
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tamás Röszer | + 2373 word(s) | 2373 | 2021-09-24 05:54:18 | | | |
| 2 | Catherine Yang | Meta information modification | 2373 | 2021-09-24 11:11:42 | | |
Video Upload Options
Adipose tissue is unique in terms of its immune effects on apoptotic cell clearance, as adipocyte apoptosis triggers inflammatory cytokine responses in macrophages, and is a potential inducer of adipose tissue inflammation. Classically, there are two types of adipose tissue in mammals: white adipose tissue (WAT) and brown adipose tissue (BAT). Adipocytes of the WAT accumulate neutral lipids in a large droplet, whereas BAT adipocytes have multilocular lipid droplets and high lipolytic activity, and oxidize fatty acids into ATP, and generate heat.
1. Introduction: The Impact of Apoptotic Cell Clearance in Fat Depots
Apoptosis is an inevitable process in tissue development, and is a key component of the necessary physiological tissue turnover for proper tissue healing (reviewed in [1]). During this process, however, apoptotic cells pollute their surroundings with damage-associated molecular patterns, potential autoantigens, modified lipids and aged molecules, which can all impair tissue integrity and initiate immune responses. The safe removal of apoptotic debris is, hence, crucial to prevent self-immunity or the development of tissue damaging inflammation. This important task is performed by tissue-resident macrophages, which clear tissues of hazardous materials from the dead or dying cells without evoking inflammation [2]. The “immunologically silent” removal of apoptotic cell contents is particularly relevant when apoptotic cells are cleared from damaged, infected or inflamed tissues. Macrophages exposed to the content of apoptotic cells adopt a pro-resolving, anti-inflammatory or tolerogenic phenotype, which is often termed alternative- or M2-macrophage activation (reviewed in [3][4]). Apoptotic cell clearance induces interleukin-10 (IL-10) and tumor growth factor beta (TGFβ) production in macrophages concomitant with the expression of various M2-activation-associated molecules such as the high affinity scavenger receptor for the hemoglobin-haptoglobin complex CD163 and the mannose receptor CD206 [5].
Adipose tissue is unique in terms of its immune effects on apoptotic cell clearance, as adipocyte apoptosis triggers inflammatory cytokine responses in macrophages, and is a potential inducer of adipose tissue inflammation [6]. Classically, there are two types of adipose tissue in mammals: white adipose tissue (WAT) and brown adipose tissue (BAT) [7][8]. Adipocytes of the WAT accumulate neutral lipids in a large droplet [9], whereas BAT adipocytes have multilocular lipid droplets and high lipolytic activity, and oxidize fatty acids into ATP, and generate heat [10][11]. Beyond their role in fat metabolism, WAT and BAT have their own, specific, endocrine and immune functions [9]. Excess development of WAT leads to obesity, which is associated with a decline in BAT mass.
2. Adipocyte Apoptosis Ignites Inflammation
Number of adipocytes, which build up the adipose tissues, is stable over a prolonged period of time, and the mature adipocytes are relatively resistant to apoptosis [12][13][14]. Albeit adipocyte apoptosis appears during development and remodeling of the adipose tissue, and the adipokine C1q /TNF-related protein 9 (CTRP9) increases the uptake of apoptotic cells by macrophages [15], adipocyte number in the WAT depots is relatively constant during infancy and childhood [16]. The lipid content of the WAT increases along postnatal development due to the expansion of the cell volume of adipocytes [17], and a notable increase of adipocyte number appears at the onset of puberty [16]. In infancy, adipocytes undergo differentiation ( Figure 1 a) and activate lipolysis in response to insulin and β-adrenergic stimulation [18][19]. Two adipocyte populations can be identified later, in puberty and in adulthood: small, still maturing cells, and larger, lipid-laden mature adipocytes ( Figure 1 a) [17][20]. The number of mature adipocytes increases in severe obesity, and newly generated adipocytes also appear. An increase in adipocyte number is termed adipocyte hyperplasia, whereas an increment in cell volume due to lipid accumulation is termed adipocyte hypertrophy [13] ( Figure 1 a).
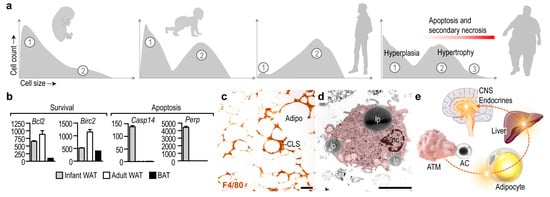
Adipocyte differentiation is associated with an increase in the expression of survival factors, such as the BCL2 apoptosis regulator and the baculoviral inhibitor of apoptosis repeat-containing 2 [12] ( Figure 1 b). WAT adipocytes are more resistant to apoptosis than BAT adipocytes [14]. Adipocyte apoptosis becomes prevalent under pathological conditions—for instance, lipodystrophy is associated with adipocyte apoptosis and necrosis [23]. Furthermore, the antiviral cytokine interferon alpha (IFNα) triggers adipocyte apoptosis both in vitro and in vivo in mice [24], and patients treated with HIV-1 protease inhibitors develop lipodystrophy due to adipocyte apoptosis [25]. In congenital generalized lipodystrophy, both WAT and BAT mass is lost due to adipocyte apoptosis and necrosis [23].
How macrophages respond to apoptotic adipocytes may depend on the immunological context in which adipocyte death occurs, the signals provided by the apoptotic cells [26], and additional immune signals such as complement and cytokines [27]. Under homeostatic conditions, when apoptotic adipocytes are scarce and Th2 cytokines are expressed in the WAT, the patrolling ATMs clear the dying adipocytes and adopt an M2-like activation state [28]. It is plausible that the first wave of apoptosis of adipocytes during the development of obesity is well controlled by the ATMs, and they are able to maintain an M2-like activation state [29]. However, the prevalence of damage-associated molecules and pro-inflammatory lipid species in the dying adipocytes can switch the function of ATMs towards the release of inflammatory cytokines and reactive oxygen species. Moreover, the capacity of ATMs to clear apoptotic cells may be exhausted in obesity [30], and the apoptotic or necrotic cell debris aggravates inflammation.
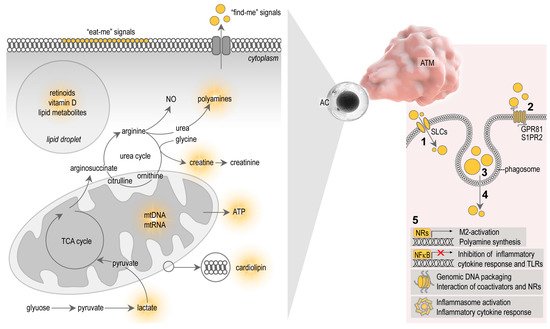
Signals that allow apoptotic cells to control macrophage behavior are complex, and involve diverse molecules and metabolites released by the apoptotic cells themselves. Apoptotic cells seem to actively elaborate cell metabolites, which serve as “find-me” signals, and display molecular patterns on the cell surfaces which serve as “eat-me” signals for the macrophages [26] ( Figure 2 ). After ingestion of the apoptotic cells, cellular contents are lysed and processed in the phagosomes of the macrophages, ultimately producing signal molecules that control macrophage functions [31] ( Figure 2 ). The study of the metabolites that shape immune response towards apoptotic cells is a new field of immunometabolism, recently termed as “efferotabolism” (efferocytosis-associated metabolism) [32]. Here, I review the possible metabolites of the apoptotic adipocytes that potentially control ATM behavior.
3. Metabolites of Apoptotic Cells and Their Effect on Macrophages
Microparticles released by apoptotic cells, and the apoptotic debris itself, contain membrane lipids and lipid mediators, which are metabolized in the macrophage phagosome to provide ligands for nuclear receptors (NRs) and trigger M2-activation (reviewed in [3][31], Figure 2 ). NRs are transcription factors that regulate gene transcription in a ligand-dependent manner [33]. Macrophages express several NRs, including peroxisome proliferator activator receptors (PPARs), liver X receptor (LXR), retinoid X receptor (RXR), retinoic acid receptor (RAR) and vitamin D receptor (VDR), and are all implicated in phagocytosis, a crucial mechanism for apoptotic cell clearance [34][35][36][37][38]. Apoptotic cells fail to inhibit pro-inflammatory cytokine responses in macrophages lacking PPARs or RXRs [37][38][39]. Furthermore, the absence of the PPARβ/δ ligand-binding domain is sufficient to impair M2-activation stimulated by apoptotic cells [37]. This suggests that PPAR/RXR signaling may be activated by lipid metabolites derived from cell membranes of the internalized apoptotic cells. However, the uptake of apoptotic cell membranes may also induce inflammasome activation in macrophages. Mitochondrial membranes, as well as entire mitochondria of the apoptotic cells, are released by apoptotic cells and trigger inflammasome activation due to the presence of damage-associated molecules in the apoptotic mitochondrial membrane, such as cardiolipin [40][41] ( Figure 2 ).
Vitamin D suppresses macrophage pro-inflammatory polarization in various settings [42][43][44]. For example, vitamin D supplementation attenuates inflammatory cytokine expression and promotes M2-macrophage traits in the epicardial adipose tissue of swine, whereas vitamin D deficiency exacerbates inflammation by increasing the number of pro-inflammatory macrophages [45]. Vitamin D also increases the expression of the antimicrobial peptide cathelicidin [46] and induces autophagy [43]. In the context of adipose tissue biology, cathelicidin is known to be expressed by the subcutaneous fat depot, and adipocyte autophagy is thought to mitigate obesity-associated diseases [47][48]. The intake of vitamin D from apoptotic adipocytes may affect ATM functions, potentially reducing pro-inflammatory cytokine expression, inducing autophagy, and aiding in the acquisition of antimicrobial traits ( Figure 2 ).
Apoptotic cells release metabolites that initiate cellular responses in neighboring cells and in macrophages that clear apoptotic debris. These signals are often called as “find-me” signals, since they promote the encounter between macrophages and apoptotic cells [26] and are released actively by dying cells through pannexin channels and possibly through other membrane channels [49].
Polyamines self-assemble with phosphate ions in the cell nucleus and generate so-called nuclear aggregates of polyamines, which interact with genomic DNA and control DNA conformation, protection and packaging [50]. Polyamines also affect the interactions of NRs with their coactivator complexes [51]. Given that NRs are important in both adipose tissue development and macrophage functions, especially in apoptotic cell removal (reviewed in [3]), it is plausible that polyamines shape macrophage functions in the process of efferocytosis at a transcriptional level ( Figure 2 ).
4. Summary and Perspective
Beyond its traditional role as a lipid storage site, adipose tissue is increasingly recognized as being pivotal in metabolic and endocrine physiology [9]. ATMs are positioned at the interface between these functions, and interactions between ATMs and adipocytes potentially affect systemic metabolism and endocrine health ( Figure 3 a). Uptake of apoptotic cells is a core function of macrophages and serves as a communication channel between adipocytes and ATMs. The rich immunometabolism of apoptotic cell contents shapes ATM function, which in turn sustains healthy metabolism by balancing the protective and destructive immune functions of ATMs. Protective immunity ensures the neutralization of apoptotic cells and favors cell metabolism and energy expenditure ( Figure 3 a). When ATMs process materials of the dying adipocytes correctly, they signal to the endocrine organs and other immune cells, which ultimately supports adipose tissue development and metabolism ( Figure 3 a). By contrast, destructive immune traits appear in response to danger signals from dying adipocytes ( Figure 3 b). The resulting ATM phenotype damages metabolism through uncontrolled inflammation or self-immunity [52], leading to a dysfunctional interplay between ATMs and adipocytes and driving the development of metabolic diseases [52][53]. ( Figure 3 b). As adipocyte death is prevalent in obese adipose tissue, the majority of the literature has focused on the role of ATMs in the setting of obesity. However, the immune functioning of ATMs begins at birth, and is crucial to sustain energy expenditure in infancy [22][54][52][55]. Adipocyte apoptosis may occur in the adipose tissue of the neonate, but we know very little about the impact of ATM-adipocyte interactions in early postnatal development, even though adipose tissue quality in infancy is contingent on ATM function and determines metabolic health in adulthood [22][56]. It is thus important to comprehend how physiological adipocyte death (i.e., during adipose tissue development) shapes ATM functions in postnatal life.
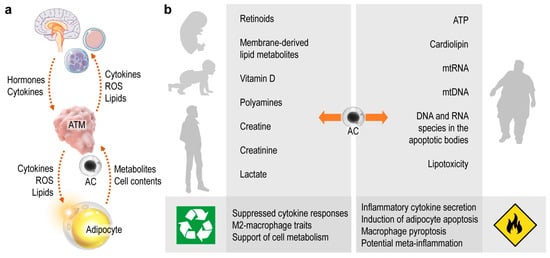
Several signals that potentially define whether ATMs safely recycle adipocyte contents or trigger inflammation are known and have been reviewed here ( Figure 3 b). In the coming decades the treatment of obesity and associated chronic diseases will likely remain a major challenge to health systems worldwide; thus, it is timely and important to understand the impact of innate immune signaling—including apoptotic adipocyte-derived signals—in the adipose tissue.
References
- Poon, I.; Lucas, C.; Rossi, A.G.; Ravichandran, K. Apoptotic cell clearance: Basic biology and therapeutic potential. Nat. Rev. Immunol. 2014, 14, 166–180.
- Kourtzelis, I.; Hajishengallis, G.; Chavakis, T. Phagocytosis of Apoptotic Cells in Resolution of Inflammation. Front. Immunol. 2020, 11, 553.
- Röszer, T. Transcriptional control of apoptotic cell clearance by macrophage nuclear receptors. Apoptosis 2016, 22, 284–294.
- Röszer, T. Signal Mechanisms of M2 Macrophage Activation. In The M2 Macrophage; Springer International Publishing: Cham, Switzerland, 2020; pp. 73–97.
- Zizzo, G.; Hilliard, B.A.; Monestier, M.; Cohen, P.L. Efficient Clearance of Early Apoptotic Cells by Human Macrophages Requires M2c Polarization and MerTK Induction. J. Immunol. 2012, 189, 3508–3520.
- Lindhorst, A.; Raulien, N.; Wieghofer, P.; Eilers, J.; Rossi, F.M.V.; Bechmann, I.; Gericke, M. Adipocyte death triggers a pro-inflammatory response and induces metabolic activation of resident macrophages. Cell Death Dis. 2021, 12, 1–15.
- Hahn, P.; Novak, M. Development of brown and white adipose tissue. J. Lipid Res. 1975, 16, 79–91.
- Hull, D. The structure and function of brown adipose tissue. Br. Med. Bull. 1966, 22, 92–96.
- Rosen, E.D.; Spiegelman, B.M. What We Talk About When We Talk About Fat. Cell 2014, 156, 20–44.
- Harms, M.; Seale, P. Brown and beige fat: Development, function and therapeutic potential. Nat. Med. 2013, 19, 1252–1263.
- Bartelt, A.; Heeren, J. Adipose tissue browning and metabolic health. Nat. Rev. Endocrinol. 2013, 10, 24–36.
- Sorisky, A.; Magun, R.; Gagnon, A. Adipose cell apoptosis: Death in the energy depot. Int. J. Obes. 2000, 24, S3–S7.
- Hirsch, J.; Faust, I.M.; Johnson, P.R. What’s New in Obesity: Current Understanding of Adipose Tissue. Morphology 1979, 385–399.
- Nisoli, E.; Cardile, A.; Bulbarelli, A.; Tedesco, L.; Bracale, R.; Cozzi, V.; Morroni, M.; Cinti, S.; Valerio, A.; Carruba, M.O. White adipocytes are less prone to apoptotic stimuli than brown adipocytes in rodent. Cell Death Differ. 2006, 13, 2154–2156.
- Song, C.-X.; Chen, J.-Y.; Li, N.; Guo, Y. CTRP9 Enhances Efferocytosis in Macrophages via MAPK/Drp1-Mediated Mitochondrial Fission and AdipoR1-Induced Immunometabolism. J. Inflamm. Res. 2021, 14, 1007–1017.
- Kuroda, M.; Sakaue, H. Adipocyte Death and Chronic Inflammation in Obesity. J. Med. Investig. 2017, 64, 193–196.
- McLaughlin, T.; Craig, C.; Liu, L.-F.; Perelman, D.; Allister, C.; Spielman, D.; Cushman, S.W. Adipose Cell Size and Regional Fat Deposition as Predictors of Metabolic Response to Overfeeding in Insulin-Resistant and Insulin-Sensitive Humans. Diabetes 2016, 65, 1245–1254.
- Björntorp, P.; Sjöström, L. The Composition and Metabolism in Vitro of Adipose Tissue Fat Cells of Different Sizes. Eur. J. Clin. Investig. 1972, 2, 78–84.
- Boulton, T.J.C.; Dunlop, M.; Court, J.M. Adipocyte growth in the first 2 years of life. J. Paediatr. Child. Health 2008, 10, 301–305.
- Dauncey, M.J.; Gairdner, D. Size of adipose cells in infancy. Arch. Dis. Child. 1975, 50, 286–290.
- Waqas, S.F.H.; Hoang, A.C.; Lin, Y.-T.; Ampem, G.; Azegrouz, H.; Balogh, L.; Thuróczy, J.; Chen, J.-C.; Gerling, I.C.; Nam, S.; et al. Neuropeptide FF increases M2 activation and self-renewal of adipose tissue macrophages. J. Clin. Investig. 2017, 127, 2842–2854.
- Yu, H.; Dilbaz, S.; Coßmann, J.; Hoang, A.C.; Diedrich, V.; Herwig, A.; Harauma, A.; Hoshi, Y.; Moriguchi, T.; Landgraf, K.; et al. Breast milk alkylglycerols sustain beige adipocytes through adipose tissue macrophages. J. Clin. Investig. 2019, 129, 2485–2499.
- Vogel, P.; Read, R.; Hansen, G.; Wingert, J.; Dacosta, C.M.; Buhring, L.M.; Shadoan, M. Pathology of congenital generalized lipodystrophy in Agpat2-/- mice. Vet. Pathol. 2011, 48, 642–654.
- Birk, R.Z.; Rubinstein, M. IFN-α induces apoptosis of adipose tissue cells. Biochem. Biophys. Res. Commun. 2006, 345, 669–674.
- Domingo, P.; Matias-Guiu, X.; Pujol, R.M.; Francia, E.; Lagarda, E.; Sambeat, M.A.; Vázquez, G. Subcutaneous adipocyte apoptosis in HIV-1 protease inhibitor-associated lipodystrophy. AIDS 1999, 13, 2261–2267.
- Ravichandran, K.S. Find-me and eat-me signals in apoptotic cell clearance: Progress and conundrums. J. Exp. Med. 2010, 207, 1807–1817.
- Benoit, M.E.; Clarke, E.; Morgado, P.; Fraser, D.A.; Tenner, A.J. Complement Protein C1q Directs Macrophage Polarization and Limits Inflammasome Activity during the Uptake of Apoptotic Cells. J. Immunol. 2012, 188, 5682–5693.
- Fischer-Posovszky, P.; Wang, Q.; Asterholm, I.W.; Rutkowski, J.; Scherer, P.E. Targeted Deletion of Adipocytes by Apoptosis Leads to Adipose Tissue Recruitment of Alternatively Activated M2 Macrophages. Endocrinology 2011, 152, 3074–3081.
- Shaul, M.E.; Bennett, G.; Strissel, K.J.; Greenberg, A.S.; Obin, M.S. Dynamic, M2-Like Remodeling Phenotypes of CD11c+ Adipose Tissue Macrophages During High-Fat Diet-Induced Obesity in Mice. Diabetes 2010, 59, 1171–1181.
- Luo, B.; Wang, Z.; Zhang, Z.-Y.; Shen, Z.; Zhang, Z. The deficiency of macrophage erythropoietin signaling contributes to delayed acute inflammation resolution in diet-induced obese mice. Biochim. Biophys. Acta (BBA)—Mol. Basis Dis. 2018, 1865, 339–349.
- Han, C.Z.; Ravichandran, K.S. Metabolic Connections during Apoptotic Cell Engulfment. Cell 2011, 147, 1442–1445.
- Yurdagul, A., Jr. Metabolic consequences of efferocytosis and its impact on atherosclerosis. Immunometabolism 2021, 3, e210017.
- Röszer, T.; Menéndez-Gutiérrez, M.P.; Cedenilla, M.; Ricote, M. Retinoid X receptors in macrophage biology. Trends Endocrinol. Metab. 2013, 24, 460–468.
- Vasina, E.M.; Cauwenberghs, S.; Feijge, M.A.H.; Heemskerk, J.W.M.; Weber, C.; Koenen, R.R. Microparticles from apoptotic platelets promote resident macrophage differentiation. Cell Death Dis. 2011, 2, e211.
- Rigamonti, E.; Chinetti-Gbaguidi, G.; Staels, B. Regulation of macrophage functions by PPAR-alpha, PPAR-gamma, and LXRs in mice and men. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1050–1059.
- Gonzalez, N.A.; Bensinger, S.J.; Hong, C.; Beceiro, S.; Bradley, M.N.; Zelcer, N.; Deniz, J.; Ramirez, C.; Díaz, M.; Castrillo, A. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity 2009, 31, 245–258.
- Mukundan, L.; Odegaard, J.I.; Morel, C.R.; Heredia, J.E.; Mwangi, J.W.; Ricardo-Gonzalez, R.R.; Goh, Y.P.S.; Eagle, A.R.; Dunn, S.E.; Chawla, A.; et al. PPAR-delta senses and orchestrates clearance of apoptotic cells to promote tolerance. Nat. Med. 2009, 15, 1266–1272.
- Röszer, T.; Menéndez-Gutiérrez, M.P.; Lefterova, M.I.; Alameda, D.; Núñez, V.; Lazar, M.A.; Fischer, T.; Ricote, M. Autoimmune kidney disease and impaired engulfment of apoptotic cells in mice with macrophage peroxisome proliferator-activated receptor gamma or retinoid X receptor alpha deficiency. J. Immunol. 2011, 186, 621–631.
- Yoon, Y.S.; Kim, S.-Y.; Kim, M.-J.; Lim, J.-H.; Cho, M.-S.; Kang, J.L. PPARgamma activation following apoptotic cell instillation promotes resolution of lung inflammation and fibrosis via regulation of efferocytosis and proresolving cytokines. Mucosal. Immunol. 2015, 8, 1031–1046.
- Kato, Y.; Park, J.; Takamatsu, H.; Konaka, H.; Aoki, W.; Aburaya, S.; Ueda, M.; Nishide, M.; Koyama, S.; Kumanogoh, A.; et al. Apoptosis-derived membrane vesicles drive the cGAS-STING pathway and enhance type I IFN production in systemic lupus erythematosus. Ann. Rheum. Dis. 2018, 77, 1507–1515.
- Zhu, M.; Barbas, A.S.; Lin, L.; Scheuermann, U.; Bishawi, M.; Brennan, T.V. Mitochondria Released by Apoptotic Cell Death Initiate Innate Immune Responses. ImmunoHorizons 2018, 2, 384–397.
- Wasnik, S.; Rundle, C.H.; Baylink, D.J.; Yazdi, M.S.; Carreon, E.E.; Xu, Y.; Qin, X.; Lau, K.-H.W.; Tang, X. 1,25-Dihydroxyvitamin D suppresses M1 macrophages and promotes M2 differentiation at bone injury sites. JCI Insight 2018, 3.
- Das, L.M.; Binko, A.M.; Traylor, Z.P.; Peng, H.; Lu, K.Q. Vitamin D improves sunburns by increasing autophagy in M2 macrophages. Autophagy 2019, 15, 813–826.
- Zhang, Y.; Leung, D.Y.M.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D Inhibits Monocyte/Macrophage Proinflammatory Cytokine Production by Targeting MAPK Phosphatase-1. J. Immunol. 2012, 188, 2127–2135.
- Gunasekar, P.; Swier, V.J.; Fleegel, J.P.; Boosani, C.; Radwan, M.M.; Agrawal, D.K. Vitamin D and macrophage polarization in epicardial adipose tissue of atherosclerotic swine. PLoS ONE 2018, 13, e0199411.
- Zhang, L.-J.; Guerrero-Juarez, C.F.; Hata, T.; Bapat, S.P.; Ramos, R.; Plikus, M.V.; Gallo, R.L. Dermal adipocytes protect against invasive Staphylococcus aureus skin infection. Science 2015, 347, 67–71.
- Fernández, Á.F.; Bárcena, C.; Martínez-García, G.G.; Tamargo-Gómez, I.; Suárez, M.F.; Pietrocola, F.; Mariño, G. Autophagy couteracts weight gain, lipotoxicity and pancreatic β-cell death upon hypercaloric pro-diabetic regimens. Cell Death Dis. 2017, 8, e2970.
- Rosa-Caldwell, M.E.; Brown, J.L.; Lee, D.E.; Blackwell, T.A.; Turner, K.W.; Brown, L.A.; Perry, R.A., Jr.; Haynie, W.S.; Washington, T.A.; Greene, N.P. Autophagy activation, not peroxisome proliferator-activated receptor γ coactivator 1α, may mediate exercise-induced improvements in glucose handling during diet-induced obesity. Exp. Physiol. 2017, 102, 1194–1207.
- Chekeni, F.B.; Elliott, M.; Sandilos, J.K.; Walk, S.F.; Kinchen, J.; Lazarowski, E.R.; Armstrong, A.J.; Penuela, S.; Laird, D.W.; Salvesen, G.S.; et al. Pannexin 1 channels mediate ‘find-me’ signal release and membrane permeability during apoptosis. Nature 2010, 467, 863–867.
- Iacomino, G.; Picariello, G.; D’Agostino, L. DNA and nuclear aggregates of polyamines. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2012, 1823, 1745–1755.
- Maeda, Y.; Rachez, C.; Hawel, L., III; Byus, C.V.; Freedman, L.P.; Sladek, F.M. Polyamines Modulate the Interaction between Nuclear Receptors and Vitamin D Receptor-Interacting Protein 205. Mol. Endocrinol. 2002, 16, 1502–1510.
- Röszer, T. (Ed.) M2 Macrophages in the Metabolic Organs and in the Neuroendocrine System. In The M2, Macrophage; Springer International Publishing: Cham, Schwitzerland, 2020; pp. 171–187.
- Chobot, A.; Górowska-Kowolik, K.; Sokołowska, M.; Jarosz-Chobot, P. Obesity and diabetes-Not only a simple link between two epidemics. Diabetes/Metab. Res. Rev. 2018, 34, e3042.
- Babaei, R.; Schuster, M.; Meln, I.; Lerch, S.; Ghandour, R.A.; Pisani, D.F.; Bayindir-Buchhalter, I.; Marx, J.; Wu, S.; Schoiswohl, G.; et al. Jak-TGFβ cross-talk links transient adipose tissue inflammation to beige adipogenesis. Sci. Signal. 2018, 11, eaai7838.
- Sun, K.; Gao, Z.; Kolonin, M.G. Transient inflammatory signaling promotes beige adipogenesis. Sci. Signal. 2018, 11, eaat3192.
- Geserick, M.; Vogel, M.; Gausche, R.; Lipek, T.; Spielau, U.; Keller, E.; Pfäffle, R.; Kiess, W.; Körner, A. Acceleration of BMI in Early Childhood and Risk of Sustained Obesity. N. Engl. J. Med. 2018, 379, 1303–1312.




