| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mariusz Gusiatin | + 956 word(s) | 956 | 2020-09-02 08:33:55 | | | |
| 2 | Nicole Yin | -3 word(s) | 953 | 2020-09-07 03:54:51 | | | | |
| 3 | Nicole Yin | Meta information modification | 953 | 2020-09-08 07:46:09 | | | | |
| 4 | Nicole Yin | Meta information modification | 953 | 2020-09-08 07:46:33 | | |
Video Upload Options
Soil contamination with heavy metals and organic pollutants poses an environmental problem due to their persistence, bioaccumulation and toxicity. Soil washing is one of the most common remediation technologies for permanent removal of undesirable pollutants. The effectiveness of soil washing is affected by many factors such as soil geochemistry, pollutant characteristics, type and chemistry of washing agents, and processing conditions. A variety of washing agents have been tested in soil washing with their different effect on pollutant removal and on the overall quality of treated soil. Development of soil washing is based on searching for an inexpensive, readily-available and environmentally-friendly agents to remediate polluted soils. Despite the unquestionable effectiveness of soil washing, spent washing solutions are generated that need further treatment. This entry presents the principles and application of soil washing technology, as well as the examples of washing agents and methods for liquid waste treatment.
Soil washing is an ex-situ technology, including physical and/or chemical processes that aim to effectively remove pollutants from soil.
1. Introduction
Soil washing can be conducted in one of three variants: physical separation, chemical extraction, or physical separation followed by chemical extraction[1]. Physical separation aims to separate the most contaminated particles from bulk soil and to reduce the volume of polluted soil to be treated. At this stage, soil washing with water allows the coarse-grained particles to separate from the polluted fine particles. Chemical extraction is based on pollutant solubilization with a washing solution (water with chemical agent) and their transfer from soil particles into the washing solution[2].
Generally, soil washing based on physical separation and chemical extraction includes six steps[3][4][5]:
- Pretreatment (removal of oversized materials from soil),
- Separation between coarse- and fine-grained soil particles in a scrubbing unit (separation of the coarse-grained soils is commonly done by using mechanical screening such as trommels, while the fine-grained soils are sorted using hydrocyclones or other methods),
- Coarse-grained treatment (usually coarse-grained soil particles >0.05 mm are likely unpolluted or minimally polluted; thus, they can be treated using surface attrition or washing with water),
- Fine-grained treatment (because fine soil particles <0.05 mm are highly contaminated, they should be treated with a suitable washing solution containing water and chemicals using sonication or mechanical agitation),
- Spent washing solution (SWS) treatment (from washing coarse and fine soil particles; necessary to reuse washing solutions or to dispose them in sewers),
- Residual management (residual materials, i.e., treated soil, and the sludge of dispersed fine particles output during soil washing; if residuals are still considered polluted, they may require further treatment before disposal).
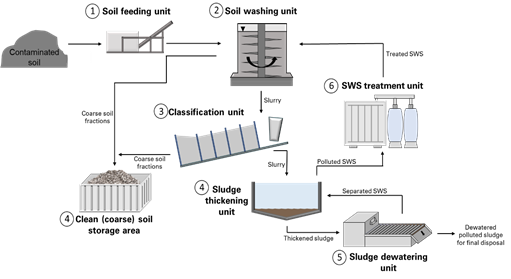
Overall, there are many different soil washing systems that are developed, and the systems can vary from site to site due to location-specific constraints for soils or pollutants. In Figure 1, an example of DEKONTA’s soil washing system is presented[6].
Figure 1. Soil washing system (adapted from www.dekonta.cz[6]). The numbers (1–7) indicate the steps of soil washing.
2. Washing agents
- Conventional: mineral acids (e.g., HCl, H2SO4, HNO3), low-molecular-weight organic acids (e.g., oxalic and citric acids), synthetic organic chelating agents (e.g., ethylenediaminetetraacetic acid (EDTA), diethylenetriaminepentaacetic acid (DTPA), and ethylenediaminedisuccinic acid (EDDS)).
- New-generation (also called as "green" washing agents): biosurfactants, dissolved organic matter (DOM), soluble humic-like substances (SHLS), soluble humic substances (SHS). The DOM, SHLS and SHS can be derived from geochemical resources, processed or unprocessed wastes.
3. Application
Soil washing is a well-established technology of soil remediation in many European countries (the Netherlands, Germany, Belgium, Switzerland), as well as in North America and Japan[2]. It can be used as stand-alone technology or in combination with other remediation systems for “hot spots” treatment, where the concentrations of pollutants are particularly high. According to Dermont et al.[1], among 37 field soil washings performed in 1989–2007, 43% were based on physical separation, 8% were based on chemical extraction, and 47% were based on physical separation and chemical extraction. Full-scale soil washing plants exist as centralized permanent treatment sites, where polluted soil is brought to the plant, or as mobile/transportable units, where polluted soil is treated on the site.
In the Netherlands, there are five permanent soil washing sites (BMR Schiedam, SBD Amersfoort, BVNN BKD Groningen, GBD Alkmaar, and BVNN BKD Heerenveen) where asbestos-contaminated soil, degraded sediments, sand from sewers, contaminated roof gravel, contaminated granulate, and stony materials are processed[7]. Soil washing is used at many large-scale cleanup sites in the USA, including Miami Harbor, Fox River (Wisconsin), and the King of Prussia Superfund Site[8]. Mobile soil washing systems are modular construction units that can be deployed at different sites. The USA and some European countries (e.g., Sweden) developed mobile soil washing systems in the 1980s and employed the devices in field sites to clean up contaminated soils[9][10]. A pilot-scale mobile soil washing system was proposed by Ko et al.[11] to treat soil polluted with As, Zn, and Ni collected from an iron mining area in Korea. The system consisted of a soil washing scrubber (drum type equipped with an inner screw blade to mix soil with washing solution), a vibrating screen, a screw feeder, a high-pressure air supply (to provide mechanical turbulence between soil and washing solution), and a ceramic filter. The mass of soil treated with individual washing solutions (HCl, H2SO4, or H3PO4) in this mobile system was 40 kg[11]. Many environmental companies offer soil washing systems. Global leaders in the field of large-scale washing of soils are Boskalis Environmental[12] and ART Engineering LLC[13].
4. Methods for treatment of spent washing solutions
- Adsorption by using various adsorbents.
- Precipitation by adjusting the pH in spent washing solution with chemicals.
- Advanced Oxidation Processes by using ozone, H2O2, sonification, or ultraviolet (UV), alone or in combination.
- Membrane Technologies by using ultrafiltration, nanofiltration, or reserve osmosis.
References
- Dermont, G.; Bergeron, M.; Mercier, G.; Richer-Laflèche, M. Soil washing for metal removal: A review of physical/chemical technologies and field applications. J. Hazard. Mater. 2008, 152, 1–31.
- Pearl, M. Understanding Soil Washing. CL AIRE Tech. Bull. 2007, 13, 1–4.
- Hubler, J.; Metz, K. Soil Washing. Available online: https://www.geoengineer.org/education/web-class-projects/cee-549-geoenvironmental-engineering-winter-2013/assignments/soil-washing (accessed on 29 May 2020).
- Anderson, W.C. Soil Washing/Soil Flushing; American Academy of Environmental Engineers: Annapolis, MD, USA, 1993.
- Griffiths, R.A. Soil-washing technology and practice. J. Hazar. Mater. 1995, 40, 175–189.
- Dekonta Transportable Soil Washing Plant. Available online: https://www.dekonta.cz/en/about-us/download.html (accessed on 29 May 2020).
- Boskalis Environmental Enhanced Soil Washing. Available online: https://environmental.boskalis.com/activities/enhanced-soil-washing.html (accessed on 29 May 2020).
- Water Front Toronto Pilot Soil Recycling Facility Fact Sheet. Available online: https://www.waterfrontoronto.ca/nbe/wcm/connect/waterfront/40e110c0-8580-485d-a555-88b10e0c79d0/soil_recycling_pilot_facility_fact_sheet___april_2012___clean_1.pdf?MOD=AJPERES&CACHEID=40e110c0-8580-485d-a555-88b10e0c79d0 (accessed on 29 May 2020).
- Liu, L.; Li, W.; Song, W.; Guo, M. Remediation techniques for heavy metal-contaminated soils: Principles and applicability. Sci. Total Environ. 2018, 633, 206–219.
- FRTR Remediation Technologies Screening Matrix and Reference Guide, Version 4.0. Available online: https://frtr.gov/matrix2/top_page.html (accessed on 29 May 2020).
- Ko, I.; Chang, Y.-Y.; Lee, C.-H.; Kim, K.-W. Assessment of pilot-scale acid washing of soil contaminated with As, Zn and Ni using the BCR three-step sequential extraction. J. Hazard. Mater. 2005, 127, 1–13.
- Boskalis Environmental. Available online: https://environmental.boskalis.com/ (accessed on 29 May 2020).
- ART Engineering LLC. Available online: https://www.art-engineering.com/index.html (accessed on 29 May 2020).