
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Piyush Kumar Gupta | + 2168 word(s) | 2168 | 2021-10-08 11:19:43 | | | |
| 2 | Vivi Li | Meta information modification | 2168 | 2021-10-19 11:34:32 | | |
Video Upload Options
Gestational diabetes mellitus (GDM) is the most frequent complication during pregnancy. This complex disease is characterized by glucose intolerance and consequent hyperglycemia that begins or is first diagnosed in pregnancy, and affects almost 7% of pregnant women. Previous reports have shown that GDM is associated with increased pregnancy complications and might cause abnormal fetal development. At present, treatments are not suitable for the prevention and management of these patients. As an alternative therapeutic opportunity and a leading scientific technique, nanotechnology has helped enlighten the health of these affected women. Theranostic nanomaterials with unique properties and small sizes (at least <100 nm in one of their dimensions) have been recently engineered for clinics and pharmaceutics. Reducing materials to the nanoscale has successfully changed their properties and enabled them to uniquely interact with cell biomolecules. Several biosensing methods have been developed to monitor glucose levels in GDM patients.
1. Introduction
2. Nanotechnology for Treatment of GDM
2.1. Use of Metallic NPs for Treatment of GDM
2.2. Use of Polymeric NPs for GDM Treatment
References
- Glovaci, D.; Fan, W.; Wong, N.D. Epidemiology of diabetes mellitus and cardiovascular disease. Curr. Cardiol. Rep. 2019, 21, 1–8.
- Zimmet, P.; Alberti, K.G.; Magliano, D.J.; Bennett, P.H. Diabetes mellitus statistics on prevalence and mortality: Facts and fallacies. Nat. Rev. Endocrinol. 2016, 12, 616–622.
- Seshiah, V.; Balaji, V.; Balaji, M.S.; Sanjeevi, C.; Green, A. Gestational diabetes mellitus in India. Japi 2004, 52, 707–711.
- Mumtaz, M. Gestational diabetes mellitus. Malays. J. Med. Sci. 2000, 7, 4–9.
- Catalano, P.M.; McIntyre, H.D.; Cruickshank, J.K.; McCance, D.R.; Dyer, A.R.; Metzger, B.E.; Lowe, L.P.; Trimble, E.R.; Coustan, D.R.; Hadden, D.R. The hyperglycemia and adverse pregnancy outcome study: Associations of GDM and obesity with pregnancy outcomes. Diabetes Care 2012, 35, 780–786.
- American Diabetes Association. Gestational diabetes mellitus. Diabetes Care 2003, 26, S103–S105.
- Metzger, B.E.; Contreras, M.; Sacks, D.; Watson, W.; Dooley, S.; Foderaro, M.; Niznik, C.; Bjaloncik, J.; Catalano, P.; Dierker, L. Hyperglycemia and adverse pregnancy outcomes. N. Engl. J. Med. 2008, 358, 1991–2002.
- Johns, E.C.; Denison, F.C.; Norman, J.E.; Reynolds, R.M. Gestational diabetes mellitus: Mechanisms, treatment, and complications. Trends Endocrinol. Metab. 2018, 29, 743–754.
- Ferrara, A. Increasing prevalence of gestational diabetes mellitus: A public health perspective. Diabetes Care 2007, 30, S141–S146.
- Mahalakshmi, M.M.; Bhavadharini, B.; Kumar Maheswari, R.M.A.; Jebarani, S.; Ninov, L.; Kayal, A.; Malanda, B.; Belton, A.; Uma, R.; Mohan, V. Current practices in the diagnosis and management of gestational diabetes mellitus in India (WINGS-5). Indian J. Endocrinol. Metab. 2016, 20, 364.
- Nelson, R.L. Oral glucose tolerance test: Indications and limitations. In Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 1988; pp. 263–269.
- Agarwal, M.M. Gestational diabetes mellitus: An update on the current international diagnostic criteria. World J. Diabetes 2015, 6, 782.
- Advertising Association. Management of diabetes in pregnancy: Standards of Medical Care in Diabetes—2018. Diabetes Care 2018, 41, S137–S143.
- Rowan, J.A.; Hague, W.M.; Gao, W.; Battin, M.R.; Moore, M.P. Metformin versus insulin for the treatment of gestational diabetes. N. Engl. J. Med. 2008, 358, 2003–2015.
- Eades, C.E.; Cameron, D.M.; Evans, J.M. Prevalence of gestational diabetes mellitus in Europe: A meta-analysis. Diabetes Res. Clin. Pract. 2017, 129, 173–181.
- Moumaris, M.; Bretagne, J.-M.; Abuaf, N. Nanomedical Devices and Cancer Theranostics. Open Nanomed. Nanotechnol. J. 2020, 6, 1–11.
- Ates, B.; Koytepe, S.; Ulu, A.; Gurses, C.; Thakur, V.K. Chemistry, Structures, and Advanced Applications of Nanocomposites from Biorenewable Resources. Chem. Rev. 2020, 120, 9304–9362.
- Shakeri-Zadeh, A.; Zareyi, H.; Sheervalilou, R.; Laurent, S.; Ghaznavi, H.; Samadian, H. Gold nanoparticle-mediated bubbles in cancer nanotechnology. J. Control. Release 2020, 330, 49–60.
- Shakeri, S.; Ashrafizadeh, M.; Zarrabi, A.; Roghanian, R.; Afshar, E.G.; Pardakhty, A.; Mohammadinejad, R.; Kumar, A.; Thakur, V.K. Multifunctional Polymeric Nanoplatforms for Brain Diseases Diagnosis, Therapy and Theranostics. Biomedicines 2020, 8, 13.
- Cash, K.J.; Clark, H.A. Nanosensors and nanomaterials for monitoring glucose in diabetes. Trends Mol. Med. 2010, 16, 584–593.
- Balasubramaniam, B.; Prateek; Ranjan, S.; Saraf, M.; Kar, P.; Singh, S.P.; Thakur, V.K.; Singh, A.; Gupta, R.K. Antibacterial and Antiviral Functional Materials: Chemistry and Biological Activity toward Tackling COVID-19-like Pandemics. ACS Pharmacol. Transl. Sci. 2021, 4, 8–54.
- Miri, A.; Beiki, H.; Najafidoust, A.; Khatami, M.; Sarani, M. Cerium oxide nanoparticles: Green synthesis using Banana peel, cytotoxic effect, UV protection and their photocatalytic activity. Bioprocess Biosyst. Eng. 2021, 44, 1891–1899.
- Miri, A.; Sarani, M.; Khatami, M. Nickel-doped cerium oxide nanoparticles: Biosynthesis, cytotoxicity and UV protection studies. RSC Adv. 2020, 10, 3967–3977.
- Nazaripour, E.; Mousazadeh, F.; Moghadam, M.D.; Najafi, K.; Borhani, F.; Sarani, M.; Ghasemi, M.; Rahdar, A.; Iravani, S.; Khatami, M. Biosynthesis of lead oxide and cerium oxide nanoparticles and their cytotoxic activities against colon cancer cell line. Inorg. Chem. Commun. 2021, 131, 108800.
- Sharma, D.; Singh, J. Long-term glycemic control and prevention of diabetes complications in vivo using oleic acid-grafted-chitosan-zinc-insulin complexes incorporated in thermosensitive copolymer. J. Control. Release 2020, 323, 161–178.
- Saï, P.; Damgé, C.; Rivereau, A.; Hoeltzel, A.; Gouin, E. Prophylactic Oral Administration of Metabolically Active Insulin Entrapped in Isobutylcyanoacrylate Nanocapsules Reduces the Incidence of Diabetes inNonobese DiabeticMice. J. Autoimmun. 1996, 9, 713–721.
- Sharma, B.; Thakur, S.; Mamba, G.; Prateek; Gupta, R.K.; Gupta, V.K.; Thakur, V.K. Titania Modified Gum Tragacanth Based Hydrogel Nanocomposite for Water Remediation. J. Environ. Chem. Eng. 2021, 9, 104608.
- Shalaby, T.I.; El-Refaie, W.M. Bioadhesive chitosan-coated cationic nanoliposomes with improved insulin encapsulation and prolonged oral hypoglycemic effect in diabetic mice. J. Pharm. Sci. 2018, 107, 2136–2143.
- Narjinary, M.; Rana, P.; Sen, A.; Pal, M. Enhanced and selective acetone sensing properties of SnO2-MWCNT nanocomposites: Promising materials for diabetes sensor. Mater. Des. 2017, 115, 158–164.
- Zhang, Y.; Zhang, P.; Gao, X.; Chang, L.; Chen, Z.; Mei, X. Preparation of exosomes encapsulated nanohydrogel for accelerating wound healing of diabetic rats by promoting angiogenesis. Mater. Sci. Eng. C 2021, 120, 111671.
- Sona, P. Nanoparticulate drug delivery systems for the treatment of diabetes. Dig. J. Nanomater. Biostruct. (DJNB) 2010, 5, 411–418.
- Kerry, R.G.; Mahapatra, G.P.; Maurya, G.K.; Patra, S.; Mahari, S.; Das, G.; Patra, J.K.; Sahoo, S. Molecular prospect of type-2 diabetes: Nanotechnology based diagnostics and therapeutic intervention. Rev. Endocr. Metab. Disord. 2021, 22, 421–451.
- Jeffery, C.J. Engineering periplasmic ligand binding proteins as glucose nanosensors. Nano Rev. 2011, 2, 5743.
- Wang, L.; Yun, X.; Stanacevic, M.; Gouma, P. An Acetone Nanosensor For Non-invasive Diabetes Detection. In Aip Conference Proceedings; American Institute of Physics: College Park, MD, USA, 2009; pp. 206–208.
- Le, L.V.; Chendke, G.S.; Gamsey, S.; Wisniewski, N.; Desai, T.A. Near-infrared optical nanosensors for continuous detection of glucose. J. Diabetes Sci. Technol. 2020, 14, 204–211.
- Zhang, W.; Zhang, H.; Wang, M.; Li, P.; Ding, C.; Zhang, W.; Wang, H.; Tang, B. Copolymer-Based Fluorescence Nanosensor for In Situ Imaging of Homocysteine in the Liver and Kidney of Diabetic Mice. Anal. Chem. 2020, 92, 16221–16228.
- Huang, C.; Hao, Z.; Qi, T.; Pan, Y.; Zhao, X. An integrated flexible and reusable graphene field effect transistor nanosensor for monitoring glucose. J. Mater. 2020, 6, 308–314.
- Wahab, A.W.; Karim, A.; La Nafie, N.; Sutapa, I.W. Synthesis of silver nanoparticles using muntingia calabura L. Leaf extract as bioreductor and applied as glucose nanosensor. Orient. J. Chem. 2018, 34, 3088.
- Chaturvedi, V.K.; Singh, A.; Singh, V.K.; Singh, M.P. Cancer nanotechnology: A new revolution for cancer diagnosis and therapy. Curr. Drug Metab. 2019, 20, 416–429.
- Eivazzadeh-Keihan, R.; Pashazadeh-Panahi, P.; Baradaran, B.; Maleki, A.; Hejazi, M.; Mokhtarzadeh, A.; de la Guardia, M. Recent advances on nanomaterial based electrochemical and optical aptasensors for detection of cancer biomarkers. TrAC Trends Anal. Chem. 2018, 100, 103–115.
- Buchanan, T.A.; Xiang, A.; Kjos, S.L.; Watanabe, R. What is gestational diabetes? Diabetes Care 2007, 30, S105–S111.
- Sheik, R. Assessment of Major Complications in Pregnancy: A Single Center Study. Asian J. Pharm. Res. Dev. 2017, 5, 1–6.
- Xu, C.; Qu, X. Cerium oxide nanoparticle: A remarkably versatile rare earth nanomaterial for biological applications. N.P.G. Asia Mater. 2014, 6, e90.
- Korsvik, C.; Patil, S.; Seal, S.; Self, W.T. Superoxide dismutase mimetic properties exhibited by vacancy engineered ceria nanoparticles. Chem. Commun. 2007, 1056–1058.
- Heckert, E.G.; Karakoti, A.S.; Seal, S.; Self, W.T. The role of cerium redox state in the SOD mimetic activity of nanoceria. Biomaterials 2008, 29, 2705–2709.
- Silva, G.A. Seeing the benefits of ceria. Nat. Nanotechnol. 2006, 1, 92–94.
- Vafaeipour, Z.; Shokrzadeh, M.; Jahani, M.; Shaki, F. Protective Effect of nanoceria against streptozotocin induced mitochondrial dysfunction in embryo of diabetic mice. J. Maz. Univ. Med. Sci. 2015, 25, 109–120.
- Najafi, R.; Hosseini, A.; Ghaznavi, H.; Mehrzadi, S.; Sharifi, A.M. Neuroprotective effect of cerium oxide nanoparticles in a rat model of experimental diabetic neuropathy. Brain Res. Bull. 2017, 131, 117–122.
- Guan, B.; Yan, R.; Li, R.; Zhang, X. Selenium as a pleiotropic agent for medical discovery and drug delivery. Int. J. Nanomed. 2018, 13, 7473.
- Rezaei-Kelishadi, M.; Ghasemi, A.; Abdolyosefi, N.N.; Zamani-Doabi, S.; Ramezani, M.; Changizi-Ashtiyani, S.; Rahimi, A. Effects of selenium nanoparticles on kidney and liver functional disorders in streptozotocin-induced diabetic rats. Physiol. Pharmacol. 2017, 21, 155–162.
- Thakur, V.K.; Thakur, M.K. Recent Advances in Graft Copolymerization and Applications of Chitosan: A Review. ACS Sustain. Chem. Eng. 2014, 2, 2637–2652.
- Hassan, I.; Ebaid, H.; Al-Tamimi, J.; Habila, M.A.; Alhazza, I.M.; Rady, A.M. Selenium nanoparticles mitigate diabetic nephropathy and pancreatopathy in rat offspring via inhibition of oxidative stress. J. King Saud Univ.-Sci. 2021, 33, 101265.
- Wang, Y.; Cheng, S.; Fan, W.; Jiang, Y.; Yang, J.; Tong, Z.; Jiang, G. Dual responsive block copolymer coated hollow mesoporous silica nanoparticles for glucose-mediated transcutaneous drug delivery. Chin. J. Chem. Eng. 2021, in press.
- Dave, P.N.; Gor, A. Natural polysaccharide-based hydrogels and nanomaterials: Recent trends and their applications. Handb. Nanomater. Ind. Appl. 2018, 36–66.
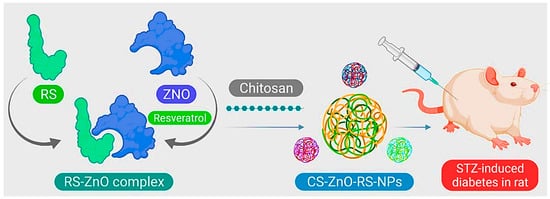
- Du, S.; Lv, Y.; Li, N.; Huang, X.; Liu, X.; Li, H.; Wang, C.; Jia, Y.-F. Biological investigations on therapeutic effect of chitosan encapsulated nano resveratrol against gestational diabetes mellitus rats induced by streptozotocin. Drug Deliv. 2020, 27, 953–963.
- Cheng, X.; Xu, Y.; Jia, Q.; Guo, N.; Wang, Z.; Wang, Y. Novel greener approached synthesis of polyacrylic nanoparticles for therapy and care of gestational diabetes. Drug Deliv. 2020, 27, 1263–1270.
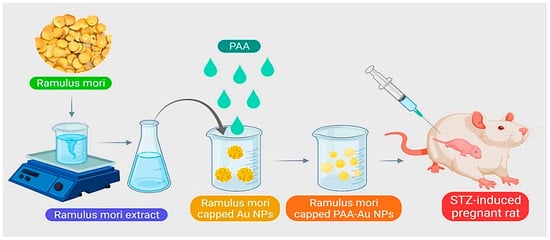
- An, Y.; Guo, Y.; Wu, H.; Huang, Y.; Xie, D.; Qin, J. Conceivable Protective Role of Murraya koenigii Leaf Extract Loaded Poly (D, L-lactic-co-glycolic acid)-Gold-Nanoparticles on the Gestational Diabetes Mellitus of Rats Induced by Streptozotocin. Sci. Adv. Mater. 2020, 12, 87–92.




