
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Remco De Bree | + 440 word(s) | 440 | 2020-11-03 08:44:45 | | | |
| 2 | Vivi Li | -9 word(s) | 431 | 2020-11-04 03:34:15 | | | | |
| 3 | Vivi Li | + 151 word(s) | 591 | 2020-11-04 03:35:49 | | | | |
| 4 | Vivi Li | Meta information modification | 591 | 2020-11-04 11:17:01 | | | | |
| 5 | Vivi Li | + 265 word(s) | 856 | 2020-11-09 09:14:30 | | |
Video Upload Options
Sentinel lymph node biopsy (SLNB) is a diagnostic staging procedure that aims to identify the first draining lymph node(s) from the primary tumor, the sentinel lymph nodes (SLN), as their histopathological status reflects the histopathological status of the rest of the nodal basin. The routine SLNB procedure consists of peritumoral injections with a technetium-99m [99mTc]-labelled radiotracer followed by lymphoscintigraphy and SPECT-CT imaging. Based on these imaging results, the identified SLNs are marked for surgical extirpation and are subjected to histopathological assessment. The routine SLNB procedure has proven to reliably stage the clinically negative neck in early-stage oral squamous cell carcinoma (OSCC). However, an infamous limitation arises in situations where SLNs are located in close vicinity of the tracer injection site. In these cases, the hotspot of the injection site can hide adjacent SLNs and hamper the discrimination between tracer injection site and SLNs (shine-through phenomenon). Therefore, technical developments are needed to bring the diagnostic accuracy of SLNB for early-stage OSCC to a higher level.
1. Introduction
In early-stage (cT1-2N0) oral squamous cell carcinoma (OSCC), occult lymph node metastases are present in 20–30% of patients, even when the status of the regional lymph nodes has been evaluated using combinations of advanced clinical diagnostic imaging modalities (i.e., ultrasound guided fine-needle aspiration (USgFNA), magnetic resonance imaging (MRI) and/or computed tomography (CT)) [1][2][3]. As watchful-waiting in these patients has been associated with a poor prognosis, especially when compared to those in whom the clinically negative neck was electively treated [1], two strategies for the clinically negative neck in early-stage OSCC are available: elective neck dissection (END) and sentinel lymph node biopsy (SLNB) [3][4][5][6]. Although END is the strategy of choice in the majority of medical centers globally [5], which has the benefit of being a single-stage procedure, SLNB is a less invasive procedure for the 70–80% of patients without metastatic neck involvement and has overall lower morbidity rates, better quality-of-life and lower health-care costs as compared to END [7][8][9][10].
The concept of SLNB is based on the premise that lymph flow from the primary tumor travels sequentially to the sentinel lymph node (SLN) and then on to the other regional lymph nodes. Hence, the SLN is the lymph node that has the highest risk of harboring metastasis [11].
2. SLNB procedure
The SLNB procedure aims to identify these first draining lymph node(s), as their histopathological status reflects the histopathological status of the rest of the nodal basin. Complementary nodal treatment (e.g., surgery, radiotherapy) should be performed in case of metastatic involvement of SLN(s). A negative SLNB, however, would justify a wait-and-scan policy [12].
In short, the routine SLNB procedure consists of preoperative peritumoral injections with a technetium-99m [99mTc; γ-emitter]-labelled radiotracer followed by planar dynamic and static lymphoscintigraphy including SPECT-CT (single photon emission computed tomography-computed tomography) imaging. Based on preoperative lymphoscintigraphy, the position of the SLN(s) is marked on the skin. The marked SLNs are surgically removed, using a portable γ-probe for intraoperative localization of SLNs. Subsequently, the harvested SLNs are subjected to meticulous histopathological assessment using step-serial-sectioning and immunohistochemistry [12,13,14,15].
SLNB has proven to reliably stage the clinically negative neck in early-stage OSCC with a pooled sensitivity and negative predictive value (NPV) of 87% and 94%, respectively [11]. However, an infamous limitation of the routine SLNB procedure arises in situations where SLNs are located in close vicinity of the tracer injection site. In these cases, the hotspot of the injection site can hide adjacent SLNs, which consequently hampers the discrimination between tracer injection site and SLNs (shine-through phenomenon; Figure 1). This shine-through phenomenon is particularly evident in patients with floor-of-mouth OSCC and sublingual, submental and submandibular SLNs, resulting in a significantly lower accuracy of SLNB in floor-of-mouth tumors (sensitivity 63%; NPV 90%) compared to other oral cavity subsites (sensitivity 86%; NPV 95%) [4][12][13][14][15][16].
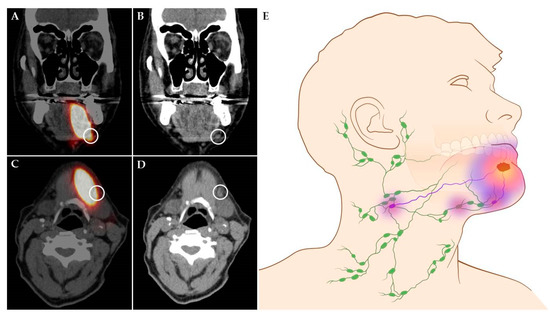
Figure 1. Shine-through phenomenon in 72-year-old patient with a cT1N0 floor-of-mouth carcinoma. (A,C) Coronal and axial SPECT-CT images: radiation flare of the tracer injection site over shines a sentinel lymph node located in cervical lymph node level Ib (white circle). (B,D) Coronal and axial low-dose CT images of same patient: (sentinel) lymph node located in cervical lymph node level Ib that could not be differentiated from the hotspot originating from tracer injection site on SPECT-CT (white circle). (E) Schematic illustration of shine-through phenomenon. (A–D) Informed consent has been obtained from this patient. (E) ©University Medical Center Groningen.
Therefore, technical developments are needed to bring the diagnostic accuracy of SLNB for early-stage OSCC to a higher level, particularly in FOM tumors. Novel SLNB imaging techniques for early-stage OSCC include MR lymphography, CT lymphography, PET lymphoscintigraphy and contrast-enhanced lymphosonography.
3. Conclusions
Novel diagnostic imaging techniques for detection of SLNs have the potential to bring the diagnostic accuracy of SLNB to a higher level for all early-stage OSCC subsites. However, technical improvements and further research of these novel techniques are required, if they are to replace the conventional SLNB procedure with [99mTc]-labelled radiotracers. Nevertheless, several of these novel techniques may already become valuable by facilitating more targeted radiotherapy; adjusting the radiation dose based on the tumor’s individual lymphatic drainage pattern.
References
- D’Cruz, A.K.; Vaish, R.; Kapre, N.; Dandekar, M.; Gupta, S.; Hawaldar, R.; Agarwal, J.P.; Pantvaidya, G.; Chaukar, D.; Deshmukh, A.D.; et al. Elective versus Therapeutic Neck Dissection in Node-Negative Oral Cancer. N. Engl. J. Med. 2015, 373, 521–529.
- Abu-Ghanem, S.; Yehuda, M.; Carmel, N.-N.; Leshno, M.; Abergel, A.; Gutfeld, O.; Fliss, D.M. Elective Neck Dissection vs Observation in Early-Stage Squamous Cell Carcinoma of the Oral Tongue with No Clinically Apparent Lymph Node Metastasis in the Neck. JAMA Otolaryngol. Neck Surg. 2016, 142, 857–865.
- De Bree, R.; Takes, R.P.; Shah, J.P.; Hamoir, M.; Kowalski, L.P.; Robbins, K.T.; Rodrigo, J.P.; Rodrigo, J.P.; Medina, J.E.; Rinaldo, A.; et al. Elective neck dissection in oral squamous cell carcinoma: Past, present and future. Oral Oncol. 2019, 90, 87–93.
- Toom, I.J.D.; Boeve, K.; Lobeek, D.; Bloemena, E.; Donswijk, M.L.; de Keizer, B.; Klop, W.M.C.; Leemans, C.; Willems, S.M.; Takes, R.P.; et al. Elective Neck Dissection or Sentinel Lymph Node Biopsy in Early Stage Oral Cavity Cancer Patients: The Dutch Experience. Cancers 2020, 12, 1783.
- Schilling, C.; Shaw, R.; Schache, A.G.; McMahon, J.; Chegini, S.; Kerawala, C.; McGurk, M. Sentinel lymph node biopsy for oral squamous cell carcinoma. Where are we now? Br. J. Oral Maxillofac. Surg. 2017, 55, 757–762. [Google Scholar] [CrossRef] [PubMed]
- Cramer, J.D.; Sridharan, S.; Ferris, R.L.; Duvvuri, U.; Samant, S. Sentinel Lymph Node Biopsy Versus Elective Neck Dissection for Stage I to II Oral Cavity Cancer. Laryngoscope 2018, 129, 162–169.
- Schiefke, F.; Akdemir, M.; Weber, A.; Akdemir, D.; Singer, S.; Frerich, B. Function, postoperative morbidity, and quality of life after cervical sentinel node biopsy and after selective neck dissection. Head Neck 2009, 31, 503–512.
- Murer, K.; Huber, G.F.; Haile, S.R.; Stoeckli, S.J. Comparison of morbidity between sentinel node biopsy and elective neck dissection for treatment of the n0 neck in patients with oral squamous cell carcinoma. Head Neck 2010, 33, 1260–1264.
- Govers, T.M.; Schreuder, W.; Klop, W.; Grutters, J.P.C.; Rovers, M.; Merkx, M.A.W.; Takes, R. Quality of life after different procedures for regional control in oral cancer patients: Cross-sectional survey. Clin. Otolaryngol. 2016, 41, 228–233.
- Govers, T.M.; Takes, R.P.; Karakullukcu, B.; Hannink, G.; Merkx, M.A.W.; Grutters, J.P.C.; Rovers, M. Management of the N0 neck in early stage oral squamous cell cancer: A modeling study of the cost-effectiveness. Oral Oncol. 2013, 49, 771–777.
- Liu, M.; Wang, S.J.; Yang, X.; Peng, H. Diagnostic Efficacy of Sentinel Lymph Node Biopsy in Early Oral Squamous Cell Carcinoma: A Meta-Analysis of 66 Studies. PLoS ONE 2017, 12, e0170322.
- Boeve, K.; Schepman, K.; Schuuring, E.; Roodenburg, J.; de Bree, R.; Boorsma, R.; de Visscher, J.; Brouwers, A.; van der Vegt, B.; Witjes, M.J.H.; et al. High sensitivity and negative predictive value of sentinel lymph node biopsy in a retrospective early stage oral cavity cancer cohort in the Northern Netherlands. Clin. Otolaryngol. 2018.
- Toom, I.J.D.; Heuveling, D.; Flach, G.B.; van Weert, S.; Karagozoglu, K.H.; van Schie, A.; Bloemena, E.; Leemans, C.R.; Jansen, F. Sentinel node biopsy for early-stage oral cavity cancer: The VU University Medical Center experience. Head Neck 2014, 37, 573–578.
- Alkureishi, L.W.T.; Ross, G.L.; Shoaib, T.; Soutar, D.S.; Robertson, A.G.; Thompson, R.; Hunter, K.D.; Sørensen, J.A.; Thomsen, J.B.; Krogdahl, A.; et al. Sentinel Node Biopsy in Head and Neck Squamous Cell Cancer: 5-Year Follow-Up of a European Multicenter Trial. Ann. Surg. Oncol. 2010, 17, 2459–2464.
- Pedersen, N.J.; Jensen, D.H.; Hedbäck, N.; Frendø, M.; Kiss, K.; Lelkaitis, G.; Mortensen, J.; Christensen, A.; Specht, L.; von Buchwald, C. Staging of early lymph node metastases with the sentinel lymph node technique and predictive factors in T1/T2 oral cavity cancer: A retrospective single-center study. Head Neck 2015, 38, E1033–E1040.
- Stoeckli, S.J.; Huebner, T.; Huber, G.F.; Broglie, M.A. Technique for reliable sentinel node biopsy in squamous cell carcinomas of the floor of mouth. Head Neck 2016, 38, 1367–1372.




