2.1. The Importance of Myo- to D-chiro-Inositol Ratio in Steroidogenesis
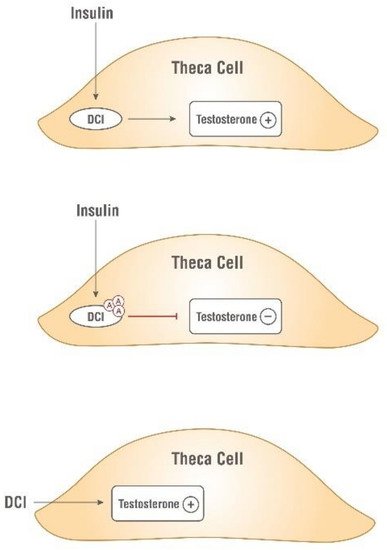
As research progresses, it is becoming clearer that, besides constituting the intracellular second messengers of insulin signaling, inositols also function as endocrine modulators, influencing steroidogenesis. In 1998 Nestler first observed that D-chiro-Ins increased testosterone levels in theca cells from women with PCOS, even though the mechanism underlying this effect remained unknown
[25]. Recently, new experiments suggested that D-chiro-Ins directly regulates the gene expression of enzymes involved in steroidogenesis in human granulosa cells, dose-dependently reducing the expression of both aromatase and cytochrome P450 side-chain cleavage genes
[29]. Therefore, D-chiro-Ins modulates estrogen levels without completely blocking their biosynthesis.
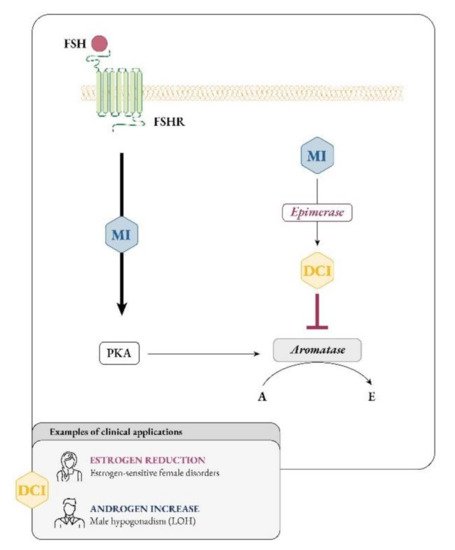
On the other hand, evidence on the effect of myo-Ins is still needed and we speculate that this isomer may affect aromatase activity in an opposite manner from D-chiro-Ins. In support to this idea, myo-Ins is involved in modulating FSH signaling, and FSH stimulates aromatase synthesis, a fundamental step for conversion of androgens to estrogens and for follicle maturation
[30]. FSH downregulation and the subsequent decrease in aromatase synthetized by granulosa cells constitute a hallmark of PCOS
[31]. Thus, while D-chiro-Ins inhibits aromatase, myo-Ins seems to enhance aromatase synthesis in granulosa cells
[32][33]. Moreover, myo-Ins could modulate ovarian steroidogenesis by rearranging cytoskeletal proteins
[34].
In this regard, higher myo-Ins/D-chiro-Ins ratios should increase aromatase activity in granulosa, fostering estrogen biosynthesis, while lower myo-Ins/D-chiro-Ins ratios promote androgen production in thecal cells
[35].
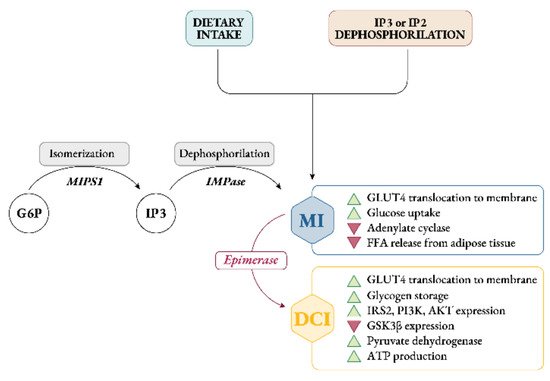
As a matter of fact, under normal homeostatic conditions, ovarian myo-Ins/D-chiro-Ins ratio ranges from 70:1 to 100:1, while in PCOS women this ratio decreased pathologically in favor of D-chiro-Ins
[21][36]. The increased D-chiro-Ins concentration promotes androgen synthesis, while myo-Ins depletion worsens the energy state of the oocytes, impairing FSH signaling and oocyte quality
[27].
These results were confirmed in a clinical trial by Nordio et al.
[37]. In this study fifty-six patients with PCOS were supplemented with different myo-Ins/D-chiro-Ins ratios (namely 0:1; 1:3.5; 2.5:1; 5:1; 20:1; 40:1 and 80:1) with the aim to restore ovulatory function, as evidenced by progesterone assay, and ameliorate abnormalities in metabolic parameters, like FSH, LH, SHBG, E2, free testosterone, HOMA index and basal and postprandial insulin. The authors observed that the 40:1 combination produced the most significant improvements, followed by the 20:1 and 80:1, while the other ratios showed less relevant outcomes. Despite these promising results, of course, future studies are mandatory to shed light on the molecular aspects of ovarian inositol activity and to investigate the beneficial effects of an ideal myo-Ins/D-chiro-Ins formulation in larger cohorts of patients, possibly with different PCOS phenotypes.
2.3. D-chiro-Inositol Dual Effects (Reduction in Estrogens in Estrogen-Sensitive Female Disorders; Increase in Androgens in Male Hypogonadism)
The discovery that D-chiro-Ins modulates the expression of the aromatase enzyme, influencing steroidogenesis and consequently the androgen/estrogen balance
[29], led to the consideration that D-chiro-Ins supplementation may be a valid approach for male and female clinical conditions that would benefit from androgen increase and/or estrogen decrease
[32][41] (
Figure 4).
Interestingly, the study by Monastra et al.
[42] showed that supplementation with 1 g/day D-chiro-Ins for 1 month to male volunteers with altered glycemia and/or hormonal status reduced serum estrone and estradiol levels (−85.0% and −14.4% respectively) and increased testosterone and dehydroepiandrosterone (+23.4% and +13.8% respectively). Besides normalizing the hormonal balance, the treatment with D-chiro-Ins decreased glycemia, insulinemia and HOMA index as well.
These results seem to confirm that D-chiro-Ins functions as an aromatase down-modulator, clearly opening new perspectives of research and therapeutic applications with this inositol.
Within the male population a potential target for D-chiro-Ins administration might be elderly men suffering from late-onset hypogonadism (LOH), who present with an impaired production of adequate levels of testosterone by testis and sperm cells, resulting in androgen deficiency
[43][44]. Decrease in sexual activity, loss of body hair, subfertility and erectile dysfunction represent the main symptoms that these patients experience, followed by an inescapable decline in their quality of life.
Testosterone replacement therapy (TRT) is widely used for the treatment of LOH, although there is still an extensive debate whether it should be administered to men with this condition
[43].
Importantly, in 2015, the FDA issued a warning about potential cardiovascular risks resulting from TRT. In addition, another concern derives from the fact that exogenous testosterone can suppress the hypothalamic-pituitary-gonadal axis through negative feedback, and TRT may lead to secondary spermatogenic failure and subsequent infertility
[45].
Since they normalize testosterone to estradiol ratio and improve sperm concentration, motility and morphology, aromatase inhibitors (AIs) and selective estrogen receptor modulators (SERMs) could represent off-label options to TRT, especially for obese individuals or those at high risk of TRT
[46]. However, these medications have not yet been established as common clinical practice.
Based on these premises and due to its proven safety
[47], D-chiro-Ins represents a valuable alternative approach for these patients.
As such, encouraging results have been obtained from a recent pilot study by Nordio and colleagues. The authors reported that, after 1 month of a daily supplementation with 1800 mg D-chiro-Ins, 10 patients showed significantly increased serum testosterone and androstenedione levels. Conversely, estradiol and estrone levels were reduced, thereby providing an important demonstration that D-chiro-Ins behaved as a molecule that could affect aromatase activity. Furthermore, the treatment with D-chiro-Ins positively impacted on insulin resistance and waist circumference, improving patients’ sexual performance and physical strength
[48].
Clearly, further studies with larger cohorts of patients should be encouraged in order to confirm these exciting data but such promising results should not be overlooked.
Notably, estrogen deficiency predisposes males to increased adiposity and metabolic dysfunction. Paradoxically, however, obesity in men has been associated with hyperestrogenism. Moreover, excessive estradiol stimulation has been postulated to play an exacerbating role in the progression of obesity and metabolic dysregulation
[49]. Obese men are often characterized by low circulating androgens but elevated levels of circulating estrone and 17β-estradiol
[50][51]. The reasons for this co-occurrence of obesity with hyperestrogenemia in men are not well defined but may include polymorphism in the aromatase gene CYP19A
[52]. It has been proposed that increased peripheral aromatization of testosterone in obese men may enhance central estradiol signaling, suppressing gonadotropin production and contributing to a sustained state of hypogonadotropic hypogonadism
[53].
From these observations, it becomes clear that obese men would greatly benefit from D-chiro-Ins supplementation, since this inositol optimizes glucose metabolism, reduces plasma insulin levels, and concomitantly, by modulating aromatase activity, promotes androgen production.
Additionally, in women, decreasing estrogen levels represents an interesting therapeutic target, particularly in the clinical management of uterine leiomyomas, also called fibroids. Indeed, estrogens, as well as progesterone, play a pivotal role in promoting cell proliferation and growth of myomas, that represent the most frequent form of benign neoplasia affecting female reproductive organs
[54]. Although leiomyomas are mostly asymptomatic and tend to resolve spontaneously after menopause
[55], some women may experiment severe symptoms, such as pelvic pain, dysmenorrhea and bleeding.
Unfortunately, up-to-date specific and safe treatments for myomas are still lacking, especially after ulipristal acetate (UPA) was withdrawn from the market in September 2020 due to the rare but serious side effect of liver failure
[56].
On the other hand, off-label pharmacological treatments including progestogens, androgens, estrogen receptors antagonists, selective progesterone receptor modulators (SPRM) and gonadotropin-releasing hormone agonists (GnRHa) have been proven effective in reducing tumor size and symptoms before surgery or, possibly, to completely avoid the surgical procedure.
Furthermore, it is noteworthy that neoplastic tissues express high levels of the aromatase enzyme compared with the lower expression observed in non-neoplastic (normal) tissues
[57]. In accordance, several groups reported that aromatase is significantly overexpressed in myoma cells compared with the adjacent normal myometrium
[58][59], leading to a high level of in situ estrogens that may contribute to the growth advantage of myomas through an intracrine/autocrine mechanism
[60]. Therefore, lowering estrogen levels could be clearly considered a valid therapeutic approach for fibroid management
[61]. Indeed, aromatase inhibitors, like Letrozole, proved to be effective in reducing myoma size and volume, ameliorating patient symptoms and improving quality of life
[62].
Since D-chiro-Ins affects aromatase expression, it can be a promising clinical option for managing uterine myomas. Montanino Oliva reported the case of two patients with leiomyomas associated with heavy menstrual bleeding, who sought pregnancy through ART
[63]. These women were supplemented daily with a combination of Epigallocatechin gallate (EGCG), vitamin D and low dose of D-chiro-Ins for 3 months.
Several studies
[64][65][66] had indeed demonstrated that natural compounds, like EGCG and vitamin D were effective in reducing myoma size and, in this contest, D-chiro-Ins may improve their efficacy in arresting myoma cell growth. Montanino Oliva observed a reduction in the fibroid volume of 73.8% and 68.4%. Moreover, a decrease in menstrual blood loss was recorded (−42.1% and −48.7%). Interestingly, 3 months after the end of the treatment, both patients underwent the ART procedure without the need for surgical intervention.
These results suggested a potentially high efficacy of this combination in reducing fibroid volume and menstrual bleeding, so that surgery could be avoided. D-chiro-Ins action on modulating aromatase activity might help to explain the important volume reduction reported in both cases, and it is tempting to speculate that vitamin D and EGCG exerted their anti-proliferative and proapoptotic effects, enhanced by the activity of D-chiro-Ins.
Certainly, these outstanding results need further confirmation on a larger sample of women.
Down-modulation in the expression of the aromatase enzyme by D-chiro-Ins, similar to the action of Letrozole, can be considered a valid approach to restore ovulation. However, because of its activity on aromatase and the subsequent androgen-rising effect, the baseline clinical condition of patients as well as the duration of supplementation should be carefully evaluated before starting therapy with D-chiro-Ins, in order to avoid a negative impact on the ovaries.
When hyperinsulinemic anovulatory women are supplemented with D-chiro-Ins, it restores ovulation, mainly functioning as an insulin-sensitizer by improving insulin signaling and decreasing the systemic hyperinsulinemia accordingly. Unfortunately, results on non-hyperinsulinemic patients are still lacking in the literature.
Based on this finding and considering that the examined cases were normo-insulinemic, it is unlikely that insulin regulation played a pivotal part in restoring ovulatory function. The authors rather concluded that in such specific cases D-chiro-Ins probably acted mainly on aromatase expression, impairing estrogen biosynthesis and contributing to FSH release.
Hopefully, controlled studies with an appropriate sample of patients will be able to confirm this preliminary observation.
2.5. The Consequences of Inositol Deficiency Due to Pharmacological Treatments, Malabsorption or Competition with Dietary Glucose
It should also be stressed that, when high doses of D-chiro-Ins or other sugar-like molecules are supplemented concomitantly with myo-Ins, they seem to negatively impact on its availability in the human body. As a matter of fact, Garzon et al. observed that D-chiro-Ins, sorbitol and maltodextrin, when provided together with myo-Ins, significantly decreased its absorption and plasma concentration, compared to when myo-Ins was supplemented alone
[7]. As a possible explanation, the authors postulated a competitive action among these molecules at inositol transporters, mainly SMIT2. Indeed, this protein exhibits similar affinity for both inositol stereoisomers, as demonstrated by the K
m values (120–150 µM for myo-Ins and 110–130 µM for D-chiro-Ins). However, considering that under physiological conditions the serum concentration of D-chiro-Ins is less than 100 nM, it is unlikely that it can interfere with myo-Ins absorption, since serum concentrations of myo-Ins typically range from 26.8 to 43.0 µM, which is significantly higher
[67][68]. The same shall apply to the formulations in which myo-Ins and D-chiro-Ins are combined in a proper ratio, such as the 40:1 formula.
On the contrary, when D-chiro-Ins or other sugars from food are consumed at high dosage (≥1 g) and/or concomitantly with myo-Ins, a strong competition for SMIT2 transporters may occur, decreasing myo-Ins absorption in the gut and consequently altering plasma myo-Ins/D-chiro-Ins ratio that possibly accounts for pathological conditions.
In addition, it should not be underestimated that some pharmacological treatments, particularly antiepileptic drugs (AEDs), such as sodium valproate (VA), and mood stabilizers for bipolar disorder, such as lithium (Li
+), can cause myo-Ins depletion. All of these medications share the “inositol-depletion hypothesis”, put forward to explain their therapeutic mechanism
[69]. According to this theory, Li
+ acts principally by inhibiting the monophosphatase (IMPase) and the inositol polyphosphatase (IPP), key enzymes in inositol synthesis, while VA inhibits the myoinositol-phosphate synthase (MIPS).
Although these drugs superficially appear beneficial to patients, since they are able to control seizures and mood disbalances, their chronic administration is not completely safe and may expose patients to side effects mostly associated with inositol depletion in peripheral tissues. Among these effects are hypothyroidism, weight gain, hyperinsulinemia, dyslipidemia, impairment of kidney function and adverse dermatological effects that are quite commonly reported
[70][71][72]. Noteworthy, PCOS is one of the most serious side-effects reported by AEDs-treated women
[72][73][74][75], who experience reduced estradiol and progesterone and increased testosterone, leading to hypogonadism, amenorrhea or oligomenorrhea, along with sexual dysfunction and infertility
[76].
Importantly, even though these conditions may sometimes spontaneously resolve after a few weeks of treatment, or revert to baseline with drug discontinuation, they certainly worsen patients’ quality of life, weakening their compliance to therapeutic protocols.
Several scientific studies have documented myo-Ins supplementation as a safe and efficient approach in treating PCOS symptoms, improving hormonal profile, hyperandrogenism, menstrual cycle, oocyte quality and psychological disturbances
[77][78]. Moreover, myo-Ins in combination with Selenium, has been reported to be effective in restoring euthyroidism in patients with subclinical hypothyroidism or autoimmune thyroiditis
[79]. Therefore, supplementing with this inositol could be a valid option to counteract the peripherical side effects connected to bipolar disorder and epileptic treatment. Clearly, the dose of myo-Ins supplemented should be properly settled so as not to increase its levels in the brain and interfere with the central pharmacological therapeutic effect.
The possible ratio between myo-Ins and D-chiro-Ins may physiologically range from 10:1 to 100:1 and the ratio 80:1 in favor of myo-Ins is particularly encouraged
[72].
From these premises, the concomitant supplementation with inositol to patients in therapy with Li+ or AEDs could represent an intriguing proposal. Interestingly, this combined treatment would safely reduce the peripheral side effects, ameliorating patients’ compliance and improving quality of life.