
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mirko Pesce | + 2110 word(s) | 2110 | 2020-11-05 04:26:24 | | | |
| 2 | Dean Liu | -837 word(s) | 1273 | 2020-11-11 09:36:14 | | | | |
| 3 | Dean Liu | Meta information modification | 1273 | 2020-12-14 04:35:18 | | |
Video Upload Options
Alzheimer’s disease (AD) is a progressive neuronal/cognitional dysfunction, leading to disability and death. Despite advances in revealing the pathophysiological mechanisms behind AD, no effective treatment has yet been provided. It urges the need for finding novel multi-target agents in combating the complex dysregulated mechanisms in AD. Amongst the dysregulated pathophysiological pathways in AD, oxidative stress seems to play a critical role in the pathogenesis progression of AD, with a dominant role of nuclear factor erythroid 2-related factor 2 (Nrf2)/Kelch-like ECH-associated protein-1 (Keap1)/antioxidant responsive elements (ARE) pathway. In the present study, a comprehensive review was conducted using the existing electronic databases, including PubMed, Medline, Web of Science, and Scopus, as well as related articles in the field. Nrf2/Keap1/ARE has shown to be the upstream orchestrate of oxidative pathways, which also ameliorates various inflammatory and apoptotic pathways. So, developing multi-target agents with higher efficacy and lower side effects could pave the road in the prevention/management of AD. The plant kingdom is now a great source of natural secondary metabolites in targeting Nrf2/Keap1/ARE. Among natural entities, phenolic compounds, alkaloids, terpene/terpenoids, carotenoids, sulfur-compounds, as well as some other miscellaneous plant-derived compounds have shown promising future accordingly. Prevailing evidence has shown that activating Nrf2/ARE and downstream antioxidant enzymes, as well as inhibiting Keap1 could play hopeful roles in overcoming AD. The current review highlights the neuroprotective effects of plant secondary metabolites through targeting Nrf2/Keap1/ARE and downstream interconnected mediators in combating AD.
1. Introduction
Amongst the dysregulated pathophysiological pathways in AD, oxidative stress seems to play a critical role in the pathogenesis progression of AD, with a dominant role of nuclear factor erythroid 2-related factor 2 (Nrf2)/Kelch-like ECH-associated protein-1 (Keap1)/antioxidant responsive elements (ARE) pathway. In the present study, a comprehensive review was conducted using the existing electronic databases, including PubMed, Medline, Web of Science, and Scopus, as well as related articles in the field. Nrf2/Keap1/ARE has shown to be the upstream orchestrate of oxidative pathways, which also ameliorates various inflammatory and apoptotic pathways. So, developing multi-target agents with higher efficacy and lower side effects could pave the road in the prevention/management of AD. The plant kingdom is now a great source of natural secondary metabolites in targeting Nrf2/Keap1/ARE. Among natural entities, phenolic compounds, alkaloids, terpene/terpenoids, carotenoids, sulfur-compounds, as well as some other miscellaneous plant-derived compounds have shown promising future accordingly. Prevailing evidence has shown that activating Nrf2/ARE and downstream antioxidant enzymes, as well as inhibiting Keap1 could play hopeful roles in overcoming AD.
2. Nrf2/Keap1/ARE Pathway and Interconnected Mediators in AD
Oxidative stress seems to be an upstream orchestrate of neurodegeneration either by activating the inflammatory and apoptotic pathways in neurodegenerative diseases. Prevailing studies have shown the critical role of overactivated reactive oxygen species (ROS)/reactive nitrogen species (RNS) in causing cell death, during pathological conditions. Consequently, the oxidative phosphorylation of mitochondria, as well as parallel dysregulated pathways, could be major sources of produced ROS/RNS. Of those parallel hallmarks of AD, amyloid-beta (Aβ) plays a major role in activating oxidative mediators[1]. On the other hand, Nrf2 is now identified as a key upstream defence mediator against oxidative pathways. Essentially, Nrf2 is a member of the cap’n’collar (CNC) family of basic region-leucine zipper transcription factors, responsible for activating the antioxidant enzymes, including NAD(P)H quinone oxidoreductase-1 (NQO1), superoxide dismutase-1 (SOD1), heme oxygenase-1 (HO-1), glutathione S-transferase (GST), catalase (CAT), and glutathione peroxidase (GPx), as downstream antioxidant enzymes[2][3] In normal condition, Keap1 is a repressor protein that binds to an E3 ubiquitin ligase complex (Rbx-1) via cullin-3 to promote the degradation of Nrf2. However, in the situation of oxidative stress, Keap1 undergoes conformational changes toward the release of Nrf2 and subsequent activation of ARE. Several interconnected mediators could activate Nrf2 through phosphorylation and proceed antioxidative responses. Amongst those mediators, PI3K [4], c-Jun N-terminal kinase (JNK), extracellular regulated kinase (ERK) and mitogen-activated protein kinases (MAPKs) [5] are key kinases that phosphorylate Nrf2 and allowing to its nucleus transportation. On the other hand, some kinases are overactivated during the pathological conditions to degrade Nrf2 via Keap1-dependent and independent manners. For instance, glycogen synthase kinase 3-beta (GSK-3β) has been shown to make proteasomal degradation of Nrf2 [6], while p38 MAPK stabilizes the Keap1/Nrf2 interaction toward oxidative responses [5] GSK-3β plays a key role in the advancement/etiology of AD. From the mechanistic point of view, GSK-3β is in a near link with Aβ deposition and tau hyper-phosphorylation, which is associated with AD pathogenesis. Additionally, GSK-3β affects the oxidative stress, as a major hypothesis in AD. In this line, growing studies have paid to build a connection between GSK-3β and Nrf2 in AD pathology. Consistently, GSK-3β suppression was found to increase Nrf2 and some downstream AREs in brain cortex during AD[7]. Nowadays, usefulness of dual GSK-3β inhibitors/Nrf2 inducers are shown in various reports against AD.
From the inflammatory point of view, nuclear factor-κB (NF-κB) has also been shown to co-transported with Keap1 into nuclei regarding trapping Nrf2 [8]. Alternatively, Nrf2, in turn, could suppress inflammatory pathways through activating anti-inflammatory mediators (e.g., IL-10) and inhibiting inflammatory ones (TNF-α, IL-6, IL-1β). Besides, Nrf2 has also shown inhibitory effects on apoptotic mediators (e.g., Bax, Bad) and stimulatory effects on antiapoptotic factors (e.g., Bcl-2) [3]. In general, the Nrf2/ARE pathway is down-regulated within hippocampal neurons during AD [9]. Therefore, inducing Nrf2/ARE could be a valuable strategy for the treatment of AD.
Considering the multiple dysregulated pathways interconnected with Nrf2, providing multi-target therapeutic agents is of great importance. Figure 1 indicates the general view of Nrf2/Keap1/ARE and related interconnected mediators in AD.
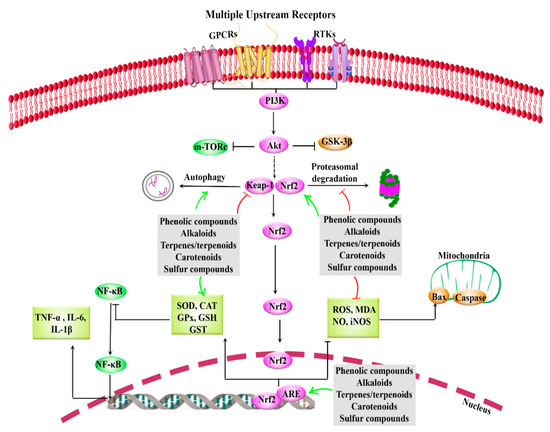
Figure 1. General overview of Nrf2/Keap1/ARE and interconnected pathways, how to be targeted by phytochemicals. Phytochemicals activate Nrf2, ARE (e.g., SOD, CAT, GPx, GSH, GST) and autophagy, while inhibits Keap1, oxidative mediators (e.g., ROS, MDA, NO, iNOS) and inflammation (IL, TNF-α, NF-κB). ↑ green: Activate or up-regulation, ⊥ red: inhibit or down-regulation, ARE: antioxidant response element, CAT: catalase, GPCRs: G protein-coupled receptors, GPx: glutathione peroxidase, GSH: glutathione, GSK-3β: glycogen synthase kinase 3-beta, GST: glutathione S-transferase, IL: interleukin, iNOS: inducible nitric oxide synthase, Keap1: Kelch-like ECH-associated protein-1, MDA: malondialdehyde, mTORc: mammalian target of rapamycin, NF-κB: nuclear factor-κB, NO: nitric oxide, Nrf2: nuclear factor erythroid 2-related factor 2, ROS: reactive oxygen species, RTKs: receptor tyrosine kinase, SOD: superoxide dismutase, TNF-α: tumor necrosis factor-α.
3. Conclusions
Compelling evidence has shown the key destructive role of oxidative stress in the pathogenesis of AD, along with the critical role of Nrf2/ARE in ameliorating neuronal/cognitional complications. The secondary metabolites of natural sources have found to be promising agents in targeting the aforementioned pathways/mediators in AD, possessing more efficacy/potency while lower side effects. In this regard, phenolic compounds, alkaloids, terpene/terpenoids, carotenoids, sulfur-compounds, as well as some other plant-derived miscellaneous compounds have been accordingly introduced as multi-target compounds in modulating several dysregulated mediators, especially those with a near interconnection with Nrf2/Keap1/ARE and related apoptotic/inflammatory pathways. In cognitive dysfunction, the aforementioned antioxidative pathway, seems to be in the upstream of either apoptotic (Bax and caspase) and inflammatory (TNF-α and ILs) mediators. So, attenuating Nrf2/Keap1/ARE could play a pivotal role in combating AD. Several clinical trials have also been provided to evaluate the therapeutic potential of phytochemicals based on their antioxidant activity. Despite their effectiveness, plant secondary metabolites often suffer from some pharmacokinetic limitations, including poor bioavailability, low solubility/selectivity, and week absorption rate, which urges the needs for developing novel delivery systems [9].
References
- Müller, W.E.; Eckert, A.; Kurz, C.; Eckert, G.P.; Leuner, K. Mitochondrial Dysfunction: Common Final Pathway in Brain Aging and Alzheimer’s Disease—Therapeutic Aspects. Mol. Neurobiol. 2010, 41, 159–171.
- Thimmulappa, R.K.; Mai, K.H.; Srisuma, S.; Kensler, T.W.; Yamamoto, M.; Biswal, S. Identification of Nrf2-regulated genes induced by the chemopreventive agent sulforaphane by oligonucleotide microarray. Cancer Res. 2002, 62, 5196–5203.
- Buendia, I.; Michalska, P.; Navarro, E.; Gameiro, I.; Egea, J.; León, R. Nrf2-ARE pathway: An emerging target against oxidative stress and neuroinflammation in neurodegenerative diseases. Pharmacol. Ther. 2016, 157, 84–104.
- Nakaso, K.; Yano, H.; Fukuhara, Y.; Takeshima, T.; Wada-Isoe, K.; Nakashima, K. PI3K is a key molecule in the Nrf2-mediated regulation of antioxidative proteins by hemin in human neuroblastoma cells. FEBS Lett. 2003, 546, 181–184.
- Keum, Y.-S.; Yu, S.; Chang, P.P.-J.; Yuan, X.; Kim, J.-H.; Xu, C.; Han, J.; Agarwal, A.; Kong, A.-N.T. Mechanism of Action of Sulforaphane: Inhibition of p38 Mitogen-Activated Protein Kinase Isoforms Contributing to the Induction of Antioxidant Response Element-Mediated Heme Oxygenase-1 in Human Hepatoma HepG2 Cells. Cancer Res. 2006, 66, 8804–8813.
- Chowdhry, S.; Zhang, Y.; McMahon, M.; Sutherland, C.; Cuadrado, A.; Hayes, J.D. Nrf2 is controlled by two distinct β-TrCP recognition motifs in its Neh6 domain, one of which can be modulated by GSK-3 activity. Oncogene 2012, 32, 3765–3781.
- Culbreth, M.; Aschner, M. GSK-3β, a double-edged sword in Nrf2 regulation: Implications for neurological dysfunction and disease. F1000Research 2018, 7, 1043.
- Yu, M.; Li, H.; Liu, Q.; Liu, F.; Tang, L.; Li, C.; Yuan, Y.; Zhan, Y.; Xu, W.; Li, W.; et al. Nuclear factor p65 interacts with Keap1 to repress the Nrf2-ARE pathway. Cell. Signal. 2011, 23, 883–892.
- Ramsey, C.P.; Glass, C.A.; Montgomery, M.B.; Lindl, K.A.; Ritson, G.P.; Chia, L.A.; Hamilton, R.L.; Chu, C.T.; Jordan-Sciutto, K.L. Expression of Nrf2 in Neurodegenerative Diseases. J. Neuropathol. Exp. Neurol. 2007, 66, 75–85.




