| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Amira Mohammed Ali | + 3290 word(s) | 3290 | 2020-09-28 05:57:51 | | | |
| 2 | Rita Xu | -1756 word(s) | 1534 | 2020-10-06 10:39:07 | | |
Video Upload Options
Apitherapy is a complementary and alternative medicine approach, which involves the therapeutic use of various bee products including apilarnil (atomized drone larva) to prevent and treat various diseases.
1. Historical background
Rock paintings from the Stone Age portray the consumption of bee products by humans [1]. The first evidence of human usage of bee products for therapeutic and cosmetic purposes dates back 6000 years in ancient Egypt and later in China, Greece, and Rome [1][2][3][4]. Current research interest is directed toward the use of bee products as potential pharmaceuticals to modify the progression of numerous diseases [5].
2. Overview of bee products
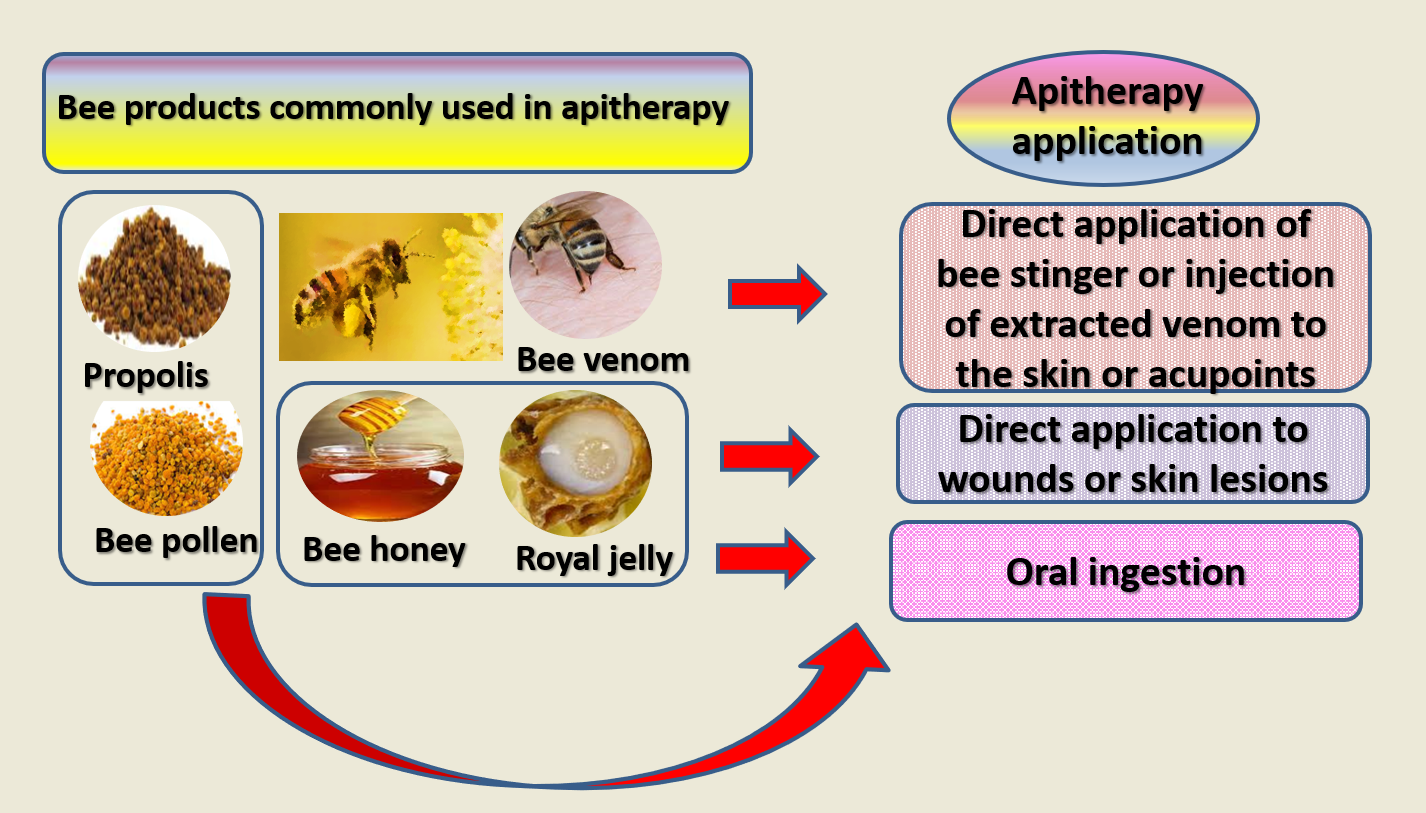
Bee workers of either Apis mellifera or Apis cerana species—the former is common in Europe, Asia, Africa, and America while the latter exists only in southern and southeastern Asia—produce and store multiple bioactive substances [6] Royal jelly, propolis, bee pollen, honey, bee venom, bee bread, and bee wax are common products of the bee hive. They all (to a varying degree) possess multiple health-promoting properties due to their high content of natural antioxidants (e.g., flavonoids, phenols, or terpenoids), peptides, rare lipids (such as those found in royal jelly), vitamins, minerals, trace elements, and several bioactive substances [3][6][7]. Research documents variability in the contents and effects of every single bee product, mainly due to variations in bee species, botanical origin, geographic location, season, extraction and handling procedures [3][8].
Figure 1. Bee products commonly involved in apitherapy along with common routes of admiration.
3. Applications of apitherapy
As shown in Figure 1, many bee products are used as pharmaceuticals. The most commonly used types are bee venom, royal jelly, honey, propolis, and bee pollen. All these products except for bee venom can be ingested to bring about health effects [9][10]. Meanwhile, propolis, royal jelly and honey can be used topically used to treat psoriasis [11], wounds and burns [12]. Given their health-promoting properties, these products are widely used as dietary supplements [9][13].
4. Bee venom therapy
Bee venom is a complex neurotoxin that exists in the stinger of honey bee workers. It comprises a large number of bioactive compounds such as polypeptides and enzymes [14]. Owing to its anti-inflammatory, antifibrotic, antioxidant, anti-microbial, and neuroprotective activities, bee venom has long been used as a traditional medicine modality, especially in the far east [15][16]. Recent research signifies positive effects of bee venom in chronic and hard-to-treat disorders such as Alzheimer’s disease and rheumatoid arthritis [17][18]. Given that bee venom exists in the stinger of living bee workers, this therapy is administered either through direct application of the stinger to certain disease-related acupoints in the skin or by subcutaneously injecting these points with extracted bee venom (Figure 1). Allergy testing is usually performed before the commence of treatment because some individuals may be allergic to phospholipase A2 in bee venom. Alternatively, treatment may be conducted only by melittin, the most copious bioactive substance in bee venom [19].
5. Apitherapy for age-related pathologies
Intracellular molecules involved in inflammatory and oxidative stresses increase with aging and contribute to cellular dysfunction—a condition described as inflammaging [8][20]. Both inflammatory mediators and free radicals threaten health span and lifespan by inducing age-related pathologies such as atherosclerosis, diabetes mellitus, obesity, neurodegenerative disease, to name a few [8]. Several lines of evidence describe anti-aging effects of royal jelly, bee pollen, and propolis both in humans and laboratory animals [8][21][22][23]. Here we give few examples of how bee products express anti-aging effects.
6. Apitherapy for neurodegenerative disorders
Alzheimer’s disease and Parkinson’s disease are the most common age-related neurodegenerative diseases. Research shows that both disorders develop as a result of a chronic inflammatory condition that involves neurons and glial cells [13][24]. Glial overactivity promotes the expression of pathogenic genes that underly Alzheimer’s disease and Parkinson’s disease such as APOE [25] and alpha-synuclein [26][27], respectively. These genes further inflammation and oxidative stress both in neurons and in the blood brain barrier promoting the passage of neurotoxic molecules (e.g., amyloid beta peptides and alpha-synuclein) from the system circulation to neurons [24][25][26][27]. Intraneuronal accumulation of the toxic amyloid beta peptides along with the uncontrolled release of inflammatory mediators and associated free radicals by dysfunctional glial cells potentiate the development of pours in mitochondrial membrane resulting in the release of molecules that activate cellular apoptosis [28][29]. Research denotes that the origin of inflammation in most disorders including Alzheimer’s disease and Parkinson’s disease is gut microbial alteration, which causes aberrations in the intestinal membrane allowing the passage of bacterial toxins (e.g., lipopolysaccharide) to the systemic circulation and subsequently to the brain to stimulate neuroinflammation [30][31][32][33].
Cumulative evidence shows that bee products such as royal jelly may prevent neurodegeneration in Alzheimer’s disease through the suppression of glial activation, protection of the blood brain barrier against leakage, improvement of neuronal energy supply, downregulation of inflammatory and oxidative signaling, improvement of signal transduction (e.g., acetylcholine), increasing the production of neurotrophic factors (e.g., brain-derived neurotrophic factor and nerve growth factor), increasing beta amyloid clearance from the brain and discouraging its accumulation inside neurons [24]. Likewise, royal jelly and propolis are reported to correct behavioral deficits in laboratory models of Parkinson’s disease via mechanisms close to those occurring in experimental models of Alzheimer’s disease [13].
7. Apitherapy for sarcopenia
Sarcopenia, also known as physical frailty, is an age-bound condition that entails loss of skeletal muscle mass, strength, and physical performance. Sarcopenia is multifactorial involving gender-related tendencies, genetics, behavioral causes (e.g., physical inactivity and inadequate dietary intake) on top of inflammaging [9]. Almost all available pharmacological treatments of sarcopenia are experimental with no approved drugs until now [34]. Existing research shows that royal jelly, bee pollen, and propolis express a potential of restoring muscle mass and strength in experimental models of aging [9]. Mechanisms contributing to such action involve inhibition of muscle atrophy genes [35], activation of myosatellite stem cells [35][36], neuronal reinnervation [37], metabolic regulation [36][38][39][40], improvement of muscular blood supply [41][42], as well as inhibition of oxidative stress, inflammation and apoptosis [43][44][45].
8. Apitherapy for insulin resistance
Insulin is a multifunctional hormone that is mainly responsible for cellular glucose uptake. Its signaling pathway interacts with a variety of other signaling cascades and affects their functioning within every single cell in the body [8]. Insulin resistance and glucose intolerance increase during old age evoking conversions in muscle fibers from type-1 to type-2 inclined in muscle loss [9]. Glucose metabolism and insulin contribute to Ach synthesis by controlling the activity of choline acetyltransferase [46]. On the other hand, cerebral insulin resistance develops in old as well as obese individuals due to depletion of adiponectin—an adipose tissue-derived adipokine that regulates neurogenesis and the metabolism of lipids and glucose [47][48]. Alterations in nutrient supply to the brain are associated with increased production of free radicals and active glycation products, which accelerate inflammation in brain cell leading to neurodegeneration [24]. Several studies show that bee products express potent hypoglycemic activities. For example, positive effects of whole royal jelly and royal jelly acid (trans-10-hydroxy-2-decenoic acid, 10-HDA) on skeletal muscle are associated improved insulin signaling and translocation of glucose transporter 4 (GLUT4) to the cell surface for glucose uptake [9][36][38][39].
9. Apitherapy for fatigue
Fatigue is a universal experience that comprises an overwhelming feeling of exhaustion, lack of energy, weariness along with a heightened sensation of physical or mental strain, even with absence of conspicuous efforts [48]. It is widely experienced by old people [48], patients undergoing exhaustive disorders such as kidney failure and cancer (up to 96% of cancer patients) [10][49], and patients with symptomatic Corona Virus Disease 2019 (COVID-19) infection [50]. It is also highly prevalent among people who exert excessive physical efforts such as athletes, and it may impede their achievement. Untreated fatigue is associated with distress, sleep loss, depression, distorted family and work relations, and poor quality of life [48]. In this regard, research denotes anti-fatigue effects of bee products (e.g., royal jelly, propolis, bee pollen, and bee honey) both in experimental animals on eccentric exercises [9][51] and in human athletes [52] as well as in patients with cancer [49].
10. Therapeutic mechanism of action of bee products
Bee products share common mechanisms of action. In particular, most of these products are strong free radical scavengers. This activity is attributed to their phenolic components, which donate an electron from their hydroxyl group [8]. They also activate the major signaling pathway nuclear respiratory factor 2 (NRF2), which contributes to the production of internal antioxidants, controls inflammatory signaling, and promotes autophagy. Through these activities, bee products foster proper cellular housekeeping where harmful molecules that buildup as a result of metabolism and other cellular activities (e.g., misfolded proteins) get properly removed [7][8][9][24][53]. Bee products possess a capacity to stimulate the regeneration and responsiveness of stem cells allowing a chance for cellular replacement and repair of damages [9][24]. Their potential to activate adenosine monophosphate activated protein kinase (AMPK) and mammalian target of rapamycin (mTOR) allows them to regulate cellular energy and other vital cellular processes [9][24].
11. Conclusion
Owing to their special composition, bee products are multifunctional compounds that are capable of counteracting most pathological mechanisms that contribute to ill-health and premature and/or dysfunctional aging such as inflammation, oxidative stress, gut dysbiosis, mitochondrial dysfunction. Existing experimental knowledge denotes the potential benefits of the therapeutic use of bee products for the treatment of age-related disorders (e.g., sarcopenia, insulin resistance, Alzheimer’s disease, etc.). However, there is an intense need for well-designed trials investigating the effects of apitherapy in human subjects.
References
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and bee bread as new health-oriented products: A review. Trends Food Sci Technol 2018, 71, 170-180, doi:https://doi.org/10.1016/j.tifs.2017.10.021.
- Nitecka-Buchta, A.; Buchta, P.; Tabenska-Bosakowska, E.; Walczynska-Dragon, K.; Baron, S. Myorelaxant effect of bee venom topical skin application in patients with RDC/TMD Ia and RDC/TMD Ib: a randomized, double blinded study. Biomed Res Int 2014, 2014, 296053, doi:10.1155/2014/296053.
- Kocot, J.; Kielczykowska, M.; Luchowska-Kocot, D.; Kurzepa, J.; Musik, I. Antioxidant Potential of Propolis, Bee Pollen, and Royal Jelly: Possible Medical Application. Oxid Med Cell Longev 2018, 2018, 7074209, doi:10.1155/2018/7074209.
- Fratini, F.; Cilia, G.; Mancini, S.; Felicioli, A. Royal Jelly: An ancient remedy with remarkable antibacterial properties. Microbiol Res 2016, 192, 130-141, doi:10.1016/j.micres.2016.06.007.
- Alvarez-Fischer, D.; Noelker, C.; Vulinovic, F.; Grunewald, A.; Chevarin, C.; Klein, C.; Oertel, W.H.; Hirsch, E.C.; Michel, P.P.; Hartmann, A. Bee venom and its component apamin as neuroprotective agents in a Parkinson disease mouse model. PLoS One 2013, 8, e61700, doi:10.1371/journal.pone.0061700.
- Cornara, L.; Biagi, M.; Xiao, J.; Burlando, B. Therapeutic Properties of Bioactive Compounds from Different Honeybee Products. Front Pharmacol 2017, 8, 412, doi:10.3389/fphar.2017.00412.
- Ali, A.M.; Hendawy, A.O. Royal Jelly Acid, 10-Hydroxy-Trans-2-Decenoic Acid, for Psychiatric and Neurological Disorders: How helpful could it be. Edelweiss Journal of Food Science and Technology 2019, 1, 1-4.
- Kunugi, H.; Ali, A.M. Royal Jelly and Its Components Promote Healthy Aging and Longevity: From Animal Models to Humans. Int. J. Mol. Sci 2019, 20, 4662, doi:https://doi.org/10.3390/ijms20194662.
- Ali, A.M.; Kunugi, H. Apitherapy for age-related skeletal muscle dysfunction (sarcopenia): A review on the effects of royal jelly, propolis, and bee pollen. Foods 2020, 9, doi:10.3390/foods9101362.
- Ali, A.M.; Kunugi, H. Potential anti-fatigue effects of bee honey, royal jelly, propolis, and bee pollen: a review. Molecules 2020, In progress.
- El-Gammal, E.; Nardo, V.D.; Daaboul, F.; Tchernev, G.; Wollina, U.; Lotti, J.; Lotti, T. Apitherapy as a New Approach in Treatment of Palmoplantar Psoriasis. Open Access Maced J Med Sci 2018, 6, 1059-1061, doi:10.3889/oamjms.2018.135.
- Martinotti, S.; Bucekova, M.; Majtan, J.; Ranzato, E. Honey: An Effective Regenerative Medicine Product in Wound Management. Curr Med Chem 2019, 26, 5230-5240, doi:10.2174/0929867325666180510141824.
- Ali, A.M.; Kunugi, H. Apitherapy for Parkinson's disease: A focus on the effects of propolis and royal jelly. Oxid Med Cell Longev 2020, https://doi.org/10.1155/2020/1727142, doi:https://doi.org/10.1155/2020/1727142.
- Uddin, M.B.; Lee, B.H.; Nikapitiya, C.; Kim, J.H.; Kim, T.H.; Lee, H.C.; Kim, C.G.; Lee, J.S.; Kim, C.J. Inhibitory effects of bee venom and its components against viruses in vitro and in vivo. J Microbiol 2016, 54, 853-866, doi:10.1007/s12275-016-6376-1.
- Shin, D.; Lee, G.; Sohn, S.H.; Park, S.; Jung, K.H.; Lee, J.M.; Yang, J.; Cho, J.; Bae, H. Regulatory T Cells Contribute to the Inhibition of Radiation-Induced Acute Lung Inflammation via Bee Venom Phospholipase A₂ in Mice. Toxins (Basel) 2016, 8, doi:10.3390/toxins8050131.
- Caramalho, I.; Melo, A.; Pedro, E.; Barbosa, M.M.; Victorino, R.M.; Pereira Santos, M.C.; Sousa, A.E. Bee venom enhances the differentiation of human regulatory T cells. Allergy 2015, 70, 1340-1345, doi:10.1111/all.12691.
- Zhang, S.; Liu, Y.; Ye, Y.; Wang, X.R.; Lin, L.T.; Xiao, L.Y.; Zhou, P.; Shi, G.X.; Liu, C.Z. Bee venom therapy: Potential mechanisms and therapeutic applications. Toxicon 2018, 148, 64-73, doi:10.1016/j.toxicon.2018.04.012.
- Somerfield, S.D.; Stach, J.L.; Mraz, C.; Gervais, F.; Skamene, E. Bee venom melittin blocks neutrophil O2- production. Inflammation 1986, 10, 175-182, doi:10.1007/bf00915999.
- Lee, Y.; Kim, S.G.; Kim, I.S.; Lee, H.D. Standardization of the Manufacturing Process of Bee Venom Pharmacopuncture Containing Melittin as the Active Ingredient. Evid Based Complement Alternat Med 2018, 2018, 2353280, doi:10.1155/2018/2353280.
- Miljkovic, N.; Lim, J.Y.; Miljkovic, I.; Frontera, W.R. Aging of skeletal muscle fibers. Ann Rehabil Med 2015, 39, 155-162, doi:10.5535/arm.2015.39.2.155.
- Eşrefoğlu, M.; Gül, M.; Ateş, B.; Erdoğan, A. The effects of caffeic acid phenethyl ester and melatonin on age-related vascular remodeling and cardiac damage. Fundam Clin Pharmacol 2011, 25, 580-590, doi:10.1111/j.1472-8206.2010.00876.x.
- Rzepecka-Stojko, A.; Stojko, J.; Kurek-Górecka, A.; Górecki, M.; Kabała-Dzik, A.; Kubina, R.; Moździerz, A.; Buszman, E. Polyphenols from Bee Pollen: Structure, Absorption, Metabolism and Biological Activity. Molecules 2015, 20, 21732-21749, doi:10.3390/molecules201219800.
- Ebadi, P.; Fazeli, M. Anti-photoaging potential of propolis extract in UVB-irradiated human dermal fibroblasts through increasing the expression of FOXO3A and NGF genes. Biomed Pharmacother 2017, 95, 47-54, doi:10.1016/j.biopha.2017.08.019.
- Ali, A.M.; Kunugi, H. Royal jelly as an intelligent anti-aging—a focus on cognitive aging and Alzheimer's disease: a review. Antioxidants 2020, 9, doi:10.3390/antiox9100937.
- Arranz, A.M.; De Strooper, B. The role of astroglia in Alzheimer's disease: pathophysiology and clinical implications. The Lancet Neurology 2019, https://doi.org/10.1016/S1474-4422(18)30490-3, doi:https://doi.org/10.1016/S1474-4422(18)30490-3.
- Rempe, R.G.; Hartz, A.M.S.; Bauer, B. Matrix metalloproteinases in the brain and blood-brain barrier: Versatile breakers and makers. J Cereb Blood Flow Metab 2016, 36, 1481-1507, doi:10.1177/0271678x16655551.
- Mori, A.; Hatano, T.; Inoshita, T.; Shiba-Fukushima, K.; Koinuma, T.; Meng, H.; Kubo, S.I.; Spratt, S.; Cui, C.; Yamashita, C., et al. Parkinson's disease-associated iPLA2-VIA/PLA2G6 regulates neuronal functions and alpha-synuclein stability through membrane remodeling. Proc Natl Acad Sci U S A 2019, 116, 20689-20699, doi:10.1073/pnas.1902958116.
- D'Orsi, B.; Mateyka, J.; Prehn, J.H.M. Control of mitochondrial physiology and cell death by the Bcl-2 family proteins Bax and Bok. Neurochem Int 2017, 109, 162-170, doi:10.1016/j.neuint.2017.03.010.
- Henderson, L.E.; Abdelmegeed, M.A.; Yoo, S.H.; Rhee, S.G.; Zhu, X.; Smith, M.A.; Nguyen, R.Q.; Perry, G.; Song, B.J. Enhanced Phosphorylation of Bax and Its Translocation into Mitochondria in the Brains of Individuals Affiliated with Alzheimer's Disease. Open Neurol J 2017, 11, 48-58, doi:10.2174/1874205x01711010048.
- Jang, S.E.; Lim, S.M.; Jeong, J.J.; Jang, H.M.; Lee, H.J.; Han, M.J.; Kim, D.H. Gastrointestinal inflammation by gut microbiota disturbance induces memory impairment in mice. Mucosal Immunol 2018, 11, 369-379, doi:10.1038/mi.2017.49.
- Gubiani, D.; Fabbretti, E.; Cestnik, B.; Lavrač, N.; Urbančič, T. Outlier based literature exploration for cross-domain linking of Alzheimer’s disease and gut microbiota. Expert Syst Appl 2017, 85, 386-396, doi:https://doi.org/10.1016/j.eswa.2017.05.026.
- Pretorius, L.; Kell, D.B.; Pretorius, E. Iron Dysregulation and Dormant Microbes as Causative Agents for Impaired Blood Rheology and Pathological Clotting in Alzheimer's Type Dementia. Front Neurosci 2018, 12, 851, doi:10.3389/fnins.2018.00851.
- Zhao, Y.; Jaber, V.; Lukiw, W.J. Secretory Products of the Human GI Tract Microbiome and Their Potential Impact on Alzheimer's Disease (AD): Detection of Lipopolysaccharide (LPS) in AD Hippocampus. Front Cell Infect Microbiol 2017, 7, 318, doi:10.3389/fcimb.2017.00318.
- Rooks, D.; Roubenoff, R. Development of Pharmacotherapies for the Treatment of Sarcopenia. J Frailty Aging 2019, 8, 120-130, doi:10.14283/jfa.2019.11.
- Okumura, N.; Toda, T.; Ozawa, Y.; Watanabe, K.; Ikuta, T.; Tatefuji, T.; Hashimoto, K.; Shimizu, T. Royal Jelly Delays Motor Functional Impairment During Aging in Genetically Heterogeneous Male Mice. Nutrients 2018, 10, 1191, doi:https://doi.org/10.3390/nu10091191.
- Niu, K.; Guo, H.; Guo, Y.; Ebihara, S.; Asada, M.; Ohrui, T.; Furukawa, K.; Ichinose, M.; Yanai, K.; Kudo, Y., et al. Royal jelly prevents the progression of sarcopenia in aged mice in vivo and in vitro. J Gerontol A Biol Sci Med Sci 2013, 68, 1482-1492, doi:10.1093/gerona/glt041.
- Yuce, S.; Cemal Gokce, E.; Iskdemir, A.; Koc, E.R.; Cemil, D.B.; Gokce, A.; Sargon, M.F. An experimental comparison of the effects of propolis, curcumin, and methylprednisolone on crush injuries of the sciatic nerve. Ann Plast Surg 2015, 74, 684-692, doi:10.1097/sap.0000000000000026.
- Takikawa, M.; Kumagai, A.; Hirata, H.; Soga, M.; Yamashita, Y.; Ueda, M.; Ashida, H.; Tsuda, T. 10-Hydroxy-2-decenoic acid, a unique medium-chain fatty acid, activates 5'-AMP-activated protein kinase in L6 myotubes and mice. Mol Nutr Food Res 2013, 57, 1794-1802, doi:10.1002/mnfr.201300041.
- Metwally Ibrahim, S.E.L.; Kosba, A.A. Royal jelly supplementation reduces skeletal muscle lipotoxicity and insulin resistance in aged obese rats. Pathophysiology 2018, https://doi.org/10.1016/j.pathophys.2018.05.001, doi:https://doi.org/10.1016/j.pathophys.2018.05.001.
- Ueda, M.; Hayashibara, K.; Ashida, H. Propolis extract promotes translocation of glucose transporter 4 and glucose uptake through both PI3K- and AMPK-dependent pathways in skeletal muscle. Biofactors 2013, 39, 457-466, doi:10.1002/biof.1085.
- Washio, K.; Kobayashi, M.; Saito, N.; Amagasa, M.; Kitamura, H. Propolis Ethanol Extract Stimulates Cytokine and Chemokine Production through NF-kappaB Activation in C2C12 Myoblasts. Evid Based Complement Alternat Med 2015, 2015, 349751, doi:10.1155/2015/349751.
- Ketkar, S.; Rathore, A.; Kandhare, A.; Lohidasan, S.; Bodhankar, S.; Paradkar, A.; Mahadik, K. Alleviating exercise-induced muscular stress using neat and processed bee pollen: oxidative markers, mitochondrial enzymes, and myostatin expression in rats. Integr Med Res 2015, 4, 147-160, doi:10.1016/j.imr.2015.02.003.
- Ozyurt, B.; Iraz, M.; Koca, K.; Ozyurt, H.; Sahin, S. Protective effects of caffeic acid phenethyl ester on skeletal muscle ischemia-reperfusion injury in rats. Mol Cell Biochem 2006, 292, 197-203, doi:10.1007/s11010-006-9232-5.
- Ozyurt, H.; Ozyurt, B.; Koca, K.; Ozgocmen, S. Caffeic acid phenethyl ester (CAPE) protects rat skeletal muscle against ischemia-reperfusion-induced oxidative stress. Vascul Pharmacol 2007, 47, 108-112, doi:10.1016/j.vph.2007.04.008.
- Shen, Y.C.; Yen, J.C.; Liou, K.T. Ameliorative effects of caffeic acid phenethyl ester on an eccentric exercise-induced skeletal muscle injury by down-regulating NF-kappab mediated inflammation. Pharmacology 2013, 91, 219-228, doi:10.1159/000348412.
- Zamani, Z.; Reisi, P.; Alaei, H.; Asghar Pilehvarian, A. Effect of Royal Jelly on spatial learning and memory in rat model of streptozotocin-induced sporadic Alzheimer's disease. Adv Biomed Res 2012, 1, 1-10, doi:10.4103/2277-9175.98150.
- Sharman, M.J.; Verdile, G.; Kirubakaran, S.; Münch, G. Inflammation in Alzheimer's Disease, and Prevention with Antioxidants and Phenolic Compounds – What Are the Most Promising Candidates? In Neurodegeneration and Alzheimer's Disease, 2019; 10.1002/9781119356752.ch8pp. 233-266.
- Ali, A.M.; Ahmed, A.H.; Smail, L. Psychological Climacteric Symptoms and Attitudes toward Menopause among Emirati Women. Int. J. Environ. Res. Public Health 2020, 17, 5028;, doi:10.3390/ijerph17145028
- Mofid, B.; Rezaeizadeh, H.; Termos, A.; Rakhsha, A.; Mafi, A.R.; Taheripanah, T.; Ardakani, M.M.; Taghavi, S.M.E.; Moravveji, S.A.; Kashi, A.S.Y. Effect of Processed Honey and Royal Jelly on Cancer-Related Fatigue: A Double-Blind Randomized Clinical Trial. Electronic physician 2016, 8, 2475-2482, doi:10.19082/2475.
- Nidadavolu, L.; Walston, J. Underlying Vulnerabilities to the Cytokine Storm and Adverse COVID-19 Outcomes in the Aging Immune System. J Gerontol A Biol Sci Med Sci 2020, 10.1093/gerona/glaa209, doi:10.1093/gerona/glaa209.
- Kwon, T.D.; Lee, M.W.; Kim, K.H. The effect of exercise training and water extract from propolis intake on the antioxidant enzymes activity of skeletal muscle and liver in rat. J Exerc Nutrition Biochem 2014, 18, 9-17, doi:10.5717/jenb.2014.18.1.9.
- Hatchett, A.; Berry, C.; Oliva, C.; Wiley, D.; St Hilaire, J.; LaRochelle, A. A Comparison between Chocolate Milk and a Raw Milk Honey Solution's Influence on Delayed Onset of Muscle Soreness. Sports (Basel) 2016, 4, doi:10.3390/sports4010018.
- Ali, A.M.; Kunugi, H. Bee honey protects astrocytes against oxidative stress: A preliminary in vitro investigation. Neuropsychopharmacol Rep 2019, 39, 312–314, doi:https ://doi.org/10.1002/npr2.12079.