
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Leonel Pereira | + 2681 word(s) | 2681 | 2020-09-14 08:00:37 | | | |
| 2 | Peter Tang | -61 word(s) | 2620 | 2020-11-08 16:48:51 | | | | |
| 3 | Ana Marta Gonçalves | + 22 word(s) | 2642 | 2021-04-06 13:45:34 | | |
Video Upload Options
Seaweeds (marine macroalgae) are autotrophic organisms capable of producing many compounds of interest. For a long time, seaweeds have been seen as a great nutritional resource, primarily in Asian countries to later gain importance in Europe and South America, as well as in North America and Australia. It has been reported that edible seaweeds have a nutritional value along with a low caloric input and with the presence of proteins, minerals (iron and calcium), vitamins, structural polysaccharides (fibers), omega-6 and omega-3 unsaturated fatty acids. Moreover, they have plenty of bioactive molecules that can be applied in nutraceutical, pharmaceutical and cosmetic areas. There are historical registers of harvest and cultivation of seaweeds but with the increment of the studies of seaweeds and their valuable compounds, their aquaculture has increased. The methodology of cultivation varies from onshore to offshore. Seaweeds can also be part of integrated multi-trophic aquaculture (IMTA), which has great opportunities but is also very challenging to the farmers. This multidisciplinary field applied to the seaweed aquaculture is very promising to improve the methods and techniques; this area is developed under the denominated industry 4.0.
Aquaculture consists in the creation or culture of aquatic organisms (in this case, Marine Macroalgae or Seaweeds), through the application of techniques designed to increase the production of these same organisms, in addition to the natural capabilities of the environment
1. Introduction
Seaweeds are benthic organisms ubiquitously distributed along coasts from tropical to polar regions. They are part of Plantae kingdom, and, as land plants, seaweeds also constitute the basis of the food chain but in aquatic ecosystems [1]. Among the major primary producers, seaweeds or benthic marine algae grow in the intertidal and sub-tidal regions of the sea and contain photosynthetic pigments, which lead them to photosynthesize and produce food.
Seaweeds are grouped in three phyla: brown algae (Ochrophyta-Phaeophyceae), red algae (Rhodophyta) and green algae (Chlorophyta). These organisms are producers of many structural molecules (primary metabolites), such as proteins, lipids and carbohydrates, and they also produce other interesting bioactive compounds (secondary metabolites) that can have applications in many sectors (food, feed, agriculture, cosmetics, pharmaceutical andbiotechnological) [2].
Since elder times, seaweeds have been used as food in some civilizations around the world [3]. Furthermore, it has been reported that edible seaweeds are rich in proteins, lipids and dietary fibers [4][5][6]. The high levels of minerals and dietary fibers, as well as low lipid levels that characterize many seaweed species, make marine algae an attractive raw material for supplying bioactive substances with a wide range of applications [5][6]. In addition, the quality of their proteins [5][7][8] and antioxidant activities, associated with their content of polyphenolic compounds [9] and pigments (e.g., fucoxanthin [10]) turn seaweeds into an interesting source of bioactive substances used especially in human and animal nutrition. Seaweeds also contain high quantities of vitamins (A, K andB12), protective pigments, minerals and trace elements that are essential for the human diet and may collaborate with many EU-approved nutritional claims (such as iron, calcium, iodine or magnesium) relative to bone health, cognitive function, maintenance of normal metabolism, normal growth and muscle function, among others [6][11][12][13][14]. Polyunsaturated fatty acids (PUFAs), mainly omega 3 (ω-3) and omega (ω-6), are the principal components of their cell membranes, so seaweed can also be a source of essential fatty acids [15][16].
Many investigations demonstrated the nutraceutical, pharmaceutical and cosmeceutical value of the seaweeds. Some of their diverse properties are anti-cancer, antiviral, antifungal, antidiabetic, antihypertensive, immuno-modulatory, cytotoxic antibiotic, anticoagulant, anti-inflammatory, anti-parasitic, antioxidant, UV-protective and neuroprotective [2][13][17][18][19][20][21][22][23]. It has also been confirmed that several species of seaweed have powerful antioxidant compounds such as phlorotannines, carotenoids and sterols, making seaweed a source of compounds with possible neuroprotective effects, useful in the treatment of neurodegenerative diseases such as Parkinson's and Alzheimer's [24][25]. Sulfated polysaccharides from seaweed have shown important potential pharmacological uses, such as their anti-ulcer effects, by preventing adhesion of the infection caused by the bacteria Helicobacter pylori [26].
These marine organisms are normally used in the cosmetics sector as bioactive extracts, coloring agents, texturing stabilizers or emulsifiers and are a source of different compounds used in skincare [27]. Due to seaweeds being photosynthetic organisms, they generate compounds that absorb UV rays, such as carotenoids and terpenes, mycosporin-like amino acids (MAAs) and phenolic compounds, which are useful photo-protective elements for the formulation of sunscreens [28].
Thus, due to all these bioactivities and potential novel applications, seaweeds have been showcased as a sustainable resource for the future, which is leading to an increased demand of these organisms' exploitation and consequently also in their production. Moreover, the biological productivity of the seaweed causes photosynthetic carbon storage. This carbon can be immobilized in sediments or moved to the depths of the sea resulting in a CO2 sink. Thus, collecting algae and using them to produce biofuels and in other industries (food, feed, pharmaceuticals and fertilizers) can help in CO2 mitigation [29]. Seaweed can be used as carbon trap and then as fuel [29][30] and can provide a sustainable alternative source of biomass for the fuel production and also for chemicals, such as bioethanoland bio-butanol [31][32][33][34]. Furthermore, high levels of dissolved inorganic nutrients, such as nitrogen, phosphorous and carbon, are taken up by seaweed leading to the algal growth and helping to alleviate eutrophication in seas and oceans [2][35].
Several seaweeds are structuring species in coastal zones, changing the environment (by modifying light, sedimentation rates and hydrodynamics) [36][37][38][39]. Seaweeds are part of food webs and give ecosystem services such as habitats, food and refuge to a diversity of associated organisms (which are of conservation and economic importance) from different trophic levels (apex predators, fishes and invertebrates) [40][41] and therefore support biodiversity [42]. In addition, marine seaweeds contribute to the coastal defense by reducing the hydrodynamic energy from waves and by maintaining a high bed-level at tidal flats, thus protecting those tidal areas from erosion [43][44].
The demand for seaweeds and their products has been growing globally and so has the interest in their production and the attraction of stakeholders to invest more widely in the production of various algal species that may fill different economic sector needs [45]. This is extremely important to suppress the need to feed a growing population, on a planet where there will not be enough land for agricultural crops, as seaweed production does not compete for inland arable land, freshwater or agriculture fertilizers [46][47][48][49]. However, it does compete with other near-shore activities such as saliculture, fish and invertebrate's aquaculture or even agriculture. Fertilizers are only used in inland cultivation system, although they present a low percentage of usage in aquaculture, being seawater rich in nutrients from other species' aquacultures normally used [46][47][48][49].
Thus, seaweed aquaculture offers a variety of opportunities to mitigate and adapt to climate change and support biodiversity. However, there may be some negative impacts, such as the unintentional introduction of non-indigenous “hitchhiker” species, including pathogens [50].
2. Seaweeds Biodiversity and Potential to Exploitation
The principal phyla of seaweed are Chlorophyta (green algae), Ochrophyta-Phaeophyceae (brown algae) and Rhodophyta (red algae). Each phylum is composed of thousands of species [51]. Food, folk remedies, dyes and fertilizers traditionally use seaweed in their confection. In the early 1900s, seaweed components were launched industrially due to the development of mass food production [52].
In the nutraceutical, pharmaceutical and biotechnological industries, there are some applications to hydrocolloids, for instance alginate, carrageenan and agar are used due to their gelling features [53][54]. However, other minor components of the seaweeds, as will be presented later in this review, could be applied in high-value products, making seaweed aquaculture even more profitable for the seaweed producers [55][56][57][58]. During the past thirty years, enthusiasm has grown in seaweed as functional foods (nutraceuticals), enabling dietary advantages superior to their macronutrient content. Furthermore, to produce therapeutic products, seaweeds have been targeted for the obtention of metabolites with biological activity [55][58].
Despite all research studies performed in this field to demonstrate the bioactivities of seaweed-derived compounds, there is not the same expression in effective products on the market [56][57]. Consequently, more research and standardized assays need to be done, where the main questions are the compound bioavailability in seaweed, the low efficiency and efficacy of the extraction and the isolation and characterization of the biomolecules [59][60][61]. Some compounds could be difficult to isolate due to their biochemical features (e.g., size, molecular weight, structural similarities or even the tendency to bind or react with other molecules) [62].
However, seaweeds are viewed as promising functional foods and as food supplements [63][64], where the lower heavy metals concentration safeguard needs to be assured. Nevertheless, there is a need for more research to clarify the seaweed state, such as their role in nutrition and disease prevention [18]. However, there are various seaweeds' compounds commercially available, where the seaweeds' polysaccharides represent a large portion of that market, used for various industries, such as food and pharmaceutical [65][66][67][68]. The seaweed polysaccharides are considered dietary fibers, although assay with vegetal jelly (carrageenan) has proven to reduce cholesterol [69][70]. In the case of proteins, the research is ambiguous regarding the digestibility, due to the interaction of the proteins with other compounds [69]. They present a low concentration of lipids, despite the amount of ω-6 and ω-3 [71][72]. Moreover, the seaweed mineral content is the most important because minerals are essential for the human cells to work properly [3][63][64][73][74].
Nevertheless, there is the need to execute further in vivo and clinical studies to guarantee that the selected raw materials maintain the great potential and are safe, as well as to perform accurate controls throughout all the production phases of industrial batches [75].
3. Seaweed Aquaculture: Global Overview
Over the past 70 years, seaweed farming technologies significantly developed in Asia, and, more recently, they have also gained position in the Americas and Europe [1][76] (see Figure 1).
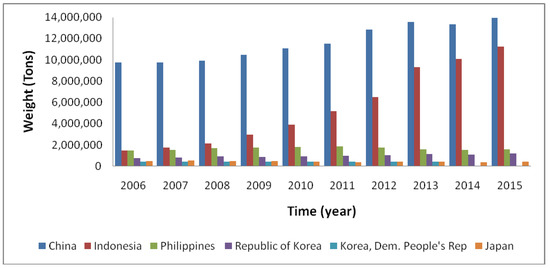
Figure 1. Global seaweed culture production by the main country producers, in tons. Adapted from FAO—The global status of seaweed production, trade and utilization, 2018 [1].
There are historical registers of large-scale cultivation of seaweeds in Asia for decades [77]; however, in Europe and in other parts of the globe, this is a recent commercial activity [78][79].
The global annual production of seaweeds does not stop growing, reaching, in 2016, 31.2 million tons (fresh weight) [1]. Of this, just 3.5% was harvested from natural populations, in the time that 96.5% was produced in aquaculture, representing 27% of the worlds' total aquaculture production [80]. The majority of this production happened in China, Indonesia and other Asian countries (47.9%, 38.7% and 12.8% of the worldwide production in 2016, respectively), mainly for human food and food additives [80]. The total aquaculture production of seaweeds exceeded more than the double in the last 20 years [1], and the total potential has been suggested to be 1000–100,000 million tons [81], but the main practice outside Asia is still to harvest natural stocks [57].
Besides the developments in seaweed aquaculture in countries such as China, Japan, Korea, Indonesia and the Philippines, there are also pilot-scale and pre-commercial farming projects for selected brown and red algae in Europe [82][83][84][85]; Latin America, for instance in Chile [86][87] and in Brazil [88]; the USA [89]; and parts of Africa [90].
Whereas the increasing global efforts to develop these farms, seaweed production and its commercialization strategies differ within the countries, as in the East there is a higher demand for edible seaweed as a direct food product, which produces higher incomes for farmers than the resources obtained from seaweeds'application in the polysaccharide industry in Western countries [91].
4. Seaweed Aquaculture: The Aquaculture 4.0
Seaweed farming has developed as one of the alternatives to not exploit natural resources. At this moment, it is economically important in Asia and has a growing importance in Europe. The widespread potential of seaweeds application areas is comparable to other natural supplies such as palm oil and cocoa. Seaweeds are applied in product areas, such as cosmetic, medicine, biopolymers, food or even as a natural source to CO2 sink and biomass energy source [92]. The worldwide requisition to produce large amounts of seaweed will grow in the next years; however, until nowadays, there is still a continuous cultivation system optimization to deliver to this growing demand, a sustainable seaweed production and of their compounds [57][87][91][93][92].
However, collaborative work between academia and the aquaculture industry through research and development centers (R&D) has led to the development of research initiatives together to find new opportunities and new technologies to improve the efficiency and productivity in the seaweed aquaculture systems, making them more eco-sustainable and fit for the blue economy [94].
Camus et al. addressed some of the main problems that have an impact in the seaweed cultivation strain selection programs: the development of new massive plantlet production independent of collecting reproductive material every cycle; disease research; research on environmental impacts of large-scale cultures; and added value to the farmed species [87].
However, in Asia, the seaweed cultivation suffered a rapid evolution at the technological advances mainly in the floating raft cultivation systems, mainly for important species to the human consumption [95][96][97]. The major problem in the offshore aquaculture is the growth of juveniles in the sexual reproduction of selected species, for example kelps and Porphyra/Pyropia sp. This problem presents an expensive cost in the production chain, where the bigger scale can make this process affordable [98]. There is a need to develop reliable technology and cultivation strategies to achieve profitability [87][98][99]. Here, kelp cultivation is the most developed cultivation methodology system, due to the high interest in alginates and for human food [84][98].
There is a real need for the optimization of the current onshore seaweed cultivation techniques for the seaweed production [91][92][94]. The existing offshore cultivation system is not yet appropriate for setting out in deep-water or in the open water area, since the conceited aquaculture system is used in sheltered areas, and thus it is not possible to support more aggressive mechanical conditions. Consequently, the current onshore and offshore cultivation systems are not yet environmentally sustainable, and they are economically unstable, because the production fluctuates very rapidly, due to the impact of abiotic and biotic factors [57][84][92].
5. Conclusions
The interest in seaweeds is increasing due to all the derived compounds and their bioactivities. They could have applications in nutraceutical and pharmaceutical products. There is an urge to produce and harvest more seaweed, in order to answer the higher demand of seaweeds and seaweed-based products. The scarce quantity of cultivated seaweed causes a real danger for wild seaweed populations due to the commercial over exploitation, causing huge marine environmental concerns. Thus, there is a need to provide more reliable aquaculture systems, in various formats (inland, nearshore andoffshore cultivations).
Several industries can use only one compound from seaweed and the science is evolving to understand how the seaweeds' metabolism works, to obtain the best amount of the compound in aquaculture. Subsequently, the seaweed aquaculture technologies have been developed dramatically over the past 70 years mostly in Asia and more recently in the Americas and Europe. However, there are still countless challenges to surpass with reverence to the science and to social acceptability. In addition, in seaweed cultivation in various points of the world, it is still hard to use feasible and sustainable methodology and be economically and productive. The seaweed aquaculture has a long road for optimization.
The present main tasks in seaweed aquaculture embrace the development of disease resistance, fast growth seaweed species, high concentration of desired molecules methodologies and technologies, and the improvement of the aquaculture systems to be more robust and cost efficient that can resist storm events and maintain the cultivation during more time.
Progress in new cultivation technologies that can be more efficient and eco-friendlier is very important, so there is a need to have a multi and interdisciplinary team to optimize the aquaculture to the perfection to reduce the risks involved in the seaweed aquaculture and enhance new and better aquaculture systems and seaweed quality. In this way, the farmer can gain more control of the aquaculture systems. However, caution is needed to not over exploit the ecosystem sustainability, due to the danger of over-dosage of aquaculture with chemical fertilizers or other compounds that can and will reduce the water quality and damage the ecosystem, thus IMTA appears to be the best solution in terms of sustainability and profit.
References
- Ferdouse, F.; Holdt, S.L.; Smith, R.; Murúa, P.; Yang, Z. The global status of seaweed production, trade and utilization. FAO Globefish Res. Programme2018, 124, 120.
- Leandro, A.; Pereira, L.; Gonçalves, A.M.M. Diverse Applications of Marine Macroalgae. Mar. Drugs2019, 18, 17.
- Pereira, L. Edible Seaweeds of the World; CRC Press: Boca Raton, FL, USA, 2016.
- Sánchez-Machado, D.I.; López-Cervantes, J.; López-Hernández, J.; Paseiro-Losada, P. Fatty acids, total lipid, protein and ash contents of processed edible seaweeds. Food Chem.2004, 85, 439–444.
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem.2007, 103, 891–899.
- Macartain, P.; Gill, C.I.R.; Brooks, M.; Campbell, R.; Rowland, I.R. Special Article Nutritional Value of Edible Seaweeds. Nutr. Rev.2007, 65, 535–543.
- Fleurence, J. Seaweed proteins. In Proteins in Food Processing; Woodhead Publishing Limited: Cambridge, UK, 2004; pp. 197–213.
- Mæhre, H.K.; Malde, M.K.; Eilertsen, K.; Elvevoll, E.O. Characterization of protein, lipid and mineral contents in common Norwegian seaweeds and evaluation of their potential as food and feed. J. Sci. Food Agric.2014, 94, 3281–3290.
- Wang, T.; Jónsdóttir, R.; Liu, H.; Gu, L.; Kristinsson, H.G.; Raghavan, S.; Ólafsdóttir, G. Antioxidant capacities of phlorotannins extracted from the brown algae fucus vesiculosus. J. Agric. Food Chem.2012, 60, 5874–5883.
- Fung, A.; Hamid, N.; Lu, J. Fucoxanthin content and antioxidant properties of Undaria pinnatifida. Food Chem.2013, 136, 1055–1062.
- Mabeau, S.; Fleurence, J. Seaweed in food products: Biochemical and nutritional aspects. Trends Food Sci. Technol.1993, 4, 103–107.
- Heo, S.J.; Park, E.J.; Lee, K.W.; Jeon, Y.J. Antioxidant activities of enzymatic extracts from brown seaweeds. Bioresour. Technol.2005, 96, 1613–1623.
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal chemodiversity and bioactivity: Sources of natural variability and implications for commercial application. Biotechnol. Adv.2011, 29, 483–501.
- Barbier, M.; Charrier, B.; Araujo, R.; Holdt, S.L.; Jacquemin, B.; Rebours, C. PEGASUS-PHYCOMORPH European Guidelines for a Sustainable Aquaculture of Seaweeds; COST Action FA1406, Roscoff, France, 2019.
- Mišurcová, L.; Ambrožová, J.; Samek, D. Seaweed lipids as nutraceuticals. Adv. Food Nutr. Res.2011, 64, 339–355.
- Pereira, H.; Barreira, L.; Figueiredo, F.; Custódio, L.; Vizetto-Duarte, C.; Polo, C.; Rešek, E.; Aschwin, E.; Varela, J. Polyunsaturated fatty acids of marine macroalgae: Potential for nutritional and pharmaceutical applications. Mar. Drugs2012, 10, 1920–1935.
- Francisco, M.E.Y.; Erickson, K.L. Ma’iliohydrin, a cytotoxic chamigrene dibromohydrin from a Philippine Laurencia species. J. Nat. Prod.2001, 64, 790–791.
- Smit, A.J. Medicinal and pharmaceutical uses of seaweed natural products: A review. J. Appl. Phycol.2004, 16, 245–262.
- Dhargalkar, V.K.; Verlecar, X.N. Southern Ocean seaweeds: A resource for exploration in food and drugs. Aquaculture2009, 287, 229–242.
- Mayer, A.M.S.; Rodríguez, A.D.; Taglialatela-Scafati, O.; Fusetani, N. Marine Pharmacology in 2009–2011 : Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms. Mar. Drugs2013, 11, 2510–2573.
- Yuan, Y.; Athukorala, Y. Red Algal Mycosporine-Like Amino Acids (MAAs) as Potential Cosmeceuticals. In Marine Cosmeceuticals; CRC Press: Boca Raton, FL, USA, 2011; pp. 143–168.
- Pereira, L. Therapeutic and Nutritional Uses of Algae; CRC Press: Boca Raton, FL, USA, 2018.
- Ruan, B. A Review of the components of seaweeds as potential candidates in cancer therapy. Anticancer. Agents Med. Chem.2018, 18, 354–366.
- Pangestuti, R.; Kim, S.K. Neuroprotective effects of marine algae. Mar. Drugs2011, 9, 803–818.
- Barbosa, M.; Valentão, P.; Andrade, P.B. Bioactive compounds from macroalgae in the new millennium: Implications for neurodegenerative diseases. Mar. Drugs2014, 12, 4934–4972.
- Besednova, N.N.; Zaporozhets, T.S.; Somova, L.M.; Kuznetsova, T.A. Review: Prospects for the use of extracts and polysaccharides from marine algae to prevent and treat the diseases caused by Helicobacter pylori. Helicobacter2015, 20, 89–97.
- Pimentel, F.; Alves, R.; Rodrigues, F.; Oliveira, M.B.P.P. Macroalgae-Derived Ingredients for Cosmetic Industry—An Update. Cosmetics2017, 5, 2.
- Guillerme, J.; Couteau, C.; Coiffard, L. Applications for marine resources in cosmetics. Cosmetics2017, 4, 35.
- Duarte, C.M.; Wu, J.; Xiao, X.; Bruhn, A.; Krause-Jensen, D. Can seaweed farming play a role in climate change mitigation and adaptation? Front. Mar. Sci.2017, 4, 1–8.
- Daroch, M.; Geng, S.; Wang, G. Recent advances in liquid biofuel production from algal feedstocks. Appl. Energy2013, 102, 1371–1381.
- Enquist-Newman, M.; Faust, A.M.E.; Bravo, D.D.; Santos, C.N.S.; Raisner, R.M.; Hanel, A.; Sarvabhowman, P.; Le, C.; Regitsky, D.D.; Cooper, S.R.; et al. Efficient ethanol production from brown macroalgae sugars by a synthetic yeast platform. Nature2014, 505, 239–243.
- Kraan, S. Mass-cultivation of carbohydrate rich macroalgae, a possible solution for sustainable biofuel production. Mitig. Adapt. Strateg. Glob. Chang. 2013, 18, 27–46.
- Potts, T.; Du, J.; Paul, M.; May, P.; Beitle, R.; Hestekin, J. The production of butanol from Jamaica Bay Macro Algae. Environ. Prog. Sustain. Energy2012, 31, 29–36.
- Wei, N.; Quarterman, J.; Jin, Y.S. Marine macroalgae: An untapped resource for producing fuels and chemicals. Trends Biotechnol.2013, 31, 70–77.
- Talep, M. Desintegra.me. Available online: https://margaritatalep.com/ (accessed on 9 March 2020).
- Reisewitz, S.E.; Estes, J.A.; Simenstad, C.A. Indirect food web interactions: Sea otters and kelp forest fishes in the Aleutian Archipelago. Oecologia2006, 146, 623–631.
- Leclerc, J.-C.; Riera, P.; Leroux, C.; Lévêque, L.; Laurans, M.; Schaal, G.; Davoult, D. Trophic significance of kelps in kelp communities in Brittany (France) inferred from isotopic comparisons. Mar. Biol.2013, 160, 3249–3258.
- Smale, D.A.; Burrows, M.T.; Moore, P.; O’Connor, N.; Hawkins, S.J. Threats and knowledge gaps for ecosystem services provided by kelp forests: A northeast Atlantic perspective. Ecol. Evol.2013, 3, 4016–4038.
- Bertocci, I.; Araújo, R.; Oliveira, P.; Sousa-Pinto, I. Potential effects of kelp species on local fisheries. J. Appl. Ecol. 2015, 52, 1216–1226.
- Almanza, V.; Buschmann, A.H. The ecological importance of Macrocystis pyrifera (Phaeophyta) forests towards a sustainable management and exploitation of chilean coastal benthic co-management areas. Int. J. Environ. Sustain. Dev.2013, 12, 341–360.
- Vásquez, J.A.; Zuñiga, S.; Tala, F.; Piaget, N.; Rodríguez, D.C.; Vega, J.M.A. Economic valuation of kelp forests in northern Chile: Values of goods and services of the ecosystem. J. Appl. Phycol.2013, 26, 1081–1088.
- Skjermo, J.; Aasen, I.M.; Arff, J.; Broch, O.J.; Carvajal, A.; Forbord, S.; Olsen, Y.; Reitan, K.I.; Rustad, T.; Sandquist, J.; et al. A new Norwegian bioeconomy based on cultivation and processing of seaweeds: Opportunities and R & D needs. SINTEF Fisheries and Aquaculture: Trondheim, Norway, 2014.
- Christianen, M.J.A.; Van Belzen, J.; Herman, P.M.J.; Van Katwijk, M.M.; Lamers, L.P.M.; Van Leent, P.J.M.; Bouma, T.J. Low-canopy seagrass beds still provide important coastal protection services. PLoS ONE2013, 8, e62413.
- Ondiviela, B.; Losada, I.J.; Lara, J.L.; Maza, M.; Galván, C.; Bouma, T.J.; van Belzen, J. The role of seagrasses in coastal protection in a changing climate. Coast. Eng.2014, 87, 158–168.
- Ashkenazi, D.Y.; Israel, A.; Abelson, A. A novel two-stage seaweed integrated multi-trophic aquaculture. Rev. Aquac.2019, 11, 246–262.
- Charrier, B.; Abreu, M.H.; Araujo, R.; Bruhn, A.; Coates, J.C.; De Clerck, O.; Katsaros, C.; Robaina, R.R.; Wichard, T. Furthering knowledge of seaweed growth and development to facilitate sustainable aquaculture. New Phytol.2017, 216, 967–975.
- Fernand, F.; Israel, A.; Skjermo, J.; Wichard, T.; Timmermans, K.R.; Golberg, A. Offshore macroalgae biomass for bioenergy production: Environmental aspects, technological achievements and challenges. Renew. Sustain. Energy Rev.2017, 75, 35–45.
- FAO.The State of World Fisheries and Aquaculture-Contributing to Food Security and Nutrition for All; FAO: Rome, Italy, 2016.
- FAO.The State of the World Fisheries and Aquaculture-Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018; Volume 3.
- Cottier-Cook, E.J.; Nagabhatla, N.; Badis, Y.; Campbell, M.; Chopin, T.; Dai, W.; Fang, J.; He, P.; Hewitt, C.; Kim, G.H.; et al. Safeguarding the future of the global seaweed aquaculture industry. In United Nations University and Scottish Association for Marine Science Policy Brief; United Nations University Institue of Water, Environment and Health: Hamilton, ON, Canada, 2016; p. 12.
- Rindi, F.; Soler-Vila, A.; Guiry, M.D. Taxonomy of Marine Macroalgae Used as Sources of Bioactive Compounds; Springer: Berlin/Heidelberg, Germany, 2011; Volume 9781461412.
- Shannon, E.; Abu-Ghannam, N. Seaweeds as nutraceuticals for health and nutrition. Phycologia2019, 58, 563–577.
- Rhein-Knudsen, N.; Ale, M.T.; Meyer, A.S. Seaweed hydrocolloid production: An update on enzyme assisted extraction and modification technologies. Mar. Drugs2015, 13, 3340–3359.
- Zollmann, M.; Robin, A.; Prabhu, M.; Polikovsky, M.; Gillis, A.; Greiserman, S.; Golberg, A. Green technology in green macroalgal biorefineries. Phycologia2019, 58, 516–534.
- Davis, G.D.J.; Vasanthi, A.H.R. Seaweed metabolite database (SWMD): A database of natural compounds from marine algae. Bioinformation2011, 5, 361–364.
- Mazarrasa, I.; Olsen, Y.S.; Mayol, E.; Marbà, N.; Duarte, C.M. Global unbalance in seaweed production, research effort and biotechnology markets. Biotechnol. Adv.2014, 32, 1028–1036.
- Buschmann, A.H.; Camus, C.; Infante, J.; Neori, A.; Israel, Á.; Hernández-González, M.C.; Pereda, S.V.; Gomez-Pinchetti, J.L.; Golberg, A.; Tadmor-Shalev, N.; et al. Seaweed production: Overview of the global state of exploitation, farming and emerging research activity. Eur. J. Phycol.2017, 52, 391–406.
- Zerrifi, S.E.A.; Khalloufi, F.E.; Oudra, B.; Vasconcelos, V. Seaweed bioactive compounds against pathogens and microalgae: Potential uses on pharmacology and harmful algae bloom control. Mar. Drugs2018, 16, 55.
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci.2010, 11, 1321–1342.
- Zubia, M.; Freile-Pelegrín, Y.; Robledo, D. Photosynthesis, pigment composition and antioxidant defences in the red alga Gracilariopsis tenuifrons (Gracilariales, Rhodophyta) under environmental stress. J. Appl. Phycol.2014, 26, 2001–2010.
- Cotas, J.; Leandro, A.; Pacheco, D.; Gonçalves, A.M.M.; Pereira, L. A comprehensive review of the nutraceutical and therapeutic applications of red seaweeds (Rhodophyta). Life2020, 10, 19.
- Mekinić, I.G.; Skroza, D.; Šimat, V.; Hamed, I.; Čagalj, M.; Perković, Z.P. Phenolic content of brown algae (Pheophyceae) species: Extraction, identification, and quantification. Biomolecules 2019, 9, 244.
- Rajapakse, N.; Kim, S.K. Nutritional and Digestive Health Benefits of Seaweed, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2011; Volume 64.
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev.2019, 77, 307–329.
- Pereira, L. A review of the nutrient composition of selected edible seaweed. In Nutrition and Food Science; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 15–47.
- Pereira, L.; Gheda, S.F.; Ribeiro-claro, P.J.A. Analysis by vibrational spectroscopy of seaweed polysaccharides with potential use in food, pharmaceutical, and cosmetic industries. Int. J. Carbohydr. Chem.2013, 2013, 7.
- Arunkumar, K. Extraction, Isolation, and Characterization of Alginate. In Industrial Applications of Marine Biopolymers; Taylor & Francis Group, Ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 19–35.
- Pereira, L. Biological and therapeutic properties of the seaweed polysaccharides. Int. Biol. Rev.2018, 2, 1–50.
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597.
- Valado, A.; Pereira, M.; Caseiro, A.; Figueiredo, J.P.; Loureiro, H.; Almeida, C.; Cotas, J.; Pereira, L. Effect of carrageenans on vegetable jelly in humans with hypercholesterolemia. Mar. Drugs2020, 18, 1–11.
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother.2002, 56, 365–379.
- Simopoulos, A.P. An increase in the Omega-6/Omega-3 fatty acid ratio increases the risk for obesity. Nutrients2016, 8, 128.
- Commitee on Diet and Health and National Research Council. Diet and Health: Implications for Reducing Chronic Disease Risk; Motulsky, A.G., Ed.; National Academies Press: Washington, DC, USA, 1989.
- Bouga, M.; Combet, E. Emergence of seaweed and seaweed-containing foods in the UK: Focus on labeling, iodine content, toxicity and nutrition. Foods2015, 4, 240–253.
- Wang, H.M.D.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresour. Technol.2015, 184, 355–362.
- Kim, J.K.; Yarish, C.; Hwang, E.K.; Park, M.; Kim, Y. Seaweed aquaculture: Cultivation technologies, challenges and its ecosystem services. Algae2017, 32, 1–13.
- Cheng, T.-H. Production of kelp—A major aspect of China’s exploitation of the sea. Econ. Bot.1969, 23, 215–236.
- FAO. The State of World Fisheries and Aquaculture—Opportunities and Challenges; FAO: Rome, Italy, 2014.
- Bostock, J.; Lane, A.; Hough, C.; Yamamoto, K. An assessment of the economic contribution of EU aquaculture production and the influence of policies for its sustainable development. Aquac. Int.2016, 24, 699–733.
- Goecke, F.; Klemetsdal, G.; Ergon, Å. Cultivar development of kelps for commercial cultivation—Past lessons and future prospects. Front. Mar. Sci.2020, 8, 1–17.
- Lehahn, Y.; Nivrutti, K.; Golberg, A. Global potential of offshore and shallow waters macroalgal biorefineries to provide for food, chemicals and energy: Feasibility and sustainability. Algal Res.2016, 17, 150–160.
- Callaway, R.; Shinn, A.P.; Grenfell, S.E.; Bron, J.E.; Burnell, G.; Cook, E.J.; Crumlish, M.; Culloty, S.; Davidson, K.; Ellis, R.P.; et al. Review of climate change impacts on marine aquaculture in the UK and Ireland. Aquat. Conserv. Mar. Freshw. Ecosyst.2012, 22, 389–421.
- Hughes, A.D.; Kelly, M.S.; Black, K.D.; Stanley, M.S. Biogas from macroalgae: Is it time to revisit the idea? Biotechnol. Biofuels2012, 5, 1–7.
- Peteiro, C.; Sánchez, N.; Martínez, B. Mariculture of the Asian kelp Undaria pinnatifida and the native kelp Saccharina latissima along the Atlantic coast of Southern Europe: An overview. Algal Res.2016, 15, 9–23.
- Stévant, P.; Rebours, C.; Chapman, A. Seaweed aquaculture in Norway: Recent industrial developments and future perspectives. Aquac. Int.2017, 25, 1373–1390.
- Buschmann, A.H.; Prescott, S.; Potin, P.; Faugeron, S.; Vásquez, J.A.; Camus, C.; Infante, J.; Hernández-gonzález, M.C.; Gutíerrez, A.; Varela, D.A. The status of kelp exploitation and marine agronomy, with emphasis on Macrocystis pyrifera, in Chile. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2014; Volume 71, pp. 161–188.
- Camus, C.; Infante, J.; Buschmann, A.H. Overview of 3 year precommercial seafarming of Macrocystis pyrifera along the Chilean coast. Rev. Aquac.2016, 3, 543–559.
- Pellizzari, F.; Reis, R.P. Seaweed cultivation on the Southern and Southeastern Brazilian Coast. Brazilian J. Pharmacogn.2011, 21, 305–312.
- Augyte, S.; Yarish, C.; Redmond, S.; Kim, J.K. Cultivation of a morphologically distinct strain of the sugar kelp, Saccharina latissima forma angustissima, from coastal Maine, USA, with implications for ecosystem services. J. Appl. Phycol.2017, 29, 1967–1976.
- Msuya, F.E. The impact of seaweed farming on the socioeconomic status of coastal communities in Zanzibar, Tanzania. World Aquac.2011, 42, 45–48.
- Hafting, J.T.; Craigie, J.S.; Stengel, D.B.; Loureiro, R.R.; Buschmann, A.H.; Yarish, C.; Edwards, M.D.; Critchley, A.T. Prospects and challenges for industrial production of seaweed bioactives. J. Phycol.2015, 51, 821–837.
- Sulaiman, O.O.; Magee, A.; Nik, W.B.W.; Saharuddin, A.H.; Kader, A.S.A. Design and Model Testing of Offshore Acquaculture Floating Structure for Seaweed Oceanic Plantation. Biosci. Biotechnol. Res. Asia2012, 9, 477–494.
- Knowler, D.; Chopin, T.; Martínez‐Espiñeira, R.; Neori, A.; Nobre, A.; Noce, A.; Reid, G. The economics of Integrated Multi‐Trophic Aquaculture: Where are we now and where do we need to go? Rev. Aquac. 2020, 8, doi:10.1111/raq.12399.
- Hafting, J.T.; Critchley, A.T.; Cornish, M.L.; Hubley, S.A.; Archibald, A.F. On-land cultivation of functional seaweed products for human usage. J. Appl. Phycol.2012, 24, 385–392.
- Pereira, R.; Yarish, C. Mass Production of Marine Macroalgae. Encycl. Ecol. 2008, 5, 2236–2247.
- Shan, T.F.; Pang, S.J.; Li, J.; Gao, S.Q. Breeding of an elite cultivar Haibao no. 1 of Undaria pinnatifida (phaeophyceae) through gametophyte clone crossing and consecutive selection. J. Appl. Phycol.2016, 28, 2419–2426.
- Zhao, X.B.; Pang, S.J.; Liu, F.; Shan, T.F.; Li, J.; Gao, S.Q.; Kim, H.G. Intraspecific crossing of Saccharina japonica using distantly related unialgal gametophytes benefits kelp farming by improving blade quality and productivity at Sanggou Bay, China. J. Appl. Phycol.2016, 28, 449–455.
- Correa, T.; Gutiérrez, A.; Flores, R.; Buschmann, A.H.; Cornejo, P.; Bucarey, C. Production and economic assessment of giant kelp Macrocystis pyrifera cultivation for abalone feed in the south of Chile. Aquac. Res.2016, 47, 698–707.
- Zuniga-Jara, S.; Marín-Riffo, M.C.; Bulboa-Contador, C. Bioeconomic analysis of giant kelp Macrocystis pyrifera cultivation (Laminariales; Phaeophyceae) in northern Chile. J. Appl. Phycol.2016, 28, 405–416.





