
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maria Lurdes Inácio | + 2281 word(s) | 2281 | 2021-09-27 08:21:32 | | | |
| 2 | Nora Tang | + 867 word(s) | 3148 | 2021-10-20 10:27:36 | | |
Video Upload Options
Pine wilt disease (PWD) is a complex disease that severely affects the biodiversity and economy of Eurasian coniferous forests. Three factors are described as the main elements of the disease: the pinewood nematode (PWN) Bursaphelenchus xylophilus, the insect-vector Monochamus spp., and the host tree, mainly Pinus spp. Nonetheless, other microbial interactors have also been considered.
1. Introduction
Pine wilt disease (PWD) has become one of the most damaging diseases to conifers worldwide and is a risk nowadays for the sustainability and profitability of forest ecosystems. PWD was detected in Japan in 1905, later spreading to other Asian countries; namely China, Korea, and Taiwan in the 1980s [1], and to Europe (Portugal and Spain in 1999 and 2010, respectively) [2][3][4]. To date, no recorded outbreaks have been identified in other European countries, despite several scientific studies alert for a high vulnerability of northern European pine forests due to the oncoming effects of climate change and the reported susceptibility of the dominating pine species to PWD [5][6]. The PWD has gradually and consistently spread between continents as a result of an increase in the global trade of wood and derivative materials. Significant economic and ecological impacts were reported in the affected countries, including a reduction in productivity and an increase in the costs of management procedures for disease control, as well as a decrease in forest biodiversity [7][8].
Effective PWD management strategies have been difficult to achieve given the complex disease infection cycle, where several organisms contribute to the infection’s overall development and severity, namely its causal agent, the pinewood nematode (PWN) Bursaphelenchus xylophilus ( Steiner & Bührer) Nickle; the PWN’s insect-vector Monochamus spp. ; and a susceptible host tree, commonly trees from the genus Pinus [1]. Recent studies have additionally identified a strong influence of the PWN-associated bacteria, and the microbiome associated with the susceptible pine, on PWD development [9][10][11]. While the description of microbial diversity has been established for the PWN, the insect-vector, and susceptible pine hosts—leading to the preliminary proposal of their functional roles in the PWD—information on the mycological diversity associated with these organisms is scarcer. In the present review, an up-to-date compilation of the published works reporting on naturally occurring fungi associated with the PWN, the host tree, and its insect-vector, is presented. A critical analysis of the summarized information further allows us to envision the functional role of associated fungi on PWD development and guide future research in this area.
2. The Complexity of PWD
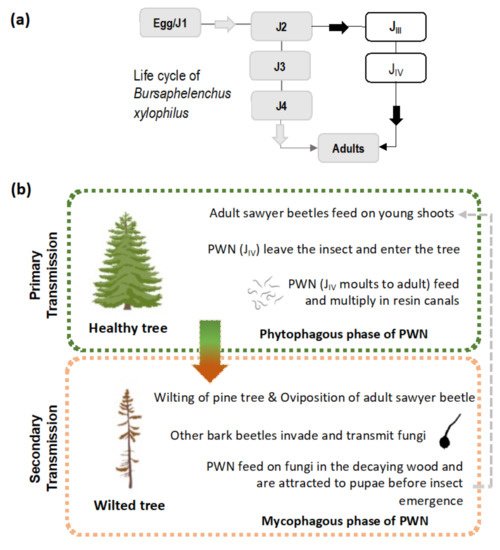
The PWN is a small plant-parasitic nematode of about 500–1000 µm in length and 22 µm in width, capable of feeding on plant (phytophagous) and fungal tissues (mycophagous). Its life cycle is comprised of four juvenile stages (J1 to J4) and both adult males and females ( Figure 1 a). In natural conditions, the PWN can quickly complete its life cycle, usually 4 days in summer conditions while, under in vitro conditions in laboratory cultured Botrytis cinerea Pers. mats, it can take between 4 to 6 days [1][12] (or slightly longer on aseptic co-cultures of in vitro pine with the PWN) [13]. Adverse environmental conditions and/or undernutrition induce morphological and physiological changes in the J3 stage, leading to the occurrence of the PWN dispersal stages, the third and fourth dispersal juvenile stages (J III and J IV ). The J IV , known as the dauer juvenile stage, is characterized by an interruption of the feeding process, the establishment of large lipidic reserves, and the production of a thick protective layer around the PWN’s body [1][14]. At this stage, the PWN is attracted to the juvenile longhorn beetles emerging from dead, or decaying, wood in forest ecosystems, invading beetles trachea. The PWN establishes these commensalism associations with members of the genus Monochamus (Coleoptera; Cerambycidae), considered the main PWN vector, with seven confirmed species vectoring the nematode in field and laboratory conditions [15][16][17], the most common being M. alternatus in East Asian forestlands [1], and M. galloprovincialis in Europe [18][19]. Colonized beetles can transmit the PWN by transporting them to (1) new beetle feeding sites during the beetle maturation stage, commonly in young branches of susceptible pine species (primary transmission), or (2) weakened or dead trees in the forest, where the matured female beetles oviposit (secondary transmission). The beetles benefit from an increase in reproduction sites at weakened or dead trees, where oviposition occurs, while the PWN takes advantage of the increase in beetle population, leading to higher rates of transmission ( Figure 1 b) [20]. Once infection occurs in a susceptible tree, the PWN quickly multiplies and begins feeding on the epithelial parenchyma cells lining the pine resin ducts, inducing extensive damage that leads to a reduction in resin production and the release of volatile terpenoids. As the infection progresses, the PWNs damage the tree’s vascular system and an embolism phenomenon begins to interrupt water transport, partly due to the build-up of volatile terpenoids [20][21]. The typical symptoms of PWD ( Figure 2 ) are visible at this stage, namely pine shoot wilting due to desiccation, chlorosis, and drooping. When external symptoms of the PWD are noticeable, the affected tree cannot recover and eventually dies. However, the symptoms caused by the PWN, including abrupt reduction in the production of resin and pine needle wilting, can be mistakenly attributed to other biotic or abiotic stress factors such as water stress [22]. In North America, endemic pines are mostly tolerant, with only exotic species expressing the strongest symptoms. In China, Japan, Korea, and Portugal, many pine species were found to be very susceptible, e.g., Pinus densiflora Sieblod & Zucc. , Pinus nigra J.F. Arnold, Pinus pinaster Aiton, Pinus radiata D. Don, Pinus sylvestris L. or Pinus thunbergii Parl., which caused extensive economic and cultural repercussions [15][23].

Despite not being able to induce PWD, the microbiome (bacteria and fungi) associated with the PWD, and its elements is considered a major biotic factor influencing the disease’s severity (further detailed in [9][10][11]). The isolation of fungi and bacteria dates back the 70s, when Tokushige and Kiyohara isolated microbial samples from dead pine trees and tested their pathogenicity in healthy pines [20]. Since then, several researchers attempted to correlate the pathogenicity of some bacterial species, like Pseudomonas spp. or Bacillus spp. , with the disease [24][25][26]. Still, the latest studies point to a less active role of the bacterial communities associated with PWD. These communities behave as opportunistic/saprophytes and/or endophytes expressing phenotypic plasticity in the PWN-wilted pine host [11][27][28][29][30][31]. The impact of fungal communities in the complexity of PWD and their elements is presented in detail in the next section.
3. Mycobiome Associated with PWD Complex
Forest trees harbour extremely complex fungal communities that play important roles on ecosystem multifunctionality and equilibrium [32]. Most of these communities are shaped by intrinsic factors of the host, like host genotype, condition, and/or development, and by external factors such as geographical location, seasonality, or even surrounding vegetation [33]. Under the presence of non-native pathogens, these ecosystems suffer disequilibrium situations that can eventually lead to considerable spatial and temporal community variation [34].
Fungal communities associated with PWD have been described since the early 80s. These culture-dependent studies worked with different culture conditions, such as different growth media (e.g., potato dextrose agar or malt extract agar) or supplementation of antibiotics ( Table 1 ). The most recent reports in fungal communities associated with the disease combine morphology with molecular identification based on fungal DNA barcode markers (i.e., primary marker ITS, internal transcribed spacer; and secondary marker TEF1-α, translational elongation factor 1 alpha) [35][36] and other protein-coding genes (i.e., beta-tubulin BT or calmodulin CAL) [37]. These conventional culturing methods are biased towards fast-growing species rather than the more specialized fungi [38], suggesting that only a limited fraction of the fungal community’s diversity has been uncovered in this complex disease. Three main phyla were identified within the Fungi kingdom, namely Ascomycota, Basidiomycota, and Mucoromycota. The most predominant phylum, Ascomycota, was represented by 6 classes (Blastomycetes, Dothideomycetes, Eurotiomycetes, Leotiomycetes, Orbiliomycetes, and Sordariomycetes) and nearly 30 families ( Table 1 ). Most of the described taxa are common saprophytes and probably not specific to the disease or associated with the PWN. This may be the case of Penicillium (Ascomycota, Eurotiomycetes, Trichocomaceae), Trichoderma (Ascomycota, Sordariomycetes, Hypocreaceae) and Aspergillus (Ascomycota, Eurotiomycetes, Aspergillaceae) among others, which are ubiquitous to all existing environments and detected in all PWD elements ( Table 1 ). The description of fungal communities has also been made in different pine species and insect-vectors from different geographical locations (e.g., China, Japan, Korea, Portugal, and the USA). The first isolations of mycoflora were conducted on symptomatic P. thunbergii (shoots, twigs, and woodchips), on the surface tissues of tunnels and pupal chambers bored by Monochamus larvae, and from the adult body of M. alternatus after its emergence [39][40]. These works reported that the genera Ceratocystis and Verticicladiella (synonym of Leptographium [41]), from Ceratocystidaceae and Ophiostomataceae families, respectively, were the only flora common to the three sampled locations apart from the saprophytic fungi Trichoderma and Penicillium. Later, Wingfield [42] isolated fungi from the cerambycid beetles M. scutellatus and M. carolinenses, also identifying Ceratocystis and Ceratocystiopis as common genera associated with adult beetles and pupal chambers from P. banksiana and P. resinosa. Curiously, Wingfield [42] could also isolate the nematode-trapping fungi Arthrobotrys cladodes var. cladodes and A. superba in the PWN. Kuroda and Iko [43] isolated fungi from healthy and wilted P. thunbergii and reported that the same species were recovered from both pine trees (i.e., Pestalotiopsis spp., Nigrospora spp. , Cladosporium spp. , and Phomopsis spp.) and that Ceratocystis sp., was only detected after PWN inoculation. This study has also shown that the composition of fungal species varied slightly among seasons [43]. In 2007, Hyun et al. [44] characterized the fungal communities associated with the PWN; the insect- vector M. alternatus and P. thunbergii in Korea. Among the 15 genera identified, Penicillium and Ophiostoma were the most frequent genera in all elements, with PWNs and insect larvae showing a smaller number of associated fungi than insect adults or infected wood. In 2015, Inácio et al. [45] described fungal communities associated with M. galloprovincialis, native to Portugal. From a total of 100 insects, species of 18 genera of filamentous fungi were reported: Acremonium, Alternaria, Arthrinium, Aspergillus, Beauveria, Bipolaris, Botryosphaeria, Botrytis, Cladosporium, Clonostachys, Eppicocum, Fusarium, Ophiostoma s. l., Paecilomyces s. l., Penicillium, Stemphylium, Trichoderma, and Trichothecium ( Table 1 ) [45]. A more detailed characterization of endophytic fungi associated with P. pinaster with, and without, PWN infection in Portugal was presented by Trindade [46]. Novel fungal species are also continuously described. Wang et al. [47] identified new species of Ophiostomatales associated with PWD in the pupal chambers of M. alternatus from infected Pinus massoniana Siebold & Zucc. and P. thunbergii in China. In this study, over 90% of all isolates were identified as Ophiostoma ips, with three novel species— Ophiostoma album sp. nov. , Ophiostoma massoniana sp. nov. and Sporothrix zhejiangensis sp. nov.—and two species whose identities remained unclear; Ophiostoma cf. deltoideosporum and Graphilbum cf. rectangulosporium [47].
| Fungal Taxonomy | Insect-Vector | Host Pine | Country | References | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ma | Ms | Mc | Mg | Pd | Pm | Pt | Pb | Pr | Pp | |||||||||||
| Phylum | Class | Family | Genus | PWN | A | L | A | L | A | A | W | Pc | Pc | W | Pc | Pc | Pc | W | ||
| Ascomycota | Blastomycetes | Crytococcaceae | Candida | • | JP | [48] | ||||||||||||||
| Dothideomycetes | Botryosphaeriaceae | Diplodia | • | • | JP | [39][48] | ||||||||||||||
| Botryosphaeria | • | PT | [46] | |||||||||||||||||
| Macrophoma | • | JP | [49] | |||||||||||||||||
| Sphaeropsis | • | JP | [50] | |||||||||||||||||
| Cladosporiaceae | Cladosporium | • | • | • | • | • | JP, PT | [39][43][45][46][48] | ||||||||||||
| Didymellaceae | Epicoccum | • | • | PT | [45][46] | |||||||||||||||
| Dothideaceae | Rhizosphaera | • | [39] | |||||||||||||||||
| Leptosphaeriaceae | Leptosphaeria | • | KR | [44] | ||||||||||||||||
| Massarinaceae | Helminthosporium | • | • | USA | [42] | |||||||||||||||
| Pleosporaceae | Alternaria | • | • | • | • | • | • | • | JP, PT | [39][45][46][48] | ||||||||||
| Bipolaris | • | PT | [45] | |||||||||||||||||
| Curvalaria | • | CH | [51] | |||||||||||||||||
| Stemphylium | • | PT | [45] | |||||||||||||||||
| Saccotheciaceae | Aureobasidium | • | • | • | • | JP, PT | [45][46][50] | |||||||||||||
| Eurotiomycetes | Aspergillaceae | Aspergillus | • | • | • | • | • | • | • | • | JP, CH, PT | [42][44][45][46][48][50][51] | ||||||||
| Trichocomaceae | Penicillium | • | • | • | • | • | • | • | • | • | • | • | • | JP, CH, PT | [39][44][45][46][48][49][50] | |||||
| Paecilomyces | • | • | PT | [45][46] | ||||||||||||||||
| Herpotrichiellaceae | Phialophora | • | • | • | JP | [42][48] | ||||||||||||||
| Rhinocladiella | • | • | USA | [42] | ||||||||||||||||
| Leotiomycetes | Sclerotinuaceae | Botrytis | • | • | • | CH, PT | [44][45][46] | |||||||||||||
| Orbiliomycetes | Orbiliaceae | Arthrobotrys | • | • | • | • | JP, CH, USA | [42][48][49] | ||||||||||||
| Dactylaria | • | JP | [48] | |||||||||||||||||
| Sordariomycetes | Amphisphaeriaceae | Pestalotia | • | JP | [39] | |||||||||||||||
| Aspiosporaceae | Arthrinium | • | • | PT | [45][46] | |||||||||||||||
| Bionectriaceae | Bionectria | • | KR | [44] | ||||||||||||||||
| Clonostachys | • | • | PT | [45][46] | ||||||||||||||||
| Boliniaceae | Camarops | • | KR | [44] | ||||||||||||||||
| Ceratocystidaceae | Ceratocystis | • | • | • | • | • | • | • | • | • | • | JP, CH, USA | [39][42][44][49][51] | |||||||
| Chaetomiaceae | Chaetomium | • | CH | [51] | ||||||||||||||||
| Cordycipitaceae | Beauveria | • | • | • | • | USA, PT | [42][45][46] | |||||||||||||
| Glomerellaceae | Colletotrichum | • | • | • | • | JP, CH | [39][51] | |||||||||||||
| Hypocreaceae | Hypocrea | • | • | KR | [44] | |||||||||||||||
| Cephalosporium | • | JP | [48] | |||||||||||||||||
| Gliocladium | • | • | • | JP, USA | [42][50] | |||||||||||||||
| Trichoderma | • | • | • | • | • | • | • | • | • | • | JP, CH, USA, PT | [39][42][44][45][46][48][49][50][51] | ||||||||
| Microascaceae | Graphium | • | JP | [48] | ||||||||||||||||
| Nectriaceae | Gibberella | • | KR | [44] | ||||||||||||||||
| Fusarium | • | • | • | • | • | • | JP, CH, KR, PT | [39][44][45][46][49][50][51] | ||||||||||||
| Mariannaea | • | JP | [48] | |||||||||||||||||
| Nectria | • | KR | [44] | |||||||||||||||||
| Ophiostomataceae | Ceratocystiopsis | • | • | • | • | • | • | USA, PT | [42][46] | |||||||||||
| Graphilbum | • | • | • | KR, PT | [46][47][52] | |||||||||||||||
| Leptographium | • | JP | [39][49] | |||||||||||||||||
| Ophiostoma | • | • | • | • | • | • | • | • | CH, KR, PT | [44][45][46][47][53] | ||||||||||
| Sporothrix | • | • | • | CH, PT | [46][47] | |||||||||||||||
| Plectosphaerellaceae | Plectosphaerella | • | KR | [44] | ||||||||||||||||
| Verticillum | • | • | JP | [49] | ||||||||||||||||
| Sordariaceae | Sordaria | • | CH | [51] | ||||||||||||||||
| Sporocadaceae | Monochaetia | • | CH | [51] | ||||||||||||||||
| Pestalotiopsis | • | • | • | • | JP, CH, PT | [43][46][48][49][50][51] | ||||||||||||||
| Trichosphaeriaceae | Nigrospora | • | • | JP, CH | [43][51] | |||||||||||||||
| Xenospadicoidaceae | Spadicoides | • | • | USA | [42] | |||||||||||||||
| Valsaceae | Phomopsis | • | • | • | JP | [48][49][51] | ||||||||||||||
| Incertae sedis | Trichothecium | • | • | PT | [45][46] | |||||||||||||||
| Basidiomycota | Agariomycetes | Irpicaceae | Irpex | • | KR | [44] | ||||||||||||||
| Ceratobasidiaceae | Rhizoctonia | • | JP | [50] | ||||||||||||||||
| Mucoromycota | Mortierellomycetes | Mortierellaceae | Mortierella | • | JP | [50] | ||||||||||||||
| Mucoromycetes | Mucoraceae | Mucor | • | • | • | KR | [44] | |||||||||||||
| Rhizopodaceae | Rhizopus | • | CH | [51] | ||||||||||||||||
The availability of high-throughput sequencing (HTS) technologies, such as amplicon metagenomics, has revolutionized ecological studies of fungal communities [37] allowing broader insight into the complexity of host-fungal interactions. The primary fungal DNA barcode ITS is commonly used on HTS-based metabarcoding. Still, caution should be taken when interpreting HTS results since, for several groups of important plant pathogens and endophytes, ITS provides insufficient resolution for species-level assignment [37]. It is recommended that researchers use the ITS2 subregion, less taxonomically biased, with lower length variation and more universal primer sites, or the full ITS region with greater taxonomic resolution and reduced amplification of dead organisms [37]. In the context of PWD, amplicon technologies were firstly applied to characterize bacterial communities (e.g., using the hypervariable regions of 16S rRNA molecule), revealing the presence of PWNs and PWD progression [54][55][52]. Chu et al. [56] described the impact of the disease on root-associated fungi (e.g., ectomycorrhizal fungi, ECMF; dark septate endophytic fungi, DSE; arbuscular mycorrhizal fungi, AMF) in different stands of Pinus tabulaeformis Carriére forest (undisturbed, moderate, and highly disturbed pine stands). The authors showed that fungal community richness and diversity, as well as soil hyphal density, decreased with the increase of disease disturbance. Basidiomycota and Ascomycota were the dominant root-associated fungi, with specific genera present in the different disturbed stands [56].
While exploring PWN growth and development by the fungi of non-infected and infected pine trees, several fungi species showed promising results in the control of nematode populations ( Table 2 ). Naturally occurring Ascomycota fungi from the genera Aureobasidium , Aspergillus, Cephalosporium, Fusarium , Gliocladium , Mucor , Mortierella , Penicillium , Rhizoctonia , and some species of Trichoderma and Verticillium, affected PWN survivability in in vitro or in vivo bioassays [40][48][49][50][57][58]. To further study the effect of Trichoderma sp. on PWN suppression and transmission by M. alternatus, Maehara et al. [59] inoculated several isolates of Trichoderma spp. into wilt-killed P. desinflora logs. Beetles from logs treated with Trichoderma sp. 3 carried less than 1000 nematodes. The authors suggested that combining the use of this fungus for PWN control with the entomopathogenic fungus Beauveria bassiana for M. alternatus control could represent a potential biocontrol application in PWD [59]. The entomopathogenic fungus B. pseudobassiana, isolated from naturally infected M. galloprovincialis in Spain, has also showed potential as a natural insect population regulator [60], and it may also be feasible in combination with other PWN control agents.
| Fungi in Interaction with PWNs | Mode of Action | References | |
|---|---|---|---|
| Growth promotors | Botrytis cinerea | Increase of PWN population growth in in vitro mycelial fungi | [49] |
| Ceratocystis sp. | Increase of PWN population growth in in vitro mycelial fungi and in pine segments of Pinus densiflora | [39][40][49] | |
| Diplodia sp. | Increase of PWN population growth in in vitro mycelial fungi | [49] | |
| Pestalotia sp. | Increase of PWN population growth in in vitro mycelial fungi and in pine segments of Pinus thunbergii | [39][40] | |
| Macrophoma sp. | Increase of PWN population growth in in vitro mycelial fungi and in pine segments of Pinus thunbergii | [39][40][61] | |
| Fusarium sp. | Increase of PWN population growth in in vitro mycelial fungi | [39][40] | |
| Ophiostoma minus | Increase of PWN population growth in wood blocks of Pinus thunbergii; Increase of PWN population growth in Pinus densiflora bolts; Increased no. of PWNs carried by emerging Monochamus alternatus | [57][58][61][62][63][64] | |
| Leptographium sp. | Increase of PWN population growth in logs of Pinus densiflora; Increase of axenic PWN population on autoclaved cuttings of Pinus thunbergii | [48][49][62] | |
| Crytosporiopsis sp. | Increase of PWN population growth in wood blocks of Pinus densiflora | [48] | |
| Sporothrix sp. | Increase of PWN population growth in in vitro mycelial fungi | [65][64] | |
| Ophiostoma ips | Increase of PWN population growth in in vitro mycelial fungi and in segments of Pinus thunbergii | [65][66][63] | |
| Leptographium pine-densiflorae | Increase of PWN population growth in in vitro mycelial fungi | [64] | |
| Trichoderma sp. 1 | Increase of PWN population growth in in vitro mycelial fungi | [48] | |
| Trichaptum abietinum Arthrobotrys sp. Gloeophyllum striatum Cryptoporus volvatus |
Increase of PWN population growth in in Pinus densiflora; Increased no. of PWNs carried by emerging Monochamus alternatus | [61] | |
| Potential for biocontrol applications | Alternaria sp. Epicoccum sp. |
Decrease of PWN population growth in in vitro mycelial fungi | [49] |
| Aureobasidium sp. Aspergillus sp. Gliocladium sp. Mucor sp. Mortierella sp. Penicillium sp. Rhizoctonia sp. |
Decrease of PWN population growth in in vitro mycelial fungi | [50] | |
| Cystidiophorus castaneus | Decrease of PWN population growth in Pinus densiflora; Decrease the no. of PWNs carried by emerging Monochamus alternatus | [61] | |
| Cephalosporium | Decrease of PWN population growth in in vitro mycelial fungi | [49][50] | |
| Fusarium sp. | Decrease of PWN population growth in in vitro mycelial fungi | [49][50] | |
| Pycnoporus coccineus | Decrease of PWN population growth in Pinus densiflora; Decrease in the no. of PWNs carried by emerging Monochamus alternatus | [61] | |
| Trichoderma sp. | Decrease of PWN population growth in in vitro mycelial fungi and in wood blocks of Pinus densiflora and P. thunbergii; Decrease in the no. of PWNs carried by emerging Monochamus alternatus | [49][57][58][61][59] | |
| Verticillium sp. | Endoparasitic fungi; Decrease of PWN population growth in in vitro mycelial fungi and in wood blocks of Pinus densiflora and P. thunbergii; Decrease in the no. of PWNs carried by emerging Monochamus alternatus | [58][61] | |
| Arthrobotrys conoides | Nematode-trapping fungi; Extracellular enzyme Ac1 with nematostatic effect on PWN | [67] | |
| Drechslerlia dactyloides | Nematode-trapping fungi | [68] | |
| Esteya vermicola | Endoparasitic fungi; Decrease of PWN population growth in in vitro feeding trials; Volatile compounds attractive to PWN | [68][69][70][71][72] | |
| Esteya floridanum | Endoparasitic fungi; Decrease of PWN population growth in in vitro feeding trials | [73] | |
| Caryospora callicarpa | Nematicidal activity of caryospomycins A to C metabolites exhibit moderate killing of PWN | [74] | |
| Geotrichum sp. AL4 | Nematicidal activity against PWN | [75] | |
| Acremonium sp. BH0531 | Nematicidal activity against PWN | [76] | |
4. Conclusions
Plant microbiome is considered a very propitious strategy for fostering plant protection against abiotic and biotic stressors [77]. Research on PWD-associated mycoflora has been slowly progressing since the first studies in the mid-seventies. HTS technologies have enriched the narrow vision of culturomic studies, and we now know that the presence of PWNs can affect the fungal diversity of the infected trees. Ophiostomatoid, or blue-stain fungi, often recovered from wilted pine trees, are considered the most determinative biotic factors for multiplication and distribution of PWNs inside the tree and in the insect-vector. Naturally occurring fungi, endophytic or nematophagous (e.g., nematode-trapping fungi D. dactyloides or the endoparasitic fungi E. vermicola ), should be further explored as new tools for PWD management.
References
- Mamiya, Y. Pathology of the pine wilt disease caused by Bursaphelenchus xylophilus. Annu. Rev. Phytopathol. 1983, 21, 201–220.
- Mota, M.M.; Braasch, H.; Bravo, M.A.; Penas, A.C.; Burgermeister, W.; Metge, K.; Sousa, E. First report of Bursaphelenchus xylophilus in Portugal and in Europe. Nematology 1999, 1, 727–734.
- Fonseca, L.; Cardoso, J.M.S.; Lopes, A.; Pestana, M.; Abreu, F.; Nunes, N.; Mota, M.; Abrantes, I. The pinewood nematode, Bursaphelenchus xylophilus, in Madeira Island. Helminthologia 2012, 49, 96–103.
- Abelleira, A.; Picoaga, A.; Mansilla, J.P.; Aguin, O. Detection of Bursaphelenchus xylophilus, causal agente of pine wilt disease on Pinus pinaster in northwestern Spain. Plant Dis. 2011, 95, 776.
- Robinet, C.; Van Opstal, N.; Baker, R.; Roques, A. Applying a spread model to identify the entry points from which the pine wood nematode, the vector of pine wilt disease, would spread most rapidly across Europe. Biol. Control 2011, 13, 2981–2995.
- Hirata, A.; Nakamura, K.; Nakao, K.; Kominami, Y.; Tanaka, N.; Ohashi, H.; Takano, K.T.; Takeuchi, W.; Matsui, T. Potential distribution of pine wilt disease under future climate change scenarios. PLoS ONE 2017, 12, e0182837.
- Webster, J.; Mota, M. Pine wilt disease: Global issues, trade and economic impact. In Pine Wilt Disease: A Worldwide Threat to Forest Ecosystems; Mota, M., Vieira, P., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 1–3.
- Vicente, C.; Espada, M.; Vieira, P.; Mota, M. Pine Wilt Disease: A threat to European forestry. Eur. J. Plant Pathol. 2012, 133, 89–99.
- Zhao, L.; Mota, M.; Vieira, P.; Butcher, R.A.; Sun, J. Interspecific communication between pinewood nematode, its insect vector, and associated microbes. Trends Parasitol. 2014, 30, 299–308.
- Nascimento, F.X.; Hasegawa, K.; Mota, M.; Vicente, C.S.L. Bacterial role in pine wilt disease development—Review and future perspectives. Environ. Microbiol. Rep. 2015, 7, 51–63.
- Proença, D.N.; Grass, G.; Morais, P.V. Understanding pine wilt disease: Roles of the pine endophytic bacteria and of the bacteria carried by the disease-causing pinewood nematode. Microbiologyopen 2017, 6, 1–20.
- Hasegawa, K.; Miwa, J. Embryology and cytology of Bursaphelenchus xylophilus. In Pine Wilt Disease, 1st ed.; Zhao, B.G., Futai, K., Sutherland, J.R., Takeuchi, Y., Eds.; Springer: Tokyo, Japan, 2008; pp. 81–104.
- Faria, J.; Sena, I.; Vieira da Silva, I.; Ribeiro, B.; Barbosa, P.; Ascensão, L.; Bennett, R.N.; Mota, M.; Figueiredo, A.C. In vitro co-cultures of Pinus pinaster with Bursaphelenchus xylophilus: A biotechnological approach to study pine wilt disease. Planta 2015, 241, 1325–1336.
- Fonseca, L.; Cardoso, J.; Abrantes, I. Nematode-plant. In Pine Wilt Disease in Europe—Biological Interactions and Integrated Management, 1st ed.; Sousa, E., Vale, F., Abrantes, I., Eds.; Federação Nacional das Associações de Proprietários Florestais: Coimbra, Portugal, 2015; pp. 35–78.
- Evans, H.F.; McNamara, D.G.; Braasch, H.; Chadoueuf, J.; Magnusson, C. Pest Risk Analysis (PRA) for the territories of the European Union (as PRA area) on Bursaphelenchus xylophilus and its vectors in the genus Monochamus. EPPO Bull. 1996, 26, 199–249.
- Sousa, E.; Naves, P.; Bonifácio, L.; Bravo, M.A.; Penas, A.C.; Pires, J.; Serrão, M. Preliminary survey for insects associated with Bursaphelenchus xylophilus in Portugal. EPPO Bull. 2012, 32, 499–502.
- Akbulut, S.; Stamps, W.T. Insect vectors of the pinewood nematode: A review of the biology and ecology of Monochamus species. For. Pathol. 2011, 42, 89–99.
- Sousa, E.; Bravo, M.A.; Pires, J.; Naves, P.; Penas, A.C.; Bonifácio, L.; Mota, M.M. Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae) associated with Monochamus galloprovincialis (Coleoptera: Cerambycidae) in Portugal. Nematology 2001, 3, 89–91.
- Naves, P.M.; Camacho, S.; De Sousa, E.M.; Quartau, J.A. Transmission of the pine wood nematode Bursaphelenchus xylophilus through feeding activity of Monochamus galloprovincialis (Col., Cerambycidae). J. Appl. Entomol. 2007, 131, 21–25.
- Futai, K. Pine wood nematode, Bursaphelenchus xylophilus. Annu. Rev. Phytopathol. 2013, 51, 61–83.
- Yazaki, K.; Takanashi, T.; Kanzaki, N.; Komatsu, M.; Levia, D.F.; Kabeya, D.; Tobita, H.; Kitao, M.; Ishida, A. Pine wilt disease causes cavitation around the resin canals and irrecoverable xylem conduit dysfunction. J. Exp. Bot. 2018, 69, 589–602.
- Mota, M.M.; Vieira, P. Pine wilt disease in Portugal. In Pine Wilt Disease, 1st ed.; Zhao, B.G., Futai, K., Sutherland, J.R., Takeuchi, Y., Eds.; Springer: Tokyo, Japan, 2008; pp. 33–38.
- Inácio, M.L.; Nóbrega, F.; Vieira, P.; Bonifácio, L.; Naves, P.; Sousa, E.; Mota, M. First detection of Bursaphelenchus xylophilus associated with Pinus nigra in Portugal, and in Europe. For. Pathol. 2015, 45, 235–238.
- Oku, H.; Shiraishi, T.; Kurozumi, S. Participation of toxin in wilting of Japanese pines caused by a nematode. Naturwissenschaften 1979, 66, 210.
- Han, Z.M.; Hong, Y.D.; Zhao, B.G. A study on pathogenicity of bacteria carried by pine wood nematodes. J. Phytopathol. 2003, 151, 683–689.
- Zhao, B.G.; Wang, H.L.; Han, S.F.; Han, Z.M. Distribution and pathogenicity of bacteria species carried by Bursaphelenchus xylophilus in China. Nematology 2003, 5, 899–906.
- Vicente, C.S.L.; Nascimento, F.X.; Espada, M.; Barbosa, P.; Hasegawa, K.; Mota, M.; Oliveira, S. Characterization of bacterial communities associated with the pine sawyer beetle Monochamus galloprovincialis, the insect vector of the pinewood nematode Bursaphelenchus xylophilus. FEMS Microbiol. Lett. 2013, 347, 130–139.
- Vicente, C.S.L.; Nascimento, F.X.; Barbosa, P.; Ke, H.M.; Tsai, I.J.; Hirao, T.; Cock, P.J.A.; Kikuchi, T.; Hasegawa, K.; Mota, M. Evidence for an Opportunistic and Endophytic Lifestyle of the Bursaphelenchus xylophilus—Associated Bacteria Serratia marcescens PWN146 Isolated from Wilting Pinus pinaster. Microb. Ecol. 2016, 72, 669–681.
- Vicente, C.S.L.; Nascimento, F.X.; Ikuyo, Y.; Cock, P.J.A.; Mota, M.; Hasegawa, K. The genome and genetics of a high oxidative stress tolerant Serratia sp. LCN16 isolated from the plant parasitic nematode Bursaphelenchus xylophilus. BMC Genom. 2016, 17, 1–15.
- Nascimento, F.; Vicente, C.; Cock, P.; Tavares, M.; Rossi, M.; Hasegawa, K.; Mota, M. From plants to nematodes: Serratia grimesii BXF1 genome reveals an adaptation to the modulation of multi-species interactions. Microb. Genom. 2018, 4, 1–13.
- Morais, P.V.; Proença, D.N.; Francisco, R.; Paiva, G. Bacteria-nematode-plant. In Pine Wilt Disease in Europe—Biological Interactions and Integrated Management, 1st ed.; Sousa, E., Vale, F., Abrantes, I., Eds.; Federação Nacional das Associações de Proprietários Florestais: Coimbra, Portugal, 2015; pp. 161–192.
- Tomao, A.; Antonio Bonet, J.; Castaño, C.; de-Miguel, S. How does forest management affect fungal diversity and community composition? Current knowledge and future perspectives for the conservation of forest fungi. For. Ecol. Manag. 2020, 457, 117678.
- Siddique, A.B.; Unterseher, M. A cost-effective and efficient strategy for Illumina sequencing of fungal communities: A case study of beech endophytes identified elevation as main explanatory factor for diversity and community composition. Fungal Ecol. 2016, 20, 175–185.
- Crous, C.J.; Burgess, T.I.; Le Roux, J.J.; Richardson, D.M.; Slippers, B.; Wingfield, M.J. Ecological disequilibrium drives insect pest and pathogen accumulation in non-native trees. AoB Plants 2017, 9, 1–16.
- Xu, J. Fungal DNA barcoding. Genome 2016, 59, 913–932.
- Meyer, W.; Irinyi, L.; Hoang, M.T.V.; Robert, V.; Garcia-Hermoso, D.; Desnos-Ollivier, M.; Yurayart, C.; Tsang, C.; Lee, C.; Woo, P.C.Y.; et al. Database establishment for the secondary fungal DNA barcode translational elongation factor 1α (TEF1α). Genome 2019, 62, 160–169.
- Nilsson, R.H.; Anslan, S.; Bahram, M.; Wurzbacher, C.; Baldrian, P.; Tedersoo, L. Mycobiome diversity: High-throughput sequencing and identification of fungi. Nat. Rev. Microbiol. 2019, 17, 95–109.
- Roberts, P.J. Protocols for an all taxa biodiversity inventory in a costa Rican conservation area. Mycologist 1998, 13, 45.
- Kobayashi, T.; Sasaki, K.; Mamiya, Y. Fungi associated with Bursaphelenchus lignicolus, the pine wood nematode. J. Jpn. For. Soc. 1974, 56, 136–145.
- Kobayashi, T.; Sasaki, T.; Mamiya, Y. Fungi associated with Bursaphelenhus lignicolus, the pine wood nematode (II). J. Jpn. For. Soc. 1975, 57, 184–193.
- Wingfield, M.J. Reclassification of Verticicladiella based on conidial development. Trans. Br. Mycol. Soc. 1985, 85, 81–93.
- Wingfield, M.J. Fungi associated with the pine wood nematode, Bursaphelenchus xylophilus, and cerambycid beetles in Wisconsin. Mycologia 1987, 79, 325–328.
- Kuroda, K.; Ito, S. Migration speed of pine wood nematodes and activities of other microbes during the development of pine wilt disease in Pinus thunbergii. Kansai Res. Cent. For. For. Prod. Res. Inst. 1992, 74, 383–389.
- Hyun, M.W.; Kim, J.H.; Suh, D.Y.; Lee, S.K.; Kim, S.H. Fungi isolated from pinewood nematode, its vector Japanese pine sawyer, and the nematode-infected Japanese black pine wood in Korea. Mycologia 2007, 35, 159–161.
- Inácio, M.L.; Nóbrega, F.; Trindade, J.; Bonifácio, L.; Naves, P.; Sousa, E.; Mota, M.; Lima, A. Fungi associated with the vector of the pinewood nematode and their influence on Pine wilt disease. In Proceedings of the XVII Congress of European Mycologists, Madeira, Portugal, 21–25 September 2015.
- Trindade, M.J.F. Estudo da População de Fungos em Pinus pinaster em Portugal. Master’s Thesis, Instituto Superior de Agronomia, Lisbon, Portugal, December 2019.
- Wang, H.M.; Lun, Y.Y.; Lu, Q.; Liu, H.X.; Decock, C.; Zhang, X.Y. Ophiostomatoid fungi associated with pines infected by Bursaphelenchus xylophilus and Monochamus alternatus in China, including three new species. MycoKeys 2018, 39, 1–27.
- Maehara, N.; Tsuda, K.; Yamasaki, M.; Shirakikawa, S.; Futai, K. Effect of fungus inoculation on the number of Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae) carried by Monochamus alternatus (Coleoptera: Cerambycidae). Nematology 2006, 8, 59–67.
- Fukushige, H. Propagation of Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae) on fungi growing in pine-shoot segments. Appl. Entomol. Zool. 1991, 26, 371–376.
- Sriwati, R.; Takemoto, S.; Futai, K. Cohabitation of the pine wood nematode Bursaphelenchus xylophilus, and fungal species in pine trees inoculated with B. xylophilus. Nematology 2007, 9, 77–86.
- Ye, W.; Zhang, Q.; Hong, S.; Zhu, D. Studies on fungi associated with Bursaphelenchus xylophilus on Pinus massoniana in Shenzen, China. In Proceedings of the International Symposium on Pine Wilt Disease Caused by the Pine Wood Nematode, Beijing, China, 31 October–5 November 1995.
- Zhang, W.; Wang, X.; Li, Y.; Liu, Z.; Li, D.; Wen, X.; Feng, Y.; Zhang, X. Pinewood Nematode Alters the Endophytic and Rhizospheric Microbial Communities of Pinus massoniana. Microb. Ecol. 2021, 81, 807–817.
- Suh, D.Y.; Hyun, M.W.; Kim, J.J.; Son, S.Y.; Kim, S.H. Ophiostoma ips from pinewood nematode vector, Japanese pine sawyer beetle (Monochamus alternatus), In Korea. Mycobiology 2013, 41, 59–62.
- Alves, M.; Pereira, A.; Matos, P.; Henriques, J.; Vicente, C.; Aikawa, T.; Hasegawa, K.; Nascimento, F.; Mota, M.; Correia, A.; et al. Bacterial community associated to the pine wilt disease insect vectors Monochamus galloprovincialis and Monochamus alternatus. Sci. Rep. 2016, 6, 23908.
- Proença, D.N.; Francisco, R.; Santos, C.V.; Lopes, A.; Fonseca, L.; Abrantes, I.M.O.; Morais, P.V. Diversity of bacteria associated with Bursaphelenchus xylophilus and other nematodes isolated from Pinus pinaster trees with pine wilt disease. PLoS ONE 2010, 5, e15191.
- Chu, H.; Wang, C.; Wang, H.; Chen, H.; Tang, M. Pine wilt disease alters soil properties and root-associated fungal communities in Pinus tabulaeformis forest. Plant Soil 2016, 404, 237–249.
- Maehara, N.; Futai, K. Factors affecting both the numbers of the pinewood nematode, Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae), carried by the Japanese pine sawyer, Monochamus alternatus (Coleoptera: Cerambycidae), and the nematode’s life history. Appl. Entomol. Zool. 1996, 31, 443–452.
- Maehara, N.; Futai, K. Effect of fungal interactions on the numbers of the pinewood nematode, Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae), carried by the Japanese pine sawyer, Monochamus alternatus (Coleoptera: Cerambycidae). Fundam. Appl. Nematol. 1997, 20, 611–617.
- Maehara, N. Reduction of Bursaphelenchus xylophilus (Nematoda: Parasitaphelenchidae) population by inoculating Trichoderma spp. into pine wilt-killed trees. Biol. Control 2008, 44, 61–66.
- Álvarez-Baz, G.; Fernández-Bravo, M.; Pajares, J.; Quesada-Moraga, E. Potential of native Beauveria pseudobassiana strain for biological control of Pine Wood Nematode vector Monochamus galloprovincialis. J. Invertebr. Pathol. 2015, 132, 48–56.
- Maehara, N.; Futai, K. Population changes of the pinewood nematode, Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae), on fungi growing in pine-branch segments. Appl. Entomol. Zool. 2000, 35, 413–417.
- Wang, Y.; Yamada, T.; Sakaue, D.; Suzuki, K. Influence of fungi on multiplication and distribution of the pinewood nematode. In Pine Wilt Disease: A Worldwide Threat to Forest Ecosystems; Springer: Dordrecht, The Netherlands, 2005; Volume 7, pp. 115–127.
- Togashi, K.; Miyauchi, O.; Kusumoto, D.; Matsushita, N. Commensal relation between Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae) and Monochamus alternatus (Coleoptera: Cerambycidae) within pine trees. Appl. Entomol. Zool. 2016, 51, 53–62.
- Zhao, L.; Ahmad, F.; Lu, M.; Zhang, W.; Wickham, J.D.; Sun, J. Ascarosides Promote the Prevalence of Ophiostomatoid Fungi and an Invasive Pathogenic Nematode, Bursaphelenchus xylophilus. J. Chem. Ecol. 2018, 44, 701–710.
- Niu, H.; Zhao, L.; Lu, M.; Zhang, S.; Sun, J. The ratio and concentration of two monoterpenes mediate fecundity of the pinewood nematode and growth of its associated fungi. PLoS ONE 2012, 7, e31716.
- Zhao, L.; Lu, M.; Niu, H.; Fang, G.; Zhang, S.; Sun, J. A native fungal symbiont facilitates the prevalence and development of an invasive pathogen-native vector symbiosis. Ecology 2013, 94, 2817–2826.
- Yang, J.; Li, J.; Liang, L.; Tian, B.; Zhang, Y.; Cheng, C.; Zhang, K.Q. Cloning and characterization of an extracellular serine protease from the nematode-trapping fungus Arthrobotrys conoides. Arch. Microbiol. 2007, 188, 167–174.
- Wang, Z.; Wang, C.Y.; Gu, L.J.; Sun, B.S.; Zhang, D.L.; Liu, L.; Lee, M.-R.; Li, Z.; Mo, E.-K.; Sung, C.-K.; et al. Variabilities of two Drechslerella dactyloides isolates in Korea and high predacity against Bursaphelenchus xylophilus. Curr. Microbiol. 2011, 62, 472–478.
- Liou, J.Y.; Shih, J.Y.; Tzean, S.S. Esteya, a new nematophagous genus from Taiwan, attacking the pinewood nematode (Bursaphelenchus xylophilus). Mycol. Res. 1999, 103, 242–248.
- Wang, C.Y.; Fang, Z.M.; Sun, B.S.; Gu, L.J.; Zhang, K.Q.; Sung, C.K. High infectivity of an endoparasitic fungus strain, Esteya vermicola, against nematodes. J. Microbiol. 2008, 46, 380–389.
- Wang, C.Y.; Wang, Z.; Fang, Z.M.; Zhang, D.L.; Gu, L.J.; Liu, L.; Sung, C.K. Attraction of pinewood nematode to endoparasitic nematophagous fungus Esteya vermicola. Curr. Microbiol. 2010, 60, 387–392.
- Lin, F.; Ye, J.; Wang, H.; Zhang, A.; Zhao, B. Host deception: Predaceous fungus, Esteya vermicola, entices pine wood nematode by mimicking the scent of pine tree for nutrient. PLoS ONE 2013, 8, e71676.
- Li, Y.; Yu, H.; Araújo, J.P.M.; Zhang, X.; Ji, Y.; Hulcr, J. Esteya floridanum sp. nov.: An Ophiostomatalean Nematophagous Fungus and Its Potential to Control the Pine Wood Nematode. Phytopathology 2021, 111, 304–311.
- Dong, J.; Zhu, Y.; Song, H.; Li, R.; He, H.; Liu, H.; Huang, R.; Zhou, Y.; Wang, L.; Cao, Y.; et al. Nematicidal resorcylides from the aquatic fungus Caryospora callicarpa YMF1.01026. J. Chem. Ecol. 2007, 33, 1115–1126.
- Li, G.H.; Yu, Z.F.; Li, X.; Wang, X.B.; Zheng, L.J.; Zhang, K.Q. Nematicidal metabolites produced by the endophytic fungus Geotrichum sp. AL4. Chem. Biodivers. 2007, 4, 1520–1524.
- Meng, Q.H.; Shi, X.X.; Meng, F.H.; Feng, X.; Sun, J.H. Isolation of an Acremonium sp. from a screening of 52 seawater fungal isolates and preliminary characterization of its growth conditions and nematicidal activity. Biotechnol. Lett. 2012, 34, 1847–1850.
- Corinne, V.; Bastien, C.; Emmanuelle, J.; Heidy, S. Trees and Insects Have Microbiomes: Consequences for Forest Health and Management. Curr. For. Rep. 2021, 7, 81–96.




