
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | MARY ANNA VENNERI | + 881 word(s) | 881 | 2020-06-03 10:12:41 | | | |
| 2 | Catherine Yang | + 12 word(s) | 893 | 2020-06-11 10:14:43 | | | | |
| 3 | Catherine Yang | + 4 word(s) | 897 | 2020-10-27 10:57:58 | | |
Video Upload Options
Infertility represents a growing health problem in industrialized countries. Thus, a greater understanding of the molecular networks involved in this disease could be critical for the development of new therapies. A recent finding revealed that circadian rhythmicity disruption is one of the main causes of poor reproductive outcome. The circadian clock system beats circadian rhythms and modulates several physiological functions such as the sleep-wake cycle, body temperature, heart rate, and hormones secretion, all of which enable the body to function in response to a 24 h cycle. This intricated machinery is driven by specific genes, called “clock genes” that fine-tune body homeostasis. Stress of modern lifestyle can determine changes in hormone secretion, favoring the onset of infertility-related conditions that might reflect disfunctions within the hypothalamic–pituitary–gonadal axis. Consequently, the loss of rhythmicity in the suprachiasmatic nuclei might affect pulsatile sexual hormones release. Herein, we provide an overview of the recent findings, in both animal models and humans, about how fertility is influenced by circadian rhythm. In addition, we explore the complex interaction among hormones, fertility and the circadian clock. A deeper analysis of these interactions might lead to novel insights that could ameliorate the therapeutic management of infertility and related disorders.
1. Introduction
The term ‘’circadian rhythm”, derived from Latin “circa dies”, is used to describe the daily oscillations in gene expression, metabolism, activity patterns and serum hormone levels that occur across 24 h. These physiological processes represent an ubiquitous feature in living organisms, from cyanobacteria to humans, and are finely regulated by the suprachiasmatic nuclei (SCN) of the anterior hypothalamus [1]. In mammals, circadian timing system is generated at cellular levels through a series of interlocked positive and negative transcription/translation feedback loops, leading to the expression of circadian rhythms. The positive loop is composed by Brain and Muscle ARNT-like 1 (BMAL1) gene and Circadian Locomotor Output Cycles Kaput (CLOCK) gene, which encode for BMAL1 and CLOCK proteins, respectively. These proteins form an heterodimer, which finally binds itself to the a cyclic AMP response element (CRE)/E-box element (E-box) of Period (PER), Cryptochrome (CRY) genes, promoting their transcription [2]. Similar to their predecessors BMAL1 and CLOCK, also PER and CRY are able to dimerize, bind to Casein Kinase 1 (CK1), and finally move into the cell nucleus, where they binds to the BMAL1/CLOCK dimer, inhibiting its transcription “closing” the loop (negative loops) [3][4]. In a second transcriptional loop, CLOCK/BMAL1 activate the transcription of genes for the nuclear receptors REV-ERB, also known as NR1D1 (nuclear receptor subfamily 1, group D, member 1). These proteins compete with the retinoic acid-related orphan receptors (ROR) for binding sites ROR-binding elements (RORE) on the BMAL1 gene, providing both positive (ROR) and negative (REV-ERB) transcriptional regulation [4] (Figure 1).
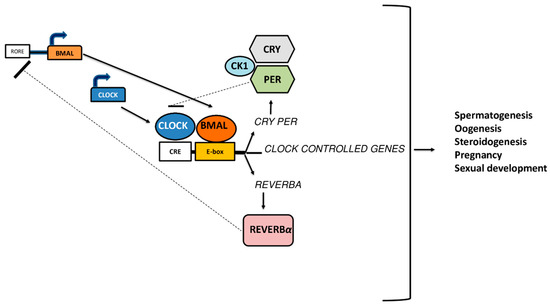
Figure 1. Autoregulatory feedback loop of clock-specific gene expressions that are involved in fertility processes.
2. Hormones and fertility from the viewpoint of the circadian system
Many data coming from animal studies have demonstrated the role of clock genes and clock-related genes in the regulation of both male and female fertility [5][6]. Indeed, alterations of biological rhythms and disrupted functions of the circadian clocks have been demonstrated to negatively impact reproductive capacity [7]. It is well known that physiological processes governing fertility need to be appropriately tight-timed orchestrated with the external environment to ensure reproductive success. Thus, a fine circadian regulation of reproductive hormones is mandatory for fertility both in males and females. It is interesting to note that the regulation of the estrus cycle, luteinizing hormone (LH) surge, sperm production and maturation, and the timing of insemination and fertilization are regulated by clock genes [6][8]. Furthermore, the timing of peripheral biological rhythm patterns is synchronized with circadian oscillation of melatonin and cortisol [9][10]. Changes in their circulating levels can indirectly impair reproduction, in which proper levels of glucocorticoids are required for normal gonadal function [11].
The aim of this review is to highlight most recent findings on the network connections among circadian rhythms, hormones and fertility.
Infertility represents one of the most important health problems following cancer and cardiovascular diseases. Few studies have explored the relation between circadian rhythms and reproduction and most of them have been made in rodents. However, the discovery of functional clock machinery in many reproductive tissues and the development of clock gene knockout mouse models have revealed the pivotal role of such genes in orchestrating reproductive processes in mammals. Clock genes affect infertility, producing low levels of sex hormones, causing embryo implantation failure and reducing newborn size both in mouse models and in shift-working women.
In this review, we assessed the connection between hormones and fertility from the viewpoint of the circadian system. Although we may not cover the whole field of disease phenotypes, we believe this review highlights the intimacy with which these three physiological aspects interact to maintain homeostasis.
Understanding how the circadian rhythms work, and how genes make them run, is becoming more and more important for human infertility. A tight net of interactors, among which clock genes and hormones are the major players, define a complex network. However, more studies are required to better fix the topic, especially regarding humans. In the years to come, the knowledge of the infertility problem could be increasingly linked to the understanding of this vast network of interactions. Chrono-therapeutical strategies that reset or modify the biological clock may contribute to restore the internal synchrony and thus counteract pathological symptoms of infertility.
References
- Thomas Dickmeis; Benjamin D. Weger; Meltem Weger; The circadian clock and glucocorticoids – Interactions across many time scales. Molecular and Cellular Endocrinology 2013, 380, 2-15, 10.1016/j.mce.2013.05.012.
- Kazuhiko Kume; Mark J Zylka; Sathyanarayanan Sriram; Lauren P Shearman; David R. Weaver; Xiaowei Jin; E S Maywood; Michael H. Hastings; S M Reppert; mCRY1 and mCRY2 are essential components of the negative limb of the circadian clock feedback loop.. Cell 1999, 98, 193-205, 10.1016/s0092-8674(00)81014-4.
- Nader, N.; Chrousos, G.P.; Kino, T. Interactions of the circadian clock system and the hpa axis. Trends Endocrinol. Metab. 2010, 21, 277–286.
- Partch, C.L.; Green, C.B.; Takahashi, J.S. Molecular architecture of the mammalian circadian clock. Trends Cell Biol. 2014, 24, 90–99.
- Kennaway, D.J.; Boden, M.J.; Varcoe, T.J. Circadian rhythms and fertility. Mol. Cell Endocrinol. 2012, 349, 56–61.
- Sen, A.; Hoffmann, H.M. Role of core circadian clock genes in hormone release and target tissue sensitivity in the reproductive axis. Mol. Cell Endocrinol. 2020, 501, 110655.
- Xiaoyue Pan; Meredith J. Taylor; Emma Cohen; Nazeeh Hanna; Samantha Mota; Circadian Clock, Time-Restricted Feeding and Reproduction. International Journal of Molecular Sciences 2020, 21, 831, 10.3390/ijms21030831.
- S.N Kalantaridou; A Makrigiannakis; E Zoumakis; G.P Chrousos; Stress and the female reproductive system. Journal of Reproductive Immunology 2004, 62, 61-68, 10.1016/j.jri.2003.09.004.
- Morris, C.J.; Aeschbach, D.; Scheer, F.A. Circadian system, sleep and endocrinology. Mol. Cell Endocrinol. 2012, 349, 91–104.
- Minnetti, M.; Hasenmajer, V.; Pofi, R.; Venneri, M.A.; Alexandraki, K.I.; Isidori, A.M. Fixing the broken clock in adrenal disorders: Focus on glucocorticoids and chronotherapy. J. Endocrinol. 2020.
- Dana N. Joseph; Shannon Whirledge; Stress and the HPA Axis: Balancing Homeostasis and Fertility. International Journal of Molecular Sciences 2017, 18, 2224, 10.3390/ijms18102224.




