| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Fatemeh Mokhtari | + 1382 word(s) | 1382 | 2020-12-29 09:22:44 | | | |
| 2 | Karina Chen | Meta information modification | 1382 | 2020-12-31 07:05:41 | | | | |
| 3 | Karina Chen | Meta information modification | 1382 | 2020-12-31 07:10:47 | | |
Video Upload Options
Bioprinting is a printing technology that prints cells together with the biomaterial, usually a hydrogel, known as a bioink . By including the capability of stimuli responsiveness, four-dimensional (4D) printing has emerged as an intriguing technology able to induce planned changes of the constructs (e.g., shape, stiffness) when triggered by a certain external stimulus , thus showing the potential of bridging the gap between the laboratory constructs and native human tissues.
1. Introducion
The main scope of tissue engineering (TE) is defined as the set of processes to develop biological structures that are able to restore, maintain, or even improve the function of tissues and/or organs [1]. Aiming to meet this challenging goal, multidisciplinary teams are required to work together in the fields of materials engineering and life sciences [2], to successfully employ scaffolds, growth factors, and cells, which represent the widely famous three pillars of TE. Initially, devices were mostly produced from bioinert materials, aiming to achieve adequate mechanical properties, inducing minimal responses from the host tissues. Then, bioengineered constructs were designed to provoke planned reactions at the implant site and follow a certain degradation rate. More recently, there has been emergent research on using smart materials to support and stimulate a better-mimicking tissue with high multicellular functionality [3][4][5][6].
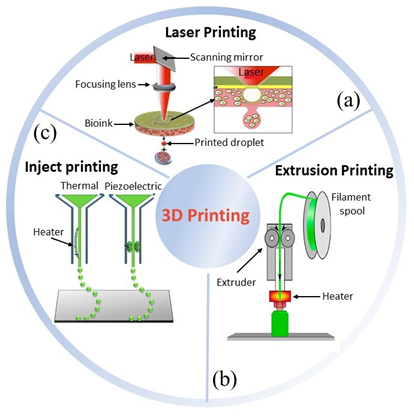
Three-dimensional (3D) printing has appeared as a useful technique for TE scaffolding to develop constructs with high control over structure geometry and architecture [7][8]. Its first appearance was reported in 1986 by Hull et al., who used a novel technique called stereolithography [9], able to fabricate 3D structures by curing a resin with a UV source in a layer-by-layer fashion. Throughout the years, several other 3D printing techniques have been developed (Figure 1), e.g., fused deposition modeling [10], selective laser melting [11], and selective laser sintering [12]. All of these share a great advantage over conventional scaffolding techniques, namely, the ability to use computer assisted design – computer assisted manufacturing (CAD–CAM) to customize constructs, and obtain replacements with topologies optimized by using medical imaging approaches [13][14]. However, conventional 3D matrices have revealed not to be the optimal approach for a number of clinical needs, e.g., tissue defects that change faster than tissue healing, regenerative processes that could be stimulated by electrical or mechanical inputs, body environments in which physicochemical conditions, such pH and temperature, may change over time.
Figure 1. Three main bioprinting technologies: (a) laser [15], (b) extrusion [16], (c) inject printing [17]. Parts of the figure are reprinted from [15][16][18]—open access under Creative Commons CC BY 4.0 license.
Smart biomaterials are at the basis of 4D printing, which, for biomedical applications, may also include printed live cells, thus being defined “bio”-printing [19]. Such materials can change shape or properties (e.g., stiffness, color, texture, transparency, energy transfer/conversion, volume) under the influence of external stimuli. Therefore, a clever use of such materials will result in 3D-built scaffolds that can adapt their features to specific applications [20][21]. As an innovative field, 4D (bio-)printing is suggested to be a promising tool to obtain effective TE therapies in a large number of clinical applications [22].
2. Four-Dimensional (Bio-)printing
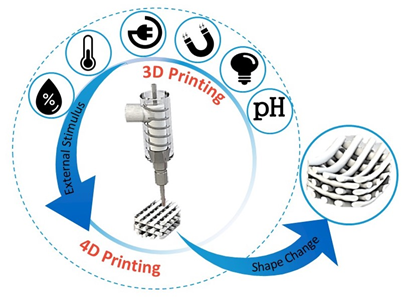
4D printing is the latest advancement in the advanced manufacturing field [23]. It was initially defined as a procedure where the shape, properties, or functionality of a 3D printed structure can change according to time and/or stimuli (Figure 2) [24]. The foundational works were developed at the Massachusetts Institute of Technology (MIT), by considering time as the fourth dimension [25]. Given this, 4D (bio-)printing indicates that for the transition, over time, the 3D printed biocompatible materials may incorporate live cells by using bioinks [22]. Thereafter, sequential approaches have been applied to the concept of 4D (bio-)printing, according to the identified stimuli concurring to defined biomedical applications. The first approach to 4D printing closely followed the MIT concept, in which the material (e.g., smart biopolymer or responsive hydrogel), under the desired stimulus, folds in a predefined 3D configuration, and cells or tissues simply follow the substrate fold and form the desired shape. The second approach is the “in vivo 4D bioprinting” in which a 3D polymer is printed, acting as a medical device, initially implanted to accommodate tissue or organ growth after the postsurgical period. When the tissue or organ gets stronger, the scaffold degrades gradually, preferably in a controlled manner, and is absorbed by the body. In this approach, tissue growth can be viewed as a leading stimulus. However, considering 3D printed scaffolds under the 4D printing umbrella because of their biodegradation property (i.e., controlled biodegradation as a shape change over time), is utmost controversial [26].
Figure 2. Illustrative diagram representing addition of a predesigned stimulus to promote a desired change at the construct. [Original image from the authors].
An et al. proposed a definition for 4D bioprinting, referring to groups of programmable self-assembling, self-folding, or self-accommodating technologies, which include three main defined or essential components: (i) man-made and not nature-made programmable design, (ii) two dimensional (2D) or 3D bioprinting process, and (iii) postprinting programmable evolution of bioprinted constructs that can be driven by cells or biomaterials and triggered by external signals [25].
The main advantageous characteristics of 4D bioprinting include additional degrees of freedom in terms of design, high precision cell placement, and the ability to produce tissues with adequate cellular density [23]. Many 3D printed scaffolds with programmable biodegradation fit within this definition, but are beyond the topic of this review. So far, a widely accepted definition of 4D printing is still missing, as from Moroni et al.: “Whether we are really witnessing 4D printing, a process that should be defined as a programmed temporal shape change occurring during the 3D manufacturing itself, or not, is still to be clarified in the field” [27]. In this scenario, many researchers are investigating different materials and approaches enabling controllable shape/property changes upon different stimuli and timeframes, which are suggested in this review under the name of 4D (bio-)printing.
A category that naturally suits the 4D (bio-)printing concept is the printing of smart hydrogels [28]. Hydrogels are polymeric networks able to form water-swollen structures thanks to different types of forces between the polymer chains, leading to semi-crystalline or amorphous materials [29][30]. Smart hydrogels are able to respond to a desired stimulus (e.g., shear stress, electricity, ionic force, light, magnetic field, pH, temperature). The interest in developing smart hydrogels has been increasing within the last years, as they should be able to provide remarkable characteristics, such as shape memory, self-healing and controllable sol-gel transition. Thus, being able to use the advantages of 3D (bio-)printing (e.g., customization), it is possible to improve the features of hydrogels by promoting self-weldability or self-unfoldability according to an external stimulus. As discussed in literature, both these characteristics, as 4D bio-origami hydrogels, may significantly contribute to achieve functional tissues [23], and to the fabrication of engineered tissues, such as liver [31] and heart [32], making a major breakthrough in the area of TE [33]. Extensive research has been carried out on the development of smart hydrogels. From a mechanical standpoint, hydrogels can show bare elastic up to viscous–elastic behavior, because of crosslinking type and density. To characterize hydrogels, Young’s modulus (E), storage modulus (G′), and loss modulus (G″), as well as other hydrogel-specific parameters, such as mesh size, polymer volume fraction in the swollen state, and average molecular weight between crosslinks, should be evaluated. Since water does act as a plasticizer among the polymer chains, hydrogels used in their swollen state, as in TE, are well above their glass transition temperature (Tg) and show viscoelasticity under loading conditions. For example, appropriate mechanical stimulation and biomechanical signals are key factors in tendon/ligament (T/L) engineering. As the native T/L is able to respond to mechanical forces by changing structure, composition, and mechanical properties [34][35][36], smart hydrogels obtained via 4D printing cold help to achieve such a critical goal. Typical mechanical values of hydrogels (e.g., ≈0.1 MPa Young’s moduli) are usually far from satisfying T/L mechanics (i.e., 1.2–1.8 GPa tensile modulus) that can be achieved only by high modulus chemically crosslinked hydrogels [37][38]. Moreover, transition zones of T/L into muscle or bone need a gradient of properties capable of responding to personalized tissue mechanics.
As the field of 4D (bio-)printing continues to grow, in the next years it is expected to rapidly expand across fields, including regenerative medicine, robotics, and medical devices [39].
References
- Miyoshi, H. Smart Design of Materials for Tissue Engineering. In Smart Materials for Tissue Engineering: Fundamental Principles; The Royal Society of Chemistry: London, UK, 2016; ISBN 9789533070797. [Google Scholar]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.P.; Leong, K.W. Scaffolding in tissue engineering: General approaches and tissue-specific considerations. Eur. Spine J. 2008, 17. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.S. Stimuli-responsive polymers: Biomedical applications and challenges for clinical translation. Adv. Drug Deliv. Rev. 2013, 65, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Stuart, M.A.C.; Huck, W.T.S.; Genzer, J.; Müller, M.; Ober, C.; Stamm, M.; Sukhorukov, G.B.; Szleifer, I.; Tsukruk, V.V.; Urban, M.; et al. Emerging applications of stimuli-responsive polymer materials. Nat. Mater. 2010, 9, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, L.; Lim, K.H.; Gonuguntla, S.; Lim, K.W.; Pranantyo, D.; Yong, W.P.; Yam, W.J.T.; Low, Z.; Teo, W.J.; et al. The Pathway to Intelligence: Using Stimuli-Responsive Materials as Building Blocks for Constructing Smart and Functional Systems. Adv. Mater. 2019, 31, 1804540. [Google Scholar] [CrossRef] [PubMed]
- Sears, N.A.; Seshadri, D.R.; Dhavalikar, P.S.; Cosgriff-Hernandez, E. A Review of Three-Dimensional Printing in Tissue Engineering. Tissue Eng. Part B Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Morouço, P.G. The Usefulness of Direct Digital Manufacturing for Biomedical Applications. In Intensification of Biobased Processes; The Royal Society of Chemistry: London, UK, 2018; pp. 478–487. [Google Scholar]
- Hull, C.W. Method of and Apparatus for Production of Three Dimensional Objects by Stereolithography. U.S. Patent No 5,236,637, 17 August 1993. [Google Scholar]
- Hutmacher, D.W.; Schantz, T.; Zein, I.; Ng, K.W.; Teoh, S.H.; Tan, K.C. Mechanical properties and cell cultural response of polycaprolactone scaffolds designed and fabricated via fused deposition modeling. J. Biomed. Mater. Res. 2001, 55, 203–216. [Google Scholar] [CrossRef]
- Mullen, L.; Stamp, R.C.; Brooks, W.K.; Jones, E.; Sutcliffe, C.J. Selective laser melting: A regular unit cell approach for the manufacture of porous, titanium, bone in-growth constructs, suitable for orthopedic applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 89, 325–334. [Google Scholar] [CrossRef]
- Nakamura, M.; Iwanaga, S.; Henmi, C.; Arai, K.; Nishiyama, Y. Biomatrices and biomaterials for future developments of bioprinting and biofabrication. Biofabrication 2010, 2, 014110. [Google Scholar] [CrossRef]
- Castro, N.J.; Meinert, C.; Levett, P.; Hutmacher, D.W. Current developments in multifunctional smart materials for 3D/4D bioprinting. Curr. Opin. Biomed. Eng. 2017, 2, 67–75. [Google Scholar] [CrossRef]
- Milazzo, M.; Contessi Negrini, N.; Scialla, S.; Marelli, B.; Farè, S.; Danti, S.; Buehler, M.J. Additive Manufacturing Approaches for Hydroxyapatite-Reinforced Composites. Adv. Funct. Mater. 2019, 29, 1903055. [Google Scholar] [CrossRef]
- Keriquel, V.; Oliveira, H.; Rémy, M.; Ziane, S.; Delmond, S.; Rousseau, B.; Rey, S.; Catros, S.; Amédée, J.; Guillemot, F.; et al. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Liu, F.; Wang, X. Synthetic Polymers for Organ 3D Printing. Polymers (Basel) 2020, 12, 1765. [Google Scholar] [CrossRef]
- Keriquel, V.; Oliveira, H.; Rémy, M.; Ziane, S.; Delmond, S.; Rousseau, B.; Rey, S.; Catros, S.; Amédée, J.; Guillemot, F.; et al. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]Liu, F.; Wang, X. Synthetic Polymers for Organ 3D Printing. Polymers (Basel) 2020, 12, 1765. [Google Scholar] [CrossRef]Vijayavenkataraman, S.; Fuh, J.Y.H.; Lu, W.F. 3D printing and 3D bioprinting in pediatrics. Bioengineering 2017, 4, 63. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Fuh, J.Y.H.; Lu, W.F. 3D printing and 3D bioprinting in pediatrics. Bioengineering 2017, 4, 63. [Google Scholar] [CrossRef]
- Groll, J.; Boland, T.; Blunk, T.; Burdick, J.A.; Cho, D.-W.; Dalton, P.D.; Derby, B.; Forgacs, G.; Li, Q.; Mironov, V.A.; et al. Biofabrication: Reappraising the definition of an evolving field. Biofabrication 2016, 8, 13001. [Google Scholar] [CrossRef]
- Leist, S.K.; Zhou, J. Current status of 4D printing technology and the potential of light-reactive smart materials as 4D printable materials. Virtual Phys. Prototyp. 2016, 2759, 1–14. [Google Scholar] [CrossRef]
- Mandon, C.A.; Blum, L.J.; Marquette, C.A. 3D-4D printed objects: New bioactive material opportunities. Micromachines 2017, 8, 102. [Google Scholar] [CrossRef]
- Gao, B.; Yang, Q.; Zhao, X.; Jin, G.; Ma, Y.; Xu, F. 4D bioprinting for biomedical applications. Trends Biotechnol. 2016, 34, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Khoo, Z.X.; Teoh, J.E.M.; Liu, Y.; Chua, C.K.; Yang, S.; An, J.; Leong, K.F.; Yeong, W.Y. 3D printing of smart materials: A review on recent progresses in 4D printing. Virtual Phys. Prototyp. 2015, 10, 103–122. [Google Scholar] [CrossRef]
- Momeni, F.M.; Mehdi Hassani, N.S.; Liu, X.; Ni, J. A review of 4D printing. Mater. Des. 2017, 122, 42–79. [Google Scholar] [CrossRef]
- An, J.; Chua, C.K.; Mironov, V. A perspective on 4D bioprinting. Int. J. Bioprinting 2016, 2, 3–5. [Google Scholar] [CrossRef]
- Morouço, P.; Lattanzi, W.; Alves, N. Four-dimensional bioprinting as a new era for tissue engineering and regenerative medicine. Front. Bioeng. Biotechnol. 2017, 5, 61. [Google Scholar] [CrossRef] [PubMed]
- Moroni, L.; Boland, T.; Burdick, J.A.; De Maria, C.; Derby, B.; Forgacs, G.; Groll, J.; Li, Q.; Malda, J.; Mironov, V.A.; et al. Biofabrication: A guide to technology and terminology. Trends Biotechnol. 2018, 36, 384–402. [Google Scholar] [CrossRef]
- Wang, S.; Lee, J.M.; Yeong, W.Y. Smart hydrogels for 3D bioprinting. Int. J. Bioprinting 2015, 1, 3–14. Available online: http://ijb.whioce.com/index.php/int-j-bioprinting/article/view/01005 (accessed on 23 October 2020). [CrossRef]
- Negrini, N.C.; Bonetti, L.; Contili, L.; Farè, S. 3D printing of methylcellulose-based hydrogels. Bioprinting 2018, 10, e00024. [Google Scholar] [CrossRef]
- Negrini, N.C.; Celikkin, N.; Tarsini, P.; Farè, S.; Święszkowski, W. Three-dimensional printing of chemically crosslinked gelatin hydrogels for adipose tissue engineering. Biofabrication 2020, 12, 25001. [Google Scholar] [CrossRef]
- Ikegami, T.; Maehara, Y. Transplantation: 3D printing of the liver in living donor liver transplantation. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 697–698. [Google Scholar] [CrossRef]
- Valverde, I.; Gomez, G.; Suarez-mejias, C.; Hosseinpour, A.; Hazekamp, M.; Roest, A.; Vazquez-jimenez, J.F.; El-rassi, I.; Uribe, S.; Gomez-cia, T. 3D printed cardiovascular models for surgical planning in complex congenital heart diseases. J. Cardiovasc. Magn. Reson. 2015, 17, P196. [Google Scholar] [CrossRef]
- Gao, G.; Cui, X. Three-dimensional bioprinting in tissue engineering and regenerative medicine. Biotechnol. Lett. 2016, 38, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.-C. Mechanobiology of tendon. J. Biomech. 2006, 39, 1563–1582. [Google Scholar] [CrossRef] [PubMed]
- Milazzo, M.; Jung, G.S.; Danti, S.; Buehler, M.J. Mechanics of mineralized collagen fibrils upon transient loads. ACS Nano 2020, 14, 8307–8316. [Google Scholar] [CrossRef] [PubMed]
- Milazzo, M.; Jung, G.S.; Danti, S.; Buehler, M.J. Wave propagation and energy dissipation in collagen molecules. ACS Biomater. Sci. Eng. 2020, 6, 1367–1374. [Google Scholar] [CrossRef]
- Ekwueme, E.C.; Patel, J.M.; Freeman, J.W.; Danti, S. Applications of bioresorbable polymers in the skeletal systems (cartilages, tendons, bones). In Bioresorbable Polymers for Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2017; pp. 391–422. [Google Scholar]
- Zhu, L.; Qiu, J.; Sakai, E. A high modulus hydrogel obtained from hydrogen bond reconstruction and its application in vibration damper. RSC Adv. 2017, 7, 43755–43763. [Google Scholar] [CrossRef]
- Li, Y.-C.; Zhang, Y.S.; Akpek, A.; Shin, S.R.; Khademhosseini, A. 4D bioprinting: The next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication 2016, 9, 12001. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-C.; Zhang, Y.S.; Akpek, A.; Shin, S.R.; Khademhosseini, A. 4D bioprinting: The next-generation technology for biofabrication enabled by stimuli-responsive materials. Biofabrication 2016, 9, 12001. [Google Scholar] [CrossRef] [PubMed]