
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Philippe Drabent | + 3264 word(s) | 3264 | 2021-07-29 08:50:35 | | | |
| 2 | Dean Liu | Meta information modification | 3264 | 2021-09-18 08:53:07 | | |
Video Upload Options
Superficial spindle cell mesenchymal tumors form a diverse group of lesions with benign and malignant entities that are often very similar clinically and/or histologically. In children, the patient’s age; the lesion site; the presentation as a mass, nodule, or plaque; and the location in the dermis or subcutis are important features that will guide the dermatologist and the pathologist.
1. Introduction
Pediatric spindle cell mesenchymal neoplasms form a very diverse group of tumors with differing prognoses. Recently, a few entities in the skin and subcutis have been described in morphological and molecular terms. There has been particular interest in CD34-positive spindle cell neoplasms, in view of their importance as a differential diagnosis for dermatofibrosarcoma protuberans (DFSP). One of the first of these tumors to have been described was medallion-like dermal dendrocyte hamartoma (MLDDH) [1], a tumor with a peculiar clinical aspect characterized by a well-circumscribed erythematous atrophic plaque and (under the microscope) a dermal proliferation of CD34-positive, S100-negative spindle cells. This tumor was first considered to be a hamartoma but has since been classified as a neoplasm of fibroblastic lineage and not of dendrocytic lineage [2]. The term “plaque-like CD34-positive dermal fibroma” (PDF) has therefore been preferred in the more recent literature, and there is a tendency to consider MLDDH and PDF as one and the same entity. MLDDH/PDF shares clinical and pathological features with fibroblastic connective tissue nevus (FCTN), a variant of connective tissue nevus first characterized in 2012 by Fletcher and de Feraudy in a series of 25 cases [3]. Although lipofibromatosis (LPF) mainly involves the subcutis, it also contains a spindle cell component admixed with adipose tissue and can be a differential diagnosis for superficial spindle cell tumors in children. In 2016, a CD34-positive spindle cell neoplasm with an interesting, recurrent genetic anomaly was described: LPF-like neural tumor (LPF-NT), which was initially associated with NTRK 1 gene fusions [4]. As indicated by its name, this tumor is very similar to LPF. It is usually located in the subcutis but infiltrates the surrounding adipose tissue, and it is composed of spindle cells arranged in fascicles. Immunohistochemically, LPF-NTs are positive for both CD34 and S100 protein. Importantly, LPF-NTs are characterized by gene fusions in the NTRK family of genes, or (less frequently) in the RAF family of genes [5] or RET [6]. The clinical course is benign in most but not all cases. The newest member of these pediatric spindle cell neoplasms is plexiform myofibroblastoma (PM), as described in 2020 by Papke and Fletcher [7]. This entity broadens the spectrum of superficial fibroblastic/myofibroblastic tumors and is particularly similar to plexiform fibrohistiocytic tumor (PFHT). However, the two must be distinguished because PFHT is a low-grade sarcoma and PM is benign.
2. The “Connective Tissue Nevus/Medallion-Like Dermal Dendrocyte Hamartoma/Plaque-Like Cd34-Positive Dermal Fibroma/Superficial Fibrous Hamartoma of Infancy” Spectrum
Fibroblastic connective tissue nevus (FCTN) was described in 2012 in a series of 25 cases. This variant of connective tissue nevus has some clinical and histological particularities [3][8]. It typically appears during the first decade of life as a slowly growing, painless, plaque-like, or nodular skin lesion, and is mainly located on the trunk, head, and neck and less frequently on the limbs. Girls are more often affected than boys (sex ratio: 0.5–0.6). The lesions range from 0.18 to 2.0 cm in size, and FCTN is rarely diagnosed on the basis of clinical signs alone. Histologically, FCTN is a poorly circumscribed dermal lesion that arises in the reticular dermis and extends to the superficial subcutis. In 70% of cases, there is overlying papillomatosis of the epidermis. In about 60% of cases, abnormally superficial adipocytes are seen in the reticular dermis. The tumor is composed of short, intersecting fascicles of bland, spindle-shaped fibroblasts/myofibroblasts with weakly eosinophilic cytoplasm and elongated nuclei with no atypia and no mitoses. It extends between the collagen bundles, around the appendages and into the subcutaneous septa. The most useful immunostaining marker is CD34, which is positive in 87% of cases—albeit often weakly and in focal sites. Smooth muscle actin (SMA) is weakly and focally present in less than 50% of cases, and S100 is always absent ( Figure 1 ).
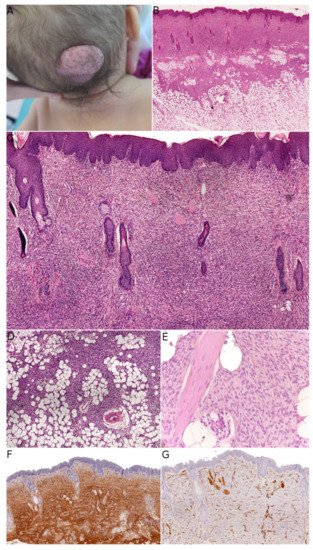
FCTN broadens the spectrum of connective tissue nevi (CTN), which are otherwise classified with regard to their most abundant component: collagen, elastin, or proteoglycans. In each of these categories, various entities have been described and differ in their clinical characteristics: inherited vs. acquired lesions, and an association with a genetic disorder (Buschke–Ollendorff syndrome, proteus syndrome, tuberous sclerosis complex, or multiple endocrine neoplasia type 1) [9]. A mixed pattern (in which both collagen and elastin are more abundant) appears to be more frequent than initially thought [8]. In line with this observation by Saussine et al. , we suggest that the “connective tissue nevi spectrum” might be even broader and overlap with other entities.
MLDDH was the first of these entities to be described, in a series of three cases in 2004 [1]. The clinical presentation was characteristic: a medallion-like, well-circumscribed, brownish, erythematous lesion with a finely wrinkled, atrophodermic surface. The lesions were initially described as congenital, situated on the neck and the upper part of the trunk, and affecting girls. Some subsequently reported cases of MLDDH occurred in boys, appeared shortly after birth, or affected the limbs [10][11][12][13]. In 2010, Kutzner and colleagues described similar lesions on the limbs in four adults and a lesion of the neck in a 9-year-old boy [2]. The name “plaque-like CD34-positive dermal fibroma” (PDF) was suggested for these lesions because they appeared after birth and were more like tumors than hamartomas. The investigators also argued that dendrocytes are known to proliferate during wound healing and that PDF/MLDDH might be reactive lesions related to trauma. Histologically, MLDDH and PDF are indistinguishable [14]. The lesion consists of a superficial dermal spindle and sometimes ovoid cell proliferation, mainly occupying the reticular dermis but occasionally infiltrating the subcutis. The degree of epidermal atrophy is variable, and elastic fibers are often diminished or fragmented in the lesion. The stroma may contain mast cells and/or venules with a dilated lumen. The proliferating cells stain positive for CD34 ( Figure 2 ). In their series of PDF cases, Kutzner et al. described a specific but inconstant pattern, with the fibroblasts in the upper part of the lesion oriented vertically to the epidermis and those in the lower part of the lesion oriented horizontally. The investigators also noted that PDF in adults never extended into the deep dermis and subcutaneous septa. Given this slight morphological distinction and the difference in clinical presentation between adults and children, it is still not clear whether PDF and MLDDH are one and the same lesion or if MLDDH is a congenital/infantile variant of PDF. It is noteworthy that MLDDH tends to extend into the subcutis, making it very challenging to differentiate between MLDDH and FCTN, as recently emphasized in a few case reports [14][15]. Both lesions may show slight infiltration clinically, although FCTN may be more irregular in shape [15]. Other differential diagnoses of MLDDH/PDF include dermatomyofibroma (which rather presents in young adults, shows fascicles preferentially oriented parallel to the epidermis, and is SMA-positive), non-pigmented cellular blue nevus (which is easily ruled out by immunophenotyping), plaque-like superficial neurofibroma (again, easily ruled out by immunophenotyping), and CD34-positive sclerotic fibroma of the skin (also known as “plywood fibroma”, featuring a more nodular or stellate-like silhouette with characteristic clefts; it is extremely rare in children and is mainly associated with Cowden syndrome).
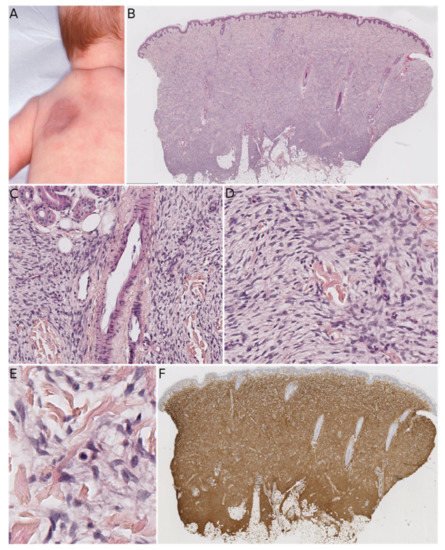
All the above-discussed lesions have similarities: they can be seen during infancy and can affect the upper back/trunk or upper limbs. Some present mostly as plaque-like lesions (MLDDH/PDF), some present as masses (FHI), and some present as both (FCTN). Under the microscope, all these lesions may show a proliferation of fibroblastic/myofibroblastic cells arranged in fascicles in the reticular dermis. For these reasons, it is important to consider all the available data (i.e., clinical; morphological; and, if necessary, molecular) when confronted with a lesion that belongs to this spectrum. In most cases, the presence of one or more of the characteristics described above will help the pathologist to make the right diagnosis (see Table 1 ). The most troublesome differential diagnosis is plaque-like DFSP, and therefore all equivocal cases should be screened for the COL 1A 1-PDGFB gene rearrangement. The literature data show that the combination of RT-PCR and FISH is the most sensitive diagnostic method [16]. For cases in which the COL 1A 1-PDGFB rearrangement is not found, our experience shows that it is best to take a closer look at the morphology: if there is any suspicion of DFSP, other gene rearrangements should be sought using RNA sequencing (RNA-seq) or next-generation sequencing techniques; if the morphology is not suggestive of DFSP, the most likely diagnosis will fall within the “FCTN-MLDDH/PDF” spectrum. Unfortunately, a characteristic, recurrent genetic abnormality has not yet been evidenced in cases of acquired CTN or MLDDH/PDF.
| Age | Sex | Site | Morphology | IHC | Genetic Abnormality | |
|---|---|---|---|---|---|---|
| FCTN | <10 y | F > M | Trunk, head and neck, limbs | Poorly delimited, reticular dermis with extension into the subcutis; short bundles of (myo)fibroblasts | CD34+ weak, multifocal (87%) S100− |
|
| MLDDH/PDF | Infants and older children | F > M | Neck, upper trunk, limbs | Poorly delimited, reticular dermis with possible extension into the subcutis; spindle and/or ovoid cells; variable epidermal atrophy, elastic fibers diminished | CD34+ S100− |
|
| Superficial FHI | <2 y | M > F | Axilla, upper limbs, upper back | Subcutis, three components: mature fibrous tissue, mature adipose tissue, and immature mesenchymal tissue; possible pseudoangiomatous or hyalinized areas | Not helpful | EGFR exon 20 ins/dup |
| DFSP | Older children | M > F | Trunk, proximal areas of the limbs, head and neck | Diffuse infiltration of the dermis and subcutis; high cellularity; spindle cells in a storiform pattern | CD34+ S100− |
COL1A1-PDGFB, other rare fusions |
3. The “Lipofibromatosis/Lipofibromatosis-Like Neural Tumor” Spectrum—The “Lipofibromatosis-Like Pattern”
In a study reported in 2018, Suurmeijer et al. described a new group of spindle cell tumors with S100 and CD34 co-expression and recurrent gene fusions. In their series of 25 cases, 8 were located in subcutaneous tissue in children. Interestingly, there were four other pediatric cases involving visceral organs (the stomach and the rectum) or bones (the maxilla and the mandible). The remaining cases were in adults and featured a variety of superficial and deep sites. All the tumors had similar features: monomorphic cytology; stromal and perivascular hyalinization; immunopositivity for S100 and CD34; and RAF 1, BRAF , or NTRK 1/2 fusions. Hence, it is likely that LPF-NT belongs to a wider group of spindle cell tumors sharing these characteristics. Moreover, some tumors showed possible signs of malignancy: scattered pleomorphic and/or multinucleate cells, highly cellular fascicular growth, or a primitive appearance. In fact, an 18-year-old patient in Suurmeijer et al. ’s series died of metastatic disease, and other patients had metastases (in the lung and other sites) or disease recurrence. This aggressive clinical course was seen both for superficial and deep tumors, and in patients as young as 4 years old. It is most likely that tumors defined by RAF 1, BRAF , or NTRK 1/2 fusions form a spectrum whose behavior can vary from benign to malignant.
In 2019, Antonescu et al. reported on six cases with tumors that resembled LPF-NT but harbored RET gene rearrangements. Five of their cases occurred in children (including four infants): three of these were diagnosed as LPF-NT, one was diagnosed as an “infantile fibrosarcoma-like” tumor (involving the chest wall), and the last was diagnosed as infantile fibrosarcoma/cellular mesoblastic nephroma (involving the kidney with bilateral lung and brain metastases). Of the three cases of LPF-NT, one was a superficial tumor of the ankle, one was a deep tumor of the foot, and the last arose in the abdominal wall. None of the LPF-NTs recurred, although the tumor had an aggressive clinical course in the two cases with malignant histological features. These interesting results broadened the spectrum of spindle cell tumors with recurrent translocations and emphasized two important features of this group: (i) the predominant value of molecular characteristics (i.e., recurrent gene fusions) in the classification of these tumors with a wide range of morphologies (from LPF-NT to infantile fibrosarcoma to MPNST-like tumors), and (ii) the probable continuum in the degree of malignancy.
This second concept was already foreseen in the first description of LPF-NT by Agaram et al., who described a case of superficial LPF-NT of the leg in a young woman who developed lung metastases [4]. This tumor had the LMNA–NTRK 1 fusion, and its possible identity as a malignant counterpart of LPF-NT was mentioned by the researchers at the time.
Besides the very similar FHI and LPF, the differential diagnoses of LPF-NT also include other entities, the most important of which is calcifying aponeurotic fibroma (CAF). This lesion is a superficial, slow-growing, ill-circumscribed tumor of the hands or feet that arises in children and adolescents. Histologically, CAF has a typically two-phase morphology that combines moderately cellular areas (with fibroblastic cells arranged in fascicles) and partly calcified nodules (with a fibrocartilage appearance, small epithelioid fibroblasts, and osteoclast-like giant cells). In 2016, a recurrent FN 1–EGF fusion was found in eight out of nine cases, either by RNA-seq, RT-PCR, or FISH [17]. This fusion results in overexpression of epidermal growth factor (EGF), which is detected using immunohistochemistry in all cases, even when a gene fusion cannot be detected. Interestingly, in a report in 2019, the FN 1–EGF fusion was found in four cases of apparently typical LPF that recurred as CAF [18]. In this study of 20 LPF cases, the other fusions involved genes that encode ligands for the EGF receptor ( EGF , TGFA , HBEGF ) or that encode RTKs as 3′-partners ( ROS 1, PDGFRB , RET ). The researchers mentioned that these findings were strongly suggestive of a link between LPF and CAF; in some cases, LPF might correspond to “early” CAF characterized by a prominent adipocytic component and no calcification. It remains to be established whether the cases with a FN 1–EGF fusion or another fusion constitute the same entity or whether the cases with FN 1–EGF fusions are CAF and the cases with other fusions are LPF. However, this interesting study highlighted potential genetic variability in LPF, with a diverse range of gene fusions and proximity to CAF. Lastly, the differential diagnoses for LPF and LPF-NT in children include DFSP, which can be distinguished by its negativity for S100 protein and the presence of the characteristic gene fusions.
4. Plexiform Myofibroblastoma
Plexiform myofibroblastoma (PM) is a newly described entity. In a series of 36 cases, Papke et al. characterized this tumor; it had a broad age range and no male or female predominance (19 females and 17 males) [7]. PM can be congenital but may appear as late as the age of 50, although the median age at onset is 9.5 years. A total of 24 of Papke et al. ’s 36 patients were children. In the pediatric population, the tumor was located (i) mostly in the neck and upper back; (ii) to a lesser extent in the lower back, axilla, chest wall and abdominal wall; and (iii) in one case in the left lower leg. The lesions were multifocal in three cases (all below the age of 2) and extended to the occiput in one case. Interestingly, two of the multifocal cases were brothers, raising the question of a genetically inherited anomaly (though not discussed by Papke et al.). None of the pediatric cases—even those with positive margins—showed any recurrence of the disease after surgical resection. Grossly, PM has a tan/white cut surface and ranges from 0.6 to 4 cm in size (in pediatric cases). Histologically, it is composed of fascicles in a plexiform pattern, which extend within the reticular dermis and subcutis. The tumor cells are fibroblastic/myofibroblastic, with a weakly eosinophilic, elongated cytoplasm and ovoid to tapered nuclei with no atypia. The characteristic collagenous stroma is reminiscent of mammary-like myofibroblastoma, with at least focal hyalinization in 35% of cases. Some cases show nodular fasciitis-like areas. The stroma may be focally myxoid. Mitoses are rare, with no more than 4 per 2 mm 2. There is no necrosis. The most frequently positive immunohistochemical markers are SMA (in 84% of cases), CD34 (in 54%), and desmin (in 43%) ( Figure 6 ). The tumor is also negative for S100 protein, and there is no nuclear translocation of beta-catenin. To date, no recurrent genetic alterations have been identified. Only one case had a missense mutation in FGFR 2, and another had a probable germline mutation in both MUTYH and BRIP 1; the significance of these genetic abnormalities remains unclear.
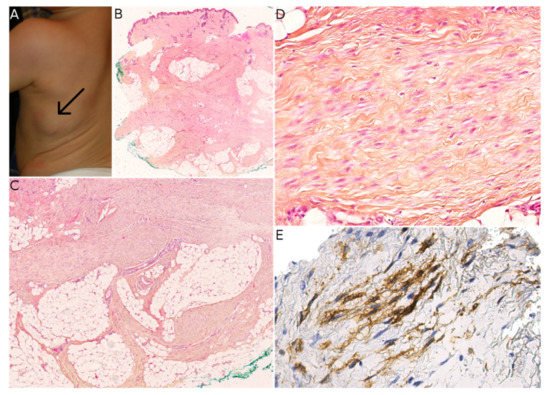
There are many potential differential diagnoses for PM. The closest mimic is the fibroblastic variant of plexiform fibrohistiocytic tumor (PFHT), a superficial tumor of children and young adults (median age at onset: ≈15) that occurs mainly in the upper limbs but also in the axilla, back, neck, and chest wall [19]. Like PM, PFHT has a characteristic plexiform architecture. Most cases have a fibrohistiocytic or mixed fibroblastic–fibrohistiocytic morphology. Cases with a predominant fibroblastic component are the hardest to differentiate from PM, especially if no osteoclast-like giant cells or histiocytoid cells are present. CD68 immunostaining is useful for highlighting the histiocytoid component. PM is usually SMA-positive (which is not helpful for the differential diagnosis), and CD34 is usually negative. As discussed by Papke et al., it is possible that cases of purely fibroblastic PFHT are in fact PM. Interestingly, there are no reports of recurrence or metastasis in purely fibroblastic PFHT [7]. It is important to differentiate between PM and PFHT; the former is benign and does not seem to recur in children, whereas the latter is a low-grade sarcoma with metastatic potential.
Other differential diagnoses for PM lack the typical plexiform pattern and can thus be ruled out quite easily: desmoid fibromatosis, nodular fasciitis, dermatomyofibroma, plaque-like myofibroblastic tumor, FCTN, and FHI.
References
- Rodríguez-Jurado, R.; Palacios, C.; Durán-McKinster, C.; Mercadillo, P.; Orozco-Covarrubias, L.; Saez-De-Ocariz, M.D.M.; Ruiz-Maldonado, R. Medallion-like dermal dendrocyte hamartoma: A new clinically and histopathologically distinct lesion. J. Am. Acad. Dermatol. 2004, 51, 359–363.
- Kutzner, H.; Mentzel, T.; Palmedo, G.; Hantschke, M.; Rütten, A.; Paredes, B.E.; Schärer, L.; Guillen, C.S.; Requena, L. Plaque-like CD34-positive Dermal Fibroma (“Medallion-like Dermal Dendrocyte Hamartoma”). Am. J. Surg. Pathol. 2010, 34, 190–201.
- Wisell, J. Fibroblastic Connective Tissue Nevus: A Rare Cutaneous Lesion Analyzed in a Series of 25 Cases. Yearb. Pathol. Lab. Med. 2013, 2013, 97–98.
- Agaram, N.P.; Zhang, L.; Sung, Y.-S.; Chen, C.-L.; Chung, C.T.; Antonescu, C.R.; Fletcher, C.D. Recurrent NTRK1 Gene Fusions Define a Novel Subset of Locally Aggressive Lipofibromatosis-like Neural Tumors. Am. J. Surg. Pathol. 2016, 40, 1407–1416.
- Suurmeijer, A.; Dickson, B.C.; Swanson, D.; Zhang, L.; Sung, Y.-S.; Cotzia, P.; Fletcher, C.D.M.; Antonescu, C.R. A novel group of spindle cell tumors defined by S100 and CD34 co-expression shows recurrent fusions involving RAF1, BRAF, and NTRK1/2 genes. Genes Chromosomes Cancer 2018, 57, 611–621.
- Antonescu, C.R.; Dickson, B.C.; Swanson, D.; Zhang, L.; Sung, Y.-S.; Kao, Y.-C.; Chang, W.-C.; Ran, L.; Pappo, A.; Bahrami, A.; et al. Spindle Cell Tumors With RET Gene Fusions Exhibit a Morphologic Spectrum Akin to Tumors With NTRK Gene Fusions. Am. J. Surg. Pathol. 2019, 43, 1384–1391.
- Papke, D.J.; Al-Ibraheemi, A.; Fletcher, C.D. Plexiform Myofibroblastoma. Am. J. Surg. Pathol. 2020, 44, 1469–1478.
- Saussine, A.; Marrou, K.; Delanoé, P.; Bodak, N.; Hamel, D.; Picard, A.; Sassolas, B.; De Prost, Y.; LeMerrer, M.; Fraitag, S.; et al. Connective tissue nevi: An entity revisited. J. Am. Acad. Dermatol. 2012, 67, 233–239.
- Arora, H.; Falto-Aizpurua, L.; Cortés-Fernandez, A.; Choudhary, S.; Romanelli, P. Connective Tissue Nevi: A Review of the Literature. Am. J. Dermatopathol. 2017, 39, 325–341.
- Marque, M.; Bessis, D.; Pedeutour, F.; Viseux, V.; Guillot, B.; Fraitag-Spinner, S. Medallion-like dermal dendrocyte hamartoma: The main diagnostic pitfall is congenital atrophic dermatofibrosarcoma. Br. J. Dermatol. 2008, 160, 190–193.
- Shah, K.N.; Anderson, E.; Junkins-Hopkins, J.; James, W.D. Medallion-Like Dermal Dendrocyte Hamartoma. Pediatr. Dermatol. 2007, 24, 632–636.
- Ducharme, E.E.; Baribault, K.E.; Husain, S.; Engler, D.E. Medallion-like dermal dendrocyte hamartoma in a 36-year-old male. J. Am. Acad. Dermatol. 2008, 59, 169–172.
- Restano, L.; Fanoni, D.; Colonna, C.; Gelmetti, C.; Berti, E. Medallion-Like Dermal Dendrocyte Hamartoma: A Case Misdiagnosed as Neurofibroma. Pediatr. Dermatol. 2010, 27, 638–642.
- Bouaoud, J.; Fraitag, S.; Soupre, V.; Mitrofanoff, M.; Boccara, O.; Galliot, C.; Bodemer, C.; Picard, A.; Khonsari, R.H. Congenital fibroblastic connective tissue nevi: Unusual and misleading presentations in three infantile cases. Pediatr. Dermatol. 2018, 35, 644–650.
- Horikawa, H.; Sato, T.; Gomi, H.; Yamazaki, K.; Ishida, Y.; Yuzaki, I.; Fukuzumi, S. Medallion-like dermal dendrocyte hamartoma: A rare congenital CD34-positive dermal lesion clinically and pathologically overlapping with fibroblastic connective tissue nevus. Pediatr. Dermatol. 2019, 36, 397–399.
- Patel, K.U.; Szabo, S.S.; Hernandez, V.S.; Prieto, V.G.; Abruzzo, L.V.; Lazar, A.; López-Terrada, D. Dermatofibrosarcoma protuberans COL1A1-PDGFB fusion is identified in virtually all dermatofibrosarcoma protuberans cases when investigated by newly developed multiplex reverse transcription polymerase chain reaction and fluorescence in situ hybridization assays. Hum. Pathol. 2008, 39, 184–193.
- Puls, F.; Hofvander, J.; Magnusson, L.; Nilsson, J.; Haywood, E.; Sumathi, V.P.; Mangham, D.C.; Kindblom, L.-G.; Mertens, F. FN1-EGFgene fusions are recurrent in calcifying aponeurotic fibroma. J. Pathol. 2016, 238, 502–507.
- Al-Ibraheemi, A.; Folpe, A.L.; Perez-Atayde, A.R.; Perry, K.; Hofvander, J.; Arbajian, E.; Magnusson, L.; Nilsson, J.; Mertens, F. Aberrant receptor tyrosine kinase signaling in lipofibromatosis: A clinicopathological and molecular genetic study of 20 cases. Mod. Pathol. 2019, 32, 423–434.
- Moosavi, C.; Jha, P.; Fanburg-Smith, J.C. An update on plexiform fibrohistiocytic tumor and addition of 66 new cases from the Armed Forces Institute of Pathology, in honor of Franz M. Enzinger, MD. Ann. Diagn. Pathol. 2007, 11, 313–319.




