| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ali Mujtaba Shah | + 1641 word(s) | 1641 | 2020-09-23 03:16:18 | | | |
| 2 | Bruce Ren | Meta information modification | 1641 | 2020-09-28 10:45:07 | | |
Video Upload Options
Hydrogen sulfide is a Janus-faced molecule with many beneficial and toxic effects on the animal health. In ruminants, rumen fermentation plays a vital role in the digestion and absorption of nutrients. During rumen fermentation, the production of hydrogen sulfide can occur, and it can be rapidly absorbed into the body of the animals through the intestinal wall. If the production of hydrogen sulfide concentration is higher in the rumen, it can cause a toxic effect on ruminants known as poliomyelitis. The production of hydrogen sulfide depends on the population of sulfate-reducing bacteria in the rumen. In rodents, H2S maintains the normal physiology of the gastrointestinal tract and also improves the healing of the chronic gastric ulcer. In the gut, H2S regulates physiological functions such as inflammation, ischemia–reperfusion injury and motility. In this review article, we summarize the toxicity occurrence in the body of animals due to high levels of hydrogen sulfide production and also recent progress in the studies of physiological function of H2S in the gut, with a special emphasis on bacteria-derived H2S is discussed in this review.
1. Introduction
The crucial life-supporting role of hydrogen sulfide (H2S) has developed from microorganisms to plants, invertebrate, vertebrate, and, lastly, to mammals [1]. H2S has only been known as a poisonous gas and ecological hazard. Earlier studies have shown that in animals, H2S is the most poisonous gaseous signaling molecule, after nitric oxide and carbon monoxide [2]. Recently, H2S was recognized as the third most physiologically significant gasotransmitter and plays a crucial role in various cell signaling pathways. H2S is an endogenous signaling molecule that may establish using protein sulfhydration to control different physiological functions. Hydrogen sulfide is also a byproduct of dietary sulfate metabolism by gut bacteria [3]. An increasing number of research studies have established hydrogen sulfide gas as an important cytoprotectant and redox modulator. In addition, H2S has a pleiotropic impact on the physiology of animals [3]. Consequently, a more important function of hydrogen sulfide was recognized as protecting from oxidative stress and ischemia–reperfusion injury by various mechanisms, for example, maintaining the level of glutathione (GSH) and directly suppressing mitochondrial reactive oxygen species (ROS) [4]. H2S also inhibits cytotoxic CD8+ T cells, though this happens at what we currently consider to be a supraphysiological level of hydrogen sulfide [2]. Production of hydrogen sulfide in mammalian tissue has been known for a long time, but it was primarily ignored as metabolic waste. A significant level of hydrogen sulfide has been detected in mammalian tissue from human, cow and rat in the range of 50–160 um [1]. Emerging studies have shown that H2S can serve as a potential barrier against antibiotics and oxidative stress [3][5]. In addition, hydrogen sulfide has been found to regulate several antioxidant enzymes such as ascorbate peroxidase (APX), catalase (CAT), super oxidase dismutase (SOD) and glutathione reductase (GR) [6]. H2S also plays a vital role as an intracellular gaseous transmitter in various physiological and pathologic mechanisms in rodents [7]. In addition, H2S plays a crucial role in the physiological function and maintenance of the gastrointestinal tract. In the rat colon, it regulates the secretion of calcium ion through activating the Ca2+ and ATP sensitive K+ [8]. In addition, rats treated with H2S improved the healing of a chronic gastric ulcer [9][10].
In any case, the natural and physiological significance of this chemical in endogenous hydrogen sulfide digestion has not been completely determined. Hydrogen sulfide is oxidized successively in the mitochondrion to thiosulfate and afterwards to sulfite, with the end result under physiological conditions being sulfate [11].
Hydrogen sulfide and its different ionic forms are highly toxic forms of sulfur. In the rumen, microbes produce sulfide—and if the diet contains more sulfate—the microbes produce a greater quantity of sulfide. In the rumen, dietary sulfur can be compressed into hydrogen sulfide. Hydrogen sulfide is a type of gas and its accumulation in ruminants may create some kind of toxic effects on the animals. It may cause some disorders, such as poliomyelitis [12]. In ruminants, poliomyelitis causes neuropathological conditions, leading to neural metabolic disorders, as, change in the level of thiamine, water deprivation, sodium ion toxicosis, higher intake of the sulfur and lead poisoning [13]. Hence it is important to regulate the production of hydrogen sulfide in the rumen.
2. Hydrogen Sulfide Toxicity in Rumen
Hydrogen sulfide’s effects on respiration are similar to that of hydrogen cyanide [14]. Hydrogen sulfide and its ionic particles are extremely lethal and have an strong effect on animals. Rumen fermentation produces hydrogen sulfide, which is quickly absorbed through the intestinal wall and making animal sensitive to the toxin hydrogen sulfide. The production of hydrogen sulfide depends on the sulfur concentration in the diet [15]. If the sulfur is provided to animals in the form of sulfate—or the concentration of sulfur in the diet is greater—the microorganisms in rumen use the sulfate to synthesize sulfides, which increases hydrogen sulfide levels in the rumen of the animals. This extreme absorption and production of sulfide in rumen causes a toxicological effect on the health of the ruminants [16]. Abe et al. (1996) [17] reported that hydrogen sulfide may act in the brain as a neuromodulator. Gould et al. (1997) [13] also stated that usually, when the concentration of hydrogen sulfide exceeded 2000 mg/L it may cause the poliomyelitis, and death may occur if animals are not cured timely. Many studies also reported that greater production of hydrogen sulfide levels could cause sulfur-induced poliomyelitis (S-PEM) in animals. Lewis et al. (1954) [18] reported that hydrogen sulfide concentration below 471.2 mg/L in rumen did not cause toxic and side effects on the health of animals.
3. Hydrogen Sulfide Production Pathway in Rumen
3.1. Hydrogen Sulfide Production in Rumen
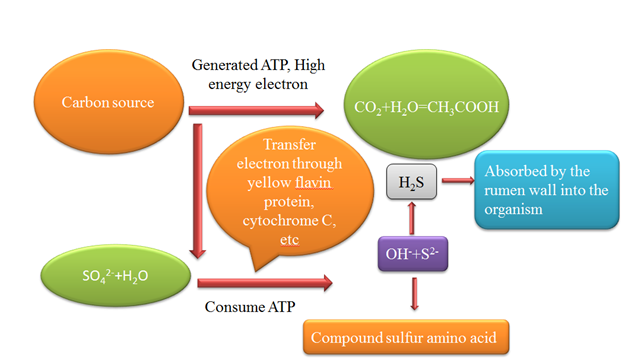
The production of H2S in the rumen is depending on the availability of sulfate reduction by ruminal sulfate-reducing bacteria. In the rumen, so many bacteria, fungi and protozoa are present, some bacteria are sulfate-reducing bacteria (SRB) and these bacteria are anaerobic. These bacteria can reduce sulfate into hydrogen sulfide in the rumen of the animals [19], and the reaction is demonstrated in Figure 1. Lewis et al. (1954) [18] verified that in the rumen of sheep, sulfate could be reduced into hydrogen sulfide. It was also confirmed in the in vitro trial that substance-reducing particles in fermentation broth could be reduced by the use of numerous sources, for example, ethanol, glucose, malic acid, citric acid and lactic acid. Though in 1960, the first strain of SRB was isolated, research on sulfate reduction in the rumen was conducted during the 1953. Desulfovibrio is SRB major strain that was found in the rumen by Howard et al. [20]. Cummings et al. (1995) [21] conducted research on steers fed high sulfate diet and concluded that there was no upsurge of SRB population, but the capability to decrease sulfate in the rumen was boosted. The source of sulfur (S) performs a vital role in the use of S and the concentration of H2S in the rumen of ruminants—especially in an increased dietary S in total mixed ration. Availability of S for ruminal reduction is more useful than total S in the diet since the differences in ruminal H2S concentration is successfully evaluated in ruminal protein sulfur and ruminal S availability (RAS) intakes instead of total intake of S [22]. Following an in vitro technique, the individual ingredients can be used to predict the coefficient of RAS. Meanwhile, the intake form can be controlled by the concentration of H2S in the ruminants. The organic source of S is methionine and can be absorbed easily as amino acids. This makes it unavailable for the reduction in the rumen to sulfide and the reverse is true; thus, the inorganic source of S is vulnerable to be metabolized in the rumen to form other dangerous compounds, including H2S. Therefore, the concept of RAS is vital to predict H2S concentration in the rumen instead of focusing the available S in the diet [23].
Figure 1. Sulfur-reducing bacteria use the sulfate to produce hydrogen sulfide.
3.2. Sulfide-Reducing Bacteria and Methanogens Relationship in the Rumen
There is an interactive and competitive relationship among methanogens and SRB. SRB and methanogens compete with each other for the requirement of the hydrogen for metabolism through anaerobic reactors [24]. Correspondingly, in the rumen, as SRB reduce sulfate to sulfide and methanogens reduces CO2 to produce CH4. SRB can also competitively attach to hydrogen ions. In the rumen, SRB’s compatibility with methanogens depends mainly on the sulfate levels [25][26]. If sulfate levels exceed a certain concentration in the rumen, the production of methane can be decreased. Nevertheless, when the concentration of sulfate becomes very low, a mutually beneficial and symbiotic association develops through hydrogen generation among methanogens and SRB, signifying ‘interspecific transfer of hydrogen. Reducing the level of sulfates in the rumen by SRB can make SRB a source of hydrogen sink [27]. Taking into account the two considerations of thermodynamics and matrix affinity, SRB has an advantage in competing with methanogens for hydrogen [28]. However, few studies have shown that methanogens cannot contest with SRB for hydrogen, though SRB and methanogens coexist [29][30]. The absolute benefit of SRB in the use of hydrogen is similarly verified by the kinetic parameters of the potential difference. The hydrogen ion is more inclined towards SRB, since the energy provided by the sulfates is greater. SRB has a low value of usable critical level for hydrogen relative to methanogens. Hence, the lower level can be used by SRB as opposed to that of methanogens. Furthermore, the temperature is also a significant aspect of the rivalry among methanogens and SRB for hydrogen. SRB dominates under moderate temperature circumstances (37 °C); however, methanogens have significant advantages in high-temperature settings (55 °C) [31]. Erin et al. (2011) reported that the level of H2S in rumen of same diet cattle were change due to the change in the SRB population in the rumen of each cattle[32].
References
- Wang, R. Physiological implications of hydrogen sulfide: A whiff exploration that blossomed. Physiol. Rev. 2012, 92, 791–896.
- Miller, T.W.; Wang, E.A.; Gould, S.; Stein, E.V.; Kaur, S.; Lim, L.; Amarnath, S.; Fowler, D.H.; Roberts, D.D. Hydrogen sulfide is an endogenous potentiator of T cell activation. J. Biol. Chem. 2012, 287, 4211–4221.
- Pal, V.K.; Bandyopadhyay, P.; Singh, A. Hydrogen sulfide in physiology and pathogenesis of bacteria and viruses. IUBMB life 2018, 70, 393–410.
- Kimura, Y.; Dargusch, R.; Schubert, D.; Kimura, H. Hydrogen sulfide protects HT22 neuronal cells from oxidative stress. Antioxid. Redox Signal. 2006, 8, 661–670.
- Shatalin, K.; Shatalina, E.; Mironov, A.; Nudler, E. H2S: A universal defense against antibiotics in bacteria. Science 2011, 334, 986–990.
- Aroca, A.; Gotor, C.; Bassham, D.C.; Romero, L.C. Hydrogen sulfNide: From a toxic molecule to a key molecule of cell life. Antioxidants 2020, 9, 621.
- Wallace, J.L.; Ferraz, J.G.; Muscara, M.N. Hydrogen sulfide: An endogenous mediator of resolution of inflammation and injury. Antioxid. Redox Signal. 2012, 17, 58–67.
- Pouokam, E.; Steidle, J.; Diener, M. Regulation of colonic ion transport by gasotransmitters. Biol. Pharm. Bull. 2011, 34, 789–793.
- Wallace, J.L.; Dicay, M.; McKnight, W.; Martin, G.R. Hydrogen sulfide enhances ulcer healing in rats. FASEB J. 2007, 21, 4070–4076.
- Magierowski, M.; Magierowska, K.; Hubalewska-Mazgaj, M.; Sliwowski, Z.; Pajdo, R.; Ginter, G.; Kwiecien, S.; Brzozowski, T. Exogenous and endogenous hydrogen sulfide protects gastric mucosa against the formation and time-dependent development of ischemia/reperfusion-induced acute lesions progressing into deeper ulcerations. Molecules 2017, 22, 295.
- Wallace, J.L.; Wang, R. Hydrogen sulfide-based therapeutics: Exploiting a unique but ubiquitous gasotransmitter. Nat. Rev. Drug Discov. 2015, 14, 329–345.
- Binversie, E.Y.; Ruiz-Moreno, M.; Carpenter, A.; Heins, B.; Crawford, G.; DiCostanzo, A.; Stern, M. Effects of dietary roughage and sulfur in diets containing corn dried distillers grains with solubles on hydrogen sulfide production and fermentation by rumen microbes in vitro. J. Anim. Sci. 2016, 94, 3883–3893.
- Gould, D.H.; Cummings, B.A.; Hamar, D.W. In vivo indicators of pathologic ruminal sulfide production in steers with diet-induced polioencephalomalacia. J. Vet. Diag. Investig. 1997, 9, 72–76.
- Beauchamp, R.; Bus, J.S.; Popp, J.A.; Boreiko, C.J.; Andjelkovich, D.A.; Leber, P. A critical review of the literature on hydrogen sulfide toxicity. CRC Crit. Rev. Toxicol. 1984, 13, 25–97.
- Drewnoski, M.; Richter, E.; Hansen, S. Dietary sulfur concentration affects rumen hydrogen sulfide concentrations in feedlot steers during transition and finishing. J. Anim. Sci. 2012, 90, 4478–4486.
- Gould, D.H. Polioencephalomalacia. J. Anim. Sci. 1998, 76, 309–314.
- Abe, K.; Kimura, H. The possible role of hydrogen sulfide as an endogenous neuromodulator. J. Neurosci. 1996, 16, 1066–1071.
- Lewis, D. The reduction of sulphate in the rumen of the sheep. Biochem. J. 1954, 56, 391.
- Huisingh, J.; McNeill, J.; Matrone, G. Sulfate reduction by a Desulfovibrio species isolated from sheep rumen. Appl. Environ. Microbiol. 1974, 28, 489–497.
- Howard, B.; Hungate, R.E. Desulfovibrio of the sheep rumen. Appl. Environ. Microbiol. 1976, 32, 598–602.
- Cummings, B.; Gould, D.; Caldwell, D.; Hamar, D. Ruminal microbial alterations associated with sulfide generation in steers with dietary sulfate-induced polioencephalomalacia. Am. J. Vet. Res. 1995, 56, 1390–1395.
- Fron, M.J.; Boling, J.A.; Bush, L.P.; Dawson, K.A. Sulfur and nitrogen metabolism in the bovine fed different forms of supplemental sulfur. J. Anim. Sci. 1990, 68, 543–552.
- Sarturi, J.; Erickson, G.; Klopfenstein, T.; Rolfe, K.; Buckner, C.; Luebbe, M. Impact of source of sulfur on ruminal hydrogen sulfide and logic for the ruminal available sulfur for reduction concept. J. Anim. Sci. 2013, 91, 3352–3359.
- Nava, G.M.; Carbonero, F.; Croix, J.A.; Greenberg, E.; Gaskins, H.R. Abundance and diversity of mucosa-associated hydrogenotrophic microbes in the healthy human colon. ISME J. 2012, 6, 57–70.
- Jiang, G.; Gutierrez, O.; Sharma, K.R.; Yuan, Z. Effects of nitrite concentration and exposure time on sulfide and methane production in sewer systems. Water Res. 2010, 44, 4241–4251.
- Singh, S.B.; Lin, H.C. Hydrogen sulfide in physiology and diseases of the digestive tract. Microorganisms 2015, 3, 866–889.
- Bryant, M.P.; Campbell, L.L.; Reddy, C.A.; Crabill, M.R. Growth of Desulfovibrio in lactate or ethanol media low in sulfate in association with H2-utilizing methanogenic bacteria. Appl. Environ. Microbiol. 1977, 33, 1162–1169.
- Zinder, S.H. Physiological ecology of methanogens. In Methanogenesis; Springer: Berlin/Heidelberg, Germany, 1993; pp 128–206.
- Stefanie, J.O.E.; Visser, A.; Pol, L.W.H.; Stams, A.J. Sulfate reduction in methanogenic bioreactors. FEMS Microbiol. Rev. 1994, 15, 119–136.
- Colleran, E.; Finnegan, S.; Lens, P. Anaerobic treatment of sulphate-containing waste streams. Antonie van Leeuwenhoek 1995, 67, 29–46.
- Colleran, S.; Pender, S. Mesophilic and thermophilic anaerobic digestion of sulphate-containing wastewaters. Water Sci. Technol. 2002, 45, 231–235.
- Richter, E.L. The Effect of Dietary Sulfur on Performance, Mineral Status, Rumen Hydrogen Sulfide, and Rumen Microbial Populations in Yearling Beef Steers. Master’s Thesis, Iowa State University, Ames, IA, USA, 2011.