1000/1000
Hot
Most Recent

Thallium, as a pharmaceutical cosmetic product, is applied for facial hair removal and fungal infections of the scalp. Thallium acetate is currently used as a catalyst in organic synthesis in the oxidation of olefins and hydrocarbons, and in epoxidation and polymerization reactions. Detection of Tl is a challenging task because its concentration in environmental samples may be at a nanogram per gram level or lower.
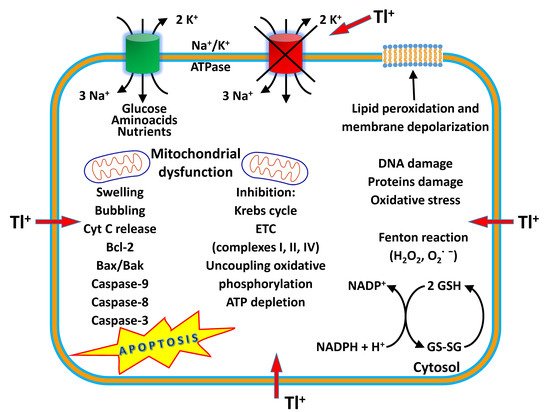
Thallium (Tl) is a rare earth bluish-white heavy metal (81 atomic number, 204.38 atomic mass, 11.85 g/cm 3), and is soft, malleable, and exists in two oxidation states (I and III). The name thallium derives from Greek thallos, a young olive-green shoot. Although thallium is present in the natural environment in low concentration, it is widely distributed in water environments [1]. The European COST Action TD1407 included Tl in the list of technology-critical elements, with associated environmental impacts and potential human health threats [2]. This element is a non-essential metal present in low concentration in human tissues but is endowed with high potential toxicity. Indeed, it has been considered one of the most toxic among the heavy metals, more toxic to humans than mercury, cadmium, lead, copper, or zinc [3][4]. Acute Tl poisoning in humans induces pathological changes in organs such as the stomach, liver, kidneys, brain, intestine, cardiovascular and nervous systems, along with chronic effects such as mental disorders or polyneuritis, and may even result in death [5]. The lethal dose of Tl for an adult human is only 8–10 mg/kg. Monovalent thallium is similar to potassium in ionic radius and electrical charge, and these factors contribute to its toxic nature. Thallium acts on several organs, interfering with cellular metabolism, affecting vital potassium-dependent processes and mitochondrial metabolism, affecting uncoupling mitochondrial oxidative phosphorylation. In addition, thallium increases reactive oxygen species (ROS) formation and phospholipid peroxidation, alters the mitochondrial membrane potential (MMP), causing mitochondrial depolarization and swelling with a release of cytochrome C from the inner mitochondrial membrane. These processes are likely to account for the neurotoxic effects of the metal [6]. Long-term, low-dose prenatal Tl exposure may cause dysfunction in the mother [7], whereas absorbed Tl can affect the developing fetus because it can cross the placental barrier [8]. Several treatment modalities have been used for thallium toxicity, but no single antidote has been shown to be effective in severe toxicity. However, combinations of different treatments have been proven to be beneficial in several cases. Prussian blue (PB) has been the most prescribed antidote to treat thallium poisoning. This chelator agent is administered by the oral route, decreasing the absorption of Tl to the enterohepatic circulation, and therefore increasing the elimination of Tl into feces [9]. Despite its effectiveness as an antidote, in severe cases of human thallotoxicosis, its administration is still ineffective. Other chelating agents have been administered alone or in combination with PB, such as sodium diethyldithiocarbamate and D-penicillamine [10][11]. The combined treatment of PB and metallothionein has proven to be a good antidotal option against thallotoxicosis [12]. Blood purification treatments are also a beneficial treatment option, especially for patients with severe thallium poisoning [13]. Despite its toxicity, known since the 1970s [14][15], this metal is applied in cardiovascular scintigraphy and as a tool for imaging malignant tumors such as lung cancer, breast cancer and osteosarcoma bone cancer [16]. Thallium-201 chloride ( 201Tl-thallous chloride) was the first radiopharmaceutical clinically used for cardiac imaging technique in the evaluation of ischemic heath disease. Short-lived radioactive thallium (emitting X-rays and gamma-rays) is administered by the intravenous route in the human body. It is obtained from metal mining, ore processing or smelting operations and is discarded as a by-product in the environment [17]. In recent years, Tl contamination incidences have been reported in many countries, mostly due to industrial activities such as the mining and smelting of Tl-rich sulfide ores, metallurgical production, coal combustion, and cement production [18]. Recent studies have shown that elevated Tl levels often occur in soils, waters, sediments, and agricultural products in the vicinity of industrial sites using Tl-bearing mineral resources [19]. Thallium can be released into the environment as waste from the production of cadmium, lead and zinc, and cement factories, and by the combustion of coal in coal-fired power plants. There is an increasing contemporary demand for this metal in the advanced technology field, in infrared spectrometers and other optical systems, electronic devices, alloys, semiconductors, and the laser industry [20][21]. High levels of thallium have been found in drinking water, vegetables, fruits and food due to anthropogenic activities [19][22]. Human beings are subjected to thallium exposure through the intake of contaminated fruits, vegetables, and other food, water, through the inhalation of polluted air, and living near industrial facilities such as cement and coal-fired power plants, mines and ore smelting. Aprea et al. reported a study which represents the most extensive human biomonitoring campaign for the evaluation of thallium exposure available at international level [22]. Staff et al. showed that urinary thallium concentrations were higher in thallium workers than in non-occupationally exposed people and general workers [23]. Therefore, the removal of this element from soil and water is vital to eradicate its health impacts. Phytoremediation is a green technology that uses plants to remove toxic and radioactive metals, and organic compounds such as pesticides and detergents from soil and water, and it is used for its cost-effectiveness and environmental friendliness [24]. Some plants are ideal for the phytoremediation process, especially when they grow fast and have a high biomass [25]. Solanum nigrum L., Brassica oleracea acephala L., Brassica napus , Brassica juncea , Iberis intermedia and Callitriche cophocarpa have been identified as good species for the phytoremediation of soil and waters contaminated by thallium [26][27][28].
| References | |
|---|---|
| Cardiac symptoms | [31][32] |
| Coma | |
| Hypotension | |
| Lethargy | |
| Tachycardia | |
| Dermatological symptoms | [32] |
| Alopecia (after about 3 weeks) | |
| Anhidrosis | |
| Hypohidrosis | |
| Mees lines on the nails (after about 1 month) | |
| Gastrointestinal symptoms | [32] |
| Diarrhea or constipation | |
| Nausea and vomiting | |
| Stool containing blood | |
| Hematologic symptoms | [32] |
| Anemia | |
| Eosinophilia | |
| Leukocytosis | |
| Neutrophilia | |
| Thrombocytopenia | |
| Neurologic symptoms (after 3–5 days) | [31][32] |
| Ataxia | |
| Convulsion | |
| Death | |
| Distal muscle weakness of the hands or feet | |
| Hallucination | |
| Headache | |
| Insomnia | |
| Paresthesia | |
| Tremor | |
| Ocular symptoms | [32] |
| Atrophy of the optic nerve | |
| Cranial nerve 7th palsy | |
| Nystagmus | |
| Lens opacity | |
| Optic neuropathy | |
| Ptosis |