| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mahroo Falah | + 4028 word(s) | 4028 | 2020-10-12 05:27:46 | | | |
| 2 | Rita Xu | -654 word(s) | 3374 | 2020-10-19 05:56:07 | | |
Video Upload Options
The ecologically-friendly inorganic materials called geopolymers are mainly produced using secondary raw materials such as fly ash, blast furnace slags, or mining residues at temperatures below 100 °C. Geopolymers are a sustainable alternative to Portland cement in the building industry. Their chemical and physical properties enable them to act as supports for photoactive species, including TiO2, Cu2O and Fe2O3, or carbon nanotubes and graphene. This review discusses the structure and synthesis process of geopolymers and the principal considerations for their use as photocatalysts. The utilisation of geopolymers as photocatalysts indicates promising applications for removing heavy metals from wastewater, purifying polluted drinking water and ecologically-friendly candidates to remediate toxic environmental pollutants.
1. Introduction
The development of materials to mitigate the effects of global warming and pollution generated by human activities is becoming a matter of increasing urgency. The ecological problems to which solutions are being sought are the increase in greenhouse gas emissions arising from the manufacture of ordinary Portland cement (OPC) and electricity generation by coal-fired power plants, especially in developing nations with increased demands for infrastructure. Ecologically-friendly alternatives to OPC which do not involve the high-temperature reaction of clay and limestone, generating large amounts of CO2, are inorganic polymers, otherwise known as geopolymers [1]. A further advantage of these materials over OPC is that they can be produced at temperatures from ambient to 80 °C by alkali activation of a range of aluminosilicate minerals [2][3][4][5][6], although geopolymers are now known to be less ecologically-friendly than previously claimed, given the production of the alkali activator and factors such as the energy-intensive processes by which some geopolymer precursors are produced [7]; these factors have often been overlooked or ignored by earlier proponents of geopolymers. Nevertheless, to date, the major interest in geopolymers has been as alternatives to OPC, although they have many other high-technology applications [8]. One such area of increasing interest is the mitigation of global environmental pollution problems, particularly air and water pollution. Over the past two or three decades, photocatalytic degradation of environmental pollutants by exploiting the conversion of solar to chemical energy has attracted considerable attention. Heterogeneous photocatalysts have many advantages over other photocatalysts, since they can be cost-effective, stable, non-toxic, strongly oxidising and effective at ambient temperature and pressure [9]. The combination of photocatalysts with geopolymers has been exploited in the construction industry to develop self-cleaning coatings for buildings, where the key factors are the aesthetic appearance and reasonable costs of cleaning maintenance [10]. Since geopolymers are environmentally friendly and can readily incorporate photocatalytically active materials such as ZnO, TiO2, CuO and Fe2O3, they are an excellent option for construction applications. Geopolymers incorporated with photoactive materials in the presence of UV and UV–visible radiation [11] can oxidize and decompose the surface pollutants on a building or roadway, allowing the products to be subsequently easily removed later by rain, cleaning or washing with water because of the hydrophilic surface of such a photocatalytic geopolymer and the low contact angle for water, allowing it to slide off [9][12][13].
A further ecological problem is associated with atmospheric pollution of waterways and the atmosphere resulting from manufacturing operations. A less well-researched but equally important environmental application of geopolymers has been as materials for the destruction of hazardous organic species in the atmosphere and in waterways. For this purpose, various photocatalytic species can be introduced into the geopolymer by exploiting its structure and chemistry, and the photocatalytic process can also be facilitated by the ability of the geopolymer to adsorb and immobilise the hazardous material in its structure. Thus, the combination of photocatalytic functionality with the environmental friendliness of geopolymers makes these potentially important materials for mitigation of climate change problems.
2. Geopolymer/TiO2 Photocatalysts
TiO2 is one of the more widely-used photocatalysts for environmental applications, such as water purification, air cleaning and self-cleaning surfaces, because of its good photocatalytic activity, chemical stability, low cost, long-term stability, ready availability and lack of toxicity [14][15][16]. One of the most challenging criteria for a suitable photocatalyst is that it must not rapidly recombine photogenerated electrons and holes. In the case of TiO2 its photocatalytic efficiency can be increased by incorporating it into a TiO2-based composite, thereby hindering its charge recombination [17]. In this way, TiO2 has been immobilized by incorporation into a number of different construction materials, including window glass, cement-based materials, bricks, ceramics and geopolymers, producing environmentally friendly self-cleaning and air purification products [18].
Strini et al. [19] demonstrated the use of TiO2-based photocatalytic geopolymers based on fly ash or metakaolin to decompose nitric oxide in air. The photocatalyst geopolymer was produced simply by mixing the geopolymer composition with P25 (a commercial mixture of rutile and anatase polymorphs of TiO2). The ideal amount of TiO2 was suggested to be 3 wt.% of the geopolymer paste, and the photocatalytic activity of the geopolymer composite depended on the type of binder and the curing conditions. The highest photocatalytic activity was found in a TiO2/fly ash-based geopolymer composite cured at room temperature, which resulted in twice the NO degradation rate compared with that of a TiO2/metakaolin geopolymer [19]. The photocatalytic activity was also found to depend on the curing parameters; curing at 60 °C produced a poorer photocatalyst, apparently due to segregation and depletion of the TiO2 in the catalyst surface [19].
Bravo et al. [20] synthesised metakaolin-based geopolymer spheres with photocatalytic activity by coating TiO2 nanoparticles on the surface of the spheres. These were produced by foaming an uncured geopolymer mixture with Polysorbate 80, and then dropping the mixture from a syringe into polyethylene glycol at 80 °C, which reduced the surface tension and resulted in the formation of beads 2–3 mm in diameter. After drying at room temperature for 24 h and then curing at 75 °C for 2 days, the beads were then coated with TiO2 nanocrystals by heating them with TiO2 at 1200 °C inside a quartz tube under high vacuum. SEM images confirmed the complete dispersion of TiO2 within the geopolymer spheres. The photocatalytic activity of the TiO2/geopolymer spheres in the degradation of methylene blue dye showed 90% degradation after 10 h of ultraviolet irradiation, compared with 4.5% degradation of the uncoated geopolymer spheres, but the brief experimental details of the photocatalytic experiments gave no indication as to how the processes of dye adsorption and photocatalysis were differentiated [20].
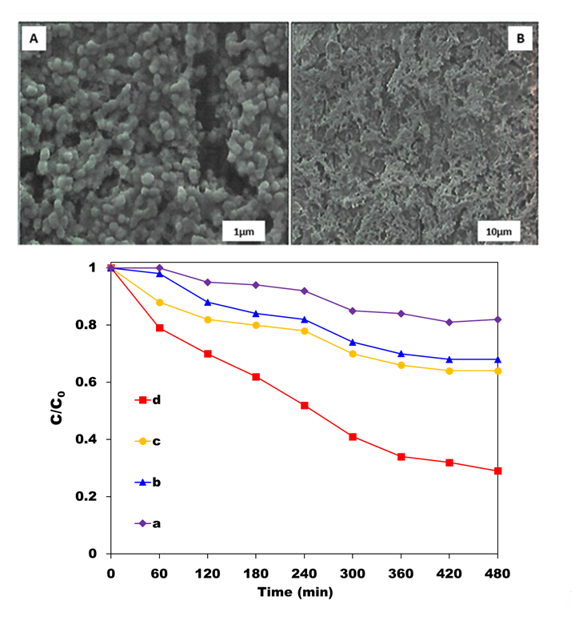
In another study, Chen et al. [18] deposited TiO2 films by a sol–gel dip-coating method on a geopolymer substrate based on 95% fly ash and 5% metakaolin cured at room temperature. The geopolymer matrix was then dip-coated in a mixture of butyl titanate in ethanol with the addition of diethanolamine to increase the stability of the sol [18][21]. The dip-coated samples were then annealed at 500, 600, 700 and 800 °C for 1 h and showed desirable photocatalytic properties for the degradation of methylene blue dye [18], especially the sample annealed at 600 °C (Figure 1C), which was shown to contain the anatase phase of TiO2 and a mesoporous morphology (Figure 1A,B). Improved photocatalytic activity of the composites could be obtained by double layer sol–gel coating, resulting in an increased specific surface area, but conversely, it may also increase the probability of electron–hole pair recombination and decrease the photocatalytic performance. A further problem observed with the sol–gel coating technique was a tendency for the films to crack upon drying, but this could be offset by the addition of 6 wt.% polyvinylpyrrolidine (PVP) [18]. Control experiments were also carried out under dark conditions to determine and correct for the degree of dye adsorption by the geopolymer alone.
Figure 1. (A). SEM images of a dip-coated TiO2 film annealed at 600 °C for 1 h. (B) Surface morphology of a typical multilayer-coated TiO2 film. (C) Residual dye concentration (C/C0) of methylene blue under UV irradiation by TiO2 films coated on a geopolymer, annealed at (a) 500 °C, (b) 800 °C, (c) 700 °C, (d) 600 °C, drawn from the data of Chen et al. [18].
Gasca-Tirado et al. [22] reported an alternative method for incorporating photoactive TiO2 into a metakaolin-based aluminosilicate inorganic polymer (geopolymer) by ion exchange with ammonium titanyl oxalate monohydrate, (NH4)2TiO(C2O4)2.H2O. The efficiency of Ti incorporation was not improved by prior conversion of the geopolymer to the NH4+ form. This ion exchange method was found to facilitate the growth of anatase-type TiO2 nanoparticles inside the geopolymer micropores, producing a photoactive geopolymer composite, demonstrated by its degradation of methylene blue (MB). Samples of the geopolymer composite, and for comparison, the geopolymer without TiO2, were equilibrated in the dark for a short time (15 min) prior to UV irradiation, and the results suggested only a small amount of dye adsorption had taken place after 80 min, whereas almost all the methylene blue was bleached by the geopolymer photocatalyst, particularly the sample cured at 90 oC prior to ion exchange [22].
Yang et al. [23] studied the degradation of MB dye by foamed fly ash-based geopolymer-TiO2 nanocomposites produced in two different ways. The use of a foamed geopolymer matrix increased its porosity, allowing the incorporation of a higher concentration of TiO2 and improved contact with the dye solution. After alkali-activation, the fly ash geopolymer was foamed by the addition of oleic acid, followed by H2O2, and then cured at 80 °C. Two methods were investigated for incorporating TiO2 in the foamed geopolymer matrix; in one case the TiO2 (P25) was directly mixed into the foamed geopolymer prior to curing, whereas in a second procedure, the cured foamed matrix was treated with a solution of TiO2 in nitric acid, and then calcined at 500 °C to crystallize the TiO2 [23]. The most efficient photocatalytic degradation of MB dye was achieved in the composite containing 5 wt.% TiO2 prepared by direct mixing, and was attained after 45 min of UV irradiation [23]. Prior to irradiation, the geopolymer photocatalyst samples for this experiment were equilibrated for 30 min in the dark to allow for adsorption effects, during which time the MB concentration was monitored, but the results show that equilibrium was not fully attained. Nevertheless, this study points to the potential of porous TiO2/geopolymer composites derived from industrial by-products such as fly ash for the treatment of industrially discharged wastewater. Table 1 compares the different synthesis methods and photodegradation applications of TiO2/geopolymer photocatalysts.
Table 1. Comparison of the different synthetic methods and TiO2/geopolymer photocatalysts.
|
Adsorbent |
Preparation Method |
TiO2 Type |
TiO2 Content |
Adsorbate |
Reference |
|
TiO2/fly ash or metakaolin geopolymer |
Mixing |
P25 |
3% |
NO and NOx |
Strini 2016 [19] |
|
TiO2/metakaolin geopolymer |
Ion-Exchange |
Anatase |
28% |
MB |
Gasca-Tirado 2012 [22] |
|
TiO2/fly ash-metakaolin geopolymer |
Sol-Gel dip coating |
Anatase, Rutile |
NA |
MB |
Chen 2017 [18] |
|
TiO2/fly ash geopolymer |
Mixing |
P25 |
10% |
MB |
Yang 2019 [23] |
|
TiO2/metakaolin geopolymer spheres |
Inside quartz tube at high temperature |
P25 |
10 mg |
MB |
Bravo 2019 [20] |
3. Geopolymer/Graphene Photocatalysts
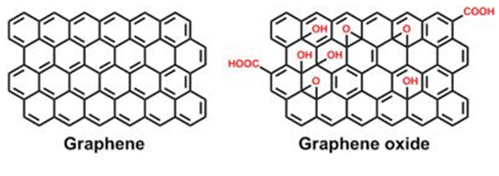
Graphene Oxide (GO) is a derivative of graphene, a two-dimensional form of carbon that is attracting increasing interest as a functional material with useful properties such as high specific surface area, high electric conductance and good thermal conductivity. GO contains functional groups containing oxygen and can be synthesized by methods such as chemical oxidation and exfoliation of graphite [24] (Figure 2). The structure of GO is based on graphene and contains a number of oxygen functional groups (surface hydrophilic hydroxyl (–OH) and epoxy (C–O–C) groups and edge carboxyl (–COOH) groups). These groups allow GO to be dispersed in water and provide many active sites for linking to other functional groups and organic molecules [25]. GO has attracted attention for adsorption and catalytic applications; in particular, its photonic properties suggest its potential for enhancing the photocatalytic properties of other materials. GO is typically suitable for the removal from water of organic dyes [26], antibiotics [27] and heavy metal ions [28].
Figure 2. Structure of graphene and graphene oxide.
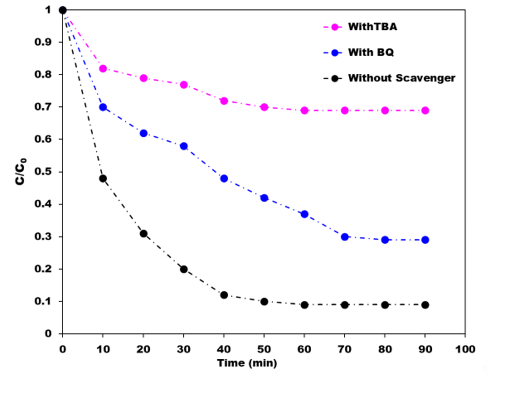
Lertcumfu et al. [24] studied the influences of GO additions on the photocatalytic properties of metakaolinite-based geopolymer composites and found that its addition can significantly improve the degradation of MB and Cr(VI) in comparison with pure geopolymer. The efficiencies of the GO/geopolymer composite for the removal of MB and Cr(VI) were 93% and 65% respectively. These authors reported that the adsorption process followed the pseudo second-order kinetic model with an R2 value > 98%. Moreover, these GO/geopolymer composites showed a potential application for waste water treatment owing to their photocatalytic activity with a poor C/C0 value of 0.6 after 2 h of UV irradiation. Zhang et al. [29] reported the photoactivity of a nanocomposite synthesized by the reaction of two-dimensional graphene with an alkali-activated granulated blast furnace slag (GBFS) geopolymer. This nanocomposite showed a high degree of methyl violet (MV) degradation under UV irradiation, especially in a composite containing 0.01 wt.% graphene, which had a degradation efficiency of 91.6 % after 110 min of UV irradiation [29]. The degradation reaction of the MV dye followed a pseudo second-order kinetic model. Zhang et al. [30] reported the photocatalytic activity of a novel electroconductive graphene/fly ash-based geopolymer composite prepared by the incorporation of 1 wt.% of graphene into the alkali-activated geopolymer matrix prior to curing at room temperature. The addition of the graphene increased the electroconductivity of the composite by 348.8 times compared with that of the geopolymer without graphene, and the synergic effect of the graphene with the matrix red-shifted the maximum absorption wavelength of the composite into the visible region [30]. Furthermore, the presence of the graphene was shown by nitrogen adsorption–desorption isotherms to effectively improve the pore structure of the composite. The photoactive composite was found to degrade indigo carmine (IC) dye with an efficiency of 90.2%, three times greater than the photocatalytic efficiency of the geopolymer matrix without graphene, and this catalytic performance for the removal of organic pollutants was unchanged after five cycling runs of UV irradiation [31]. Furthermore, the graphene structure was shown to be unchanged after the dye photodegradation cycles. The proposed mechanism involves interactions of the graphene with Fe2O3 particles from the fly ash geopolymer matrix in which the photogenerated electrons from the former are rapidly transferred to the π-conjugated system of the graphene, efficiently separating the photogenerated electron–hole pairs and allowing them to oxidize the H2O molecules adsorbed on the iron oxide surfaces. The resulting hydroxyl radicals oxidize and degrade the dye molecules adsorbed on the iron oxide surfaces [30]. In experiments to further confirm this mechanism, it was found that the addition of benzoquinone, which traps hydroxyl radicals, reduced the dye degradation efficiency from 91.6% to 70.8%, whereas the addition of tertiary butyl alcohol, which traps superoxide radicals, reduced the degradation efficiency to 35.1% (Figure 3). These experiments suggest that graphene can act as an electron acceptor to enhance the oxidation degradation capacities of geopolymers.
Figure 3. The effects of tertiary butyl alcohol (TBA) and benzoquinone (BQ) radical scavengers on the residual dye concentration (C/C0) of indigo carmine dye, drawn from the data of Zhang et al [30].
Table 2, which summarises the reported investigations of photoactive geopolymers containing graphene or graphene oxide, highlights the varying experimental conditions used by different researchers, making a direct comparison of the photocatalytic results impossible. Although the available research data on these graphene/geopolymer photocomposites are very promising, considerably more theoretical and experimental research is required on these systems.
Table 2. Summary of the different studies on graphene and graphene oxide geopolymer photocatalysts.
|
Matrix |
Preparation |
Graphene content (%) |
Adsorbate |
Reference |
|
graphene/fly ash-based geopolymer |
Mixing |
0.1, 0.4, 0.7 and 1 |
Indigo carmine |
Zhang 2009 [32] |
|
graphene oxide/calcined kaolinite-based geopolymer |
Mixing |
0, 2.5, 5, and 10 |
Methylene blue |
Lertcumfu 2020 [24] |
|
graphene/blast furnace slag-based geopolymer |
Mixing |
0.01 |
Methyl violet |
Zhang 2016 [29] |
4. Geopolymer/Cu2O Photocatalysts
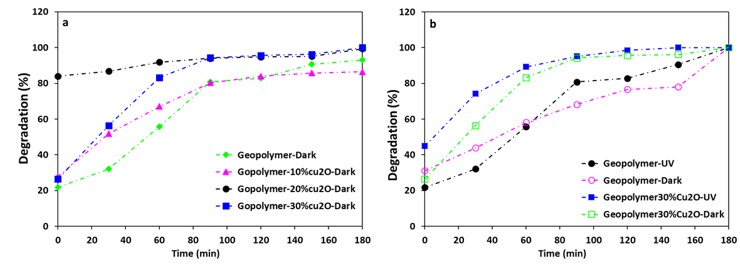
Cu2O, a prominent p-type semiconductor, acts as a photocatalyst under visible light irradiation ( ≤ 600 nm). Its direct bandgap energy of 2.2 eV is readily excited by wavelengths in the visible region, giving it a significant absorption coefficient (up to ≈104 cm-1) in this region. This suggests that Cu2O should act as a stable photocatalyst for the photochemical decomposition of H2O with the generation of O2 and H2 under visible light irradiation, and that it should also be a suitable candidate for the photocatalytic degradation of organic pollutants under visible light irradiation. The photocatalytic possibilities of Cu2O were investigated by Huang et al. [33] who demonstrated its photodegradation of methyl orange, but reported that it was readily deactivated by photocorrosion, especially when in the form of nanoparticles [33]. An improvement in its catalytic performance was obtained with larger microparticles which underwent photocorrosion more slowly, and its performance was also improved by the addition of 0.1 mmol/L of methanol which acted as a hole scavenger [33]. The photocatalytic behaviour of the different well-formed crystal facets of Cu2O microcrystals with well-formed facets was investigated by Zheng et al. [33]. They observed that during the photodegradation of methyl orange, the {100} and {110} faces gradually transformed into the {111} facets of nanosheets which exhibit stable photocatalytic activity [32]. These results, and the low toxicity, low cost and environmental friendliness of Cu2O suggest its use as a possible alternative to other common photocatalysts such as TiO2 for the photocatalytic degradation of organic contaminants, particularly when combined with ecologically-friendly geopolymers. These considerations led Fallah et al. [34] to investigate a novel photoactive inorganic polymer composite containing copper(I) oxide nanoparticles. The Cu2O nanoparticles were synthesized by the solution precipitation method, producing cubic crystallites of nanometre size. Metakaolin-based Cu2O/geopolymer composites were prepared by mixing 10–30 wt.% of pre-synthesized Cu2O nanoparticles with the geopolymer paste and curing for 12 h under ambient temperature. The photocatalytic activity of the Cu2O/geopolymer composite in the degradation of methylene blue (MB) was studied under the UV irradiation [34]. In a preliminary experiment, the adsorption of MB dye by geopolymer composites of varying Cu2O content was determined in the dark (Figure 4a), indicating that at 20% Cu2O content, adsorption approached equilibrium at 30 min. With higher Cu2O content, ingress of the dye solution into the pores of the substrate became progressively impeded, although the nano-Cu2O itself did not adsorb the methylene blue dye. Under UV radiation, the composites removed the MB dye from solution with a combination of adsorption and photodegradation (Figure 4b). In the dark, photocatalysis cannot occur and the process is by adsorption alone, evidenced by essentially similar behaviour of the geopolymer matrix under dark and UV conditions. By contrast, the geopolymer containing Cu2O showed improved removal of the dye under UV irradiation without deterioration of the geopolymer structure or the photoactive Cu2O component, as shown by 63Cu NQR spectroscopy [34]. This study suggested that these geopolymer composites should function as useful new materials for the removal of organic pollutants from water or the atmosphere. The degradation of the MB dye followed a pseudo-second order kinetic model [34].
Figure 4. (a) Removal of methylene blue dye upon exposure to the geopolymer matrix (GP) and the Cu2O-geopolymer composites of various compositions versus time in the dark; (b) comparison of the removal of methylene blue dye by the geopolymer matrix and the 30 wt.% Cu2O-geopolymer composite in the dark and under UV illumination. Drawn from the data from Falah et al [34].
5. Geopolymer/Carbon Nanotube Photocatalysts
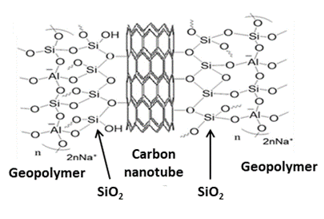
Carbon nanotubes (CNTs) are graphite sheets rolled up into cylinders with partly one-dimensional nanostructures. CNTs have diameters of a few nanometres and lengths of some millimetres. CNTs can occur as single-walled nanotubes (SWCNTs), double-walled nanotubes (DWCNTs) and multi-walled nanotubes (MWCNTs), the latter consisting of multiple layers of graphite arranged in concentric cylinders [31]. CNTs have been successfully used as catalyst-supporting materials with properties superior to those of other regular catalyst supports. CNTs have large specific surface areas and have excellent capacities for absorbing toxic materials such as nitrogen oxides and polluted waste water [35][36]. They are also useful for reinforcing geopolymers. Bi et al. [37] synthesised metakaolin-based geopolymer nanocomposites containing CNTs by ultrasonically dispersing 0.1, 0.25, and 0.5 vol.% of CNTs into a mixture of NaOH and sodium silicate solution, followed by blending with metakaolin powder, curing at 40 °C for 2 h and aging at 60 °C for 24 h. Before use, the CNTs were given a surface silica coating by treatment with a mixture of H2SO4 and HNO3, followed by a mixture of TEOS and NH4OH. The resulting good distribution of the CNTs in the geopolymer matrix and the interfacial interaction between the SiO2 coating and the geopolymer matrix (Figure 5) were found significantly to improve the mechanical properties of the geopolymer nanocomposites [37] and suggested their application as a self-sensing structural material with ultrahigh sensitivity. Although the possible use of these composites as photoactive materials has not yet been investigated, the photoactive properties of the closely-related graphene/geopolymer composites suggest an investigation of the CNT/geopolymer composites would be worthwhile.
Figure 5. Schematic representation of the interface between a SiO2-treated carbon nanotube surface and a geopolymer matrix.
References
- Davidovits, J. Geopolymers. J. Therm. Anal. Calorim. 1991, 37, 1633–1656, doi:10.1007/bf01912193.
- Barbosa, V.F.; MacKenzie, K.J.; Thaumaturgo, C. Synthesis and characterisation of materials based on inorganic polymers of alumina and silica: Sodium polysialate polymers. Int. J. Inorg. Mater. 2000, 2, 309–317, doi:10.1016/s1466-6049(00)00041-6.
- MacKenzie, K.J.D.; Komphanchai, S.; Vagana, R. Formation of inorganic polymers (geopolymers) from 2:1 layer lattice aluminosilicates. J. Eur. Ceram. Soc. 2008, 28, 177–181, doi:10.1016/j.jeurceramsoc.2007.06.004.
- Lemougna, P.N.; MacKenzie, K.J.D.; Melo, U.C. Synthesis and thermal properties of inorganic polymers (geopolymers) for structural and refractory applications from volcanic ash. Ceram. Int. 2011, 37, 3011–3018, doi:10.1016/j.ceramint.2011.05.002.
- Xu, G.; Shi, X. Characteristics and applications of fly ash as a sustainable construction material: A state-of-the-art review. Resour. Conserv. Recycl. 2018, 136, 95–109, doi:10.1016/j.resconrec.2018.04.010.
- Temuujin, J.; Minjigmaa, A.; Bayarzul, U.; Kim, D.S.; Lee, S.-H.; Lee, H.J.; Ruescher, C.H.; MacKenzie, K.J. Properties of geopolymer binders prepared from milled pond ash. Mater. Constr. 2017, 67, 134, doi:10.3989/mc.2017.07716.
- Turner, L.K.; Collins, F. Carbon dioxide equivalent (CO2- e) emissions: A comparison between geopolymer and OPC cement concrete. Constr. Build. Mater. 2013, 43, 125–130, doi:10.1016/j.conbuildmat.2013.01.023.
- MacKenzie, K.J.D. Inorganic Polymers (Geopolymers); in Encyclopedia of Polymer Science and Technology, Wiley: New Jersey, NJ, USA, 2017; pp. 1–31, doi:10.1002/0471440264.pst165.pub2. ISBN9780471440260.
- Santhi, K.; Rani, C.; Karuppuchamy, S. Synthesis and characterization of a novel SnO/SnO2 hybrid photocatalyst. J. Alloy. Compd. 2016, 662, 102–107, doi:10.1016/j.jallcom.2015.12.007.
- Boonen, E.; Beeldens, A. Photocatalytic roads: From lab tests to real scale applications. Eur. Transp. Res. Rev. 2012, 5, 79–89, doi:10.1007/s12544-012-0085-6.
- Falah, M. Synthesis of New Composites of Inorganic Polymers (Geopolymers) with Metal Oxide Nanoparticles and Their Photodegradation of Organic Pollutants, PhD thesis, Victoria University of Wellington 2015. Available online: http://researcharchive.vuw.ac.nz/handle/10063/4847.
- Shen, S.; Kronawitter, C.; Kiriakidis, G. An overview of photocatalytic materials. J. Materiomics 2017, 3, 1–2, doi:10.1016/j.jmat.2016.12.004.
- Karthikeyan, C.; Arunachalam, P.; Ramachandran, K.; Al-Mayouf, A.M.; Karuppuchamy, S. Recent advances in semiconductor metal oxides with enhanced methods for solar photocatalytic applications. J. Alloy. Compd. 2020, 828, 154281, doi:10.1016/j.jallcom.2020.154281.
- Hanus, M.J; Harris, A.T. Nanotechnology innovations for the construction industry, Prog. Mater. Sci. 2013, 58, 1056–1102. doi:10.1016/j.pmatsci.2013.04.001.
- Pacheco-Torgal, F.; Jalali, S. Nanotechnology: Advantages and drawbacks in the field of construction and building materials. Constr. Build. Mater. 2011, 25, 582–590, doi:10.1016/j.conbuildmat.2010.07.009.
- Zheng, K.; Chen, L.; Gbozee, M. Thermal stability of geopolymers used as supporting materials for TiO2 film coating through sol-gel process: Feasibility and improvement. Constr. Build. Mater. 2016, 125, 1114–1126, doi:10.1016/j.conbuildmat.2016.09.007.
- Fan, W.; Lai, Q.; Zhang, Q.; Wang, Y. Nanocomposites of TiO2 and reduced graphene oxide as efficient photocatalysts for hydrogen evolution. J. Phys. Chem. C 2011, 115, 10694–10701, doi:10.1021/jp2008804.
- Chen, L.; Zheng, K.; Liu, Y. Geopolymer-supported photocatalytic TiO2 film: Preparation and characterization. Constr. Build. Mater. 2017, 151, 63–70, doi:10.1016/j.conbuildmat.2017.06.097.
- Strini, A.; Roviello, G.; Ricciotti, L.; Ferone, C.; Messina, F.; Schiavi, L.; Corsaro, D.; Cioffi, R. TiO2-based photocatalytic geopolymers for nitric oxide degradation. Materials 2016, 9, 513, doi:10.3390/ma9070513.
- Bravo, P.I.; Malenab, R.A.; Shimizu, E.; Yu, D.E.; Promentilla, M.A. Synthesis of geopolymer spheres with photocatalytic activity. MATEC Web Conf. 2019, 268, 04007, doi:10.1051/matecconf/201926804007.
- Samuneva, B.; Kozhukharov, V.; Trapalis, C.; Kranold, R. Sol-gel processing of titanium-containing thin coatings-Part I. Preparation and structure. J. Mater. Sci. 1993, 28, 2353–2360. doi:10.1007/BF01151665.
- Gasca-Tirado, J.; Manzano-Ramírez, A.; Villaseñor-Mora, C.; Muñiz-Villarreal, M.; Zaldivar-Cadena, A.; Rubio-Avalos, J.; Borrás, V.A.; Mendoza, R.N. Incorporation of photoactive TiO2 in an aluminosilicate inorganic polymer by ion exchange. Microporous Mesoporous Mater. 2012, 153, 282–287, doi:10.1016/j.micromeso.2011.11.026.
- Yang, X.; Liu, Y.; Yan, C.; Peng, R.; Wang, H. Geopolymer-TiO2 nanocomposites for photocsatalysis: Synthesis by one-step adding treatment versus two-step acidification calcination. Minerals 2019, 9, 658, doi:10.3390/min9110658.
- Lertcumfu, N.; Jaita, P.; Thammarong, S.; Lamkhao, S.; Tandorn, S.; Randorn, C.; Tunkasiri, T.; Rujijanagul, G. Influence of graphene oxide additive on physical, microstructure, adsorption, and photocatalytic properties of calcined kaolinite-based geopolymer ceramic composites. Colloids Surf. A: Physicochem. Eng. Asp. 2020, 602, 602 125080, doi:10.1016/j.colsurfa.2020.125080.
- Gupta, K.; Khatri, O.P. Reduced graphene oxide as an effective adsorbent for removal of malachite green dye: Plausible adsorption pathways. J. Colloid Interface Sci. 2017, 501, 11–21, doi:10.1016/j.jcis.2017.04.035.
- Gao, Y.; Wu, J.-C.; Chen, C.; Tan, X.; Hayat, T.; Alsaedi, A.; Cheng, C.; Chen, C. Impact of graphene oxide on the antibacterial activity of antibiotics against bacteria. Environ. Sci. Nano 2017, 4, 1016–1024, doi:10.1039/C7EN00052A.
- Gopalakrishnan, A.; Krishnan, R.; Thangavel, S.; Venugopal, G.; Kim, S.-J.; Arthi, G.; Rajasekar, K.; Sakthivel, T.; Gunasekaran, V. Removal of heavy metal ions from pharma-effluents using graphene-oxide nanosorbents and study of their adsorption kinetics. J. Ind. Eng. Chem. 2015, 30, 14–19, doi:10.1016/j.jiec.2015.06.005.
- Shamsaei, E.; De Souza, F.B.; Yao, X.; Benhelal, E.; Akbari, A.; Zhuang, J. Graphene-based nanosheets for stronger and more durable concrete: A review. Constr. Build. Mater. 2018, 183, 642–660, doi:10.1016/j.conbuildmat.2018.06.201.
- Zhang, Y.J.; Yang, M.Y.; Zhang, L.; Zhang, K.; Kang, L. A new graphene/geopolymer nanocomposite for degradation of dye wastewater. Integr. Ferroelectr. 2016, 171, 38–45, doi:10.1080/10584587.2016.1171178.
- Zhang, Y.J.; He, P.Y.; Zhang, Y.X.; Chen, H. A novel electroconductive graphene/fly ash-based geopolymer composite and its photocatalytic performance. Chem. Eng. J. 2018, 334, 2459–2466, doi:10.1016/j.cej.2017.11.171.
- Saleh, T.A. Syntheses and Applications of Carbon Nanotubes and Their Composites; InTech Open: London, UK, 2013. doi:10.5772/3377.
- Zheng, Z.; Huang, B.; Wang, Z.; Guo, M.; Qin, X.; Zhang, X.; Wang, P.; Dai, Y. Crystal Faces of Cu2O and Their stabilities in photocatalytic reactions. J. Phys. Chem. C 2009, 113, 14448–14453, doi:10.1021/jp904198d.
- Huang, L.; Peng, F.; Yu, H.; Wang, H. Preparation of cuprous oxides with different sizes and their behaviors of adsorption, visible-light driven photocatalysis and photocorrosion. Solid State Sci. 2009, 11, 129–138, doi:10.1016/j.solidstatesciences.2008.04.013.
- Falah, M.; MacKenzie, K.J.D.; Hanna, J.V.; Page, S.J. Novel photoactive inorganic polymer composites of inorganic polymers with copper (I) oxide nanoparticles. J. Mater. Sci. 2015, 50, 7374–7383, doi:10.1007/s10853-015-9295-3.
- Kumar, M. Effect of Nanomaterials on the Properties of Geopolymer Mortars and Concrete; Elsevier BV, Amsterdam, USA, 2018; Volume 5, pp. 9035–9040.
- Mahmoodi, N.M. Synthesis of magnetic carbon nanotube and photocatalytic dye degradation ability. Environ. Monit. Assess. 2014, 186, 5595–5604, doi:10.1007/s10661-014-3805-7.
- Bi, S.; Liu, M.; Shen, J.; Hu, X.; Zhang, L. Ultrahigh self-sensing performance of geopolymer nanocomposites via unique interface engineering. ACS Appl. Mater. Interfaces 2017, 9, 12851–12858, doi:10.1021/acsami.7b00419.