| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hyeon-Ju Oh | + 976 word(s) | 976 | 2020-10-04 15:28:04 | | | |
| 2 | Hyeon-Ju Oh | Meta information modification | 976 | 2020-10-09 13:33:59 | | | | |
| 3 | Hyeon-Ju Oh | -75 word(s) | 901 | 2020-10-09 20:48:04 | | | | |
| 4 | Vicky Zhou | Meta information modification | 901 | 2020-10-10 05:06:19 | | | | |
| 5 | Vicky Zhou | Meta information modification | 901 | 2020-10-26 10:12:36 | | |
Video Upload Options
Characterize particulate matter (PM) concentrations released during the application of cosmetic powders, estimate the respiratory dosage for the different cosmetic powder types, and evaluate the sustainability based on the environmental and health effects.
1. Introduction
Cosmetics are attractive to consumers of various ages [1][2][3] because they enhance or change appearance without affecting the structure or function of the body [4]. According to the Environmental Working Group, women use an average of 12 products per day, containing 168 different chemicals [5][6]. Over 3000 chemicals are used to formulate a large range of fragrances used in consumer products worldwide [7]. In the United States alone, there are approximately 12,500 unique chemical ingredients approved for use in the manufacture of personal care products. The combination of active ingredients in cosmetics is essential for improving skin health in areas such as anti-aging, moisturizing, and acne treatment [8], and cosmetic manufacturers are constantly developing new products to meet the needs of users.
European Regulation (EC) No. 1223/2009 states that “cosmetics are any substance or mixture intended to come into contact with the external parts of the human body (epidermis, hair system, nails).” In particular, the EC regulations, which took effect 11 July 2013, stipulate that cosmetics found in the European market must be safe for consumer health. Some commonly used cosmetics include products that can be rinsed immediately (e.g., shampoo, toothpaste), but each type of product includes a body emulsion that can touch the skin for several hours, especially a powder or spray that can be directly exposed to the body’s respiratory system.
2. Effect of Cosmetics on Respiratory System
When using consumer products, airborne particles can be released as a respirable fraction of the health-related particle size [9][10][11]. Moreover, people may be exposed to nanosized and unsafe ingredients. For example, Zn [12][13], TiO2 [14][15][16][17][18][19][20][21][22][23][24][25][26][27][28][29], SiO2 [30][31][32], Ag [33][34][35][36] and Au substances [37][38] were included in respirable particles, and these airborne particles could penetrate deep into the gas-exchange regions of the lungs [39][40][41][42][43][44].
Currently, nano-cosmetics are an area of particular interest because nanomaterials can be used to create new types of products. Since nanotechnology is a key driver of product innovation, it has the potential to change many parts of everyday consumer goods, especially cosmetics, and a variety of products are already on the market. However, respirable-sized particles (including nanoparticles) in cosmetics have been used as unstable particles or insoluble particles that are decomposed into molecular components [45][46][47][48][49][50], and consumers must be assured about the safety of the particles used [51]. Ingredients present in cosmetics are used as antioxidants, preservatives, emollients, surfactants, pigments, fragrances, and ultraviolet absorbers, but are strictly regulated by relevant laws in many countries [52]. In addition, some toxicity studies have tested manufactured nanomaterial products in different exposure situations to demonstrate the adverse effects of nanoparticles containing metal that can induce significant neurotoxicity, cytotoxicity, and genotoxicity in human cell cultures, which is critical for assessing the health risk to human or animal cells. However, these studies only observed a dose-dependent and time-dependent relationship at the highest dose at 24 h and 48 h post-exposure in cultured human cells and did not consider a realistic exposure approach [53][54][55][56][57][58][59].
These issues raise concerns about potential consumer exposure to penetrating particles in the lungs, including the use of cosmetics and personal care products, and, in particular, the toxic inorganic components contained within them [52]. At the same time, relatively little is known about the quantitative exposure assessment to inhalable aerosols when using cosmetics in general. In a recent study investigating the potential inhalation exposure and lung deposition resulting from the use of cosmetic powders, including nanoparticle formulations [60], it was found that particles of respirable size (< 10 µm) were released and potentially inhaled. In the case of respirable-sized cosmetics, once inhaled, these particles settle in all areas of the respiratory system from the head airways to the alveoli. To this end, it is necessary to conduct a quantitative exposure assessment of aerosols generated when using cosmetics and to investigate possible toxic substances contained in the exposed aerosols.
The respiratory exposure characteristics depend on the chemical manufacturing route, the manufacturing process, and the conflicting views of manufacturers [61]. The report “Our Common Future” highlighted the need for a sustainable way of life [62]. Since then, the world has changed dramatically socially and economically, raising consumer awareness of environmental and social issues regarding resources consumed at an unsustainable rate. Based on these issues, the main decision to purchase cosmetics still depends on personal preferences, but environmental and ethical considerations are becoming increasingly important [63].
Another factor driving the cosmetics industry to a more sustainable path is the availability of raw materials that are advantageous from a sustainability perspective [64]. Some countries in the European Union have already banned the use of microplastics in cosmetics [65]. The use of plastic microbeads has decreased by 97.6% between 2012 and 2017; 4250 tons of plastic has been replaced [66]. From another sustainability perspective, some countries mandate the labeling of environmental indicators and impose various environmental taxes such as a packaging tax or landfill tax, which affects purchasing behaviors [67].
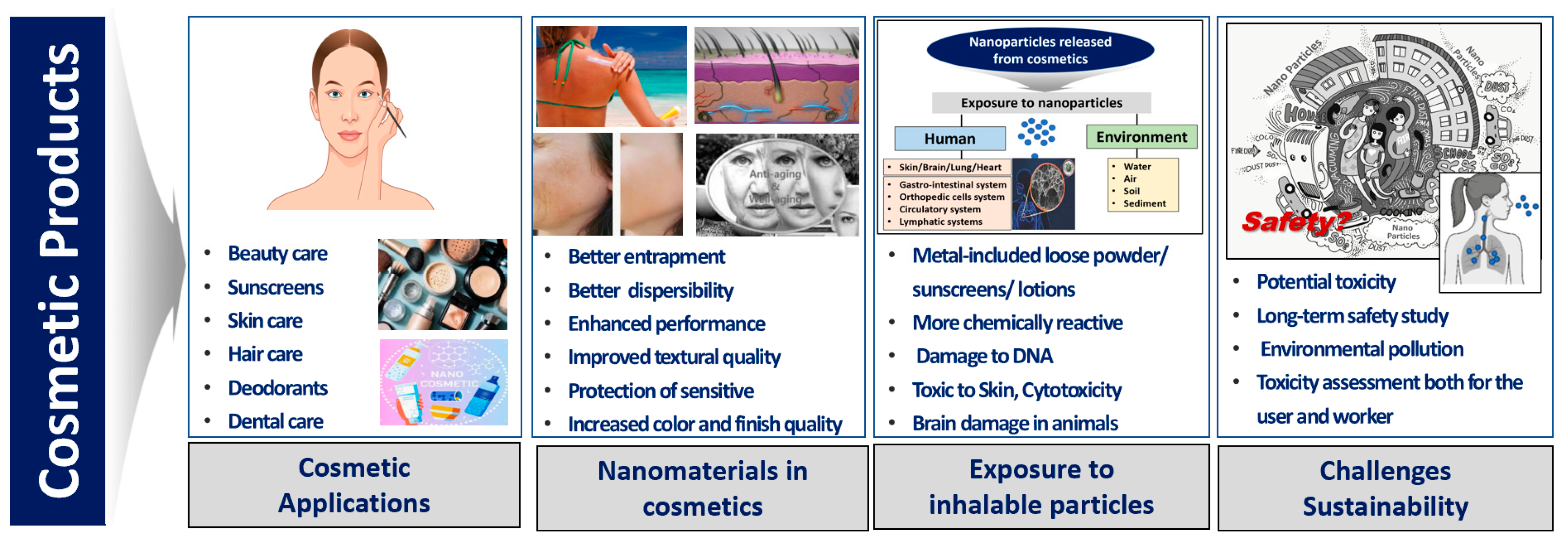
Furthermore, the cosmetic supply chain can have an impact on sustainability, so every step in the product lifecycle must be considered from the initial design and sourcing of raw materials to manufacturing, packaging, and distribution. The term “eco-innovation” is crucially an overall assessment of the environmental impacts and risks [68], which can be a tool to support assessment within the environmental dimension of cosmetic sustainability [69] (Figure 1).
Figure 1. Cosmetic applications, exposure to inhalable particles released from cosmetics and sustainability challenges of future strategies [8][50][69].
References
- Ficheux, A.S.; Dornic, N.; Bernard, A.; Chevillotte, G.; Roudot, A.C. Probabilistic assessment of exposure to cosmetic products by French children aged 0-3 years. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2016, 94, 85–92.
- Biesterbos, J.W.; Dudzina, T.; Delmaar, C.J.; Bakker, M.I.; Russel, F.G.; von Goetz, N.; Scheepers, P.T.; Roeleveld, N. Usage patterns of personal care products: Important factors for exposure assessment. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2013, 55, 8–17.
- Gomez-Berrada, M.P.; Ficheux, A.S.; Guillou, S.; Berge, C.; de Javel, D.; Roudot, A.C.; Ferret, P.J. Consumption and exposure assessment to cosmetic products for children under 2 years old. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2017, 105, 151–160.
- Nourmoradi, H.; Foroghi, M.; Farhadkhani, M.; Vahid Dastjerdi, M. Assessment of lead and cadmium levels in frequently used cosmetic products in Iran. J. Environ. Public Health 2013, 2013, 962727.
- Lupkin, S. Women Put an Average of 168 Chemicals on Their Bodies Each Day, Consumer Group Says. Available online: https://abcnews.go.com/Health/women-put-average-168-chemicals-bodies-day-consumer/story?id=30615324 (accessed on 10 August 2020).
- Chow, L. Women Apply an Average of 168 Chemicals on Their Bodies Every Day. Available online: https://www.ecowatch.com/women-apply-an-average-of-168-chemicals-on-their-bodies-every-day-1882041568.html (accessed on 5 September 2020).
- Jones, O.; Sellinger, B. The Chemistry of Cosmetics. Australian Academy of Science. Available online: https://www.science.org.au/curious/people-medicine/chemistry-cosmetics (accessed on 13 August 2020).
- Kumud, M.; Sanju, N. Nanotechnology Driven Cosmetic Products: Commercial and Regulatory Milestones. Appl. Clin. Res. Clin. Trials Regul. Aff. 2018, 5, 112–121.
- Baan, R.A. Carcinogenic hazards from inhaled carbon black, titanium dioxide, and talc not containing asbestos or asbestiform fibers: Recent evaluations by an IARC Monographs Working Group. Inhal. Toxicol. 2007, 19, 213–228.
- CCOHS How Do Particulates Enter the Respiratory System? Available online: https://www.ccohs.ca/oshanswers/chemicals/how_do.html (accessed on 26 August 2020).
- IARC. Carbon Black, Titanium Dioxide, and Talc; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC Press, International Agency for Research on Cancer: Lyon, France, 2010; Volume 93.
- Guan RK, T.; Lu, F.; Zhang, Z.; Shen, H.; Liu, M. Cytotoxicity, oxidative stress, and genotoxicity in human hepatocyte and embryonic kidney cells exposed to ZnO nanoparticles. Nanoscale Res. Lett. 2012, 7, 602–608.
- Alarifi, S.; Ali, D.; Alhadlaq, H.A. Induction of oxidative stress, DNA damage, and apoptosis in a malignant human skin melanoma cell line after exposure to zinc oxide nanoparticles. Int. J. Nanomed. 2013, 8, 983–993.
- Donner, E.M.; Myhre, A.; Brown, S.C.; Boatman, R.; Warheit, D.B. In vivo micronucleus studies with 6 Titanium Dioxide materials (3 pigment-grade & 3 nanoscale) in orally-exposed rats. Regul Toxicol Pharm. 2016, 74, 64–74.
- Guichard, Y.S.J.; Darne, C.; Gaté, L.; Goutet, M.; Rousset, D.; Binet, S. Cytotoxicity and genotoxicity of nanosized and microsized Titanium Dioxide and Iron Oxide particles in Syrian Hamster embryo cells. Ann. Occup. Hyg. 2015, 56, 631–644.
- Hamzeh, M.; Sunahara, G.I. In vitro cytotoxicity and genotoxicity studies of titanium dioxide (TiO2) nanoparticles in Chinese hamster lung fibroblast cells. Toxicol. In Vitro 2013, 27, 864–873.
- Jomini, S.L.J.; Bauda, P.; Pagnout, C. Modifications of the bacterial reverse mutation test reveals mutagenicity of TiO2 nanoparticles and byproducts from a sunscreen TiO2-based nanocomposite. Toxicol Lett. 2012, 215, 54–61.
- Landsiedel, R.M.-H.L.; Van Ravenzwaay, B.; Schulz, M.; Wiench, K.; Champ, S.; Oesch, F. Gene toxicity studies on titanium dioxide and zinc oxide nanomaterials used for UV-protection in cosmetic formulations. Nanotoxicology 2010, 4, 364–381.
- Lindberg, H.K.; Falk, G.-M.; Catalán, J.; Koivisto, A.J.; Suhonen, S.; Järventaus, H.; Norppa, H. Genotoxicity of inhaled nanosized TiO2 in mice. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2012, 745, 58–64.
- Naya, M.; Kobayashi, N.; Ema, M.; Kasamoto, S.; Fukumuro, M.; Takami, S.; Nakanishi, J. In vivo genotoxicity study of titanium dioxide nanoparticles using comet assay following intratracheal instillation in rats. Regul. Toxicol. Pharm. 2012, 62, 1–6.
- Sadiq, R.; Bhalli, J.A.; Yan, J.; Woodruff, R.S.; Pearce, M.G.; Li, Y.; Chen, T. Genotoxicity of TiO2 anatase nanoparticles in B6C3F1 male mice evaluated using Pig-a and flow cytometric micronucleus assays. Mutat. Res. 2012, 745, 65–72.
- Tavares, A.M.; Louro, H.; Antunes, S.; Quarré, S.; Simar, S.; De Temmerman, P.-J.; Silva, M.J. Genotoxicity evaluation of nanosized titanium dioxide, synthetic amorphous silica and multi-walled carbon nanotubes in human lymphocytes. Toxicol. In Vitro 2014, 28, 60–69.
- Wang, S.; Hunter, L.A.; Arslan, Z.; Wilkerson, M.G.; Wickliffe, J.K. Chronic exposure to nanosized, anatase titanium dioxide is not cyto- or genotoxic to Chinese hamster ovary cells. Environ. Mol. Mutagen. 2011, 52, 614–622.
- Woodruff, R.S.; Li, Y.; Yan, J.; Bishop, M.; Jones, M.Y.; Watanabe, F.; Chen, T. Genotoxicity evaluation of titanium dioxide nanoparticles using the Ames test and Comet assay. J. Appl. Toxicol. 2012, 32, 934–943.
- Mavon, A.; Miquel, C.; Lejeune, O.; Payre, B.; Moretto, P. In vitro percutaneous absorption and in vivo stratum corneum distribution of an organic and a mineral sunscreen. Skin Pharmacol. Physiol. 2007, 20, 10–20.
- Bernardeschi, M.; Guidi, P.; Scarcelli, V.; Frenzilli, G.; Nigro, M. Genotoxic potential of TiO2 on bottlenose dolphin leukocytes. Anal. Bioanal. Chem. 2010, 396, 619–623.
- Osman, I.F.; Baumgartner, A.; Cemeli, E.; Fletcher, J.N.; Anderson, D. Genotoxicity and cytotoxicity of zinc oxide and titanium dioxide in HEp-2 cells. Nanomedicine 2010, 5, 1193–2203.
- Shukla, R.K.; Sharma, V.; Pandey, A.K.; Singh, S.; Sultana, S.; Dhawan, A. ROS-mediated genotoxicity induced by titanium dioxide nanoparticles in human epidermal cells. Toxicol. In Vitro 2011, 25, 231–241.
- Sycheva, L.P.; Zhurkov, V.S.; Iurchenko, V.V.; Daugel-Dauge, N.O.; Kovalenko, M.A.; Krivtsova, E.K.; Durnev, A.D. Investigation of genotoxic and cytotoxic effects of micro- and nanosized titanium dioxide in six organs of mice in vivo. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2011, 726, 8–14.
- Kwon, J.Y.; Koedrith, P.; Seo, Y.R. Current investigations into the genotoxicity of zinc oxide and silica nanoparticles in mammalian models in vitro and in vivo: Carcinogenic/genotoxic potential, relevant mechanisms and biomarkers, artifacts, and limitations. Int. J. Nanomed. 2014, 9, 271–286.
- Liu, X.; Keane, M.J.; Zhong, B.Z.; Ong, T.M.; Wallace, W.E. Micronucleus formation in V79 cells treated with respirable silica dispersed in medium and in simulated pulmonary surfactant. Mutat. Res. 1996, 361, 89–94.
- Yang, H.; Liu, C.; Yang, D.; Zhang, H.; Xi, Z. Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: The role of particle size, shape and composition. J. Appl. Toxicol. 2009, 29, 69–78.
- Asharani, P.V.; Mun, L.G.K.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and Genotoxicity of Silver Nanoparticles in Human Cells. ACS Nano 2009, 3, 279–290.
- Kaiser, J.P.; Roesslein, M.; Diener, L.; Wichser, A.; Nowack, B.; Wick, P. Cytotoxic effects of nanosilver are highly dependent on the chloride concentration and the presence of organic compounds in the cell culture media. J. Nanobiotechnol. 2017, 15, 1–11.
- Dobrzyńska, M.M.; Gajowik, A.; Radzikowska, J.; Lankoff, A.; Dušinská, M.; Kruszewski, M. Genotoxicity of Silver and Titanium Dioxide nanoparticles in bone marrow cells of rats in vivo. Toxicology 2014, 315, 86–91.
- Plotnikov, E.; Zhuravkov, S.; Gapeyev, A.; Plotnikov, V.; Martemianova, I.; Martemianov, D. Comparative study of genotoxicity of Silver and Gold nanoparticles prepared by the electric spark dispersion method. J. Appl. Pharm. Sci. 2017, 7, 35–39.
- Jia, Y.-P.; Ma, B.-Y.; Wei, X.-W.; Qian, Z.-Y. The in vitro and in vivo toxicity of gold nanoparticles. Chin. Chem. Lett. 2017, 28, 691–702.
- De Alteriis, E.; Falanga, A.; Galdiero, S.; Guida, M.; Maselli, C.; Galdiero, E. Genotoxicity of gold nanoparticles functionalized with indolicidin towards Saccharomyces cerevisiae. J. Environ. Sci. 2017, 66, 138–145.
- Kreyling, W.G.; Semmler, M.; Erbe, F.; Mayer, P.; Takenaka, S.; Schulz, H.; Oberdorster, G.; Ziesenis, A. Translocation of ultrafine insoluble iridium particles from lung epithelium to extrapulmonary organs is size dependent but very low. J. Toxicol. Environ. Health Part A 2002, 65, 1513–1530.
- Bérubé, V.; Radtke, G.; Dresselhaus, M.; Chen, G. Size effects on the hydrogen storage properties of nanostructured metal hydrides: A review. Int. J. Energy Res. 2007, 31, 637–663.
- Warheit, D.B.; Webb, T.R.; Reed, K.L.; Frerichs, S.; Sayes, C.M. Pulmonary toxicity study in rats with three forms of ultrafine-TiO2 particles: Differential responses related to surface properties. Toxicology 2007, 230, 90–104.
- Gate, L.; Disdier, C.; Cosnier, F.; Gagnaire, F.; Devoy, J.; Saba, W.; Brun, E.; Chalansonnet, M.; Mabondzo, A. Biopersistence and translocation to extrapulmonary organs of titanium dioxide nanoparticles after subacute inhalation exposure to aerosol in adult and elderly rats. Toxicol. Lett. 2017, 265, 61–69.
- IARC. Titanium Dioxide Group 2B; IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC Press, International Agency for Research on Cancer: Lyon, France, 2006; Volume 9.
- OEHHA. Public Hearing to Consider Amendments to the Ambient Air Quality Standards for Particulate Matter and Sulfates; Air Resources Board, California Environmental Protection Agency: Sacramento, CA, USA, 2002.
- Nanosystems, P. Lipid Nanoparticles. Available online: http://www.precisionnanosystems.com/areas-of-interest/formulations/lipid-nanoparticles (accessed on 13 August 2020).
- Muzzalupo, R.; Tavano, L. Niosomal drug delivery for transdermal targeting: Recent advances. Res. Rep. Transdermal Dug Deliv. 2015, 4, 23–33.
- Pulicharla, R.; Zolfaghari, M.; Brar, S.K.; Cledon, M.; Drogui, P.; Surampalli, R.Y. Cosmetic Nanomaterials in Wastewater: Titanium Dioxide and Fullerenes. J. Hazard. Toxic. Radioact. Waste 2016, 20, B4014005.
- Al-Bedairy, M.; Alshamsi HA, H. Environmentally Friendly Preparation of Zinc Oxide, Study Catalytic Performance of Photodegradation by Sunlight for Rhodamine B Dye. Eurasian J. Anal. Chem. 2018, 13, 1–9.
- Kaul, S.; Gulati, N.; Verma, D.; Mukherjee, S.; Nagaich, U. Role of Nanotechnology in Cosmeceuticals: A Review of Recent Advances. J. Pharm. 2018, 2018, 1–19.
- Chaudhri, N.; Soni, G.C.; Prajapati, S.K. Nanotechnology: An Advance Tool for Nano-cosmetics Preparation. Int. J. Pharm. Res. Rev. 2015, 4, 28–40.
- Nanda, S.; Arun, N.; Lohan, S.; Kaur, R.; Singh, B. Nanobiomaterials in Galenic Formulations and Cosmetics; William Andrew: Norwich, NY, USA, 2016; Volume 10.
- Borowska, S.; Brzoska, M.M. Metals in cosmetics: Implications for human health. J. Appl. Toxicol. 2015, 35, 551–572.
- Wang, J.J.; Sanderson, B.J.; Wang, H. Cyto- and genotoxicity of ultrafine TiO2 particles in cultured human lymphoblastoid cells. Mutat. Res. 2007, 628, 99–106.
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839.
- Colvin, V.L. The potential environmental impact of engineered nanomaterials. Nat. Biotechnol. 2003, 21, 1166–1170.
- Chen, J.; Dong, X.; Zhao, J.; Tang, G. In vivo acute toxicity of titanium dioxide nanoparticles to mice after intraperitioneal injection. J. Appl. Toxicol. 2009, 29, 330–337.
- Liu, H.; Ma, L.; Zhao, J.; Liu, J.; Yan, J.; Ruan, J.; Hong, F. Biochemical toxicity of nano-anatase TiO2 particles in mice. Biol. Trace Elem. Res. 2009, 129, 170–180.
- Zhao, J.; Li, N.; Wang, S.; Zhao, X.; Wang, J.; Yan, J.; Ruan, J.; Wang, H.; Hong, F. The mechanism of oxidative damage in the nephrotoxicity of mice caused by nano-anatase TiO2. J. Exp. Nanosci. 2010, 5, 447–462.
- Song, B.; Liu, J.; Feng, X.; Wei, L.; Shao, L. A review on potential neurotoxicity of titanium dioxide nanoparticles. Nanoscale Res. Lett. 2015, 10, 342–358.
- Nazarenko, Y.; Zhen, H.; Han, T.; Lioy, P.J.; Mainelis, G. Potential for inhalation exposure to engineered nanoparticles from nanotechnology-based cosmetic powders. Environ. Health Perspect. 2012, 120, 885–892.
- Adawi, H.I.; Newbold, M.A.; Reed, J.M.; Vance, M.E.; Feitshans, I.L.; Bickford, L.R.; Lewinski, N.A. Nano-enabled personal care products: Current developments in consumer safety. NanoImpact 2018, 11, 170–179.
- Environment & Society Portal. Report of the World Commission on Environment and Development: Our Common Future. Available online: http://www.environmentandsociety.org/mml/un-world-commission-environment-and-development-ed-report-world-commission-environment-and (accessed on 27 August 2020).
- Liobikienė, G.; Bernatonienė, J. Why determinants of green purchase cannot be treated equally? The case of green cosmetics: Literature review. J. Clean. Prod. 2017, 162, 109–120.
- Krauter, J. In-Cosmetics 2018: Evonik Focuses on Naturalness. Available online: https://www.cosmeticsdesign-europe.com/Article/2018/10/16/Evonik-hones-in-on-naturals-and-sustainability (accessed on 28 August 2020).
- Xanthos, D.; Walker, T.R. International policies to reduce plastic marine pollution from single-use plastics (plastic bags and microbeads): A review. Mar. Pollut. Bull. 2017, 118, 17–26.
- Cosmetics Europe. Over 97% of Plastic Microbeads already Phased out from Cosmetics–Cosmetics Europe Announces. Available online: https://cosmeticseurope.eu/news-events/over-97-plastic-microbeads-already-phased-out-cosmetics-cosmetics-europe-announces (accessed on 28 August 2020).
- Sahota, A. Sustainability: How the Cosmetics Industry Is Greening Up; Wiley: West Sussex, UK, 2014; Volume 1.
- Motta, W.H.; Issberner, L.-R.; Prado, P. Life cycle assessment and eco-innovations: What kind of convergence is possible? J. Clean. Prod. 2018, 187, 1103–1114.
- Krishna, M., IV; Manickam, V. Environmental Risk Assessment; Elsevier: Oxford, UK, 2017; Volume 1, pp. 135–150.