
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | André Silva | + 2958 word(s) | 2958 | 2020-12-01 09:52:49 | | | |
| 2 | Lily Guo | + 190 word(s) | 3148 | 2020-12-21 07:35:34 | | | | |
| 3 | Lily Guo | + 190 word(s) | 3148 | 2020-12-21 07:36:10 | | |
Video Upload Options
The Internet of medical things (IoMT) is an area within the IoT that directly focuses on medical applications and deals with the acquisition, processing, transmission and storage of medical information through the amalgamation of specific devices (things) built to ensure patient safety and data security. IoMT builds over a general IoT system. Different design concepts have been shown both using existent protocols, as well as commonly used bands, i.e., industrial, scientific and medical (ISM) bands, which includes both Bluetooth and ZigBee. Additionally, novel and emerging protocols for wireless sensor body network (WSBN) are being investigated.
1. Introduction
The healthcare sector is rapidly changing. Many countries have implemented or are implementing value-based healthcare. The European Union supports the shift toward digital healthcare and a patient-centric approach through several funding programs[1]. Digital Healthcare and Domiciliary hospitalization are some important components of future healthcare. This is due to several reasons. First, hospital beds are scarce[2] and costly. Second, the world is getting older[3]. In 2025 the world is expected to have twice elderly as it had in 2010[4]. Third, For those living in isolated zones, daily access to healthcare is challenging[5]. Chronic diseases are one example of medical monitoring at a distance; the current demographic dictates the number of people who are fighting with chronic diseases is increasing[6]. Quality of life is sensible and could increase, regarding the physical aspect, for people living with these conditions if they are monitored 24 h per day. The national health expenditures (NHE) of the United States of America are expected to almost double from 2012 to 2024 [7]. This situation can pose a strain on the country’s economy for the near future and the lack of access to health for part of the population[8].
Domiciliary hospitalization is a model of hospitalization in which the patient receives hospital-level care while staying at home. One important tool for a wider application of this hospitalization model is wireless patient monitoring. In the last decade, rapid advances were made on architectures for the Internet of things (IoT) and the Internet of medical things (IoMT), including compact and low consumption processing and communication chips. In addition, emerging technologies for the implementation of wearable patches have rapidly evolved. This includes technologies in printed and stretchable electronics and e-textile, printed sensors, including, as well, printed batteries, energy harvesting and supercapacitors. IoMT is expected to have a market of $322.2 billion by 2025, growing 29.9% from 2019 to 2020[9]. Still, the Internet of medical things corresponds to only 7% of the Internet of things devices[10].
The Internet of things is the amalgamation of devices including sensors, actuators and software, all together constitute a network that exchanges data over the Internet. The Internet of Things concept was first discussed more than 20 years ago[11], but recent years observed an increasing interest in research and commercial investment in this field, for different applications[12][13], and increased from 19 billion devices in 2013 to 40 billion in 2019[12]. The Internet of medical things (IoMT) is an area within the IoT that directly focuses on medical applications and deals with the acquisition, processing, transmission and storage of medical information through the amalgamation of specific devices (things) built to ensure patient safety and data security[14]. IoMT builds over a general IoT system. Different design concepts have been shown both using existent protocols, as well as commonly used bands, i.e., industrial, scientific and medical (ISM) bands[15], which includes both Bluetooth and ZigBee. Additionally, novel and emerging protocols for wireless sensor body network (WSBN) are being investigated. Some of the protocols were also implemented specifically for medical use, such as the medical implant communications service (MICS). The acceptance of the IoMT system among the population is generally more challenging than other IoT systems, as it deals with highly sensitive and personal data. Due to the sensitivity of these types of data, security and patient privacy are two important factors in IoMT design. On the sensing layer, as these devices are in direct contact with the patient body, it is desired to have systems that are soft, and stretchable, biocompatible, electrically safe, and comfortable for long-term use. In addition, as these are generally mobile devices, special attention is given to low-energy devices in order to enable long-time monitoring without the hassle of battery recharging.
2. Wearable Bioelectronics for an IoMT System
The first part of wearable bioelectronics is body-interfacing devices that acquire and transmit the electrophysiological data. These devices are generally composed of electronic components for acquisition, digital conversion, processing, and communication of data. Wearable biomonitoring devices are a category of wireless monitoring, which in addition to electronics and signal processing, includes research on materials and fabrication techniques that provide conformable and comfortable wearable monitoring [16][17][18]. Traditional wearables, such as smartwatches and fitness trackers, are widely used wearable monitoring devices. Nevertheless, these are generally limited both in terms of the location that can be applied (e.g., wrist or chest) and the type of signal that they can monitor. For instance, as these devices cannot be applied over the throat, they are not able to monitor throat activity. Soft and stretchable biomonitoring stickers do not have these limitations. They can be applied anywhere on the body, and as they conform better to the human soft tissues, they can provide a better signal, less prone to noise and human motion[19]. Other examples of futuristic patches are “electronic tattoos”, that are ultrathin films populated with microelectronics that are transferred to the human skin, similar to the temporary decorative tattoos. This category of devices is further explained in Section 3.1.
In this section, an overview of the parameters that can be measured by wearable bioelectronics systems will be presented. Next, the general structural design for an IoMT system will be presented, including sensing, communication, and application layers.
2.1. Skin Interfacing Electrophysiological Sensing: What Can Be Measured?
The human body emits various types of signals that contain important information about human health. These signals can be divided into various categories, based on the type of signal and the transducers that are necessary to measure them. The most common types of these signals include bio-electric, bio-Impedance, bio-acoustics, and bio-optical signals, mechanical and motion sensing, temperature sensing, and electrochemical measurements for sweat monitoring.
Bio-potential signals are parameters based on an electric signal produced by the body. This includes biopotentials that are generated by muscles, heart, and brain, i.e., electrocardiogram (ECG) for monitoring of heart, electroencephalogram (EEG) for brain signal acquisition, and electromyography (EMG) to measure the muscular activity.
Bioimpedance is the measurement of the impedance of a specific part of the body. This is usually measured as the response of the body upon the introduction of a small current[20]. Examples of an application include smart scales that measure and report body fat and body mass index. Other than body composition information, this can be as well used to measure heart pulse and other vital signs as blood flow or breathing rate[21]. Another example of an application is to monitor the skin impedance changes to analyze the galvanic skin response (GSR) as an indicator of human emotions. As a response to emotional responses, sweat glands activity increase, changing the conductance of the skin[22].Bioacoustics are sounds produced by organs or other body components. This includes heart valve sounds, blood flow, lung inflation sounds and bowel movement[23][24]. Bioacoustics can be used, as well, to monitor the throat sounds. This can be used to monitor the throat vibration during talking[25], eating (to monitor daily food intake)[26], and coughs[27]. The application of the latter is under study for the possible classification of Covid-19 patients[28]. Some implementations also allow bio-acoustic sounds to be used as a gesture controller[29].Bio-optical signals are produced using a light source or body imaging. This includes pulse oximetry to measure the blood oxygen and heart rate, using an LED and a photodiode that monitor changes on the light reflection resulting from the oxyhemoglobin concentration in the blood[30][31]. Another example of application includes using the optical spectra of the skin to study cancer cells[32].Body temperature constitutes one important health indicator and can be measured with various techniques using an optical signal, using thermal cameras[33] or by contact sensors, including digital thermometers and thermistors[34] [49].Mechanical movements of the body can be used as well in monitoring the daily activity in general or in the analysis of motion and vibration of a specific part of the body. This is generally performed using accelerometers and gyroscopes[35]. Mechanical sensors can also be exploited for measuring muscular movements through force myography as an alternative to electromyography[36].Other types of bio-signals are emerging as well. Chemical and electrochemical signals are the measure of the chemical composition of bio-fluids [37], such as sweat. Sweat analysis can be done with wearable and non-invasive methods and allow for easy tracking of bio-markers in their composition for the detection of genetic conditions and other kinds of diseases[38] [53]. For more information on types of bioelectronics signals for biomonitoring, or HMIs, one can consult related review articles[39][40].Figure 1, and Table 1, summarizes the main electrophysiological parameters that can be monitored by non-invasive bioelectronics devices.
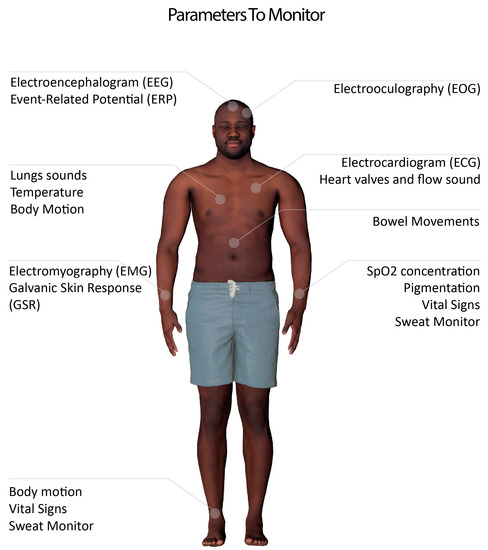
Figure 1. Parameters that can be monitored using an Internet of medical things (IoMT) system and wearable biomonitoring.
Table 1. Types of Bio-signals, and most common tests.
|
Biopotential |
Bioimpedance |
Conductance |
Acoustics |
Optical |
Others |
|
Muscles (EMG) |
Emotions |
GSR |
Voice |
SpO2 Concentration |
Temperature |
|
Heart (ECG) |
Body fat |
|
Food intake |
Pigmentation changes |
Mechanical (myography) |
|
Brain (EEG) |
|
|
Digestive System |
|
Chemical (e.g., sweat) |
|
Eyes (EOG) |
|
|
Coughs |
|
|
|
|
|
|
Heart murmurs |
|
|
2.2. IoMT System for Patient Monitoring and Domiciliary Hospitalization
Over the last years, different types of IoMT architectures were proposed and investigated[13]. These are generally multilayer systems that intend to assure safe data transmission and communication. Recent implementations generally propose three main layers, a wearable sensing layer, an intermediary data acquisition and transmission layer and a cloud computing layer[41][42]. The first two layers are sometimes combined, but the objective of the intermediary layer is to reduce the necessary components over the sensing layer and build a robust middle layer that is more flexible in data transmission to the cloud. With those implementations in mind, we discuss a simple general design for an IoMT system that can be adapted for specific uses.
Figure 2 demonstrates the general structural design of an IoMT system for wireless patient monitoring and domiciliary hospitalization. Every implementation of an IoMT will vary; however, we can generalize three layers that help understand the general direction of the information:
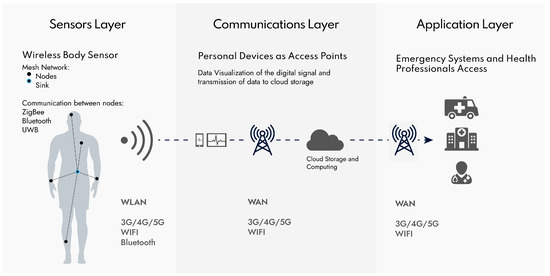
First layer (sensors)—when several different sensors are implemented at various locations of the body, it is common to build a wireless sensor body network (WSBN) or wireless sensor network (WSN) as well. WSBN is defined as a network of various sensors connected to each other. Usually, in a mesh arrangement, even though other typologies are possible[15]. Sensors can be implantable, wearable and mobile (hold in hands or inside pockets). The sensors’ communications have a low range (~2 m) and have low power needs. Active research tries to improve the efficiency of the protocols to consume the least possible energy[43]. The band used for this low power network is mostly the common industrial, scientific and medical (ISM) band, or may be more specific, such as medical implants communications service (MICS) band. Both may be used for direct communication of a single sensory device, to an external device, or in a WSBN architecture. These bands allow for common technologies as Zigbee or Bluetooth to be used, but also some new approaches like fat–intrabody communication. The sensors layer was given that name for simplicity since some actuators are also possible to implement. Most of the challenges of this layer, though, belong to the sensors themselves. We will discuss wearable sensors challenges in Section 3. The mesh nodes from the WSBN connect to a central master, also called the sink. The master controls the communications with the sensors and communicates with an exterior gateway to the wireless local area network (WLAN)[44]. Energy and security of the data transmitted are of great importance.
Second layer (communications)—The master of the WSBN will communicate to the WLAN via common technologies like mobile communications, WIFI or Bluetooth. The power requirements of these types of networks are still important; however, not as limited as the sensors layer. In this case, it is characterized by the existence of a local gateway that can be a personal mobile device like a smartphone or tablet. The IoMT system gets easier and faster to implement, without the need for unnecessary gateway components. It can also be an ad-hoc device, specially built for this purpose. The mid-layer gateway can communicate both with the sensing layer and with the exterior network WAN, for instance, through 5G, WIFI or GPR. This serves as an intermediate layer for data storage and communication. Ideally, the sensor layer components are not always transmitting information, only being awake from time-to-time to transmit the fundamental information. For this reason, the data that arrives at the gateway is not yet treated a first raw part of information can be displayed and treated in the phone. However, after transmitting the data to the cloud server, it has fewer limitations in terms of computing power and energy, so it stores the data and can also be used to process the data applying algorithms of data-mining. The cloud server is located at the border with the application layer.
Third layer (applications)—As the cloud server manages the communications to the gateway system, it also handles data processing and data application. This way, it also needs to handle data access to health professionals or the patient through a website or smartphone application, as well as implement ambulatory emergency services to request medical attention in a domiciliary hospitalization system. For this reason, the application layer connects via WAN to personal devices as to other services like the ambulatory or other hospital storage center service. The biggest concern for the application layer is the security of the data. As it manages communications with different terminals, it is more vulnerable to exposition. Even though difficult, security is being researched with new approaches dealing with blockchain for a secure and decentralized implementation of the IoMT[45].
2.3. Technologies for Wireless Communications
Bluetooth was created to replace RS-232 cable connections with a wireless alternative. As such, continuous communication by Bluetooth requires 1 W of power[46] and is capable of 1–3 Mbit/s data communication over 500 m is possible (100 m advisable)[47] [62]. Bluetooth low energy (BLE), originally from Nokia in 2006, was later integrated with Bluetooth 4.0 release in 2009 to address the necessity of very low power communication for IoT devices. The data transfer was slightly affected (1 Mbit/s data rate), but the power consumption was significantly reduced (0.05–0.1 W). This tremendous increase makes the BLE low energy, an excellent candidate for the use in IoMT devices. The newer version of Bluetooth is the 5; it was created with IoT applications in mind, thus allowing the use of Bluetooth low energy while promising increasing speed and range[48].
ZigBee was created with the objective of a low power alternative for implementation in home automation personal area network (PAN) while being less expensive. Zigbee has a slow data rate (250 kbit/s) but is very power efficient (consuming as low as 9.3 mA while in working mode) [49]. It uses mostly the bands of 2.4 GHz (915 MHz and 868 MHz for different zones) with mesh typology. Implementing a Zigbee network is low cost and power-efficient, becoming an excellent alternative for WSBN implementation, plus it allows for theoretical 65,000 devices connected in the same network. ZigBee is, however, less popular than Bluetooth. Because Bluetooth is widely and easily available, it is implemented in almost all smartphones and tablets, while the ZigBee market goes around industrial applications and small home IoT applications. Green energy is also a feature of ZigBee, allowing the possibility of energy harvesting options in the stack protocol. It is designed specifically for uses with no possibility of external power and work together with extremely low power silicon devices. This way, ultra-low power communication is possible, with five or more times lower power than normal ZigBee [50]. One example case is that it is possible to use a small photovoltaic cell powered by indoor lights to send a message every minute.
Another less popular alternative is ultra wide band (UWB), which operates with pulses of bigger frequencies of the spectrum (3.1–10.6 GHz). The first consequence of this is the short-range, ideal for WSBN, but not good enough for long-distance communication in the IoT and IoMT last layers of the architecture. Ant is also an interesting alternative, with an ultra-low power design is capable of consuming less power than Bluetooth and ZigBee[49].
Table 2 summarizes these and some other common communication protocols/technologies.
Table 2. Most common wireless communication standards.
|
Recent Version |
Range (m) |
Data Rate |
Frequency |
Band |
Standard |
Energy Consumption |
||
|
Bluetooth [63] |
5.2 (2020) |
<10–500+ |
1–3 Mbit/s |
2.402–2.480 GHz |
ISM |
IEEE 802.15.1 |
<30 mA |
|
|
Bluetooth Low energy |
- |
500+ |
125 kbit/s-2 Mbit/s |
2.400–2.4835 GHz |
ISM |
- |
<15 mA |
|
|
ZigBee [73] |
2015 |
10–300+ |
250 Kbit/s |
2.40 GHz |
ISM |
IEEE 802.15.4 |
<16 mA |
|
|
UWB [74] |
- |
short |
675 Mbit/s |
3.1–10.6 GHz (500 MHz channels) |
- |
IEEE 802.15.6-2012 |
|
|
|
ANT [75] |
ANT+ |
30 |
60 Kbit/s |
2.4 GHz |
ISM |
- |
<60µA |
|
|
RuBee [76] |
- |
20 |
1200 kB/s |
131 kHz |
IEEE 1902.1 |
|||
|
Sensium [77] (HR monitor) |
>3 |
160 kb/s |
900 MHz |
<3 mA |
||||
|
Zarlink [78] (implants) |
ZL70101 ZL70081 ZL70250 |
<2 |
<800 kb/s |
402–405 MHz; |
MICS/ ISM |
|
<6 mA |
|
|
Z-Wave [79], [80] (Homecare) |
Z-Wave Plus V2 |
10–100 |
100 Kbit/s |
2.4 GHz and 900 MHz |
ISM |
IEEE 802.15.4 |
<38.8 mA (13 dB) <12.9 mA (0 dB) |
|
|
NFC [81] |
- |
0.1 |
424 Kbit/s |
13.56 MHz |
ISM |
ISO/IEC 18000-3 |
- |
|
|
RFID [82] |
- |
<12 (100) |
- |
120–150 kHz; 13.56–928 MHz; 2.45–5.8 GHz |
ISM |
ISO/IEC 18000 |
- |
|
|
Mobile technology [83] |
5G |
- |
100–900 Mbit/s |
600–700 MHz |
- |
- |
- |
|
Novel and emerging communication protocols are as well being developed specifically for WSBN. One popular example is using the human body as the communication medium, also called Fat-Intrabody communication (Fat-IBC). The layer of fat that exists between the skin and the muscles has very different dielectric properties than the skin and muscle. Hence, the layer of fat acts as a parallel plate waveguide, being possible to pass a radio frequency of 2.45 GHz with manageable losses, achieving low power communication between two parts of the body depending on a fat layer[51]. It should be mentioned that to implement this communication system, two electrodes need to be implemented inside the skin in contact with the fat tissue[43], and the blood vessel orientation can also affect the signal[52]. Another way to communicate using the human body is to use the body as a group of capacitors to which two electrodes are connected in contact (galvanic coupling) or separated (capacitive coupling). This way, a signal of 1–100 MHz can be transmitted for a simple and low power communication; the common name given to his method is Intra-body communication (IBC)[53][54].
The use of passive RFID tags is another energy-efficient solution, as passive RFID tags do not require a power supply to transfer data; instead, they harvest their energy using their antenna. However, the range and data transfer are very limited. Communications using this backscattering technique has been shown for a distance of 50 cm[55].
References
- European Commission. Uptake of Digital Solutions in the Healthcare Industry. No. January, 2017. Available online: https://ec.europa.eu/growth/tools-databases/dem/monitor/sites/default/files/DTM_Uptake of digital solutions v1.pdf (accessed on 24 November 2020).
- Remuzzi, A.; Remuzzi, G. COVID-19 and Italy: What next? Lancet 2020, 395, 1225–1228, doi:10.1016/s0140-6736(20)30627-9.
- Ahmadi, H.; Arji, G.; Shahmoradi, L.; Safdari, R.; Nilashi, M.; Alizadeh, M. The application of internet of things in healthcare: A systematic literature review and classification. Univers. Access Inf. Soc. 2019, 18, 837–869, doi:10.1007/s10209-018-0618-4.
- Campbell, P. Population Projections: States, 1995–2025; Census Bureau: Suitland-Silver Hill, MD, USA, 1997; pp. 1–6.
- Gouda, K.; Okamoto, R. Current status of and factors associated with social isolation in the elderly living in a rapidly aging housing estate community. Environ. Health Prev. Med. 2012, 17, 500–511, doi:10.1007/s12199-012-0282-x.
- Megari, K. Quality of Life in Chronic Disease Patients. Health Psychol. Res. 2013, 1, 27, doi:10.4081/hpr.2013.e27.
- Keehan, S.P.; Cuckler, G.A.; Sisko, A.M.; Madison, A.J.; Smith, S.D.; Stone, D.A.; Poisal, J.A.; Wolfe, C.J.; Lizonitz, J.M. National Health Expenditure Projections, 2014–2024: Spending Growth Faster Than Recent Trends. Health Aff. 2015, 34, 1407–1417, doi:10.1377/hlthaff.2015.0600.
- Jost, T.S. Our Broken Health Care System and How to Fix It: An Essay on Health Law and Policy. Wake For. L. Rev. 2006, 41, 537. Available online: https://heinonline.org/HOL/Page?handle=hein.journals/wflr41&id=547&div=&collection= (accessed on 31 May 2020).
- Internet of Things (IoT) in Healthcare Market is Expected to Grow at a CAGR of 29.9% to Reach $322.2 billion by 2025: Meticulous Research®. Available online: https://www.globenewswire.com/news-release/2020/03/19/2003195/0/en/Internet-of-Things-IoT-in-Healthcare-Market-is-Expected-to-Grow-at-a-CAGR-of-29-9-to-Reach-322-2-billion-by-2025-Meticulous-Research.html (accessed on 31 May 2020).
- Onag, G. IoT Developers to Focus more on Smart Healthcare Post-COVID-19. FutureIOT, 2020. Available online: https://futureiot.tech/iot-developers-to-focus-more-smart-healthcare-post-covid-19/ (accessed on 20 April 2020).
- That ‘Internet of Things’ Thing—RFID Journal. Available online: https://www.rfidjournal.com/that-internet-of-things-thing (accessed on 31 May 2020).
- Adam Thierer, A.C. Economic Perspectives Projecting the Growth and Economic Impact of the Internet of Things; George Mason University, Mercatus Center: Arlington, Virginia, 2015.
- Irfan, M.; Ahmad, N. Internet of medical things: Architectural model, motivational factors and impediments. In Proceedings of the 2018 15th Learning and Technology Conference (L&T), Jeddah, Saudi Arabia, 25–26 February 2018; pp. 6–13.
- Gatouillat, A.; Badr, Y.; Massot, B.; Sejdić, E. Internet of Medical Things: A Review of Recent Contributions Dealing With Cyber-Physical Systems in Medicine. IEEE Internet Things J. 2018, 5, 3810–3822, doi:10.1109/jiot.2018.2849014.
- Ullah, S.; Higgins, H.; Braem, B.; Latre, B.; Blondia, C.; Moerman, I.; Saleem, S.; Rahman, Z.; Kwak, K.-S. A Comprehensive Survey of Wireless Body Area Networks. J. Med Syst. 2012, 36, 1065–1094, doi:10.1007/s10916-010-9571-3.
- Ray, T.R.; Choi, J.; Bandodkar, A.J.; Krishnan, S.; Gutruf, P.; Tian, L.; Ghaffari, R.; Rogers, J.A. Bio-Integrated Wearable Systems: A Comprehensive Review. Chem. Rev. 2019, 119, 5461–5533, doi:10.1021/acs.chemrev.8b00573.
- Trung, T.Q.; Lee, N. Flexible and Stretchable Physical Sensor Integrated Platforms for Wearable Human-Activity Monitoringand Personal Healthcare. Adv. Mater. 2016, 28, 4338–4372, doi:10.1002/adma.201504244.
- Amjadi, M.; Kyung, K.-U.; Park, I.; Sitti, M. Stretchable, Skin-Mountable, and Wearable Strain Sensors and Their Potential Applications: A Review. Adv. Funct. Mater. 2016, 26, 1678–1698, doi:10.1002/adfm.201504755.
- Lopes, P.A.; Gomes, D.V.; Marques, D.G.; Faia, P.; Góis, J.; Patrício, T.F.; Coelho, J.F.J.; Serra, A.C.; De Almeida, A.T.; Majidi, C.; et al. Soft Bioelectronic Stickers: Selection and Evaluation of Skin‐Interfacing Electrodes. Adv. Healthc. Mater. 2019, 8, e1900234, doi:10.1002/adhm.201900234.
- Bera, T.K. Bioelectrical Impedance Methods for Noninvasive Health Monitoring: A Review. J. Med Eng. 2014, 2014, 1–28, doi:10.1155/2014/381251.
- Havlík, J.; Fousek, O.; Ložek, M. Patient Monitoring Using Bioimpedance Signal. Comput. Vis. 2012, 7451, 171–172, doi:10.1007/978-3-642-32395-9_15.
- Villarejo, M.V.; Garcia-Zapirain, B.; Mendez-Zorrilla, A. A Stress Sensor Based on Galvanic Skin Response (GSR) Controlled by ZigBee. Sensors 2012, 12, 6075–6101, doi:10.3390/s120506075.
- Hadjileontiadis, L.J.; Rekanos, I.T.; Panas, S.M. Bioacoustic Signals. In Wiley Encyclopedia of Biomedical Engineering; Wiley: Hoboken, NJ, USA, 2006.
- Cotur, Y.; Kasimatis, M.; Kaisti, M.; Olenik, S.; Georgiou, C.; Güder, F. Stretchable Composite Acoustic Transducer for Wearable Monitoring of Vital Signs. Adv. Funct. Mater. 2020, 30, 1910288, doi:10.1002/adfm.201910288.
- Shahina, A.; Yegnanarayana, B. Mapping Speech Spectra from Throat Microphone to Close-Speaking Microphone: A Neural Network Approach. EURASIP J. Adv. Signal Process. 2007, 2007, 087219, doi:10.1155/2007/87219.
- Makeyev, O.; Lopez-Meyer, P.; Schuckers, S.; Besio, W.; Sazonov, E. Automatic food intake detection based on swallowing sounds. Biomed. Signal Process. Control. 2012, 7, 649–656, doi:10.1016/j.bspc.2012.03.005.
- Xiao, Y.; Carson, D.; Boris, L.; Mabary, J.; Lin, Z.; Nicodème, F.; Cuttica, M.; Kahrilas, P.J.; Pandolfino, J.E.; Carlson, D. The acoustic cough monitoring and manometric profile of cough and throat clearing. Dis. Esophagus 2013, 27, 5–12, doi:10.1111/dote.12038.
- Jeong, H.; Rogers, J.; Xu, S. Continuous on-body sensing for the COVID-19 pandemic: Gaps and opportunities. Sci. Adv. 2020, 6, eabd4794, doi:10.1126/sciadv.abd4794.
- Laput, G.; Xiao, R.; Harrison, C. ViBand. In Proceedings of the 29th Annual Symposium on User Interface Software and Technology; Association for Computing Machinery (ACM), New York, NY, USA, 16-20 October 2016; pp. 321–333.
- Sinex, J.E. Pulse oximetry: Principles and limitations. Am. J. Emerg. Med. 1999, 17, 59–66, doi:10.1016/s0735-6757(99)90019-0.
- Kim, J.; Gutruf, P.; Chiarelli, A.M.; Heo, S.Y.; Cho, K.; Xie, Z.; Banks, A.; Han, S.; Jang, K.-I.; Lee, J.W.; et al. Miniaturized Battery-Free Wireless Systems for Wearable Pulse Oximetry. Adv. Funct. Mater. 2017, 27, doi:10.1002/adfm.201604373.
- Nayak, G.; Puttamadappa, C.; Davide, O. Classification of Bio Optical signals using K- Means Clustering for Detection of Skin Pathology. Int. J. Comput. Appl. 2010, 1, 112–116, doi:10.5120/32-141.
- O’hara, G.J.; Phillips, D.B.; Diego, S. Method and Apparatus for Measuring Internal Body Temperature Utilizing Infrared Emissions. U.S. Patent 4,602,642, 23 October 1984.
- Togawa, T. Body temperature measurement. Clin. Phys. Physiol. Meas. 1985, 6, 83–108, doi:10.1088/0143-0815/6/2/001.
- He, Y.; Li, Y.; Bao, S.-D. Fall detection by built-in tri-accelerometer of smartphone. In Proceedings of the Proceedings of the 2012 IEEE-EMBS International Conference on Biomedical and Health Informatics, Hong Kong, China, 5–7 January 2012; pp. 184–187.
- Xiao, Z.G.; Menon, C. A Review of Force Myography Research and Development. Sensors 2019, 19, 4557, doi:10.3390/s19204557.
- Min, J.; Sempionatto, J.R.; Teymourian, H.; Wang, J.; Gao, W. Wearable electrochemical biosensors in North America. Biosens. Bioelectron. 2020, 172, 112750, doi:10.1016/j.bios.2020.112750.
- Chung, M.; Fortunato, G.; Radacsi, N. Wearable flexible sweat sensors for healthcare monitoring: A review. J. R. Soc. Interface 2019, 16, 20190217, doi:10.1098/rsif.2019.0217.
- Schmidt, A. Biosignals in human-computer interaction. Interactions 2015, 23, 76–79, doi:10.1145/2851072.
- Jakobsson, M. The Big Picture: Bioengineering Signals and Systems. Academic Press, 2012, Pages 3-33, ISBN 9780123849823.
- Moosavi, S.R.; Gia, T.N.; Nigussie, E.; Rahmani, A.M.; Virtanen, S.; Tenhunen, H.; Isoaho, J. End-to-end security scheme for mobility enabled healthcare Internet of Things. Future Gener. Comput. Syst. 2016, 64, 108–124, doi:10.1016/j.future.2016.02.020.
- Rahmani, A.; Gia, T.N.; Negash, B.; Anzanpour, A.; Azimi, I.; Jiang, M.; Liljeberg, P. Exploiting smart e-Health gateways at the edge of healthcare Internet-of-Things: A fog computing approach. Future Gener. Comput. Syst. 2018, 78, 641–658, doi:10.1016/j.future.2017.02.014.
- Khan, R.A.; Pathan, A.-S.K. The state-of-the-art wireless body area sensor networks: A survey. Int. J. Distrib. Sens. Netw. 2018, 14, doi:10.1177/1550147718768994.
- Hyun, W.; You, I.; Jang, J.; Leu, F.-Y. A Wireless Body Sensor Network and Its Applications: Rehearsal with a Smartphone. In Proceedings of the 2016 10th International Conference on Innovative Mobile and Internet Services in Ubiquitous Computing (IMIS), Fukuoka, Japan, 6–8 July 2016; pp. 415–418.
- Ellouze, F.; Fersi, G.; Jmaiel, M. Blockchain for Internet of Medical Things: A Technical Review. In International Conference on Smart Homes and Health Telematics; Springer: Cham, Switzerland, 2020; pp. 259–267.
- Lee, J.-S.; Su, Y.-W.; Shen, C.-C. A Comparative Study of Wireless Protocols: Bluetooth, UWB, ZigBee, and Wi-Fi. In Proceedings of the IECON 2007—33rd Annual Conference of the IEEE Industrial Electronics Society, Taipei, Taiwan, 5–8 November 2007; pp. 46–51.
- Rutronik. Determination of Practical Extremes of Bluetooth Low Energy: Throughput, Energy Consumption and Maximum Range. Available online: https://www.rutronik.com/fileadmin/Rutronik/Downloads/printmedia/products/06_wireless/bluetooth5.pdf (accessed on 24 November 2020).
- Woolley, M.; Schmidt, S. Bluetooth 5 Go Faster. Go Further; Bluetooth SIG: Kirkland, WA, USA, 2016; p. 25.
- Dementyev, A.; Hodges, S.; Taylor, S.; Smith, J. Power consumption analysis of Bluetooth Low Energy, ZigBee and ANT sensor nodes in a cyclic sleep scenario. In Proceedings of the 2013 IEEE International Wireless Symposium (IWS), Beijing, China, 14–18 April 2013; pp. 1–4.
- Kelsch, N. Green Power. Water Wastes Dig. 2013, 53, 1–7.
- Asan, N.B.; Velander, J.; Redzwan, S.; Perez, M.; Hassan, E.; Blokhuis, T.J.; Voigt, T.; Augustine, R. Effect of Thickness Inhomogeneity in Fat Tissue on In-Body Microwave Propagation. In Proceedings of the 2018 IEEE International Microwave Biomedical Conference (IMBioC), Philadelphia, PA, USA, 14–15 June 2018; pp. 136–138.
- Asan, N.B.; Hassan, E.; Perez, M.D.; Shah, S.R.M.; Velander, J.; Blokhuis, T.J.; Voigt, T.; Augustine, R. Assessment of Blood Vessel Effect on Fat-Intrabody Communication Using Numerical and Ex-Vivo Models at 2.45 GHz. IEEE Access 2019, 7, 89886–89900, doi:10.1109/access.2019.2926646.
- Ma, C.; Huang, Z.; Wang, Z.; Zhou, L.; Li, Y. An Energy Efficient Technique Using Electric Active Shielding for Capacitive Coupling Intra-Body Communication. Sensors 2017, 17, 2056, doi:10.3390/s17092056.
- Kang, T.; Oh, K.-I.; Park, H.; Kang, S. Review of capacitive coupling human body communications based on digital transmission. ICT Express 2016, 2, 180–187, doi:10.1016/j.icte.2016.11.002.
- Thielens, A.; Baumbauer, C.; Anderson, M.G.; Ting, J.; Arias, A.C.; Rabaey, J.M. Feasability of On-Body Backscattering in the UHF-RFID Band using Screen-Printed Dipole Antennas. In Proceedings of the 2019 13th International Symposium on Medical Information and Communication Technology (ISMICT), Oslo, Norway, 8–10 May 2019; pp. 1–5.




