| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Roberta Barrasso | + 2783 word(s) | 2783 | 2020-06-24 09:53:59 | | | |
| 2 | Camila Xu | -7 word(s) | 2776 | 2020-06-28 04:34:47 | | | | |
| 3 | Camila Xu | -173 word(s) | 2603 | 2020-10-28 08:49:32 | | |
Video Upload Options
Fungal infections are potentially zoonotic and represent challenging cases to accurately diagnose. The current study documents a rare case of Geotrichum candidum dermatomycosis in a horse, which caused chronic skin disease and significant morbidity, partly attributed to incorrect clinical management.
1. Introduction
Skin disease can adversely affect the health and welfare of horses, and disease can be exacerbated by delays in diagnosis and commencement of suitable treatment regimens, also not helped by clinical signs often being similar despite causes being different (infectious versus non-infectious) [1]. Infectious skin disease can be caused by bacteria, parasites, and fungi [2]. Skin diseases caused by fungi are called mycoses and can be categorized as dermatophytosis and dermatomycosis. Dermatophytosis involves infection of the keratinized tissues including hair and stratum corneum, caused by fungi such as Microsporum, Trichophyton, or Epidermophyton. Dermatomycoses are fungal infections of hair, claw, or skin caused by non-dermatophyte fungi, not classified in the aforementioned genera [3]. Equine fungal diseases can also be classified into superficial, cutaneous, subcutaneous, and deep mycoses [4]. Cutaneous infections are the most common fungal skin infections in horses, with dermatophytoses and onychomycoses commonly reported [5]. Dermatophytoses are superficial, cutaneous mycoses caused by dermatophytes and these diseases are considered as zoonoses. Geotrichum candidum is an etiological agent of equine dermatomycosis. From the authors’ knowledge, detailed descriptions of generalized G. candidum dermatitis has not been previously reported. Fungal infections are potentially zoonotic and represent challenging cases to accurately diagnose. The current study documents a rare case of Geotrichum candidum dermatomycosis in a horse, which caused chronic skin disease and significant morbidity, partly attributed to incorrect clinical management. The goal of this case report is to educate horse owners, trainers and veterinarians to consider utilizing various diagnostic tools to enable accurate diagnosis of disease, especially where the initial clinical response to treatment is poor.
At the beginning of the winter season, an eleven-year-old gelded bay saddle horse exhibited well-circumscribed, non-pruritic nodules, 1-2 cm in diameter, on the dorsal part of the neck. Those lesions appeared one week after standing surgery to remove a fracture from the lateral splint bone for which the patient was placed on prophylactic post-surgical antibiotic treatment (gentamicin 6 mg/kg BW; and procaine penicillin 8.000 U.I./kg BW for 7 days). On development of these skin lesions, the owner did not consult with a veterinarian, as the lesions were suspected to represent an allergic reaction, so the owner administered corticosteroids (dexamethasone) per os for a week. The owner monitored the horse during treatment and no skin improvement was noted, and the horse lost weight (about 50 Kg BW and one point in Body Condition Score (BCS) over 3 weeks), the latter attributed to reduced appetite. One week after cessation of oral steroids, a veterinarian was consulted. The veterinarian reported alopecic skin nodules and well-circumscribed, non-pruritic alopecia on the upper lateral region of the front legs. Parental corticosteroids (prednisolone, 1 mg/kg BW) and antihistamine (chlorphenamine, 5 mg/kg BW) were prescribed for a week, as an allergic reaction was still clinically suspected. During this second treatment regimen and continuing for an additional two weeks, dermatitis spread to the head, shoulders and legs (Figure 1), the horse remained inappetant and became lethargic, with clinical signs of colic (i.e., pawing, stamping) which were then treated with flunixin meglumine (1.1 mg/kg BW iv). At this time, a second veterinarian was consulted, approximately one month after the initial nodules developed over the neck.

Figure 1. Distribution of alopecia and crusting seen during the second veterinary examination. (a) Alopecic areas, with thick keratin-rich crusts, distributed over the head; (b) alopecia of the neck (with sparing over the trachea/ventrum) and shoulders; (c) skin erosion on the left knee (palmar and lateral).
During the physical examination performed by the second veterinarian, the horse was lethargic, pyretic (40 °C), with increased heart and respiratory rates (54 bpm and 28 bpm, respectively) and hair was shedding/easily epilated over the head, neck, shoulder, chest and all four legs, with severe swelling of the hind limbs and carpal region. Yellow exudate could be easily expressed from the surface of a skin erosion (2 cm) on the left knee (palmar and lateral). The distribution of alopecia involved the head (severe on the cheek and forelock regions, with sparing over the nose, nostril and muzzle regions), neck (widespread with sparing over the trachea/ventrum) and circumferentially around all four lower limbs and shoulders, most severe between the neck and shoulder regions. Large white, loosely adherent scale coated alopecic areas, with thick keratin-rich crusts, ranging from 0.1 to 1 cm (Figure 1). Blood was collected via the external jugular vein into two tubes with ethylenediaminotetracetic-acid (EDTA) and without anticoagulant of the vacutainer system (Becton Dickinson, Franklin Lakes, NJ, USA), stored at 4 °C and sent to the Laboratory of Internal Medicine (University of Bari, Italy). Hair and skin scraping samples were collected from lesions (at the neck level) using a sterile scalpel and pliers and placed into plastic vials containing 70% ethanol and submitted for parasitological analysis to the Laboratory of Parasitology of the Department of Veterinary Medicine (DVM) (University of Bari, Italy). Hair was collected by brush and also by tweezers making sure to collect hairs with bulbs. Hair and skin scraping samples were placed into a sterile petri dish and sent to the Mycology laboratory of DVM. Anti-inflammatory drugs were administered (flunixin meglumine 1.1 mg/Kg iv, once daily for three days). A week after this initial physical examination, additional samples were collected. Three skin biopsies were performed from three different sites over the neck and included a margin of a recently developed lesion. Biopsy samples were placed into a sterile tube and sent at 4 °C to the Laboratories of Pathology and Bacteriology, DVM for histopathology and culture. The same day, blood samples and hairs were collected and sent for bacteriological and mycological analysis to the Laboratories of Bacteriology and Mycology of DVM.The BCC (Blood cell counts) parameters were within the normal range, apart from a minimal increase in leucocytes due to neutrophilia. LDH, bilirubin, urea, and triglycerides levels were mildly increased. Total proteins were mildly elevated, due to a mild increase in globulins representing hypergammaglobulinemia as per electrophoresis. Alpha 1 fraction was moderately decreased and both beta fractions were mildly to moderately increased. Skin biopsies revealed normal contaminant microflora and the blood culture sample tested negative for bacterial growth after 48 h of incubation and negative for fungi after 14 days. Parasitological analyses were negative. Geotrichum candidum was cultured from skin scale (Figure 2).
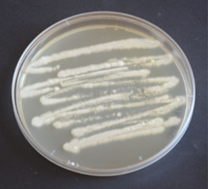 (a)
(a) 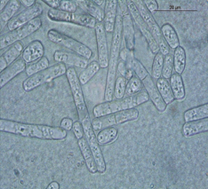 (b)
(b)Figure 2. Macroscopic (a) and microscopic (b) features of Geotrichum candidum. (a): White, dry, powdery to cottony colonies of Geotrichum candidum. (b): Hyaline septate hyphae breaking up into chains of hyaline, smooth, one-celled, sub-globose to cylindrical arthroconidia (6-12 x 3-6 µm in size).
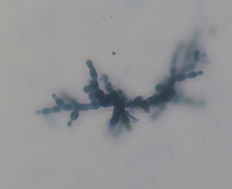
The fungal culture was repeated twice from hair samples collected on two different days and Geotrichum candidum was the only isolated in pure culture. Cytology revealed a mass of branching segmented hyphae forming rectangular or oval-shaped arthroconidia which stained blue (M.G.) and varied in length from 3 to 6 µm (Figure 3).
Figure 3. Cytology impression smear from hair and skin sample collected from a horse with severe dermatitis; May-Grünwald Giemsa stained specimen revealing a mass of hyphae (M.G. 100 x, oil objective).
A single biopsy sample of haired skin exhibited mild orthokeratotic hyperkeratosis with mild epidermal hyperplasia due to acanthosis. There was a minimal degree of interface dermatitis, with low numbers of leucocytes, which were difficult to further identify, crossing the epidermis. The superficial dermis exhibited a mild perivascular inflammatory pattern with small lymphocytes admixed with fewer neutrophils. Endothelium was plump and reactive and small caliber dermal capillaries were moderately dilated. Hair follicles were in different stages of the hair cycle with retained hairs and anagen bulbs seen (Figures 4 and 5). PAS and GMS did not reveal any fungal organisms. No surface crust material was submitted. Mild perivascular to interface lymphocytic, hyperplastic dermatitis (Figures 4 and 5).
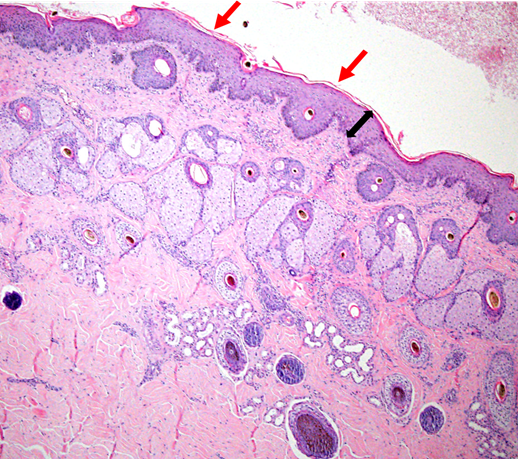
Figure 4. Skin histopathology from a horse with severe dermatitis; mild lymphocytic, hyperplastic dermatitis (H.E. 4x objective). Black arrow indicates hyperplasia (acanthosis) of the epidermis. Red arrows indicate mildly increased surface keratin.
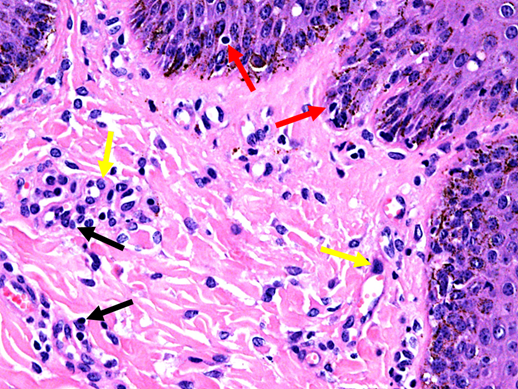
Figure 5. Skin histopathology from a horse with severe dermatitis, showing mild lymphocytic, hyperplastic dermatitis (H.E. 40x objective). Black arrows highlight perivascular inflammatory cells; yellow arrows highlight plump endothelial cells of dermal capillaries; red arrows highlight leucocytes in epithelium.
Therapy using a topical solution (2% of concentration) of miconazole and chlorhexidine[6], applied once a day for a week, then every other day for 14 days, and finally twice a week for 28 days, was recommended[5]. The patient’s diet was supplemented with vitamins C (ascorbic acid, 30 g/daily per os) and E (5000 IU/daily per os). The indoor stable and horse equipment were disinfected (Virkon S Farmaceutici Gellini, Aprilia, Italy)[7] and the management was changed, providing at least 6 h/day of grazing without a rug. Three-weeks following topical treatment, the horse had gained weight (about 80 Kg difference using a BW tape) and BCS (manually evaluated), alopecic regions showed new hair growth with fewer crusts, hematological and biochemical parameters returned to the normal range[8][9]. The antifungal treatment was continued twice a week for three more months. The horse recovered completely, and antioxidant supplementation was recommended as an ongoing dietary supplementation as prescribed.
This case report documents a case of dermal geotrichosis in a horse previously treated with prophylactic, post-surgical antibiotic and corticosteroid therapy. The horse was housed in a single box for 24 h wearing a winter blanket and then improperly treated with corticosteroids for three weeks (by the owner for a week and then by the first of two veterinarians, for two weeks). Corticosteroid treatment may have impaired the immune system of this horse, making the horse suspectable to contracting fungal infection. This case report highlights the importance of collecting appropriate samples to enable a thorough clinical workup of skin diseases in horses. Due to the initial lack of appropriate treatment when the skin disease first developed, this horse experienced prolonged sufferance for more than two months, developing chronic, spreading skin disease, with systemic signs of illness including weight loss, inappetence, fever and colic. Equine skin diseases are often challenging to diagnose and successfully treat, but this case demonstrates that appropriate laboratory tests should always be conducted as part of the basic principles of responsible pet ownership and proper veterinary duty of care, which are important to safeguard animal health and well-being.
The failure to identify the disease early in the disease course played a role in the development of extensive skin disease in this case, as dermatomycoses, are generally considered to represent opportunistic disease[5]. It was difficult to decide whether all the clinical signs revealed at the second clinical examination were due to the dermal geotrichosis, or whether a systemic disease process was complicating the clinical picture. Only skin bacteriological and parasitological tests were run, and those possible skin diseases were excluded because negative. However, since specific tests to detect systemic infections were not performed, it would be impossible to be certain whether dermal geotrichosis was the primary condition or represented an opportunistic infection with a horse with co-morbidity.
The biochemical profile supported chronicity of inflammation due to the elevated globulins, low albumin/globulin ratio (useful as a biomarker of inflammation), with inflammation further supported by neutrophilia [9]. Serum electrophoresis was consistent for a polyclonal gammopathy, likely due to increased IgG and IgM. The increased β1 fraction was assumed to be due to increased IgM and isotypes of IgG which can migrate into this region. Increased β2 fraction was likely due to isotypes of IgG, such as IgGT, and/or IgM, which can both spill over into this fraction. The decreased α1 fraction is unexpected as this fraction is represented mostly by positive acute phase proteins. The decrease may indicate the chronicity of the case, confirming the late diagnosis. This decrease may also indicate protein loss from skin exudation and/or loss from the gut, which may also have contributed to the low albumin value, although albumin is typically reduced in inflammatory states [9]. The increase in LDH, urea, and triglycerides and low magnesium mirror the negative energy status of the horse, which was refusing to eat and showed mild signs of colic the day before blood sampling [8].
Geotrichum spp. are yeast-like fungi, commonly isolated from soil, air, water, milk, silage, and plant tissues and can be isolated from the digestive tract, skin, vagina, and mouth of humans and other mammals [10][11]. In some cases, this fungus can be pathogenic, producing disseminated or localized skin disease in both humans and animals [12][13][14]. There are 13 species of Geotrichum but Geotrichum candidum is one of the most important species reported to cause infection in animals [10][11]. Figueredo et al. [7] reported that the head and neck regions were the most common regions affected with fungal dermatitis, whilst legs and tails were less likely affected. The current horse had severe alopecia particularly affecting the head and neck regions, which likely represented primary areas of infection, in agreement with the literature [7] and then, the fungus likely migrated or was spread to other parts of the body.
2. Conclusion
This study is therefore useful to educate veterinarians on how to diagnose generalized Geotrichum candidum infection in horses. Geotrichosis causes alopecia with desquamation and more rarely erythema, hyperkeratosis and pruritus in horses [7]. The clinical signs in the current horse at the beginning were in agreement with the literature, with the horse becoming inappetant and pyretic. Likely, the extent of dermatitis and the lack of prompt treatment (i.e., chronic form) were enough to create the clinical picture presented: fever, lethargy and inappetence. Geotrichum species have rarely been reported as a cause of sepsis or disseminated infection in any species, with severe systemic infections affecting immunosuppressed or patients with severe, chronic co-morbidities in humans. However, as mentioned, it is impossible to say whether all the clinical signs in the current case were due to dermal geotrichosis or there was systemic infection also present since the immune system of the horse may have been adversely affected by the previous corticosteroid therapy.
The horse responded well to the topical antimycotic treatment confirming that dermal geotrichosis was a significant cause of morbidity. The presence of fungi was initially suspected by cytological examination and was confirmed using routine mycological procedures. Geotrichum can be easy to identify using cytology, as it forms true hyphae that segment into rectangular arthroconidia which vary in length (4-10 µm). However, in this case, cytology revealed only the presence of yeast-like organisms not clearly attributable to Geotrichum [15]. The successful culture in pure growth, of Geotrichum candidum from different skin specimens collected at two different time points confirmed the etiology of this skin disease. The lack of fungus seen on histology is attributed to the lack of crusts submitted for examination, as this fungus is known to be keratinophilic [16] and possibly also to the limited number of biopsy samples taken from not ideal sites. The authors recommend for all cases of multifocal or generalized skin disease, multiple skin biopsy samples and inclusion of crusts be collected as a routine practice. This study confirms the risk of developing generalized fungal infections, such as Geotrichum infection, in horses after prolonged treatment with corticosteroids, which is a known risk factor for developing secondary infections in both human and equine veterinary medicine [7]. This case also confirms the efficacy of topical antifungal treatments as a useful therapy to resolve fungal skin infections. Moreover, it highlights the potential clinical usefulness of antioxidant therapy in enhancing the immune response [17] in affected animals and of changing the management as a coadjuvant of treatment. Finally, this case report may be useful to enhance the knowledge of horse owners, trainers, and veterinarians not only on geotrichosis but also on the importance of prompt and correct diagnosis of equine skin disease.
References
- Scott, D.W.; Miller, W.H. Bacterial skin diseases. In Equine Dermatology, 2nd ed.; Scott, D.W., Miller,W.H., Eds.; Elsevier Saunders: St Louis, MO, USA, 2011; pp. 130-170.
- Lloyd, D.H.; Littlewood, J.D.; Craig, J.M.; Thomsett, L.R. Practical Equine Dermatology, 1st ed.;Wiley-Blackwell: Oxford, UK, 2003.
- Ainsworth, G.C.; Austwick, P.K.C. Fungal Disease of Domestic Animals. In Commonwealth Agricultural Bureau, 2nd ed.; Farnham Royal: London, UK, 1973.
- De Hoog, G.S.; Guarro, J.; Gene, J.; Figueras, M.J. Atlas of Clinical Fungi. In Centraal Bureau Voor Schimmel Cultures; Centraalbureau voor Schimmelcultures (CBS): Utrecht, The Netherland, 2000; p. 1126.
- Cafarchia, C.; Figueredo, L.A.; Otranto, D. Fungal diseases of horses. Vet. Microbiol. 2013, 167, 215-234.
- Sidhu, R.K.; Singh, K.B.; Jand, S.K.; Joshi, D.V. Cutaneous geotrichosis in a dog and its handler - A case report. Indian J. Anim. Health 1993, 32, 75.
- Figueredo, L.A.; Cafarchia, C.; Otranto, D.Geotrichum candidum as etiological agent of horse dermatomycosis. Vet. Microbiol. 2011, 148, 368-371.
- Weiss, D.J.; Wardrop, K.J. Shalm’s Veterinary Hematology, 6th ed.; Wiley Blackwell: Ames, IA, USA, 2010; p. 1206.
- Dietz, O.; Huskamp, B. Handbuch Pferdepraxis, 3rd ed.; Enke: Stuttgart, Germany, 1999; pp. 623-650.
- Pottier, I.; Gente, S.; Vernoux, J.P.; Gueguen, M. Safety assessment of dairy microorganisms: Geotrichum candidum. Int. J. Food Microbiol. 2008, 126, 327-332.
- Miceli, M.H.; Diaz, J.A.; Lee, S.A. Emerging opportunistic yeast infections. Lancet Infect. Dis. 2011, 11, 142-151.
- Dolensek, E.P.; Napolitano, R.L.; Kazimiroff, J. Gastrointestinal geotrichosis in six adult gorillas. J. Am. Vet. Med. Assoc. 1977, 171, 975-976.
- Chahota, R.; Katoch, R.; Mahajan, A.; Verma, S. Clinical bovine mastitis caused by Geotrichum candidum. Vet. Arhiv. 2001, 171, 197-201.
- Henrich, T.J.; Marty, F.M.; Milner, D.A., Jr.; Thorner, A.R. Disseminated Geotrichum candidum infection in a patient with relapsed acute myelogenous leukemia following allogeneic stem cell transplantation and review of the literature. Transpl. Infect. Dis. 2009, 11, 458-462.
- Koneman, E.W. Koneman’s Color Atlas and Textbook of Diagnostic Microbiology, 6th ed.; Koneman, E.W., Ed.; LippincottWilliams & Wilkins (LWW): Philadelphia, PA, USA, 2006.
- Rajak, R.C.; Parwekar, S.; Malviya, H.; Hasija, S.K. Keratin degradation by fungi isolated from gelatin factory in Jabalpur. Mycopathologia 1991, 114, 83-87.
- Fekete, S.G.; Kellems, R.O. Interrelationship of feeding with immunity and parasitic infection: A review. Vet. Med. 2007, 52, 131-143.