
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ibraheem Al-Naib | + 756 word(s) | 756 | 2021-10-19 10:44:49 | | | |
| 2 | Vicky Zhou | Meta information modification | 756 | 2021-10-30 03:41:32 | | |
Video Upload Options
Glucose monitoring is essential to control diabetes and avoid long-term complications. Diabetics suffer on a daily basis with the traditional glucose monitors currently in use, which are invasive, painful, and cost-intensive. Therefore, the demand for non-invasive, painless, economical, and reliable approaches to monitor glucose levels is increasing.
1. Introduction
As no cure has been found for diabetes yet, regular monitoring and control of glucose concentration in the blood is the only solution to optimize the lifestyle of diabetics and prevent them from experiencing severe complications [1]. Various glucose monitoring techniques have been developed recently. These technologies are classified based on their mechanism as invasive (IN), minimally invasive (MI), and non-invasive (NI). Figure 1 maps most of the glucose-sensing technologies [2][3][4].
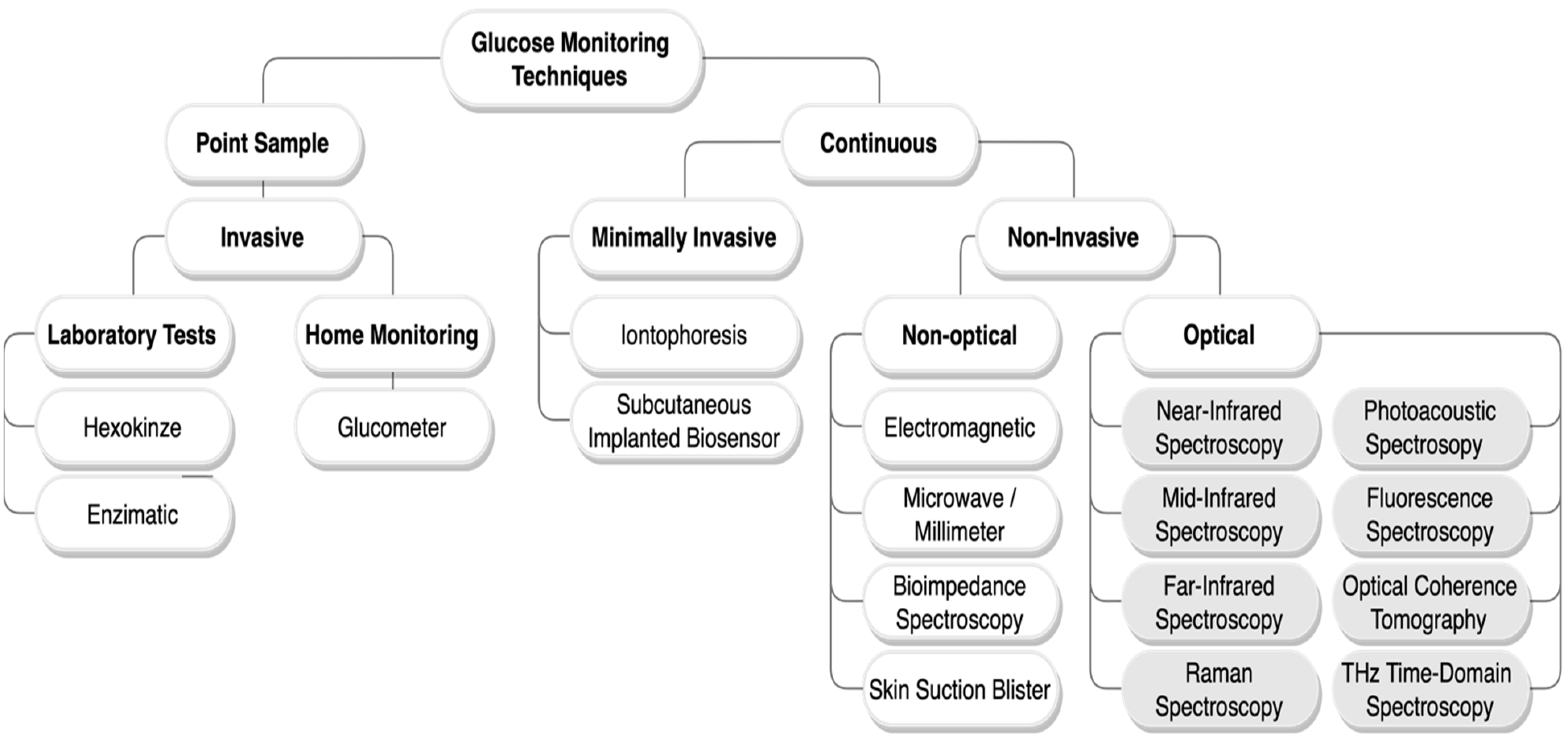
Figure 1. Glucose monitoring techniques classification chart. Herein, only the non-invasive optical methods with a grey background are reviewed.
The invasive methods are the gold standard and the most broadly employed for blood glucose measurements [2]. Conventional devices currently in use, such as the self-monitoring blood glucose (SMBG) devices, so-called glucometer, and the continuous-glucose-monitoring (CGM) devices, follow the invasive and minimally invasive methods, respectively [5][6][7]. They are both based on electrochemical biosensors. The SMBG sensors require drawing a drop of blood to be tested through finger-pricking, while the CGM sensors are based on a needle implanted subcutaneously. Although these techniques give high accuracy measurements, they have multiple drawbacks for being painful, promoting infection, using cost-intensive blood glucose testing supplies, and deterioration of accuracy over time. Accordingly, scientists have conducted studies on non-invasive glucose sensors that are reliable, fast, painless, and cost-effective for the convenience of patients to monitor their glucose level frequently; hence, reducing diabetes complications. Various types of non-invasive techniques have been proposed during the last two decades, including non-optical and optical techniques as illustrated in Figure 1. Among the non-invasive glucose monitoring techniques, the optical methods give the best measurements [8]. Optical technologies such as near-infrared, mid-infrared, or Raman spectroscopy have great selectivity for glucose sensing given the complexity of the blood/tissue properties. Furthermore, the targeted biological tissue in optical approaches is less exposed to irritation [8].
2. Comparison of Current Non-Invasive Optical Techniques
| Technology | Wavelength | Selectivity | Measurement Site | Merits | Drawbacks |
|---|---|---|---|---|---|
| NIR spectroscopy |
750–2500 nm | Good | Ear lobe, finger, forearm, cheek, lip mucosa, oral mucosa, and tongue |
|
|
| MIR spectroscopy |
2500–10,000 nm | Good, superior to NIR |
Finger, skin, and oral mucosa |
|
|
| FIR spectroscopy |
10–1000 μm | Good | ISF |
|
|
| Raman spectroscopy |
Visible light | Excellent | Eye, human skin |
|
|
| THz-TDS | 30 µm to 3 mm | Good | ISF |
|
|
| Fluorescence | Ultraviolet light, visible light | Excellent | Tears, human skin |
|
|
| PA spectroscopy |
Ultraviolet light, NIR, and MIR | Good | Finger, forearm, and aqueous humor |
|
|
3. Conclusions
References
- Bruen, D.; Delaney, C.; Florea, L.; Diamond, D. Glucose sensing for diabetes monitoring: Recent developments. Sensors 2017, 17, 1866.
- Dinani, S.T.; Zekri, M.; Kamali, M. Regulation of blood glucose concentration in type 1 diabetics using single order sliding mode control combined with fuzzy on-line tunable gain, a simulation study. J. Med. Signals Sens. 2015, 5, 131–140.
- Cryer, P.E. Minireview: Glucagon in the Pathogenesis of Hypoglycemia and Hyperglycemia in Diabetes. Endocrinology 2012, 153, 1039–1048.
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843.
- Ajjan, R.; Slattery, D.; Wright, E. Continuous Glucose Monitoring: A Brief Review for Primary Care Practitioners. Adv. Ther. 2019, 36, 579–596.
- Li, K.; Daniels, J.; Liu, C.; Herrero, P.; Georgiou, P. Convolutional Recurrent Neural Networks for Glucose Prediction. IEEE J. Biomed. Health Inform. 2020, 24, 603–613.
- Jernelv, I.L.; Milenko, K.; Fuglerud, S.S.; Hjelme, D.R.; Ellingsen, R.; Aksnes, A. A review of optical methods for continuous glucose monitoring. Appl. Spectrosc. Rev. 2019, 54, 543–572.
- Sim, J.; Ahn, C.; Jeong, E.; Kim, B. In vivo Microscopic Photoacoustic Spectroscopy for Non-Invasive Glucose Monitoring Invulnerable to Skin Secretion Products. Sci. Rep. 2018, 8, 1059.
- Rachim, V.P.; Chung, W.Y. Wearable-band type visible-near infrared optical biosensor for non-invasive blood glucose monitoring. Sens. Actuators B Chem. 2019, 286, 173–180.
- Kasahara, R.; Kino, S.; Soyama, S.; Matsuura, Y. Noninvasive glucose monitoring using mid-infrared absorption spectroscopy based on a few wavenumbers. Biomed. Opt. Express 2018, 9, 289.
- Kang, J.W.; Park, Y.S.; Chang, H.; Lee, W.; Singh, S.P.; Choi, W.; Galindo, L.H.; Dasari, R.R.; Nam, S.H.; Park, J.; et al. Direct observation of glucose fingerprint using in vivo Raman spectroscopy. Sci. Adv. 2020, 6, eaay5206.
- Zhou, J.; Wang, X.; Wang, Y.; Huang, G.; Yang, X.; Zhang, Y.; Xiong, Y.; Liu, L.; Zhao, X.; Fu, W. A novel THz molecule-selective sensing strategy in aqueous environments: THz-ATR spectroscopy integrated with a smart hydrogel. Talanta 2021, 228, 122213.
- Aloraefy, M.; Pfefer, T.; Ramella-Roman, J.C.; Sapsford, K.E.; Joshua Pfefer, T.; Ramella-Roman, J.C.; Sapsford, K.E. In vitro evaluation of fluorescence glucose biosensor response. Sensors 2014, 14, 12127–12148.
- Chen, T.-L.; Lo, Y.-L.; Liao, C.-C.; Phan, Q.-H. Noninvasive measurement of glucose concentration on human fingertip by optical coherence tomography. J. Biomed. Opt. 2018, 23, 047001.
- Lundsgaard-Nielsen, S.M.; Pors, A.; Banke, S.O.; Henriksen, J.E.; Hepp, D.K.; Weber, A. Critical-depth Raman spectroscopy enables home-use non-invasive glucose monitoring. PLoS ONE 2018, 13, e0197134.




