
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mirre De Bondt | + 1648 word(s) | 1648 | 2020-06-28 13:57:46 | | | |
| 2 | Catherine Yang | -2 word(s) | 1646 | 2025-09-05 05:14:22 | | | | |
| 3 | Catherine Yang | + 2 word(s) | 1648 | 2025-09-05 05:15:02 | | |
Video Upload Options
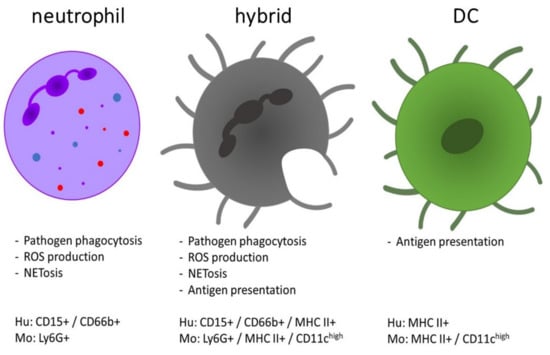
Neutrophils are the most abundant circulating and first-responding innate myeloid cells and have so far been underestimated in the context of multiple sclerosis (MS). MS is the most frequent, immune-mediated, inflammatory disease of the central nervous system. MS is treatable but not curable and its cause(s) and pathogenesis remain elusive. The involvement of neutrophils in MS pathogenesis has been suggested by the use of preclinical animal disease models, as well as on the basis of patient sample analysis. In this review, we provide an overview of the possible mechanisms and functions by which neutrophils may contribute to the development and pathology of MS. Neutrophils display a broad variety of e ector functions enabling disease pathogenesis, including (1) the release of inflammatory mediators and enzymes, such as interleukin-1 , myeloperoxidase and various proteinases, (2) destruction and phagocytosis of myelin (as debris), (3) release of neutrophil extracellular traps, (4) production of reactive oxygen species, (5) breakdown of the blood–brain barrier and (6) generation and presentation of autoantigens. An important question relates to the issue of whether neutrophils exhibit a predominantly proinflammatory function or are also implicated in the resolution of chronic inflammatory responses in MS.
1. Introduction
2. The Role of Neutrophils
References
- Opdenakker, G.; Fibbe, W.E.; Van Damme, J. The Molecular Basis of Leukocytosis. Immunol. Today 1998, 19, 182–189.
- Kolaczkowska, E.; Kubes, P. Neutrophil Recruitment and Function in Health and Inflammation. Nat. Rev. Immunol. 2013, 13, 159–175.
- Lahoz-Beneytez, J.; Elemans, M.; Zhang, Y.; Ahmed, R.; Salam, A.; Block, M.; Niederalt, C.; Asquith, B.; Macallan, D. Human Neutrophil Kinetics: Modeling of Stable Isotope Labeling Data Supports Short Blood Neutrophil Half-Lives. Blood 2016, 127, 3431–3438.
- Lok, L.S.C.; Dennison, T.W.; Mahbubani, K.M.; Saeb-Parsy, K.; Chilvers, E.R.; Clatworthy, M.R. Phenotypically Distinct Neutrophils Patrol Uninfected Human and Mouse Lymph Nodes. Proc. Natl. Acad. Sci. USA 2019, 116, 19083–19089.
- Miralda, I.; Uriarte, S.M.; McLeish, K.R. Multiple Phenotypic Changes Define Neutrophil Priming. Front. Cell. Infect. Microbiol. 2017, 7, 217.
- Metzemaekers, M.; Gouwy, M.; Proost, P. Neutrophil Chemoattractant Receptors in Health and Disease: Double-Edged Swords. Cell. Mol. Immunol. 2020, 17, 433–450.
- Van Damme, J.; Van Beeumen, J.; Opdenakker, G.; Billiau, A. A Novel, NH2-Terminal Sequence-Characterized Human Monokine Possessing Neutrophil Chemotactic, Skin-Reactive, and Granulocytosis-Promoting Activity. J. Exp. Med. 1988, 167, 1364–1376.
- Cowland, J.B.; Borregaard, N. The Individual Regulation of Granule Protein MRNA Levels during Neutrophil Maturation Explains the Heterogeneity of Neutrophil Granules. J. Leukoc. Biol. 1999, 66, 989–995.
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil Extracellular Traps Kill Bacteria. Science 2004, 303, 1532–1535.
- Steinbach, K.; Piedavent, M.; Bauer, S.; Neumann, J.T.; Friese, M.A. Neutrophils Amplify Autoimmune Central Nervous System Infiltrates by Maturing Local APCs. J. Immunol. 2013, 191, 4531–4539.
- Megiovanni, A.M.; Sanchez, F.; Robledo-Sarmiento, M.; Morel, C.; Gluckman, J.C.; Boudaly, S. Polymorphonuclear Neutrophils Deliver Activation Signals and Antigenic Molecules to Dendritic Cells: A New Link between Leukocytes Upstream of T Lymphocytes. J. Leukoc. Biol. 2006, 79, 977–988.
- Takashima, A.; Yao, Y. Neutrophil Plasticity: Acquisition of Phenotype and Functionality of Antigen-Presenting Cell. J. Leukoc. Biol. 2015, 98, 489–496.
- Reinisch, W.; Tillinger, W.; Lichtenberger, C.; Gangl, A.; Willheim, M.; Scheiner, O.; Steger, G. In Vivo Induction of HLA-DR on Human Neutrophils in Patients Treated with Interferon-Gamma. Blood 1996, 87, 3068.
- Vono, M.; Lin, A.; Norrby-Teglund, A.; Koup, R.A.; Liang, F.; Loré, K. Neutrophils Acquire the Capacity for Antigen Presentation to Memory CD4(+) T Cells in Vitro and Ex Vivo. Blood 2017, 129, 1991–2001.
- Matsushima, H.; Geng, S.; Lu, R.; Okamoto, T.; Yao, Y.; Mayuzumi, N.; Kotol, P.F.; Chojnacki, B.J.; Miyazaki, T.; Gallo, R.L.; et al. Neutrophil Differentiation into a Unique Hybrid Population Exhibiting Dual Phenotype and Functionality of Neutrophils and Dendritic Cells. Blood 2013, 121, 1677–1689.
- Geng, S.; Matsushima, H.; Okamoto, T.; Yao, Y.; Lu, R.; Page, K.; Blumenthal, R.M.; Ward, N.L.; Miyazaki, T.; Takashima, A. Emergence, Origin, and Function of Neutrophil-Dendritic Cell Hybrids in Experimentally Induced Inflammatory Lesions in Mice. Blood 2013, 121, 1690–1700.
- Ostanin, D.V.; Kurmaeva, E.; Furr, K.; Bao, R.; Hoffman, J.; Berney, S.; Grisham, M.B. Acquisition of Antigen-Presenting Functions by Neutrophils Isolated from Mice with Chronic Colitis. J. Immunol. 2012, 188, 1491–1502.
- Radsak, M.; Iking-Konert, C.; Stegmaier, S.; Andrassy, K.; Hänsch, G.M. Polymorphonuclear Neutrophils as Accessory Cells for T-Cell Activation: Major Histocompatibility Complex Class II Restricted Antigen-Dependent Induction of T-Cell Proliferation. Immunology 2000, 101, 521–530.
- Abi Abdallah, D.S.; Egan, C.E.; Butcher, B.A.; Denkers, E.Y. Mouse Neutrophils Are Professional Antigen-Presenting Cells Programmed to Instruct Th1 and Th17 T-Cell Differentiation. Int. Immunol. 2011, 23, 317–326.
- Fites, J.S.; Gui, M.; Kernien, J.F.; Negoro, P.; Dagher, Z.; Sykes, D.B.; Nett, J.E.; Mansour, M.K.; Klein, B.S. An Unappreciated Role for Neutrophil-DC Hybrids in Immunity to Invasive Fungal Infections. PLoS Pathog. 2018, 14.
- Gosselin, E.J.; Wardwell, K.; Rigby, W.F.; Guyre, P.M. Induction of MHC Class II on Human Polymorphonuclear Neutrophils by Granulocyte/Macrophage Colony-Stimulating Factor, IFN-Gamma, and IL-3. J. Immunol. 1993, 151, 1482–1490.
- Iking-Konert, C.; Ostendorf, B.; Sander, O.; Jost, M.; Wagner, C.; Joosten, L.; Schneider, M.; Hansch, G.M. Transdifferentiation of Polymorphonuclear Neutrophils to Dendritic-like Cells at the Site of Inflammation in Rheumatoid Arthritis: Evidence for Activation by T Cells. Ann. Rheum. Dis. 2005, 64, 1436–1442.
- Lin, A.; Loré, K. Granulocytes: New Members of the Antigen-Presenting Cell Family. Front. Immunol. 2017, 8, 1781.
- Mudzinski, S.P.; Christian, T.P.; Guo, T.L.; Cirenza, E.; Hazlett, K.R.; Gosselin, E.J. Expression of HLA-DR (Major Histocompatibility Complex Class II) on Neutrophils from Patients Treated with Granulocyte-Macrophage Colony-Stimulating Factor for Mobilization of Stem Cells. Blood. 1995, 86, 2452–2453.
- Spagnoli, G.C.; Juretic, A.; Rosso, R.; Van Bree, J.; Harder, F.; Heberer, M. Expression of HLA-DR in Granulocytes of Polytraumatized Patients Treated with Recombinant Human Granulocyte Macrophage—Colony-Stimulating Factor. Hum. Immunol. 1995, 43, 45–50.




