiplex sensor systems have recently drawn increasing attention in chemosensors, which help enable or enhance the detection capability for complex or mixture samples with the assistance of algorithm data analytics like machine learning.
2. Physicochemical and Optical Properties of PIs
Bulk-phase PIs have been widely used in various optoelectronics devices [29][30][31], including thin-film transistors [32][33], photovoltaics [34][35], liquid crystals [36][37] and chemiresistive sensors [38], etc. Performance of optical chemosensors (as well the devices fabricated therefrom) depends primarily on the physicochemical and optical properties of the sensing materials, here specifically the fluorophores or chromophores, which in turn are tightly correlated with the molecular design and structure engineering [39]. For PIs-based materials, there are three unique features that render their excellent applications in optical chemosensors (especially for vapor detection). Firstly, the PI molecule possesses a large, planar and rigid π-conjugation system based on the central perylene skeleton, which is of π-electron deficient aromatic nature, making it a strong electron acceptor (n-type) with good environmental chemical stability. Due to the highly efficient p-p transition, PIs exhibit strong visible light absorption and fluorescence (close to unit quantum yield in good organic solvents), well suited for application in optical sensors based on fluorescence or colorimetric modulation . Secondly, molecules of PIs are flexible for structural modification through facile organic synthesis by changing the substitution at the imide position or bay area, which in turn can tune the solubility, spectroscopic and redox properties, as well as the self-assembly of PIs. Moreover, the imide (nitrogen) position on PI is a node in the π-orbital molecular wavefunction, meaning that modification with different substitutions at the position will not change the electronic (light absorption and fluorescence) properties. This provides a number of viable pathways for controlling the molecular structure of PIs so as to achieve the best sensor performance. Nonetheless, the bay-area substitution usually brings significant changes of the optical and electronic properties of PIs due to the direct interruption to the π-conjugation [40]. Lastly, PIs are one of the most studied classes of n-type semiconductor molecules for self-assembly that form structure and morphology defined nanomaterials. Particularly, owing to the planar rigid π-conjugation geometry, PIs favor one-dimensional assembly, forming shape-defined nanofibers driven by the strong π-π stacking between perylene planes. There are enormous options for the one-dimensional molecular assembly to be optimized for both structure and morphology through changing the side substitutions at the imide or bay positions. The co-facial π-π stacking (in the format of H-aggregate) results in fluorescence quenching and a hypsochromic shift of the absorption [41]. While the nanofibers of PIs have been extensively studied as optical and chemiresistive sensors, the aggregation-disaggregation induced fluorescence change of PIs have also been developed into fluorescence sensors for detecting inorganic or organic ions in liquid-phase environment. However, for vapor analytes sensing, PIs have to be fabricated in the form of films or other formats of solid substrate, for which it is imperative to retain sufficient fluorescence of PIs in solid state (i.e., minimizing the fluorescence quenching caused by π-π stacking). In most cases, significantly increased fluorescence can be achieved by introducing large bulky moieties at either the imide position or the bay area; bulky moieties provide steric hindrance, thus tilting the co-facial π-π stacking, which in turn weakens the fluorescence quenching effect [42]. In addition, it has been demonstrated by our group and others that the construction and performances of PIs-based optical chemosensors are closely related to the different dimensions and morphologies of PIs assemblies [43][44][45][46]. For example, porous films with 1D PIs nanofibers showed significantly enhanced sensing performances in comparison to those with irregular morphologies. As mentioned above, relying on the good tradeoff between ordered molecular assembly and retaining strong fluorescence in solid state, our group initially explored the PMI-based fluorescent nanofiber sensor for trace vapor detection of organic amines. Following the initial work, Che and co-workers [47] have successfully fabricated thermally stable bilayer nanocoils based on an asymmetric PDI molecule, which demonstrate great sensor performance in detecting amines like aniline and phenethylamine.
Combination of the excellent physicochemical and optical properties mentioned above makes PIs an ideal fluorophore and chromophore materials for being developed as efficient chemosensors that are capable of vapor defection, targeting a wide range of environmental pollutants, explosives and biomarkers of some food and diseases, including organic amines, BTEX (benzene, toluene, ethylbenzene, o-xylene, m-xylene and p-xylene), organic peroxides, phenols, nitroaromatics, etc. Beside molecule design and synthesis, many efforts have been laid on the fabrication (and nanoscale engineering) of optically active thin (often porous) films from PIs. The sensor performance against environmental factors such as temperature, humidity and concentration, and in correlation with the thermodynamic parameters of analytes like boiling point, has also been studied in detail, aiming to further improve the robustness and sustainability of sensor system as desired in practical use. The environmental factors may also affect the intermolecular association of PIs aggregates, thus causing change in sensing performance [48][49].
3. Fabrication of PIs-Based Optical Chemosensors
Optical sensors fabricated from PIs are mainly developed in the aspects of fluorescent film construction, array technology and sensing platform. There are many methods for constructing thin-film based fluorescent sensors as described in this review, such as physical coating technology, Langmuir-Blodgett (LB) technology [50] and molecular gel technology [51]. Among them, physical coating technology, including spin coating, dip coating and spraying, is the most commonly used method for constructing fluorescent thin film due to its easy operation. However, this method requires strict instrumentation control in order to obtain high-quality films. Conversely, LB technology is relatively easy to operate and control, and the films obtained usually have highly organized structure, and to some extent, such structure can also be controlled or modified upon various needs. Molecular gels, on the other hand, mainly form three-dimensional (3D) network structure through interactions between molecules such as Van der Waals force, hydrogen bonding, π-π stacking, solvophobic vs. solvophilic interactions and so on [52]. Formation of molecular gels normally exhibits stimulus phase change (shear stimulation and temperature change stimulation); therefore, the gels can be considered as physical gels, involving small molecules as gelling agents.
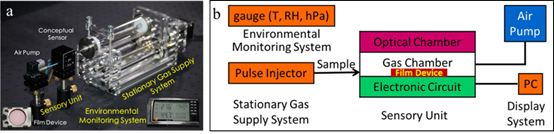
As for a complete sensor system or testing platform, it is typically composed of a sensor unit (material), a sampling part including gas (vapor) supply under precise control of flow rate, a signal readout device like photodiode for measuring the light intensity and an environmental chamber holding all the parts in a closed space where temperature, humidity and other experimental conditions can be controlled (Figure 2a,b). The gas supply system is designed to work in a stationary manner to adjust sample volume or flow rate so as to control the vapor concentration mixed in ambient or zero air. The environmental chamber is usually required and critical for assessing the sensor performance against real world ambient conditions, with varying temperature and humidity, as well as presence of other interference chemicals or gases. While the sensor unit is often based on a single sensor material or sensing mode, nowadays the array-based sensing approach has become more popular and attracted increasing interest, particularly because the recent advancement in electronic circuit and on-chip data processing makes the incorporation of multiple sensors on a chip to be more feasible [53]. An array with multiple sensors or sensing modes integrated can not only selectively identify a single analyte, but also enable or enhance the capability of differentiating multiple analytes in a mixed sample, which are actually more common in real world detections. For example, a colorimetric sensor array has recently been reported that effectively discriminates ten explosives [54]. Particularly for colorimetric or fluorescence sensing mode, varying materials and molecules (e.g., dyes, nanomaterials and polymers) have been developed and incorporated into an array to target a broad range of chemical vapor analytes [55][56]. Taking advantage of the specific, strong chemical reactions (not just physical interaction) between the sensor and analytes, these sensors in an array can provide unprecedented sensitivity and selectivity in detection of different analytes within complexed or mixed samples.
Figure 2. (a) The picture of the gaseous analyte detection platform based on the film device; (b) Schematic description of the home-made sensing platform.
References
- Elke, K.; Jermann, E.; Begerow, J.; Dunemann, L. Determination of benzene, toluene, ethylbenzene and xylenes in indoor air at environmental levels using diffusive samplers in combination with headspace solid-phase microextraction and high-resolution gas chromatography–flame ionization detection. J. Chromatogr. A 1998, 826, 191–200, doi:10.1016/s0021-9673(98)00736-5.
- Caygill, J.S.; Davis, F.; Higson, S.P. Current trends in explosive detection techniques. Talanta 2012, 88, 14–29, doi:10.1016/j.talanta.2011.11.043.
- Toal, S.J.; Trogler, W.C. Polymer sensors for nitroaromatic explosives detection. J. Mater. Chem. 2006, 16, 2871–2883, doi:10.1039/b517953j.
- Lee, J.H.; Hwang, S.M.; Lee, D.W.; Heo, G.S. Determination of Volatile Organic Compounds (VOCs) Using Tedlar Bag/Solid-phase Microextraction/Gas Chromatography/Mass Spectrometry (SPME/GC/MS) in Ambient and Workplace Air. Bull. Korean Chem. Soc. 2002, 23, 488–496, doi:10.5012/bkcs.2002.23.3.488.
- Guan, X.; Zhao, Z.; Cai, S.; Wang, S.; Lu, H. Analysis of volatile organic compounds using cryogen-free thermal modulation based comprehensive two-dimensional gas chromatography coupled with quadrupole mass spectrometry. J. Chromatogr. A 2019, 1587, 227–238, doi:10.1016/j.chroma.2018.12.025.
- Mirzaei, A.; Leonardi, S.; Neri, G. Detection of hazardous volatile organic compounds (VOCs) by metal oxide nanostructures-based gas sensors: A review. Ceram. Int. 2016, 42, 15119–15141, doi:10.1016/j.ceramint.2016.06.145.
- Prebihalo, S.E.; Berrier, K.L.; Freye, C.E.; Bahaghighat, H.D.; Moore, N.R.; Pinkerton, D.K.; Synovec, R.E. Multidimensional Gas Chromatography: Advances in Instrumentation, Chemometrics, and Applications. Anal. Chem. 2018, 90, 505–532, doi:10.1021/acs.analchem.7b04226.
- Al-Rubaye, A.F.; Hameed, I.H.; Kadhim, M.J. A Review: Uses of Gas Chromatography-Mass Spectrometry (GC-MS) Technique for Analysis of Bioactive Natural Compounds of Some Plants. Int. J. Toxicol. Pharmacol. Res. 2017, 9, 81–85, doi:10.25258/ijtpr.v9i01.9042.
- Sayago, I.; Fernández, M.; Fontecha, J.; Horrillo, M.C.; Vera, C.; Obieta, I.; Bustero, I. New sensitive layers for surface acoustic wave gas sensors based on polymer and carbon nanotube composites. Sens. Actuators B: Chem. 2012, 175, 67–72, doi:10.1016/j.snb.2011.12.031.
- Khot, L.R.; Panigrahi, S.; Lin, D. Development and evaluation of piezoelectric-polymer thin film sensors for low concentration detection of volatile organic compounds related to food safety applications. Sens. Actuators B Chem. 2011, 153, 1–10, doi:10.1016/j.snb.2010.05.043.
- Lipatov, A.; Varezhnikov, A.; Wilson, P.; Sysoev, V.; Kolmakov, A.; Sinitskii, A. Highly selective gas sensor arrays based on thermally reduced graphene oxide. Nanoscale 2013, 5, 5426–5434, doi:10.1039/c3nr00747b.
- Massie, C.; Stewart, G.; McGregor, G.; Gilchrist, J.R. Design of a portable optical sensor for methane gas detection. Sens. Actuators B Chem. 2006, 113, 830–836, doi:10.1016/j.snb.2005.03.105.
- Wu, N.; Wang, C.; Bunes, B.R.; Zhang, Y.; Slattum, P.M.; Yang, X.; Zang, L. Chemical Self-Doping of Organic Nanoribbons for High Conductivity and Potential Application as Chemiresistive Sensor. ACS Appl. Mater. Interfaces 2016, 8, 12360–12368, doi:10.1021/acsami.6b03151.
- Azzouz, A.; Vikrant, K.; Kim, K.-H.; Ballesteros, E.; Rhadfi, T.; Malik, A.K. Advances in colorimetric and optical sensing for gaseous volatile organic compounds. TrAC Trends Anal. Chem. 2019, 118, 502–516, doi:10.1016/j.trac.2019.06.017.
- García, J.M.; García, F.C.; Serna, F.; De La Peña, J.L. Fluorogenic and Chromogenic Polymer Chemosensors. Polym. Rev. 2011, 51, 341–390, doi:10.1080/15583724.2011.616084.
- Liu, L.; Li, Z.; Liu, Y.; Zhang, S.X.-A. Recent advances in film-based fluorescence sensing. Sci. Sin. Chim. 2019, 50, 39–69, doi:10.1360/ssc-2019-0077.
- Chen, S.; Slattum, P.; Wang, C.; Zang, L. Self-Assembly of Perylene Imide Molecules into 1D Nanostructures: Methods, Morphologies, and Applications. Chem. Rev. 2015, 115, 11967–11998, doi:10.1021/acs.chemrev.5b00312.
- Díez-Pérez, I.; Li, Z.; Guo, S.; Madden, C.; Huang, H.; Che, Y.; Yang, X.; Zang, L.; Tao, N. Ambipolar Transport in an Electrochemically Gated Single-Molecule Field-Effect Transistor. ACS Nano 2012, 6, 7044–7052, doi:10.1021/nn302090t.
- Chen, S.; Xue, Z.; Gao, N.; Yang, X.; Zang, L. Perylene Diimide-Based Fluorescent and Colorimetric Sensors for Environmental Detection. Sensors 2020, 20, 917, doi:10.3390/s20030917.
- Gao, R.; Cao, D.; Guan, Y.; Yan, D. Flexible Self-Supporting Nanofibers Thin Films Showing Reversible Photochromic Fluorescence. ACS Appl. Mater. Interfaces 2015, 7, 9904–9910, doi:10.1021/acsami.5b01996.
- Che, Y.; Yang, X.; Loser, S.; Zang, L. Expedient Vapor Probing of Organic Amines Using Fluorescent Nanofibers Fabricated from an n-Type Organic Semiconductor. Nano Lett. 2008, 8, 2219–2223, doi:10.1021/nl080761g.
- Fu, Y.; Xu, W.; He, Q.; Cheng, J. Recent progress in thin film fluorescent probe for organic amine vapour. Sci. China Ser. B Chem. 2015, 59, 3–15, doi:10.1007/s11426-015-5498-3.
- Chen, S.; Yu, J.; Zang, L. Fluorescent chemosensor based on organic nanofibril thin-films. Sci. Sin. Chim. 2019, 50, 78–91, doi:10.1360/ssc-2019-0110.
- Wang, Z.; Liu, K.; Chang, X.; Qi, Y.; Shang, C.; Liu, T.; Liu, J.; Ding, L.; Fang, Y. Highly Sensitive and Discriminative Detection of BTEX in the Vapor Phase: A Film-Based Fluorescent Approach. ACS Appl. Mater. Interfaces 2018, 10, 35647–35655, doi:10.1021/acsami.8b13747.
- Shang, C.; Wang, L.; An, Y.; Chen, P.; Chang, X.; Qi, Y.; Kang, R.; Fang, Y. Langmuir–Blodgett films of perylene bisimide derivatives and fluorescent recognition of diamines. Phys. Chem. Chem. Phys. 2017, 19, 23898–23904, doi:10.1039/c7cp04235c.
- Liu, Q.; Liu, T.; Fang, Y. Perylene Bisimide Derivative-Based Fluorescent Film Sensors: From Sensory Materials to Device Fabrication. Langmuir 2020, 36, 2155–2169, doi:10.1021/acs.langmuir.9b03919.
- Chen, S.; Gao, N.; Bunes, B.R.; Zang, L. Tunable nanofibril heterojunctions for controlling interfacial charge transfer in chemiresistive gas sensors. J. Mater. Chem. C 2019, 7, 13709–13735, doi:10.1039/c9tc04659c.
- Singh, P.; Sharma, P.; Kaur, N.; Mittal, L.; Kumar, K. Perylene diimides: Will they flourish as reaction-based probes? Anal. Methods 2020, 12, 3560–3574.
- Würthner, F. Perylene bisimide dyes as versatile building blocks for functional supramolecular architectures. Chem. Commun. 2004, 14, 1564–1579.
- Newman, C.R.; Frisbie, C.D.; da Silva Filho, D.A.; Brédas, J.-L.; Ewbank, P.C.; Mann, K.R. Introduction to Organic Thin Film Transistors and Design of n-Channel Organic Semiconductors. Chem. Mater. 2004, 16, 4436–4451, doi:10.1021/cm049391x.
- Law, K.Y. Organic photoconductive materials: Recent trends and developments. Chem. Rev. 1993, 93, 449–486, doi:10.1021/cr00017a020.
- Jones, B.A.; Ahrens, M.J.; Yoon, M.-H.; Facchetti, A.; Marks, T.J.; Wasielewski, M.R. High-mobility air-stable n-type semiconductors with processing versatility: dicyanoperylene-3,4:9,10-bis(dicarboximides). Angew. Chem. Int. Ed. Engl. 2004, 43, 6363–6366.
- Chesterfield, R.J.; McKeen, J.C.; Newman, C.R.; Ewbank, P.C.; da Silva Filho, D.A.; Brédas, J.-L.; Miller, L.L.; Mann, K.R.; Frisbie, C.D. Organic Thin Film Transistors Based onN-Alkyl Perylene Diimides: Charge Transport Kinetics as a Function of Gate Voltage and Temperature. J. Phys. Chem. B 2004, 108, 19281–19292, doi:10.1021/jp046246y.
- Schmidt-Mende, L.; Fechtenkötter, A.; Müllen, K.; Moons, E.; Friend, R.H.; MacKenzie, J.D. Self-Organized Discotic Liquid Crystals for High-Efficiency Organic Photovoltaics. Science 2001, 293, 1119–1122, doi:10.1126/science.293.5532.1119.
- Kitazume, K.; Chen, Y.; Itou, M.; Araki, Y.; Uchida, S.; Ito, O. Photoinduced electron-transfer processes between phthalocyanines and perylene derivatives. J. Porphyrins Phthalocyanines 2006, 10, 1190–1196, doi:10.1142/s1088424606000557.
- Struijk, W.; Sieval, B.; Dakhorst, J.; Van Dijk, M.; Kimkes, P.; Koehorst, M.; Donker, H.; Schaafsma, J.; Picken, J.; Van de Craats, M.; et al. Liquid crystalline perylene diimides: Architecture and charge carrier mobilities. J. Am. Chem. Soc. 2000, 122, 11057–11066.
- Rohr, U.; Schilichting, P.; Bohm, A.; Gross, M.; Meerholz, K.; Brauchle, C.; Mullen, K. Liquid crystalline coronene derivatives with extraordinary fluorescence properties. Angew. Chem. Int. Ed. Engl. 1998, 37, 1434–1437.
- Huang, H.; Chou, C.-E.; Che, Y.; Li, L.; Wang, C.; Yang, X.; Peng, Z.; Zang, L. Morphology Control of Nanofibril Donor–Acceptor Heterojunction To Achieve High Photoconductivity: Exploration of New Molecular Design Rule. J. Am. Chem. Soc. 2013, 135, 16490–16496, doi:10.1021/ja407024u.
- Zhang, B.-T.; Liu, H.; Liu, Y.; Teng, Y. Application trends of nanofibers in analytical chemistry. TrAC Trends Anal. Chem. 2020, 131, 115992, doi:10.1016/j.trac.2020.115992.
- Huang, C.; Barlow, S.; Marder, S.R. Perylene-3,4,9,10-tetracarboxylic Acid Diimides: Synthesis, Physical Properties, and Use in Organic Electronics. J. Org. Chem. 2011, 76, 2386–2407, doi:10.1021/jo2001963.
- Wang, Q.; Li, Z.; Tao, D.-D.; Zhang, Q.; Zhang, P.; Guo, D.-P.; Jiang, Y.-B. Supramolecular aggregates as sensory ensembles. Chem. Commun. 2016, 52, 12929–12939, doi:10.1039/c6cc06075g.
- Sun, J.; Li, Z.; Sun, Y.; Zhong, L.; Huang, J.; Zhang, J.; Liang, Z.; Chen, J.; Jiang, L. Uniform and reproducible plasmon-enhanced fluorescence substrate based on PMMA-coated, large-area Au@Ag nanorod arrays. Nano Res. 2018, 11, 953–965, doi:10.1007/s12274-017-1708-y.
- Zang, L.; Che, Y.; Moore, J.S. One-Dimensional Self-Assembly of Planar π-Conjugated Molecules: Adaptable Building Blocks for Organic Nanodevices. Accounts Chem. Res. 2008, 41, 1596–1608, doi:10.1021/ar800030w.
- Balakrishnan, K.; Datar, A.; Oitker, R.; Chen, H.; Zuo, J.; Zang, L. Nanobelt Self-Assembly from an Organic n-Type Semiconductor: Propoxyethyl-PTCDI. J. Am. Chem. Soc. 2005, 127, 10496–10497, doi:10.1021/ja052940v.
- Balakrishnan, K.; Datar, A.; Naddo, T.; Huang, J.; Oitker, R.; Yen, M.; Zhao, J.; Zang, L. Effect of Side-Chain Substituents on Self-Assembly of Perylene Diimide Molecules: Morphology Control. J. Am. Chem. Soc. 2006, 128, 7390–7398, doi:10.1021/ja061810z.
- Chen, Y.; Feng, Y.; Gao, J.; Bouvet, M. Self-assembled aggregates of amphiphilic perylene diimide–based semiconductor molecules: Effect of morphology on conductivity. J. Colloid Interface Sci. 2012, 368, 387–394, doi:10.1016/j.jcis.2011.10.076.
- Yue, E.; Ma, X.; Zhang, Y.; Zhang, Y.; Duan, R.; Ji, H.; Li, J.; Che, Y.; Zhao, J. Fluorescent bilayer nanocoils assembled from an asymmetric perylene diimide molecule with ultrasensitivity for amine vapors. Chem. Commun. 2014, 50, 13596–13599, doi:10.1039/c4cc06915c.
- Wang, C.; Wang, J.; Wu, N.; Xu, M.; Yang, X.; Lu, Y.; Zang, L. Donor–acceptor single cocrystal of coronene and perylene diimide: Molecular self-assembly and charge-transfer photoluminescence. RSC Adv. 2017, 7, 2382–2387, doi:10.1039/c6ra25447k.
- Datar, A.; Oitker, R.; Zang, L. Surface-assisted one-dimensional self-assembly of a perylene based semiconductor molecule. Chem. Commun. 2006, 1649, doi:10.1039/b518060k.
- Roberts, G. An applied science perspective of Langmuir-Blodgett films. Adv. Phys. 1985, 34, 475–512, doi:10.1080/00018738500101801.
- Weiss, R.G. The Past, Present, and Future of Molecular Gels. What Is the Status of the Field, and Where Is It Going? J. Am. Chem. Soc. 2014, 136, 7519–7530, doi:10.1021/ja503363v.
- Sangeetha, N.M.; Maitra, U. Supramolecular gels: Functions and uses. Chem. Soc. Rev. 2005, 34, 821–836, doi:10.1039/b417081b.
- Hu, W.; Wan, L.; Jian, Y.; Ren, C.; Jin, K.; Su, X.; Bai, X.; Haick, H.; Yao, M.; Wu, W. Electronic Noses: From Advanced Materials to Sensors Aided with Data Processing. Adv. Mater. Technol. 2019, 4, 1800488, doi:10.1002/admt.201800488.
- Zhao, M.; Yu, H.; He, Y. A dynamic multichannel colorimetric sensor array for highly effective discrimination of ten explosives. Sens. Actuators B Chem. 2019, 283, 329–333, doi:10.1016/j.snb.2018.12.061.
- Nelson, T.L.; O’Sullivan, C.; Greene, N.T.; Maynor, M.S.; Lavigne, J.J. Cross-Reactive Conjugated Polymers: Analyte-Specific Aggregative Response for Structurally Similar Diamines. J. Am. Chem. Soc. 2006, 128, 5640–5641, doi:10.1021/ja060589n.
- Yang, X.; Zhou, J.; Li, Y.; Yan, M.; Cui, Y.; Sun, G. A reaction-based sensing scheme for volatile organic amine reagents with the chromophoric-fluorogenic dual mode. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 240, 118539, doi:10.1016/j.saa.2020.118539.