Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alexandre Harlé | + 1370 word(s) | 1370 | 2021-10-21 04:17:47 | | | |
| 2 | Catherine Yang | Meta information modification | 1370 | 2021-10-22 03:19:43 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Harlé, A. Damage-Specific DNA-Binding Protein 2. Encyclopedia. Available online: https://encyclopedia.pub/entry/15251 (accessed on 08 February 2026).
Harlé A. Damage-Specific DNA-Binding Protein 2. Encyclopedia. Available at: https://encyclopedia.pub/entry/15251. Accessed February 08, 2026.
Harlé, Alexandre. "Damage-Specific DNA-Binding Protein 2" Encyclopedia, https://encyclopedia.pub/entry/15251 (accessed February 08, 2026).
Harlé, A. (2021, October 21). Damage-Specific DNA-Binding Protein 2. In Encyclopedia. https://encyclopedia.pub/entry/15251
Harlé, Alexandre. "Damage-Specific DNA-Binding Protein 2." Encyclopedia. Web. 21 October, 2021.
Copy Citation
Damage-specific DNA-binding protein 2 (DDB2) was originally identified as a DNA damage recognition factor that facilitates global genomic nucleotide excision repair (GG-NER) in human cells. DDB2 also contributes to other essential biological processes such as chromatin remodeling, gene transcription, cell cycle regulation, and protein decay.
DDB2
DNA repair
cancers
1. Introduction
DDB2 (damage-specific DNA-binding protein 2, also known as p48 subunit) is a 48-kDa protein exclusively localized in the nucleus of mammalian cells. DDB2 is ubiquitously present in human tissues, albeit differentially expressed. High DDB2 expression level has been described in liver, thymus, kidney, and testes, and a low expression level has been described in brain, lung, skin, heart, and muscles [1]. DDB2 is composed of seven WD40 repeat domains and a N-terminal helix–loop–helix motif [2]. The WD40 domains play a role as a module for sequence-specific protein–DNA or protein–protein interactions. Both domains appear critical for the biological functions of DDB2.
Numerous overlapping mechanisms have been found to be involved in the regulation of DDB2 expression. DDB2 basal expression is cell cycle-dependent in normal dividing cells with a DDB2 level that gradually increases in the mid-G1 phase and reaches a maximum at the G1/S boundary before dropping in the S-phase [3]. DDB2 transcription was shown to be transiently increased after UV-induced DNA damage in a p53-dependent manner [1]. The p53 protein cooperates with BRCA1 (Breast Cancer Associated protein 1) for its binding to the ddb2 promoter and inducing ddb2 transactivation [1] (Figure 1). The TAp63γ (Tumor protein 63 isoform gamma) isoform of the p63 protein that belongs to the p53 family and shares strong structural similarity with p53 is also able to activate DDB2 expression through recognition of the same region upstream of the transcription initiation site [4]. Structural analysis of the ddb2 promoter shows multiple Sp1 (Transcription factor Sp1) -specific binding sites as usually found in gene promoters with a G–C rich sequence lacking a TATA box and suggesting a critical role for Sp1 for the basal expression of DDB2 [5] (Figure 1). NF1 (Neurofibromin 1) and E2F (Transcription factor E2F) elements are also identified, although probably having a smaller impact on DDB2 regulation [5]. Moreover, DDB2 activity is finely adjusted through post-transcriptional and post-translational mechanisms. An IRES (internal ribosome entry site) element located at the 5′ end of DDB2 mRNA stimulates the translational process of DDB2 in stress conditions such as serum starvation or exposure to doxorubicin [6]. Moreover, the 3′ untranslated region (3′UTR) of DDB2 mRNA harbors an uracil-rich sequence enabling its prompt export to the cytoplasm and thus DDB2 translational upregulation. During the DNA repair process, the Cul4A (Cullin4A) protein regulates DDB2 protein lifespan by means of its ubiquitin ligase functions [3]. The PARylation (Protein poly ADP-ribosylation) of the DDB2 protein in a PARP1-dependent (Poly [ADP-ribose] polymerase 1) manner stabilizes DDB2 and delays its degradation [7]. A PIP (PCNA-interacting protein) box sequence located in the N-terminal region of DDB2 enables the interaction between DDB2 and PCNA (proliferating cell nuclear antigen) for DDB2 proteolytic degradation even in the absence of DNA damage [8][9].
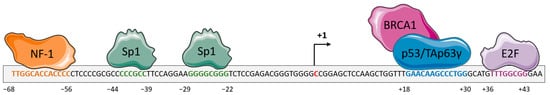
Figure 1. Schematic representation of the regulation of the gene encoding the DDB2 (Damage-specific DNA-binding protein 2) protein. The proximal promoter of ddb2 gene harbors response elements for the transcription factors NF-1 (Neurofibromin 1) (orange) and Sp1 (Transcription factor Sp1) (green) upstream of the transcription initiation site. The proximal promoter also contains a response element for p53, in association with BRCA1 (Breast cancer type 1 susceptibility protein), or Tap63γ (Tumor protein 63 isoform gamma) (blue) proteins and the E2F (Transcription factor E2F) transcription factor downstream of the transcription initiation site. The binding of these proteins leads to the regulation of ddb2 gene expression.
2. DDB2: A New Potent Tumor Suppressor?
DDB2 was first considered as a novel tumor suppressor based on the findings that mutations in the ddb2 gene result in an impairment in DDB2–DNA or DDB2–DDB1 interactions and subsequent NER activity defects [2]. Such deficiencies are observed in a subset of Xeroderma pigmentosum hereditary disease (Xeroderoma pigmentosum group E, XP-E) that displays an extreme sensitivity to UV radiation and a high predisposition to skin cancers [7]. Moreover, DDB2-deficient mice are prone to developing a wide panel of tumors, even in the absence of UV exposition [10]. Several studies highlighted an altered DDB2 expression, compared to non-malignant tissues, in many types of cancers [11], including prostate [12], colorectal [13][14], skin [15], head and neck [16], and ovarian [17] cancers. Furthermore, a correlation between low DDB2 expression level and poor outcomes was established among patients with melanoma, colon, ovarian, lung, breast, brain or head, and neck cancers, suggesting a critical role for DDB2 in tumor suppression [13][16][17][18].
3. DDB2 Has a Dual Activity on Cancer Cell Proliferation
High levels of DDB2 protein and mRNA are reported in ER (Estrogen receptor)-positive and non-invasive breast cancer models compared to ER-negative aggressive breast cancer cells and mammary non-malignant cells [19]. Enhanced DDB2 expression in DDB2-low level models upregulates in vitro cancer cell growth rate and clonogenicity. Such effects are abrogated by DDB2 knockdown in DDB2-overexpressed breast cancer models, suggesting the oncogenic role of DDB2 in mammary cancer cell growth [19]. DDB2 facilitates cell cycle progression, especially the entry in the S-phase, through the binding of the C-terminal domain and the co-activation of the transcription factor E2F1 [3]. By means of E2F1 transcriptional abilities, DDB2 indirectly regulates the expression of key genes involved in DNA replication and G1/S transition. The stimulating effect of DDB2 on cancer cell growth also involves the downregulation of manganese superoxide dismutase (MnSOD) [19]. MnSOD is a mitochondrial enzyme that detoxifies reactive oxygen species (ROS) to protect cells from oxidative damage. DDB2 interacts with the proximal SOD2 (Superoxide dismutase 2) promoter, resulting in the loss of H3 histone acetylation, and in the recruitment of the AP-2 transcription factor, which is well known in the repression of the SOD2 gene and downregulation of the encoded MnSOD [20]. By this mechanism, DDB2 attenuates the elimination of ROS that are known to activate several signaling pathways involved in breast cancer cell growth [20].
In contrast, DDB2 has shown antiproliferative properties in ovarian and prostate cancers in vitro. In an ovarian cancer cell model, DDB2 is reported to negatively regulate NEDD4L (Neural precursor cell expressed developmentally downregulated gene 4-like) cellular levels by inducing histone H3 trimethylation at the NEDD4L promoter region [21]. This limits the NEDD4L-dependent proteolytic degradation of the effector proteins SMAD2 (Mothers against decapentaplegic homolog) and SMAD3 and enhances the TGF-β (Transforming growth factor beta) signal transduction downstream, finally contributing to the inhibition of cancer cell proliferation [21].
NRIP/DCAF6 (Nuclear receptor-interacting protein 1/DDB1- and CUL4-associated factor 6) and DDB2 proteins are both members of the DDB1 and CUL4-associated factors DCAF family and androgen receptor (AR)-interacting proteins that physiologically compete to maintain AR expression level. DDB2 mediates the contact between AR and CUL4A–DDB1 E3 ligase complex for AR ubiquitination and proteasomal degradation in a p53-independent manner [22], while NRIP displaces DDB2 from the complex and stabilizes AR [12]. As the DDB2 expression level is found to be lower in prostate cancer tissues compared to non-neoplastic ones, this could interfere with AR homeostasis and induce subsequent AR-dependent prostate cancer growth.
Deregulation in the Wnt signaling pathway is usually co-opted during colon cancer development and is sought to be a driver event in this process. Negative regulatory effects of DDB2 in the Wnt signaling are mediated by the recruitment of β-catenin and the H3K27 methylase EZH2 (Enhancer of zeste homolog 2) to Rnf43 (Ring Finger protein 43) promoter. This facilitates the activation of the RNF43 enzyme that eliminates Wnt receptors at the cell surface and downregulates Wnt signaling in colorectal cancer cells [23].
4. DDB2 Affects Cancer Stem Cell Populations
Some studies reveal the protective role of DDB2 for ovarian cancer progression and recurrence by limiting the cancer stem cell (CSC) population. High levels of DDB2 are shown to halt the formation of ovarian tumor xenografts in vivo [17]. DDB2 exerts this regulatory function through the increase of IκBα protein levels, which results in the repression of the NF-κB signaling pathway and impinges the capacity of CSC to self-renew [17]. DDB2 also represses ovarian cancer cell dedifferentiation by inhibiting the transcription of aldehyde dehydrogenase 1 family member A1 (ALDH1A1) [24]. The impact of DDB2 on cancer stem cell populations is still under investigation in other cancer models and need to be explored.
References
- Tan, T.; Chu, G. p53 Binds and Activates the Xeroderma Pigmentosum DDB2 Gene in Humans but Not Mice. Mol. Cell. Biol. 2002, 22, 3247–3254.
- Scrima, A.; Konícková, R.; Czyzewski, B.K.; Kawasaki, Y.; Jeffrey, P.D.; Groisman, R.; Nakatani, Y.; Iwai, S.; Pavletich, N.P.; Thomä, N.H. Structural basis of UV DNA-damage recognition by the DDB1-DDB2 complex. Cell 2008, 135, 1213–1223.
- Nag, A.; Bondar, T.; Shiv, S.; Raychaudhuri, P. The xeroderma pigmentosum group E gene product DDB2 is a specific target of cullin 4A in mammalian cells. Mol. Cell. Biol. 2001, 21, 6738–6747.
- Liu, J.; Lin, M.; Zhang, C.; Wang, D.; Feng, Z.; Hu, W. TAp63γ enhances nucleotide excision repair through transcriptional regulation of DNA repair genes. DNA Repair 2012, 11, 167–176.
- Nichols, A.F.; Itoh, T.; Zolezzi, F.; Hutsell, S.; Linn, S. Basal transcriptional regulation of human damage-specific DNA-binding protein genes DDB1 and DDB2 by Sp1, E2F, N-myc and NF1 elements. Nucleic Acids Res. 2003, 31, 562–569.
- Dai, W.; Ma, W.; Li, Q.; Tao, Y.; Ding, P.; Zhu, R.; Jin, J. The 5′-UTR of DDB2 harbors an IRES element and upregulates translation during stress conditions. Gene 2015, 573, 57–63.
- Luijsterburg, M.S.; Lindh, M.; Acs, K.; Vrouwe, M.G.; Pines, A.; van Attikum, H.; Mullenders, L.H.; Dantuma, N.P. DDB2 promotes chromatin decondensation at UV-induced DNA damage. J. Cell Biol. 2012, 197, 267–281.
- Cazzalini, O.; Perucca, P.; Mocchi, R.; Sommatis, S.; Prosperi, E.; Stivala, L.A. DDB2 association with PCNA is required for its degradation after UV-induced DNA damage. Cell Cycle Georget. Tex 2014, 13, 240–248.
- Perucca, P.; Sommatis, S.; Mocchi, R.; Prosperi, E.; Stivala, L.A.; Cazzalini, O. A DDB2 mutant protein unable to interact with PCNA promotes cell cycle progression of human transformed embryonic kidney cells. Cell Cycle 2015, 14, 3920–3928.
- Yoon, T.; Chakrabortty, A.; Franks, R.; Valli, T.; Kiyokawa, H.; Raychaudhuri, P. Tumor-prone phenotype of the DDB2-deficient mice. Oncogene 2005, 24, 469–478.
- Bagchi, S.; Raychaudhuri, P. Damaged-DNA Binding Protein-2 Drives Apoptosis Following DNA Damage. Cell Div. 2010, 5, 3.
- Chen, H.-H.; Fan, P.; Chang, S.W.; Tsao, Y.P.; Huang, H.P.; Chen, S.L. NRIP/DCAF6 stabilizes the androgen receptor protein by displacing DDB2 from the CUL4A-DDB1 E3 ligase complex in prostate cancer. Oncotarget 2017, 8, 21501–21515.
- Yang, H.; Liu, J.; Jing, J.; Wang, Z.; Li, Y.; Gou, K.; Feng, X.; Yuan, Y.; Xing, C. Expression of DDB2 Protein in the Initiation, Progression, and Prognosis of Colorectal Cancer. Dig. Dis. Sci. 2018, 63, 2959–2968.
- Liu, J.; Li, H.; Sun, L.; Feng, X.; Wang, Z.; Yuan, Y.; Xing, C. The Differential Expression of Core Genes in Nucleotide Excision Repair Pathway Indicates Colorectal Carcinogenesis and Prognosis. BioMed Res. Int. 2018, 2018, 9651320.
- Stoyanova, T.; Roy, N.; Bhattacharjee, S.; Kopanja, D.; Valli, T.; Bagchi, S.; Raychaudhuri, P. p21 cooperates with DDB2 protein in suppression of ultraviolet ray-induced skin malignancies. J. Biol. Chem. 2012, 287, 3019–3028.
- Bommi, P.V.; Ravindran, S.; Raychaudhuri, P.; Bagchi, S. DDB2 regulates Epithelial-to-Mesenchymal Transition (EMT) in Oral/Head and Neck Squamous Cell Carcinoma. Oncotarget 2018, 9, 34708–34718.
- Han, C.; Zhao, R.; Liu, X.; Srivastava, A.; Gong, L.; Mao, H.; Qu, M.; Zhao, W.; Yu, J.; Wang, Q.E. DDB2 suppresses tumorigenicity by limiting the cancer stem cell population in ovarian cancer. Mol. Cancer Res. 2014, 12, 784–794.
- De Sousa, J.F.; Torrieri, R.; Serafim, R.B.; Di Cristofaro, L.F.; Escanfella, F.D.; Ribeiro, R.; Zanette, D.L.; Paçó-Larson, M.L.; da Silva, W.A., Jr.; Tirapelli, D.P.; et al. Expression signatures of DNA repair genes correlate with survival prognosis of astrocytoma patients. Tumor Biol. 2017, 39, 1010428317694552.
- Kattan, Z.; Marchal, S.; Brunner, E.; Ramacci, C.; Leroux, A.; Merlin, J.L.; Domenjoud, L.; Dauça, M.; Becuwe, P. Damaged DNA binding protein 2 plays a role in breast cancer cell growth. PLoS ONE 2008, 3, e2002.
- Becuwe, P.; Ennen, M.; Klotz, R.; Barbieux, C.; Grandemange, S. Manganese superoxide dismutase in breast cancer: From molecular mechanisms of gene regulation to biological and clinical significance. Free Radic. Biol. Med. 2014, 77, 139–151.
- Zhao, R.; Cui, T.; Han, C.; Zhang, X.; He, J.; Srivastava, A.K.; Yu, J.; Wani, A.A.; Wang, Q.E. DDB2 modulates TGF-β signal transduction in human ovarian cancer cells by downregulating NEDD4L. Nucleic Acids Res. 2015, 43, 7838–7849.
- Chang, S.W.; Su, C.H.; Chen, H.H.; Huang, C.W.; Tsao, L.P.; Tsao, Y.; Chen, S.L. DDB2 is a novel AR interacting protein and mediates AR ubiquitination/degradation. Int. J. Biochem. Cell Biol. 2012, 44, 1952–1961.
- Huang, S.; Fantini, D.; Merrill, B.J.; Bagchi, S.; Guzman, G.; Raychaudhuri, P. DDB2 Is a Novel Regulator of Wnt Signaling in Colon Cancer. Cancer Res. 2017, 77, 6562–6575.
- Cui, T.; Srivastava, A.K.; Han, C.; Wu, D.; Wani, N.; Liu, L.; Gao, Z.; Qu, M.; Zou, N.; Zhang, X.; et al. DDB2 represses ovarian cancer cell dedifferentiation by suppressing ALDH1A1. Cell Death Dis. 2018, 9, 561.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
799
Revisions:
2 times
(View History)
Update Date:
22 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




