| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kamila Białkowska | + 1880 word(s) | 1880 | 2020-08-28 10:28:07 | | | |
| 2 | Vicky Zhou | -76 word(s) | 1804 | 2020-09-01 11:02:06 | | | | |
| 3 | Conner Chen | Meta information modification | 1804 | 2020-09-02 10:40:49 | | | | |
| 4 | Vicky Zhou | -21 word(s) | 1783 | 2020-10-27 09:53:59 | | |
Video Upload Options
Cell cultures are very important for testing materials and drugs, and in the examination of cell biology and special cell mechanisms. The most popular models of cell culture are two-dimensional (2D) as monolayers, but this does not mimic the natural cell environment. Cells are mostly deprived of cell–cell and cell–extracellular matrix interactions. A much better in vitro model is three-dimensional (3D) culture. Because many cell lines have the ability to self-assemble, one 3D culturing method is to produce spheroids. There are several systems for culturing cells in spheroids, e.g., hanging drop, scaffolds and hydrogels, and these cultures have their applications in drug and nanoparticles testing, and disease modeling.
1. Introduction
Cell cultures are very important for testing materials and drugs, and in the examination of cell biology and special cell mechanisms. The most popular models of cell culture are two-dimensional (2D) as monolayers, but this does not mimic the natural cell environment. Cells are mostly deprived of cell–cell and cell–extracellular matrix interactions. A much better in vitro model is three-dimensional (3D) culture. Because many cell lines have the ability to self-assemble, one 3D culturing method is to produce spheroids. There are several systems for culturing cells in spheroids, e.g., hanging drop, scaffolds and hydrogels, and these cultures have their applications in drug and nanoparticles testing, and disease modeling.
Cells have been cultured since the 1940s [1], and are generally in use to examine cell biology and molecular mechanisms [2]. Cells are taken directly from a tissue and, after suitable preparation, transferred into an artificial environment or they are obtained from a cell line already adopted by others. Cells grow in a medium containing the required nutrients, growth factors, and hormones, in an incubator. Cultures are kept in special dishes placed in strictly controlled temperature conditions, normally a 37°C [3]. Cells are attached to a flat surface as a substrate, glass or plastic, mainly in two dimensions, as monolayers. This method of cell culturing is most popular because it is simple and convenient; it has been an invaluable method providing important knowledge as models of variety diseases [4][5]. However, forcing cells to grow on flat surfaces can change their metabolism and functioning [4].
In 2D cell cultures, the cell–cell and cell–extracellular matrix interactions are reduced, and the level of cellular responsiveness is limited [2][6]. Moreover, cell culture environment can have an effect on the phenotype of cells and hence affect the cellular response to added substances, e.g., drugs [1]. All cells in the body live in 3D environment, which is crucial for their metabolism and growth. The phenotype and functions of each cell are highly dependent on elaborated interactions with neighboring cells, the extracellular matrix (ECM) and proteins [6]. Those cell–cell and cell–ECM interactions differ from 2D to 3D cultures and also between cell layers in spheroids structures, and this can affect cytotoxicity results [7]. For these reasons, testing the toxicity of materials and substances on 2D cell cultures is not exactly predictive of that which might be expected in the body [6][8]. 3D cell cultures more precisely mimic the natural cell microenvironment. The morphology and physiology of cells in 3D cultures are different from cells in 2D cultures, showing responses that correspond in some ways more like in vivo behavior [8]. In 2D models, molecules can be secreted into the culture medium, and, therefore, changing the medium will remove these substances and might disturb some analysis. For example, in 2D models of Alzheimer disease, removing the medium will mean that secreted amyloid beta (Aß) is discarded and, therefore, change the analysis of Aß aggregation. 3D cell cultures can limit the diffusion of Aß into the culture medium [5].
2. Applications of 3D Cell Cultures
2.1. Drug Testing and Nanoparticles Examination
2.1.1. Drug Testing
A generation of new cancer drugs is based on three approaches, referred to as: (i) high throughput drug screening (HTS), (ii) expansion of analogs of existing drugs, and (iii) rational drug design. These involve assays based on measuring cell viability, proliferation, and clonogenicity in an in vitro environment [9]. Cell cultures help assess drug safety and indicate their possible mechanism of action. Test substances are added to the culture medium and their activity investigated [10]. Presently, 2D cell cultures remain very useful in drug investigation. However, as already mentioned, 2D models do not mimic well the physiological environment of living cells [11]. For instance, cells of the colon cancer cell line, HCT-116 wt, cultured as spheroids were more resistant to some of the tested drugs compared with them cultured as a monolayer [9][12].
Cells grown in 3D cultures can be maintained longer than as 2D monolayers. The 3D aggregates can be kept for 4 weeks, whereas cells in 2D cultures last approximately 1 week before reaching confluence. For this reason, 3D cell cultures make a better model for studying long-term effects of drugs. Tumor cells in a monolayer proliferate faster than in 3D aggregates and are more sensitive to agents used during chemotherapy or radiation therapy [3].
2.1.2. Nanoparticle Examination
Testing of nanomaterials and their effects on cells is important because of possible therapeutic application. Nanomaterials are considered as safe gene carriers in gene therapy. Gene therapy is a potential method for fighting diseases, such as cancer, when traditional treatment is poor [13].
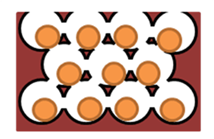
Because of the lack of the nanoparticle (NP) transport through cell layers in 2D cell cultures, 3D cell culture offers a better model. Techniques used for testing nanoparticle toxicity are the same as the methods for drug examination, although the toxic mechanism can be different [6]. Lee et al. (2009) have introduced 3D spheroid-culture-based NP toxicity testing system (Figure 1) using human hepatocarcinoma (HepG2) cells, because the liver is the main organ for NP accumulation. As a substrate, they used transparent and nonadhesive polyacrylamide hydrogel to measure the toxic effects of cadmium telluride (CdTe) and gold (Au) nanoparticles. Morphology, metabolic activity, membrane activity, and mechanism of cell death were explored, comparing the results from 3D cultures with those from 2D cultures. Cell number and spheroid diameter were crucial parameters to get repeatable results. They also showed that the activity of a spheroid depends on its size. They found significant differences between the morphology of cells in 2D and 3D cell cultures after treatment with CdTeNPs, with more death in 2D than in 3D cultures. Cell toxicity assays also confirmed that the toxic effects of NPs were reduced in 3D compared to 2D cultures [6].
Figure 1. Model of hydrogel inverted colloidal crystal (ICC) scaffold [6].
2.2. Models for Neurodegenerative Diseases
The 3D cell cultures are widely used in medical studies [3], e.g., research on neurodegenerative diseases [14].
2.2.1. Alzheimer Disease
The most widespread neurodegenerative disease in the world is Alzheimer Disease (AD), for which there is no effective therapy, only some symptomatic treatment [15][16]. It is characterized by a progressive cognitive decline and involves memory deterioration. Orientation, judgments, and reasoning are also disturbed [17]. There are two characteristic features of AD, namely plaques of ß-amyloid and neurofibrillary tangles of tau protein [14][16][18]. Aß is generated from amyloid precursor protein (APP) during the process caused by two enzymes, ß-secretase and γ-secretase [15]. The hypothesis that Aß accumulation is the initial event in AD, leading to the next pathological events is called the “amyloid cascade hypothesis” [19].
Transgenic mice have now been used as models for studies on AD, but unfortunately, they do not exhibit important features occurring in humans [16]. Additional phenotypes of mice can also occur, which are not related to AD [20]. For these reasons, therapies for AD that are effective in mouse models probably do not work on humans [16][21]. In transgenic mice, there is also no amyloid cascade [19][22]. According to Choi et al. (2016) the Matrigel-based 3D cell culture system is a more appropriate model for AD testing as Aß plaques are present, and these are not present in the mouse model [16].
SH-SY5Y is a neuronal-like cell line that is artificially differentiated to neural cells. This cell line came from the bone marrow of a patient with neuroblastoma [14]. Characteristic features of the cells are activities of dopamine-ß-hydroxylase and tyrosine hydroxylase, some level of noradrenaline (NA) release, and the presence of choline acetyltransferase, acetylcholinesterase, and butyrylcholinesterase [23]. Seidel et al. (2012) used spheroids of human neuroblastoma cell line (SH-SY5Y) which overexpress EGFP-fused tau as a model to study the pathologies of tau protein in AD. They obtained 3 variants of SH-SY5Y over expressing tau (0N4R), namely wild type (WT), a variant with single point mutation P301L (which is used in common) and K280q (which is 4-fold gene mutation in the tau protein gene DK280, P301L, V337M, R406W), which was used to enhance tauopathy. Generally, differentiation of SHSY5Y cells took place by using several agents, e.g., phorbol esters and retinoic acid, growth factors (like brain derived neurotrophic factor, BDNF), nerve growth factor (NGF) or cholesterol [24]. However, differentiation agents influence cell metabolism and could probably affect the induction of tauopathy. The 3D cell cultures might help in eliminating the problem with differentiation agents [25].
2.2.2. Parkinson Disease
Parkinson disease (PD) is a neurodegenerative illness characterized by a loss of cells in the substantia nigra in the midbrain. The loss of these dopaminergic neurons is related to motor dysfunction [26][27], resting tremors, bradykinesia, postural instability and rigidity [28][29]. There is no representative in vitro model to study this neurodegenerative dysfunction. Animal models are not sufficient to predict responses occurring in humans [26]. Since there is the possibility of obtaining most major cell types from the human brain during differentiating induced-pluripotent stem cells—iPSCs [30], this research model seems to show promise as an accurate human model for PD [26].
Moreno et al. (2015) obtained human neuroepithelial cells from iPSCs and finally differentiated them as receiving dopaminergic neurons, cultured within 3D microfluidic cell culture bioreactors. After 30 days, those neurons had characteristic features of dopaminergic neurons and were active [31]. The 3D culture bioreactors were described by Trietsch et al. (2013), who proposed a platform that mimics tissue and perfusion excluding spatial separation [32]. Neighboring lanes of gels and liquids reproduced tissue heterogeneity. A single bioreactor is made from a row of cells settled in hydrogel, and one or more neighboring lanes of liquid flowing laminarly. To shape liquids flowing into the bioreactor, each pair of lanes is separated using a phaseguide. Phaseguide technology makes it possible to control the filling and emptying of a range of types of microfluidic constructions [33][34]. Cells are mixed with replacement ECM, which is subsequently distributed into a well plugged to the phaseguide delimited lane. Finally, the fresh portion of medium is added to the well, which is combined with the medium lane neighboring with cells in hydrogel [31]. Moreno et al. (2015) confirmed that using this technique allows them to obtain dopaminergic neurons and proved its usefulness in calcium imaging and immunofluorescence. Moreover, analysis of 3D images showed neurons with long neurites [31].
References
- Goodman, T.T.; Ng, C.P.; Pun, S.H. 3-D tissue culture systems for the evaluation and optimization of nanoparticle-based drug carriers. Bioconjug. Chem. 2008, 19, 1951–1959, doi:10.1021/bc800233a.
- Do Amaral, J.B.; Rezende-Teixeira, P.; Freitas, V.M.; Machado-Santelli, G.M. MCF-7 cells as a three-dimensional model for the study of human breast cancer. Tissue Eng. Part C Methods 2011, 17, 1097–1107, doi:10.1089/ten.tec.2011.0260.
- Antoni, D.; Burckel, H.; Josset, E.; Noel, G. Three-dimensional cell culture: A breakthrough in vivo. Int. J. Mol. Sci. 2015, 16, 5517–5527, doi:10.3390/ijms16035517.
- Andersen, T.; Auk-Emblem, P.; Dornish, M. 3D cell culture in alginate hydrogels. Microarrays 2015, 4, 133–161, doi:10.3390/microarrays4020133.
- Centeno, E.G.Z.; Cimarosti, H.; Bithell, A. 2D versus 3D human induced pluripotent stem cell-derived cultures for neurodegenerative disease modeling. Mol. Neurodegener. 2018, 13, 27, doi:10.1186/s13024-018-0258-4.
- Lee, J.; Lilly, G.D.; Doty, R.C.; Podsiadlo, P.; Kotov, N.A. In vitro toxicity testing of nanoparticles in 3D cell culture. Small 2009, 5, 1213–1221, doi:10.1002/smll.200801788.
- Moshksayan, K.; Kashaninejad, N.; Warkiani, M.E.; Lock, J.G.; Moghadas, H.; Firoozabadi, B.; Saidi, M.S.; Nguyen, N.T. Spheroids-on-a-chip: Recent advances and design considerations in microfluidic platforms for spheroid formation and culture. Sens. Actuators B Chem. 2018, 263, 151–176, doi:10.1016/j.snb.2018.01.223.
- Edmondson, R.; Broglie, J.J.; Adcock, A.F.; Yang, L.J. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. Assay Drug Dev. Technol. 2014, 12, 207–218, doi:10.1089/adt.2014.573.
- Karlsson, H.; Fryknas, M.; Larsson, R.; Nygren, P. Loss of cancer drug activity in colon cancer HCT-116 cells during spheroid formation in a new 3-D spheroid cell culture system. Exp. Cell Res. 2012, 318, 1577–1585, doi:10.1016/j.yexcr.2012.03.026.
- Amelian, A.; Wasilewska, K.; Megias, D.; Winnicka, K. Application of standard cell cultures and 3D in vitro tissue models as an effective tool in drug design and development. Pharmacol. Rep. 2017, 69, 861–870, doi:10.1016/j.pharep.2017.03.014.
- Souza, G.R.; Molina, J.R.; Raphael, R.M.; Ozawa, M.G.; Stark, D.J.; Levin, C.S.; Bronk, L.F.; Ananta, J.S.; Mandelin, J.; Georgescu, M.M.; et al. Three-dimensional tissue culture based on magnetic cell levitation. Nat. Nanotechnol. 2010, 5, 291–296, doi:10.1038/nnano.2010.23.
- Fang, Y.; Eglen, R.M. Three-dimensional cell cultures in drug discovery and development. SLAS Discov. 2017, 22, 456–472, doi:10.1177/1087057117696795.
- Wu, J.Y.; Huang, W.Z.; He, Z.Y. Dendrimers as Carriers for siRNA Delivery and Gene Silencing: A Review. Sci. World J. 2013, doi:10.1155/2013/630654.
- Słońska, A.; Cymerys, J. Zastosowanie trójwymiarowych hodowli komórek nerwowych w badaniach mechanizmów przebiegu chorób neurodegeneracyjnych. Postȩpy Hig. Med. Doświadczalnej 2017, 71, 510–519, doi:10.5604/01.3001.0010.3832.
- Bertram, L.; Tanzi, R.E. Thirty years of Alzheimer’s disease genetics: The implications of systematic meta-analyses. Nat. Rev. Neurosci. 2008, 9, 768–778, doi:10.1038/nrn2494.
- Choi, S.H.; Kim, Y.H.; Quinti, L.; Tanzi, R.E.; Kim, D.Y. 3D culture models of Alzheimer’s disease: A road map to a “cure-in-a-dish”. Mol. Neurodegener. 2016, 11, 75, doi:10.1186/s13024-016-0139-7.
- Tanzi, R.E.; Bertram, L. Twenty years of the Alzheimer’s disease amyloid hypothesis: A genetic perspective. Cell 2005, 120, 545–555, doi:10.1016/j.cell.2005.02.008.
- D’Avanzo, C.; Aronson, J.; Kim, Y.H.; Choi, S.H.; Tanzi, R.E.; Kim, D.Y. Alzheimer’s in 3D culture: Challenges and perspectives. Bioessays 2015, 37, 1139–1148, doi:10.1002/bies.201500063.
- Armstrong, R.A. A critical analysis of the ‘amyloid cascade hypothesis’. Folia Neuropathol. 2014, 52, 211–225, doi:10.5114/fn.2014.45562.
- Sasaguri, H.; Nilsson, P.; Hashimoto, S.; Nagata, K.; Saito, T.; De Strooper, B.; Hardy, J.; Vassar, R.; Winblad, B.; Saido, T.C. APP mouse models for Alzheimer’s disease preclinical studies. EMBO J. 2017, 36, 2473–2487, doi:10.15252/embj.201797397.
- De Strooper, B. Lessons from a failed gamma-secretase Alzheimer trial. Cell 2014, 159, 721–726, doi:10.1016/j.cell.2014.10.016.
- Mudher, A.; Lovestone, S. Alzheimer’s disease—Do tauists and baptists finally shake hands? Trends Neurosci. 2002, 25, 22–26, doi:10.1016/S0166-2236(00)02031-2.
- Xicoy, H.; Wieringa, B.; Martens, G.J.M. The SH-SY5Y cell line in Parkinson’s disease research: A systematic review. Mol. Neurodegener. 2017, 12, 10, doi:10.1186/s13024-017-0149-0.
- Agholme, L.; Lindstrom, T.; Kagedal, K.; Marcusson, J.; Hallbeck, M. An in vitro model for neuroscience: Differentiation of SH-SY5Y cells into cells with morphological and biochemical characteristics of mature neurons. J. Alzheimer’s Dis. 2010, 20, 1069–1082, doi:10.3233/JAD-2010-091363.
- Seidel, D.; Krinke, D.; Jahnke, H.G.; Hirche, A.; Kloss, D.; Mack, T.G.A.; Striggow, F.; Robitzki, A. Induced tauopathy in a novel 3D-culture model mediates neurodegenerative processes: A real-time study on biochips. PLoS ONE 2012, 7, e49150, doi:10.1371/journal.pone.0049150.
- Bolognin, S.; Fossepre, M.; Qing, X.B.; Jarazo, J.; Scancar, J.; Moreno, E.L.; Nickels, S.L.; Wasner, K.; Ouzren, N.; Walter, J.; et al. 3D cultures of Parkinson’s disease-specific dopaminergic neurons for high content phenotyping and drug testing. Adv. Sci. 2018, 6, 1800927, doi:10.1002/advs.201800927.
- Mahlknecht, P.; Seppi, K.; Poewe, W. The concept of prodromal Parkinson’s disease. J. Parkinson’s Dis. 2015, 5, 681–697, doi:10.3233/JPD-150685.
- Meissner, W.G.; Frasier, M.; Gasser, T.; Goetz, C.G.; Lozano, A.; Piccini, P.; Obeso, J.A.; Rascol, O.; Schapira, A.; Voon, V.; et al. Priorities in Parkinson’s disease research. Nat. Rev. Drug Discov. 2011, 10, 377–393, doi:10.1038/nrd3430.
- Antony, P.M.A.; Diederich, N.J.; Kruger, R.; Balling, R. The hallmarks of Parkinson’s disease. FEBS J. 2013, 280, 5981–5993, doi:10.1111/febs.12335.
- Parr, C.J.C.; Yamanaka, S.; Saito, H. An update on stem cell biology and engineering for brain development. Mol. Psychiatry 2017, 22, 808–819, doi:10.1038/mp.2017.66.
- Moreno, E.L.; Hachi, S.; Hemmer, K.; Trietsch, S.J.; Baumuratov, A.S.; Hankemeier, T.; Vulto, P.; Schwamborn, J.C.; Fleming, R.M.T. Differentiation of neuroepithelial stem cells into functional dopaminergic neurons in 3D microfluidic cell culture. Lab Chip 2015, 15, 2419–2428, doi:10.1039/c5lc00180c.
- Trietsch, S.J.; Israëls, G.D.; Joore, J.; Hankemeier, T.; Vulto, P. Microfluidic titer plate for stratified 3D cell culture. Lab Chip 2013, 13, 3548–3554, doi:10.1039/c3lc50210d.
- Yildirim, E.; Trietsch, S.J.; Joore, J.; van den Berg, A.; Hankemeier, T.; Vulto, P. Phaseguides as tunable passive microvalves for liquid routing in complex microfluidic networks. Lab Chip 2014, 14, 3334–3340, doi:10.1039/c4lc00261j.
- Vulto, P.; Podszun, S.; Meyer, P.; Hermann, C.; Manz, A.; Urban, G.A. Phaseguides: A paradigm shift in microfluidic priming and emptying. Lab Chip 2011, 11, 1596–1602, doi:10.1039/c0lc00643b.