| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Samuel Oluwafemi | + 1643 word(s) | 1643 | 2020-10-19 09:30:36 | | | |
| 2 | Dean Liu | Meta information modification | 1643 | 2020-11-02 08:33:56 | | |
Video Upload Options
The clinical translation of quantum dots (QDs) has gained much importance for the biomedical application. But the most used conventional QDs contains toxic heavy metals such as Pb, Cd and Hg. Hence, there is a need for an alternative material with similar optical properties and low cytotoxicity. Among these materials, CuInS2 (CIS) QDs has attracted a lot of interest due to its direct bandgap in the infrared region, large optical absorption coefficient and low toxic composition. All these make it a good material for biomedical application. For further research on the material, there is a need for a critical review which summarise the various synthetic strategies, surface modification and Biological evaluation involved with CIS QDs.
1. Introduction
Semiconductor nanoparticles also referred to as zero-dimensional material or quantum dots (QDs), have gained significant interest in many areas of applications ranging from water to energy, sensing and biological applications due to their quantum confinement effect.[1][2][3][4] Compared to conventional organic dyes, QDs exhibit high photostability and tunable optical properties by changing the size and composition.[5] The development/research on binary QDs has received considerable interest, and the field has been explored extensively; however, only a few commercial products have been developed due to their toxic composition.[6] Therefore, alternative, low or non-toxic material is imperative, especially for biological application.[7][8][9][10] The II-VI group and IV-VI group-based materials have been extensively studied, while other materials such as binary III-V and low-toxic ternary I-III-VI are less explored[11]. The I-III-VI quantum dots, which contain group I (Cu, Ag), group III (Al, Ga, In, Ti) and group IV (S, Se, Te), have been reported as less toxic compared to II-VI binary QDs. The replacement of group II in II-VI binary QDs by the group I and group III cations has led to the production of Cd- and Pb-free ternary nanocrystals NCs/QDs (e.g., CuInS2, AgInS2).
2. Basic Synthetic Method
The basic synthetic method for the formation of ternary QDs is based on the initial formation of nuclei, which are combined to form multinary NCs, or through the nuclei, which can also act as seeds and facilitate cation exchange or ionic diffusion.[9][12] For example, binary copper chalcogenides (e.g., Cu2S) can be subjected to a partial cationic exchange which can partly replace copper ions with other cations (e.g., Zn2+, Sn4+).[13] The limitation of this method is the long reaction times and polydispersity. Therefore, to obtain a monodisperse confined NCs with controlled composition, the cationic reaction should be finely balanced. Thus, using stabilizing agents to control the reactivity of the precursors has been proposed.[14][15][16][17][18] In order to tune the optical properties of the ternary NCs, the control on composition and phase homogeneities are very important. The desired phase of the crystal depends on the nature of cation and control on the site occupancy by tuning the precursors, surfactant and experimental conditions (Figure 1). CuInS2 (CIS) is a direct semiconductor with a bandgap of around 1.45 eV, a large extinction coefficient, good radiation hardness and excellent defect tolerance. The Bohr exciton radius of CIS NCs is reported to be around 4.1 nm, and the quantum confinement in this crystal can be observed until it reaches 8 nm in diameter.[19] There are reports on the tuning of its emission position as the diameter of the CIS changes [20][21], but with a broad full width and half maxima (FWHM) (90–120 nm) compared to binary QDs. The initial assessment of this broadness was focused on the size distribution of the as-synthesized material.[20] However, after various reports, this broadness of photoluminescence (PL) spectra has been shown to be independent of the size distribution in the case of CIS-based QDs and can be attributed to the distribution of vibrational states.[21][22] This shows that quantum size effects are not the only factors that can influence the bandgap of the ternary NCs[7][20][22], as the composition of anions and cations also plays a major factor.[23][24] Regarding the composition tuning, the valence band of the CIS (Cu 3d and S 3p orbital) can be lowered when the Cu content is reduced. The tuning of composition leads to the stoichiometric deviation, which, in turn, generates a large number of donor-acceptor states within the bandgap. These high densities of states lead to a high probability of radiative recombination of donor-acceptor and result in broad photoluminescence (PL) spectra, long PL lifetime and a shift in stokes.[10][25] The time-resolved PL(TRPL) analysis is used to understand the charge recombination in these QDs. For the ternary QDs, the PL decay is usually reported to be double/triple exponential analysis. The average PL lifetime for CIS QDs is reported to be between 100–300 ns, which is much longer than the binary QDs. As reported, the PL decay also depends on the composition of the CIS NCs.[21] Thus, the bandgap cannot be determined by merely accessing the PL spectra. Moreover, the large volumes of defects lower the quantum yield (QY). From this, we can conclude that the optical property of ternary QDs depends not only on the size but also on the composition, crystal phase, ordering and surface passivation.
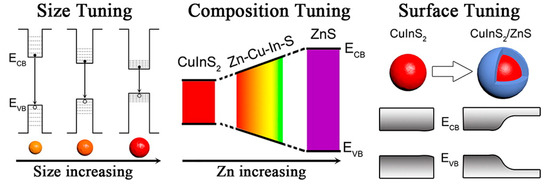
Figure 1. The strategies (size tuning, composition tuning, and surface tuning) that are applied to synthesize the highly luminescent CuInS2 NCs.[26]
Biological application (in vivo and in vitro) using QDs has become an attractive alternative over the conventional fluorophores. Higher quantum yield, photoluminescence efficiency, stability and biocompatibility are the desired characteristics for bioimaging application. Studies have shown that incidence of visible light (400–650 nm) for bio-imaging will be absorbed by the collagen, hemoglobin and lipids, thus making tissue imaging impossible in this spectral region. Therefore, a material with a near-infrared (NIR) window (650 to 1350 nm), where light is expected to have maximum penetration depth in tissue and less autofluorescence, is preferred.[27] The preferred intake of quantum dots by cells is through transfection or receptor-mediated endocytosis rather than through microinjection. Therefore, proper surface conjugation or charge is necessary for their bio application. In addition, before biological application of quantum dots, it is important to tune the surface reactivity, stability, lifetime and biocompatibility of the QDs. This review focuses on the various synthetic strategies (organic and aqueous), formation mechanism, biological applications (in vitro and in vivo) and future perspectives of CuInS-based QDs (Scheme 1).
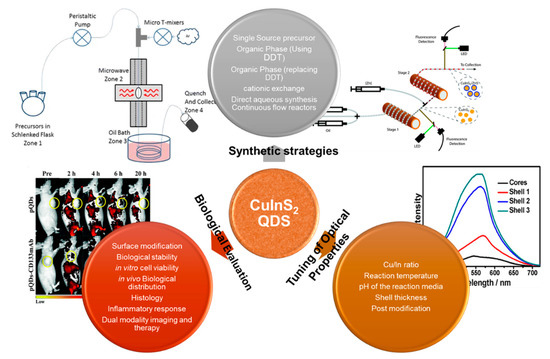
Scheme 1. A schematic representation of the different synthetic methodology and evaluation of CuInS-based QDs.
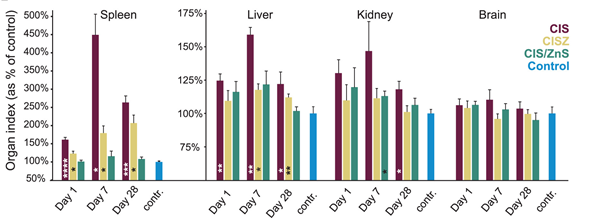
The significant biological evaluation was conducted by Kays et al. [28] in which they used the murine model to study the in vivo biodistribution and cytotoxicity of CuInS2 (CIS), Zn-doped CuInS2 (CISZ) and CuInS2/ZnS QDs for different days (1,7 and 28 days). They observed a quick clearance, and less than 25% of the material was found in the organs after 28 days. They also studied the in vivo toxicity of the QDs by recording organ indexes (Figure 2). For CIS and CISZ, there was a significant increase in the liver and spleen organ index, but there were no significant changes observed for CIS/ZnS QDs. The kidney size variation was inconsistent, and only CIS and CIS/ZnS showed a significant change. From the histopathology studies, they observed geographic necrosis with the CIS QDs. For the CISZ, there was only small inflammation. In the case of the spleen, CIS- and CISZ-treated cells exhibited multinucleation, which was the result of an inflammatory response. In addition, disruption of spleen architecture was observed with CIS QDs. However, no significant toxicity to the kidney was observed for all the materials. From the results, they confirmed that with the ZnS coating, they were able to minimize the degradation and subsequent toxicity of the material.
Figure 2. Organ index (organ weight/total body weight) plotted as the percent of control values for four major organs[28]
3. Conclusions and Future Outlook
CuInS2 (CIS) has been reported as an alternative to Pb-, As-, Cd- and Hg-based QDs due to its toxic-free composition. Moreover, its PL can be easily tuned towards NIR regions by varying its composition. The CIS/ZnS QDs synthesized through organic method have excellent PLQY, but their effective application is hindered due to the hydrophobic nature and toxicity of the reagents used. However, the hydrophobic QDs can be effectively transferred into aqueous media through the ligand exchange process, which can facilitate their use in biomedical applications. However, the ligand exchange process has a great effect on the PL properties of the material, such as a decrease in PLQY and blue shift in the emission position. Therefore, direct synthesis in water is preferred with postmodification with polymers and other capping agents which can improve the solubility, biocompatibility and target specificity of the material. The surface-modified CIS core and CIS/ZnS core/shell QDs have been tested in vivo and in vitro in various cell lines and animals for tumour imaging, lymph node imaging, etc., which shows the excellent applicability of the material for a clinical trial.
Even though there are various green synthesis approaches reported for the synthesis of CIS-based QDs, a complete green synthesis has not been fully established. The authors recommend thorough research in the designing of a green synthesis protocol for CIS QDs. The tuning of optical properties by mere adjustment of the ratio between metal precursors will not be enough. Since the formation of CIS QDs also depends on other parameters such as the nature of the capping agent, the concentration of the precursors, the effect of pH, the nature of shell source, and reaction temperature, a detailed optimization of parameters is required when a new precursor or capping agent is chosen. Most of the studies also reported CIS and CIS/ZnS QDs as “nontoxic”, which was based on an average observation and histology. More investigations are needed in this area using different cytotoxicity assays for a concrete report on the toxicity of this material. Further studies are also required in the modification of synthetic strategies towards large scale synthesis, without any significant change in the PL properties of the material when compared to those synthesize in the laboratory.
References
- Rodney Maluleke; El Hadji Mamour Sakho; Oluwatobi S. Oluwafemi; Aqueous synthesis of glutathione-capped CuInS2/ZnS quantum dots-graphene oxide nanocomposite as fluorescence “switch OFF” for explosive detection. Materials Letters 2020, 269, 127669, 10.1016/j.matlet.2020.127669.
- R. Jose Varghese; Sundararajan Parani; Olufemi O. Adeyemi; V. R. Remya; El Hadji Mamour Sakho; Rodney Maluleke; Sabu Thomas; Oluwatobi S. Oluwafemi; Green Synthesis of Sodium Alginate Capped -CuInS2 Quantum Dots with Improved Fluorescence Properties. Journal of Fluorescence 2020, 0, 1-5, 10.1007/s10895-020-02604-0.
- R. Jose Varghese; Sundararajan Parani; V.R. Remya; Rodney Maluleke; Sabu Thomas; Samuel Oluwatobi Oluwafemi; Sodium alginate passivated CuInS2/ZnS QDs encapsulated in the mesoporous channels of amine modified SBA 15 with excellent photostability and biocompatibility. International Journal of Biological Macromolecules 2020, 161, 1470-1476, 10.1016/j.ijbiomac.2020.07.240.
- Nawzad Nadhim Jawhar; Ehsan Soheyli; Ahmet Faruk Yazici; Evren Mutlugun; Reza Sahraei; Preparation of highly emissive and reproducible Cu–In–S/ZnS core/shell quantum dots with a mid-gap emission character. Journal of Alloys and Compounds 2020, 824, 153906, 10.1016/j.jallcom.2020.153906.
- Guang Sun; Wanying Xing; Ren Xing; Liu Cong; Sun Tong; Siyao Yu; Targeting breast cancer cells with a CuInS2/ZnS quantum dot-labeled Ki-67 bioprobe. Oncology Letters 2017, 15, 2471-2476, 10.3892/ol.2017.7615.
- Zahra Ranjbar-Navazi; Yadollah Omidi; Morteza Eskandani; Soodabeh Davaran; Cadmium-free quantum dot-based theranostics. TrAC Trends in Analytical Chemistry 2019, 118, 386-400, 10.1016/j.trac.2019.05.041.
- Peter M. Allen; Moungi G. Bawendi; Ternary I−III−VI Quantum Dots Luminescent in the Red to Near-Infrared. Journal of the American Chemical Society 2008, 130, 9240-9241, 10.1021/ja8036349.
- Daocheng Pan; Lijia An; Zhongming Sun; William Hou; Yang Yang; Zhengzhong Yang; Yunfeng Lu; Synthesis of Cu−In−S Ternary Nanocrystals with Tunable Structure and Composition. Journal of the American Chemical Society 2008, 130, 5620-5621, 10.1021/ja711027j.
- Maria Ibáñez; Reza R. Zamani; Wenhua Li; A. Shavel; Jordi Arbiol; Juan Ramón Morante; Andreu Cabot; Extending the Nanocrystal Synthesis Control to Quaternary Compositions. Crystal Growth & Design 2012, 12, 1085-1090, 10.1021/cg201709c.
- Dmitry Aldakov; Aurélie Lefrançois; Peter Reiss; Ternary and quaternary metal chalcogenide nanocrystals: synthesis, properties and applications. Journal of Materials Chemistry C 2013, 1, 3756-3776, 10.1039/c3tc30273c.
- H. Zhong; T. Mirkovic; G.D. Scholes; Nanocrystal Synthesis. Comprehensive Nanoscience and Technology 2011, 1-5, 153-201, 10.1016/b978-0-12-374396-1.00051-9.
- Reza R. Zamani; Maria Ibáñez; Martina Luysberg; Nuria Garcia-Castello; Lothar Houben; Joan Daniel Prades; Vincenzo Grillo; Rafal E. Dunin-Borkowski; Juan Ramón Morante; Andreu Cabot; et al.Jordi Arbiol Polarity-Driven Polytypic Branching in Cu-Based Quaternary Chalcogenide Nanostructures. ACS Nano 2014, 8, 2290-2301, 10.1021/nn405747h.
- Vladimir Lesnyak; Chandramohan George; Alessandro Genovese; Mirko Prato; Alberto Casu; S. Ayyappan; Alice Scarpellini; Liberato Manna; Alloyed Copper Chalcogenide Nanoplatelets via Partial Cation Exchange Reactions. ACS Nano 2014, 8, 8407-8418, 10.1021/nn502906z.
- Yanyan Chen; Shenjie Li; Lijian Huang; Daocheng Pan; Green and Facile Synthesis of Water-Soluble Cu–In–S/ZnS Core/Shell Quantum Dots. Inorganic Chemistry 2013, 52, 7819-7821, 10.1021/ic400083w.
- Ling Zhu; Kishan Das; Tabasum Akhter; Rahul Ranjan; Rajan Patel; H. B. Bohidar; Eco-friendly synthesis of CuInS2 and CuInS2@ZnS quantum dots and their effect on enzyme activity of lysozyme. RSC Advances 2018, 8, 30589-30599, 10.1039/c8ra04866e.
- Anila Arshad; Raheel Akram; Saleem Iqbal; Fozia Batool; Bushra Iqbal; Bilal Khalid; Arif Ullah Khan; Aqueous synthesis of tunable fluorescent, semiconductor CuInS2 quantum dots for bioimaging. Arabian Journal of Chemistry 2019, 12, 4840-4847, 10.1016/j.arabjc.2016.10.002.
- Ward Van Der Stam; Anne C. Berends; Freddy T. Rabouw; Tom Willhammar; Xiaoxing Ke; Johannes D. Meeldijk; Sara Bals; Celso De Mello Donegá; Luminescent CuInS2 Quantum Dots by Partial Cation Exchange in Cu2–xS Nanocrystals. Chemistry of Materials 2015, 27, 621-628, 10.1021/cm504340h.
- Ncediwe Tsolekile; Sara Nahle; Nkosingiphile Zikalala; Sundararajan Parani; El Hadji Mamour Sakho; Olivier Joubert; Mangaka C. Matoetoe; Sandile P. Songca; Oluwatobi S. Oluwafemi; Cytotoxicity, fluorescence tagging and gene-expression study of CuInS/ZnS QDS - meso (hydroxyphenyl) porphyrin conjugate against human monocytic leukemia cells. Scientific Reports 2020, 10, 1-13, 10.1038/s41598-020-61881-8.
- Joanna Kolny-Olesiak; Horst Weller; Synthesis and Application of Colloidal CuInS2Semiconductor Nanocrystals. ACS Applied Materials & Interfaces 2013, 5, 12221-12237, 10.1021/am404084d.
- Haizheng Zhong; Shun S. Lo; Tihana Mirkovic; Yunchao Li; Yuqin Ding; Yongfang Li; Gregory D. Scholes; Noninjection Gram-Scale Synthesis of Monodisperse Pyramidal CuInS2 Nanocrystals and Their Size-Dependent Properties. ACS Nano 2010, 4, 5253-5262, 10.1021/nn1015538.
- Hai‐Zheng Zhong; Yi Zhou; Mingfu Ye; YouJun He; Jianping Ye; Chang He; Chunhe Yang; Yongfang Li; Controlled Synthesis and Optical Properties of Colloidal Ternary Chalcogenide CuInS2Nanocrystals. Chemistry of Materials 2008, 20, 6434-6443, 10.1021/cm8006827.
- Tzung-Luen Li; Hsisheng Teng; Solution synthesis of high-quality CuInS2 quantum dots as sensitizers for TiO2 photoelectrodes. Journal of Materials Chemistry 2010, 20, 3656-3664, 10.1039/b927279h.
- Daocheng Pan; Xiaolei Wang; Z. Hong Zhou; Wei Chen; Chuanlai Xu; Yunfeng Lu; Synthesis of Quaternary Semiconductor Nanocrystals with Tunable Band Gaps. Chemistry of Materials 2009, 21, 2489-2493, 10.1021/cm900439m.
- Deniz Bozyigit; Vanessa Wood; Challenges and solutions for high-efficiency quantum dot-based LEDs. MRS Bulletin 2013, 38, 731-736, 10.1557/mrs.2013.180.
- Luca De Trizio; Mirko Prato; Alessandro Genovese; Alberto Casu; Mauro Povia; Roberto Simonutti; Marcelo J. P. Alcocer; Cosimo D’Andrea; Francesco Tassone; Liberato Manna; et al. Strongly Fluorescent Quaternary Cu–In–Zn–S Nanocrystals Prepared from Cu1-xInS2 Nanocrystals by Partial Cation Exchange. Chemistry of Materials 2012, 24, 2400-2406, 10.1021/cm301211e.
- Haizheng Zhong; Zelong Bai; Bingsuo Zou; Tuning the Luminescence Properties of Colloidal I–III–VI Semiconductor Nanocrystals for Optoelectronics and Biotechnology Applications. The Journal of Physical Chemistry Letters 2012, 3, 3167-3175, 10.1021/jz301345x.
- Feng Ding; Yibei Zhang; Xiaoju Lu; Yao Sun; Recent advances in near-infrared II fluorophores for multifunctional biomedical imaging. Chemical Science 2018, 9, 4370-4380, 10.1039/c8sc01153b.
- Joshua C. Kays; Alexander M. Saeboe; Reyhaneh Toufanian; Danielle E. Kurant; Allison M. Dennis; Shell-Free Copper Indium Sulfide Quantum Dots Induce Toxicity in Vitro and in Vivo. Nano Letters 2020, 20, 1980-1991, 10.1021/acs.nanolett.9b05259.