
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jan Kiebist | + 2872 word(s) | 2872 | 2021-09-26 08:08:17 | | | |
| 2 | Jason Zhu | Meta information modification | 2872 | 2021-09-29 05:18:50 | | | | |
| 3 | Jason Zhu | + 1 word(s) | 2873 | 2021-10-27 12:00:33 | | |
Video Upload Options
Antithrombotic thienopyridines, such as clopidogrel and prasugrel, are prodrugs that undergo a metabolic two-step bioactivation for their pharmacological efficacy. In the first step, a thiolactone is formed, which is then converted by cytochrome P450-dependent oxidation via sulfenic acids to the active thiol metabolites. These metabolites are the active compounds that inhibit the platelet P2Y12 receptor and thereby prevent atherothrombotic events.
1. Introduction
Clopidogrel (Plavix®, Iscover®) and prasugrel (Efient®) are antithrombotic prodrugs of the thienopyridine family that ignite their intrinsic effect after metabolic bioactivation and irreversibly inhibit the platelet P2Y12 receptor resulting in prevention of atherothrombotic events [1][2][3]. The two-step enzymatic bioactivation required for both prodrugs differs in its initial reactions: In the case of clopidogrel, the hydroxylation of the thiophene ring, required for the spontaneous formation of the thiolactone metabolite, 2-oxo-clopidogrel (Scheme 1), is catalyzed by cytochrome P450 (CYP) monooxygenases [4][5]. In case of prasugrel, the corresponsive thiolactone (R-95913) is formed by hydrolysis of its ester functionality, which seems to be mainly catalyzed by human carboxylesterases hCE2[6]. The final transformation of these thiolactone derivatives into the respective active thiol metabolites is formally a thioester hydrolysis, since the oxidation state remains unchanged. In fact, thioesterases such as paraoxonase-1 (PON-1) are thought to accomplish this kind of hydrolysis, but the reactions studied led to the biologically inactive endo-isomer, in which the double bond has migrated into the piperidine ring[7][8]. Recent studies disclosed the formation of the active metabolite as a multistep biochemical process that also involves CYP enzymes, while the exact chemical nature of the intermediates is still under discussion. The most plausible metabolic process describes the formation of thiolactone sulfoxides during the oxidative activation, the hydrolytic opening of which leads to sulfenic acids that are efficiently reduced by ascorbate, phosphines, reductases or glutathione to give the active thiol metabolites (Scheme 1) [9][10][11][12].
The bioactivation of clopidogrel to form 2-oxo-clopidogrel and the active thiol metabolite can be catalyzed by several CYP enzymes. Kinetic and inhibition studies, however, revealed that just CYP2C19 contributes substantially to both oxidative steps and that CYP3A4 is essential for the formation of the active metabolite. Furthermore, CYP1A2, CYP2B6 and CYP2C9 may be involved in the oxidative cascade[5]. Several studies revealed that polymorphism in the gene encoding CYP2C19 contributes to the variability of platelet response to clopidogrel in patients. Individuals who carry a reduced-function CYP2C19 allele had significantly lower levels of the active metabolite of clopidogrel, diminished platelet inhibition and a higher rate of major adverse cardiovascular events [13][14][15][16]. In addition, clopidogrel undergoes an extensive first-pass metabolism in the human liver where most of the prodrug is hydrolyzed to inactive clopidogrel carboxylic acid by carboxylesterase hCE1, followed by glucuronidation and renal excretion [16]. To address these causes for the low plasma level of active metabolites, prasugrel was developed replacing the ester functionality with a metabolically stable ketone and the introduction of an ester group in the thiophene 2-position that switched the first-step activation from CYP2C19 to carboxylesterase hCE2 [17].
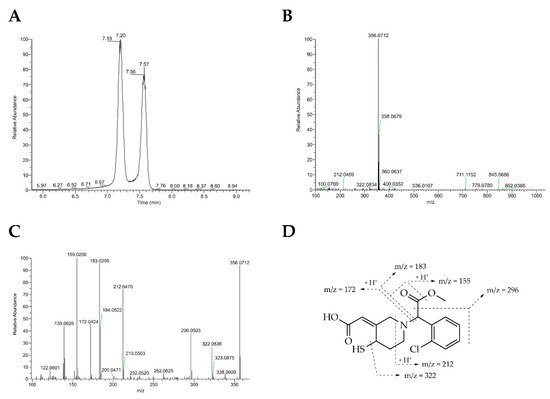
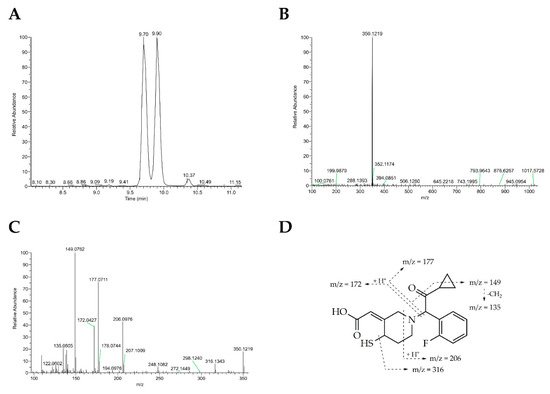
In vitro metabolism studies showed that metabolic activation of clopidogrel results in a mixture of four stereoisomers (H1–H4, Figure 1). The H1 and H2 isomers are trans isomers, whereas the exocyclic double bond between C3 and C16 in H3 and H4 appears in cis configuration. Analysis of clinical samples demonstrated that only H3 and H4 are formed in vivo, with only H4 being active. Therefore, the configuration of the thiol group at C4 seems to be crucial for antiplatelet activity. The reactive free thiol group of the active metabolites leads to high instability in plasma samples, which is why they were stabilized by derivatization with 2-bromo-3’-methoxyacetophenone to be subsequently quantified by HPLC-MS [18][19]. For the unambiguous structural elucidation of metabolites H1–H4, syntheses for both the stabilized and free thiol metabolites have been described in the literature with yields below 3%[20][21]. Synthesis of active metabolites using human liver microsomes is hampered by low yield as well and by complicated purification protocols, and hence is not suitable for use on a preparative scale [18]. The active metabolite of prasugrel (PAM, R-138727) found in human plasma samples consists of four stereoisomers, (R, S)-, (R, R)-, (S, S)- and (S, R)-isomers (the first letter stands for the configuration of the thiol group and the second for that at the benzylic position), with 84% found as (R, S)- and (R, R)-, representing the most potent isomers. This underlines the importance of the R-configuration of the thiol group for bioactivity [22][23][24]. A chemical synthesis of PAM (R-138727) has not been reported so far.
Herein, researchers report biocatalytic syntheses of the bioactive metabolites of clopidogrel and prasugrel by unspecific peroxygenases (UPOs, EC 1.11.2.1) secreted by fungi. UPOs have arisen to ‘dream catalysts’ for oxyfunctionalization reactions, since they incorporate selectively oxygen into nonactivated hydrocarbons generating hydroxylations and epoxidations that are difficult to attain by chemical methods [25][26][28][27]. These extracellular glycosylated heme-thiolate proteins are activated by hydrogen peroxide and work independently of electron donors, transport proteins and additional cofactors. Their catalytic cycle combines characteristics of heme peroxidases and cytochrome P450s, which is why UPOs and P450 enzymes have partly overlapping reaction portfolios [28,29]. UPOs have already been used for the conversion of various pharmaceuticals, e.g., volixibat (N-dealkylation), cyclophosphamide (aliphatic hydroxylation), propranolol (aromatic hydroxylation), testosterone (epoxidation) and corticosteroids (C–C fission) [30,31,32,33,34]. In this context, it was found that, depending on the catalyzed reactions, the most relevant parameters in respect to reaction optimization were pH, H2O2 concentration and dosing rate as well as organic solvents and their content. The effective generation of authentic samples of drug metabolites is important for their structural confirmation and for LC-MS recovery, for evaluation of their potential safety risk, for investigation of drug–drug interactions, for pharmacological testing and for detailed pharmacokinetic and pharmacodynamic analysis [29][30][31].
2. Research
| Entry | Substrate | Enzyme(s) | pH | 2-Oxo Metabolite (%) 2 |
Active Metabolite (%) 2 |
|---|---|---|---|---|---|
| 1 | CPG | AaeUPO | 7.0 | 29.0 ± 0.5 | <2 |
| 2 | CPG | CraUPO | 7.0 | 12.7 ± 0.3 | <2 |
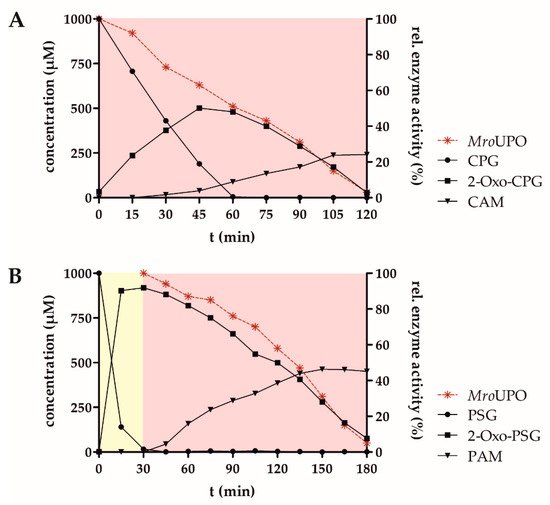
| 3 | CPG | MroUPO | 7.0 | 10.6 ± 0.2 | 8.6 ± 0.2 |
| 4 | CPG | MroUPO | 5.5 | 46.6 ± 0.5 | 3.6 ± 0.2 |
| 5 | CPG | CglUPO | 7.0 | 3.7 ± 0.1 | <2 |
| 6 | CPG | MweUPO | 7.0 | 8.1 ± 0.9 | 6.4 ± 0.1 |
| 7 | CPG | MweUPO | 5.5 | 31.6 ± 1.2 | <2 |
| 8 | PSG | PLE/AaeUPO | 7.0 | 87.4 ± 3.0 | 5.8 ± 0.6 |
| 9 | PSG | PLE/CraUPO | 7.0 | 87.2 ± 0.4 | <2 |
| 10 | PSG | PLE/MroUPO | 7.0 | 39.1 ± 2.0 | 34.2 ± 0.4 |
| 11 | PSG | PLE/CglUPO | 7.0 | 19.0 ± 0.5 | 28.2 ± 1.2 |
| 12 | PSG | PLE/MweUPO | 7.0 | 46.5 ± 2.7 | 31.0 ± 0.3 |
| Entry | Substrate | Solvent | Reductant | 2-Oxo Metabolite (%) 2 |
Active Metabolite (%) 2 |
|---|---|---|---|---|---|
| 13 (3) | CPG | Buffer/acetone 90:10 |
Ascorbate | 10.6 ± 0.2 | 8.6 ± 0.2 |
| 14 | CPG | Buffer/acetone 90:10 |
Glutathione | 26.2 ± 1.9 | <2 |
| 15 | CPG | Buffer/acetone 90:10 |
None | <2 | <2 |
| 16 | CPG | Buffer/MeCN 3 90:10 |
Ascorbate | 3.2 ± 0.1 | 4.0 ± 0.1 |
| 17 | CPG | Buffer/MeOH 3 90:10 |
Ascorbate | 31.1 ± 0.3 | <2 |
| 18 | CPG | Buffer/DMSO 3 90:10 |
Ascorbate | 16.9 ± 1.8 | <2 |
| 19 | CPG | Buffer/DMF 3 90:10 |
Ascorbate | 25.5 ± 0.5 | 4.5 ± 0.1 |
| 20 | CPG | Buffer | Ascorbate | 11.5 ± 0.2 | <2 |
| 21 | CPG | Buffer/acetone 80:20 |
Ascorbate | 8.7 ± 0.6 | 9.8 ± 0.5 |
| 22 | CPG | Buffer/acetone 60:40 |
Ascorbate | 6.3 ± 0.4 | <2 |
3. Conclusions
References
- Jarvis, B.; Simpson, K. Clopidogrel. Drugs 2000, 60, 347–377. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Braunwald, E.; McCabe, C.H.; Montalescot, G.; Ruzyllo, W.; Gottlieb, S.; Neumann, F.-J.; Ardissino, D.; De Servi, S.; Murphy, S. Prasugrel Versus Clopidogrel in Patients with Acute Coronary Syndromes. N. Engl. J. Med. 2007, 357, 2001–2015. [Google Scholar] [CrossRef]
- Farid, N.A.; Kurihara, A.; Wrighton, S.A. Metabolism and Disposition of the Thienopyridine Antiplatelet Drugs Ticlopidine, Clopidogrel, and Prasugrel in Humans. J. Clin. Pharmacol. 2010, 50, 126–142. [Google Scholar] [CrossRef]
- Pereillo, J.-M.; Maftouh, M.; Andrieu, A.; Uzabiaga, M.-F.; Fedeli, O.; Savi, P.; Pascal, M.; Herbert, J.-M.; Maffrand, J.-P.; Picard, C. Structure and Stereochemistry of the Active Metabolite of Clopidogrel. Drug Metab. Dispos. 2002, 30, 1288–1295. [Google Scholar] [CrossRef]
- Kazui, M.; Nishiya, Y.; Ishizuka, T.; Hagihara, K.; Farid, N.A.; Okazaki, O.; Ikeda, T.; Kurihara, A. Identification of the Human Cytochrome P450 Enzymes Involved in the Two Oxidative Steps in the Bioactivation of Clopidogrel to Its Pharmacologically Active Metabolite. Drug Metab. Dispos. 2010, 38, 92–99. [Google Scholar] [CrossRef]
- Williams, E.T.; Jones, K.O.; Ponsler, G.D.; Lowery, S.M.; Perkins, E.J.; Wrighton, S.A.; Ruterbories, K.J.; Kazui, M.; Farid, N.A. The Biotransformation of Prasugrel, a New Thienopyridine Prodrug, by the Human Carboxylesterases 1 and 2. Drug Metab. Dispos. 2008, 36, 1227–1232. [Google Scholar] [CrossRef]
- Dansette, P.M.; Rosi, J.; Bertho, G.; Mansuy, D. Paraoxonase-1 and Clopidogrel Efficacy. Nat. Med. 2011, 17, 1040–1041. [Google Scholar] [CrossRef] [PubMed]
- Dansette, P.M.; Rosi, J.; Bertho, G.; Mansuy, D. Cytochromes P450 Catalyze Both Steps of the Major Pathway of Clopidogrel Bioactivation, Whereas Paraoxonase Catalyzes the Formation of a Minor Thiol Metabolite Isomer. Chem. Res. Toxicol. 2012, 25, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Dansette, P.M.; Libraire, J.; Bertho, G.; Mansuy, D. Metabolic Oxidative Cleavage of Thioesters: Evidence for the Formation of Sulfenic Acid Intermediates in the Bioactivation of the Antithrombotic Prodrugs Ticlopidine and Clopidogrel. Chem. Res. Toxicol. 2009, 22, 369–373. [Google Scholar] [CrossRef]
- Dansette, P.M.; Thébault, S.; Bertho, G.; Mansuy, D. Formation and Fate of a Sulfenic Acid Intermediate in the Metabolic Activation of the Antithrombotic Prodrug Prasugrel. Chem. Res. Toxicol. 2010, 23, 1268–1274. [Google Scholar] [CrossRef]
- Hagihara, K.; Kazui, M.; Kurihara, A.; Iwabuchi, H.; Ishikawa, M.; Kobayashi, H.; Tanaka, N.; Okazaki, O.; Farid, N.A.; Ikeda, T. Biotransformation of Prasugrel, a Novel Thienopyridine Antiplatelet Agent, to the Pharmacologically Active Metabolite. Drug Metab. Dispos. 2010, 38, 898–904. [Google Scholar] [CrossRef]
- Dansette, P.M.; Levent, D.; Hessani, A.; Bertho, G.; Mansuy, D. Thiolactone Sulfoxides as New Reactive Metabolites Acting as Bis-Electrophiles: Implication in Clopidogrel and Prasugrel Bioactivation. Chem. Res. Toxicol. 2013, 26, 794–802. [Google Scholar] [CrossRef]
- Mega, J.L.; Close, S.L.; Wiviott, S.D.; Shen, L.; Hockett, R.D.; Brandt, J.T.; Walker, J.R.; Antman, E.M.; Macias, W.; Braunwald, E. Cytochrome P-450 Polymorphisms and Response to Clopidogrel. N. Engl. J. Med. 2009, 360, 354–362. [Google Scholar] [CrossRef]
- Shuldiner, A.R.; O’Connell, J.R.; Bliden, K.P.; Gandhi, A.; Ryan, K.; Horenstein, R.B.; Damcott, C.M.; Pakyz, R.; Tantry, U.; Gibson, Q. Association of Cytochrome P450 2c19 Genotype with the Antiplatelet Effect and Clinical Efficacy of Clopidogrel Therapy. JAMA 2009, 302, 849–857. [Google Scholar] [CrossRef]
- Gong, I.Y.; Crown, N.; Suen, C.M.; Schwarz, U.I.; Dresser, G.K.; Knauer, M.J.; Sugiyama, D.; DeGorter, M.K.; Woolsey, S.; Tirona, R.G. Clarifying the Importance of Cyp2c19 and Pon1 in the Mechanism of Clopidogrel Bioactivation and in Vivo Antiplatelet Response. Eur. Heart J. 2012, 33, 2856–2864. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.-J.; Wang, X.; Gawronski, B.E.; Brinda, B.J.; Angiolillo, D.J.; Markowitz, J.S. Carboxylesterase 1 as a Determinant of Clopidogrel Metabolism and Activation. J. Pharmacol. Exp. Ther. 2013, 344, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Zetterberg, F.; Svensson, P. State of Affairs: Design and Structure–Activity Relationships of Reversible P2y12 Receptor Antagonists. Bioorg. Med. Chem. Lett. 2016, 26, 2739–2754. [Google Scholar] [CrossRef]
- Tuffal, G.; Roy, S.; Lavisse, M.; Brasseur, D.; Schofield, J.; Touchard, N.D.; Savi, P.; Bremond, N.; Rouchon, M.-C.; Hurbin, F. An Improved Method for Specific and Quantitative Determination of the Clopidogrel Active Metabolite Isomers in Human Plasma. Thromb. Haemost. 2011, 105, 696–705. [Google Scholar] [PubMed]
- Karaźniewicz-Łada, M.; Danielak, D.; Teżyk, A.; Żaba, C.; Tuffal, G.; Główka, F. HPLC–MS/MS Method for the Simultaneous Determination of Clopidogrel, Its Carboxylic Acid Metabolite and Derivatized Isomers of Thiol Metabolite in Clinical Samples. J. Chromatogr. B 2012, 911, 105–112. [Google Scholar] [CrossRef]
- Shaw, S.A.; Balasubramanian, B.; Bonacorsi, S.; Cortes, J.C.; Cao, K.; Chen, B.-C.; Dai, J.; Decicco, C.; Goswami, A.; Guo, Z. Synthesis of Biologically Active Piperidine Metabolites of Clopidogrel: Determination of Structure and Analyte Development. J. Org. Chem. 2015, 80, 7019–7032. [Google Scholar] [CrossRef]
- Bluet, G.; Blankenstein, J.; Brohan, E.; Prevost, C.; Cheve, M.; Schofield, J.; Roy, S. Synthesis of the Stabilized Active Metabolite of Clopidogrel. Tetrahedron 2014, 70, 3893–3900. [Google Scholar] [CrossRef]
- Wickremsinhe, E.R.; Tian, Y.; Ruterbories, K.J.; Verburg, E.M.; Weerakkody, G.J.; Kurihara, A.; Farid, N.A. Stereoselective Metabolism of Prasugrel in Humans Using a Novel Chiral Liquid Chromatography-Tandem Mass Spectrometry Method. Drug Metab. Dispos. 2007, 35, 917–921. [Google Scholar] [CrossRef]
- Hasegawa, M.; Sugidachi, A.; Ogawa, T.; Isobe, T.; Jakubowski, J.A.; Asai, F. Stereoselective Inhibition of Human Platelet Aggregation by R-138727, the Active Metabolite of Cs-747 (Prasugrel, Ly640315), a Novel P2y12 Receptor Inhibitor. Thromb. Haemost. 2005, 94, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Farid, N.A.; Smith, R.L.; Gillespie, T.A.; Rash, T.J.; Blair, P.E.; Kurihara, A.; Goldberg, M.J. The Disposition of Prasugrel, a Novel Thienopyridine, in Humans. Drug Metab. Dispos. 2007, 35, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- Sigmund, M.-C.; Poelarends, G.J. Current State and Future Perspectives of Engineered and Artificial Peroxygenases for the Oxyfunctionalization of Organic Molecules. Nat. Catal. 2020, 3, 690–702. [Google Scholar] [CrossRef]
- Kiebist, J.; Hofrichter, M.; Zuhse, R.; Scheibner, K. Oxyfunctionalization of Pharmaceuticals by Fungal Peroxygenases. In Pharmaceutical Biocatalysis; Jenny Stanford Publishing: Singapore, 2019; Volume 5, pp. 643–680. [Google Scholar]
- Hobisch, M.; Holtmann, D.; Gomez de Santos, P.; Alcalde, M.; Hollmann, F.; Kara, S. Recent Developments in the Use of Peroxygenases–Exploring Their High Potential in Selective Oxyfunctionalisations. Biotechnol. Adv. 2020, 51, 107615. [Google Scholar] [CrossRef]
- Hofrichter, M.; Kellner, H.; Herzog, R.; Karich, A.; Liers, C.; Scheibner, K.; Kimani, V.W.; Ullrich, R. Fungal Peroxygenases: A Phylogenetically Old Superfamily of Heme Enzymes with Promiscuity for Oxygen Transfer Reactions. In Grand Challenges in Fungal Biotechnology; Springer: Berlin/Heidelberg, Germany, 2020; pp. 369–403. [Google Scholar]
- Hofrichter, M.; Kellner, H.; Pecyna, M.J.; Ullrich, R. Fungal Unspecific Peroxygenases: Heme-Thiolate Proteins That Combine Peroxidase and Cytochrome P450 Properties. Adv. Exp. Med. Biol. 2015, 851, 341–368. [Google Scholar]
- Kiebist, J.; Holla, W.; Heidrich, J.; Poraj-Kobielska, M.; Sandvoss, M.; Gröbe, G.; Atzrodt, J.; Hofrichter, M.; Scheibner, K. One-Pot Synthesis of Human Metabolites of SAR548304 by Fungal Peroxygenases. Bioorg. Med. Chem. 2015, 23, 4324–4332. [Google Scholar] [CrossRef]
- Steinbrecht, S.; Kiebist, J.; König, R.; Thiessen, M.; Schmidtke, K.-U.; Kammerer, S.; Küpper, J.-H.; Scheibner, K. Synthesis of Cyclophosphamide Metabolites by a Peroxygenase from Marasmius Rotula for Toxicological Studies on Human Cancer Cells. AMB Express 2020, 10, 1–13. [Google Scholar] [CrossRef] [PubMed]




