
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anayt Ulla | + 7062 word(s) | 7062 | 2021-08-16 05:37:53 | | | |
| 2 | Vivi Li | Meta information modification | 7062 | 2021-09-08 08:49:24 | | |
Video Upload Options
Skeletal muscle atrophy is the decrease in muscle mass and strength caused by reduced protein synthesis/accelerated protein degradation. Various conditions, such as denervation, disuse, ageing, chronic diseases, heart disease, obstructive lung disease, diabetes, renal failure, AIDS, sepsis, cancer, and steroidal medications, can cause muscle atrophy. Mechanistically, inflammation, oxidative stress, and mitochondrial dysfunction are among the major contributors to muscle atrophy, by modulating signalling pathways that regulate muscle homeostasis. To prevent muscle catabolism and enhance muscle anabolism, several natural and synthetic compounds have been investigated. Recently, polyphenols (i.e., natural phytochemicals) have received extensive attention regarding their effect on muscle atrophy because of their potent antioxidant and anti-inflammatory properties. Numerous in vitro and in vivo studies have reported polyphenols as strongly effective bioactive molecules that attenuate muscle atrophy and enhance muscle health.
1. Introduction
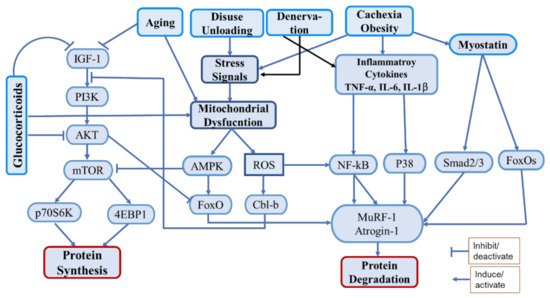
2. Polyphenols in Managing Muscle Atrophy and Muscle Health
2.1. Phenolic Acids
| Class | Sub-Class | Compound/Derivatives/Compounds Mixture | Model | Effects | References |
|---|---|---|---|---|---|
| Phenolic Acid | Hydroxybenzoic Acid | Gallic Acid (GA) | C2C12 Myotubes | Increased Mitochondrial Function and mitochondrial biogenesis, Enhanced myosin heavy chain expression |
[29] |
| EC, EGC and GA | Normal and oxidative stress-induced C2C12 cells | Increased myotube density upregulated genetic expression of myogenic factors |
[30] | ||
| Ellagic acid | CCL4-induced muscle injury in rats | Reduced muscle tissue damage induced caspase-3, Nrf-2 and antioxidant enzymes suppressed inflammatory markers |
[31] | ||
| Ellagic Acid | Cuprizone-induced multiple sclerosis model in mice | Protects muscle tissue prevented mitochondrial dysfunction and oxidative stress |
[32] | ||
| Urolithin A | C2C12 cells, young and HF-induced aged mice | Induced autophagy and mitophagy both invitro and in vivo increased muscle function improved exercise capacity |
[33] | ||
| Urolithin A | Mouse model of DMD | Induced mitophagy and improved muscle function and MuSCs regeneration increased skeletal muscle respiratory capacity |
[34] | ||
| Urolithin A | C57BL/6 mice | Strengthen skeletal muscle and angiogenesis Increase ATP and NAD+ level Upregulates angiogenic pathways |
[35] | ||
| Urolithin B | C2C12 myotubes and denervation-induced mice | Enhanced growth and differentiation of C2C12 myotubes and muscle hypertrophy Increased protein synthesis and suppressed UPS |
[36] | ||
| Pomegranate extract | TNF-α induced muscle atrophy in mice | Prevented muscle wasting suppressed cytokines and NF-kB level activated protein synthesis pathway |
[37] | ||
| Hydroxycinnamic Acid | Ferulic acid | Mouse C2C12 myotubes | Regulates muscle fiber type formation activated SIRT1/AMPK pathway Increased PGC-1α expression |
[38] | |
| Ferulic Acid | Corticosteroid-Induced Rat Myopathy | Induced growth of fast glycolytic and slow oxidative muscle fiber suppressed myostatin and oxidative stress |
[39] | ||
| Ferulic Acid | Zebrafish model | Enhanced muscle mass and MyHC fast type Increased myogenic transcriptional factors activated zTOR/p70S6K/4EBP1 |
[40] | ||
| Chlorogenic acid | Resistance training-induced rat model | improved muscle strength by promoting mitochondrial function and cellular energy metabolism | [41] | ||
| Caffeic acid phenethyl ester | Eccentric exercise-induced skeletal muscle injury in rats | Protected skeletal muscle damage down-regulated NF-κB activation |
[42] | ||
| Caffeic acid | Human fibroblast cell line | Decreased spinal muscular atrophy increased SMN2 transcripts |
[43] | ||
| Coffee | In-vitro and in-vivo model | Skeletal muscle hypertrophy and myoblast differentiation | [44] | ||
| P-Coumaric acid | C2C12 myotubes | Reduce differentiation of muscle cells by reducing MyoD and Myogenin. | [45] |
2.2. Flavonoids
| Class | Sub-Class | Compound/Derivatives | Model | Effects | References |
|---|---|---|---|---|---|
| Flavonoids | Flavanols | EC | Young and old C57BL/6 mice and human tissue samples | Decreased myostatin and β-galactosidase Increased follistatin and markers of muscle growth and differentiation |
[52] |
| EC | Young and old C57BL/6 mice | Increased survival of aged mice prevented muscle wasting |
[53] | ||
| EC | HLS-induced muscle atrophy in mice | Counteracts muscle degradation maintains muscle angiogenesis and mitochondrial biogenesis |
[54] | ||
| EC | Denervation-induced muscle atrophy in rats | Reduced muscle wasting down regulated Foxo1a, Atrgoin-1 and MuRF-1 |
[55] | ||
| EC | Ambulatory adults with Becker Muscular Dystrophy | Induced mitochondrial biogenesis and muscle regeneration factors increased follistatin decreased myostatin |
[56] | ||
| EC | C2C12 myotubes | Stimulates mitochondrial biogenesis and cell growth through GPER | [57] | ||
| EC | C2C12 cells | Enhanced myogenic differentiation and MyoD activity | [58] | ||
| EC | HF-induced muscle damage in aged mice | Improved physical performance increased follistatin and MEF2A decreased Foxo1a and MuRF-1 |
[59] | ||
| EC, ECG, EGCG | 3D-clinorotation induced C2C12 atrophy | Suppressed Atrogin-1 and MuRF-1 dephosphorylated the ERK signaling |
[60] | ||
| EGCG | Aging-induced sarcopenic rat model | Attenuated muscle atrophy and protein degradation Increased protein synthesis |
[61] | ||
| EGCG | Aged mice and late passaged C2C12 cells | Attenuated muscle atrophy and protein degradation upregulated miR-486-5p |
[62] | ||
| EGCG | Starvation and TNF-α-induced C2C12 atrophy | Attenuation of muscle wasting inhibited protein degradation and activated protein synthesis |
[63] | ||
| EGCG | Tumor induced LLC and C57Bl/6 mice atrophy | Attenuates muscle atrophy inhibits NF-κB, atrogin-1 and MuRF-1 |
[64] | ||
| EGCG | Mouse model of Duchenne muscular dystrophy | Protected from muscle necrosis improved muscle functions |
[65] | ||
| EGCG | HLS-induced muscle atrophy in aged rats | Improved Plantaris muscle weight and fiber size reduced pro-apoptotic signaling |
[66] | ||
| EGCG | HLS-induced muscle atrophy in aged rats | Maintained autophagy signaling in disuse muscle prevented autophagy and apoptosis during reloading |
[67] | ||
| EGCG | Dex-induced C2C12 myotubes and tail-suspended mice | Prevented muscle atrophy suppressed MuRF1 |
[68] | ||
| Green tea extracts (EGCG) |
HF diet-induced muscle atrophy in SAMP8 mice | Ameliorated HF-induced muscle wasting Decreased insulin resistance and LECT2 expression |
[69] | ||
| EGCG | Live skeletal muscle fibers model | Promoted nuclear efflux of Foxo1 activated PI3K/Akt pathway |
[70] | ||
| Catechin (ECG+EGCG) | C2C12 cells and Cardiotoxin induced C56BL/6 mice | Stimulated muscle stem cell activation and differentiation for muscle regeneration. | [71] | ||
| Catechin | Downhill running-induced muscle damage in ICR mice | Attenuated downhill running-induced muscle damage suppressed oxidative stress and inflammation |
[72] | ||
| catechins | Tail-suspension induced muscle atrophy in mice | Minimized contractile dysfunction and muscle atrophy decreased oxidative stress |
[73] | ||
| Flavanones | Hesperidin | Human skeletal muscle cell and mice | Reverted aging-induced decrease in muscle fiber size increased mitochondrial function and running performance reduced oxidative stress |
[74] | |
| Naringenin | L6 and C2C12 cells | Delays skeletal muscle differentiation | [75] | ||
| 8-Prenylnaringe-nin (8-PN) | Denervated Mice | Prevented muscle atrophy suppressed Atrogin-1 Phosphorylated Akt |
[76] | ||
| 8-PN | C2C12 myotubes and casting-induced muscle atrophy in mice | Reversed casting-induced loss of tibialis anterior muscle activated PI3K/Akt/mTOR pathway |
[77] | ||
| Flavones | Apigenin | Aged Mice | Relieved muscle atrophy and increased myofiber size inhibited hyperactive mitophagy/ autophagy and apoptosis. |
[78] | |
| Apigenin | Obesity-induced muscle atrophy in Mice | Attenuated muscle atrophy and mitochondrial dysfunctions. | [79] | ||
| Apigenin | Denervated-induced muscle atrophy in mice | Protected muscle loss Upregulated MyHC reduced TNF-α and MuRF-1 level |
[80] | ||
| Apigenin | C57BL/6 mice and C2C12 cells | Promotes skeletal muscle hypertrophy enhanced myogenic differentiation upregulated Prmt7-PGC-1α-GPR56 pathway |
[81] | ||
| Flavones | LPS-induced muscle atrophy in C2C12 myotube | Prevented myotube atrophy suppressed Atrgoin-1 inhibited JNK phosphorylation |
[82] | ||
| Luteolin | LLC-induced muscle atrophy in mice | Prevented muscle atrophy downregulated MuRF-1, Atrgoin-1, cytokines/inflammatory markers |
[83] | ||
| Luteolin | Dex-Induced muscle atrophy in mice | Prevented muscle atrophy Induced antioxidant and antiapoptotic activity |
[84] | ||
| Isoflavones | Isoflavone (genistein and daidzein) | TNF-α induced C2C12 myotubes | Suppressed MuRF1 promoter activity and myotube atrophy | [85] | |
| Isoflavone | Tumor-induced muscle atrophy in mice | Prevented muscle wasting suppressed ubiquitin ligases expression |
[86]. | ||
| Isoflavone | Denervation-induced muscle atrophy in mice | Prevented muscle fiber atrophy decreased apoptosis-dependent signaling. |
[87] | ||
| Genistein | Denervation-induced muscle atrophy in mice | Mitigated soleus muscle atrophy | [88] | ||
| Genistein | C2C12 myoblast | Enhanced proliferation and differentiation downregulated miR-222 |
[89] | ||
| Daidzein | Young female mice | Down-regulated ubiquitin-specific protease 19 increased soleus muscle mass |
[90] | ||
| Daidzein | Cisplatin-induced LLC bearing mice |
Alleviated skeletal muscle atrophy prevented protein degradation |
[91] | ||
| Daidzein | C2C12 cells | Promotes myogenic differentiation and myotube hypertrophy | [92] | ||
| Soy protein | Rats | Improved muscle function | [93] | ||
| Formononetin (isoflavone) | CKD rats and TNF-α-induced C2C12 myotubes atrophy | Prevented muscle wasting suppressed MuRF-1, MAFbx and myostatin expression Phosphorylated PI3K, Akt and FoxO3a |
[94] | ||
| Glabridin | Dex-induced atrophy (in vitro and in vivo) | Inhibited protein degradation and muscle atrophy in vitro and in vivo | [95] | ||
| Flavonols | Quercetin | HF-diet induced muscle atrophy in mice | Protected muscle mass and muscle fiber size reduced ubiquitin ligases and inflammatory cytokines |
[96] | |
| Quercetin | TNF-α induced myotube and Obesity induced mice muscle atrophy | Averted muscle atrophy Upregulated HO-1 and Nrf2 Inactivates NF-kB |
[97] | ||
| Quercetin | Murine C26 cancer-cachexia model | Prevented body and muscle weight loss tended to decrease Atrgoin-1 and MuRF-1 |
[98] | ||
| Quercetin | A549 cells injected tumor model in mice | Prevented loss of GM and protein degradation Increased MyHC level |
[99] | ||
| Quercetin | Dex-Induced C2C12 cell injury | Increased C2C12 cell viability exerted antiapoptotic effects reduce oxidative stress regulates mitochondrial membrane potential |
[100] | ||
| Quercetin | Dex-induced-muscle atrophy in mice | Prevented muscle loss reduced myostatin, atrgoin-1 and MuRF-1 increased Akt phosphorylation |
[101] | ||
| Quercetin | Tail-suspension induced muscle atrophy in mice | Prevented GM loss suppressed ubiquitin ligases and lipid peroxidation |
[102] | ||
| Quercetin | Denervation-induced muscle atrophy in mice | Prevented muscle atrophy suppressed mitochondrial hydrogen peroxide generation elevated mitochondrial biogenesis |
[103] | ||
| Quercetin Glycosides | Male C57BL/6J aged mice | Improved motor performance Increased muscle mass |
[104] | ||
| Quercetin | Mice | Mitochondrial biogenesis Increased endurance and running capacity |
[105] | ||
| Morin | LLC cell-bearing mice and C2C12 myotubes atrophy | Suppressed muscle wasting and myofiber size reduction by binding to ribosomal protein S10 |
[106] | ||
| Morin | Dex- induced atrophy of C2C12 skeletal myotubes | Prevented protein degradation reduced oxidative stress, Atrogin-1, MuRF-1 and Cbl-b Phosphorylated Foxo3a |
[107] | ||
| Anthocyanins | Delphinidin | Dex-induced C2C12 atrophy and tail-suspension induced atrophy in mice | Suppressed MuRF-1 expression Prevented muscle weight loss upregulated miR-23a and NFATc3 |
[108] | |
| Delphinidin | Dex-induced C2C12 atrophy and tail-suspension induced atrophy in mice | Suppressed disused-muscle loss suppressed Cbl-b and stress related gene |
[109] | ||
| Delphinidin | LPS-induced atrophy in C2C12 myotubes | Reduced atrogin-1 expression insignificantly | [82] | ||
| Cyanidin | Dystrophic alpha-sarcoglyan (Sgca) null mice | Reduced progression of muscular dystrophy Reduced inflammation and fibrosis |
[110] |
2.3. Flavanols
2.4. Flavanones
2.5. Flavones
2.6. Isoflavones
2.7 Flavonols
2.8. Anthocyanins
3. Conclusion
References
- Frontera, W.R.; Ochala, J. Skeletal Muscle: A Brief Review of Structure and Function. Calcif. Tissue Int. 2015, 96, 183–195.
- Cohen, S.; Nathan, J.; Goldberg, A. Muscle wasting in disease: Molecular mechanisms and promising therapies. Nat. Rev. Drug Discov. 2015, 14, 58–74.
- Schakman, O.; Kalista, S.; Barbé, C.; Loumaye, A.; Thissen, J.P. Glucocorticoid-induced skeletal muscle atrophy. Int. J. Biochem. Cell Biol. 2013, 45, 2163–2172.
- Glass, D.J. PI3 kinase regulation of skeletal muscle hypertrophy and atrophy. Curr. Top. Microbiol. Immunol. 2010, 346, 267–278.
- Kang, S.H.; Lee, H.A.; Kim, M.; Lee, E.; Sohn, U.D.; Kim, I. Forkhead box O3 plays a role in skeletal muscle atrophy through expression of E3 ubiquitin ligases MuRF-1 and atrogin-1 in Cushing’s syndrome. Am. J. Physiol. Endocrinol. Metab. 2017, 312, E495–E507.
- Milan, G.; Romanello, V.; Pescatore, F.; Armani, A.; Paik, J.H.; Frasson, L.; Seydel, A.; Zhao, J.; Abraham, R.; Goldberg, A.L.; et al. Regulation of autophagy and the ubiquitin-proteasome system by the FoxO transcriptional network during muscle atrophy. Nat. Commun. 2015, 6, 6670.
- Costamagna, D.; Costelli, P.; Sampaolesi, M.; Penna, F. Role of Inflammation in Muscle Homeostasis and Myogenesis. Mediat. Inflamm. 2015, 2015, 805172.
- Braun, T.P.; Marks, D.L. The regulation of muscle mass by endogenous glucocorticoids. Front. Physiol. 2015, 6, 12.
- Hyatt, H.; Deminice, R.; Yoshihara, T.; Powers, S.K. Mitochondrial dysfunction induces muscle atrophy during prolonged inactivity: A review of the causes and effects. Arch. Biochem. Biophys. 2019, 662, 49–60.
- Webster, J.M.; Kempen, L.; Hardy, R.S.; Langen, R. Inflammation and Skeletal Muscle Wasting during Cachexia. Front. Physiol. 2020, 11, 597675.
- Ábrigo, J.; Elorza, A.A.; Riedel, C.A.; Vilos, C.; Simon, F.; Cabrera, D.; Estrada, L.; Cabello-Verrugio, C. Role of Oxidative Stress as Key Regulator of Muscle Wasting during Cachexia. Oxidative Med. Cell. Longev. 2018, 2018, 2063179.
- Li, H.; Malhotra, S.; Kumar, A. Nuclear factor-kappa B signaling in skeletal muscle atrophy. J. Mol. Med. 2008, 86, 1113–1126.
- Rodriguez, J.; Vernus, B.; Chelh, I.; Cassar-Malek, I.; Gabillard, J.C.; Hadj Sassi, A.; Seiliez, I.; Picard, B.; Bonnieu, A. Myostatin and the skeletal muscle atrophy and hypertrophy signaling pathways. Cell. Mol. Life Sci. 2014, 71, 4361–4371.
- Huang, Z.; Fang, Q.; Ma, W.; Zhang, Q.; Qiu, J.; Gu, X.; Yang, H.; Sun, H. Skeletal Muscle Atrophy Was Alleviated by Salidroside through Suppressing Oxidative Stress and Inflammation during Denervation. Front. Pharmacol. 2019, 10, 997.
- Meng, S.J.; Yu, L.J. Oxidative stress, molecular inflammation and sarcopenia. Int. J. Mol. Sci. 2010, 11, 1509–1526.
- Uchida, T.; Sakashita, Y.; Kitahata, K.; Yamashita, Y.; Tomida, C.; Kimori, Y.; Komatsu, A.; Hirasaka, K.; Ohno, A.; Nakao, R.; et al. Reactive oxygen species upregulate expression of muscle atrophy-associated ubiquitin ligase Cbl-b in rat L6 skeletal muscle cells. Am. J. Physiol. Cell Physiol. 2018, 314, C721–C731.
- Nakao, R.; Hirasaka, K.; Goto, J.; Ishidoh, K.; Yamada, C.; Ohno, A.; Okumura, Y.; Nonaka, I.; Yasutomo, K.; Baldwin, K.M.; et al. Ubiquitin ligase Cbl-b is a negative regulator for insulin-like growth factor 1 signaling during muscle atrophy caused by unloading. Mol. Cell. Biol. 2009, 29, 4798–4811.
- Wang, J.; Leung, K.S.; Chow, S.K.; Cheung, W.H. Inflammation and age-associated skeletal muscle deterioration (sarcopaenia). J. Orthop. Transl. 2017, 10, 94–101.
- Londhe, P.; Guttridge, D.C. Inflammation induced loss of skeletal muscle. Bone 2015, 80, 131–142.
- Liu, J.; Peng, Y.; Wang, X.; Fan, Y.; Qin, C.; Shi, L.; Tang, Y.; Cao, K.; Li, H.; Long, J.; et al. Mitochondrial Dysfunction Launches Dexamethasone-Induced Skeletal Muscle Atrophy via AMPK/FOXO3 Signaling. Mol. Pharm. 2016, 13, 73–84.
- Yan, W.; Diao, S.; Fan, Z. The role and mechanism of mitochondrial functions and energy metabolism in the function regulation of the mesenchymal stem cells. Stem Cell Res. Ther. 2021, 12, 140.
- Hernández-Hernández, J.M.; García-González, E.G.; Brun, C.E.; Rudnicki, M.A. The myogenic regulatory factors, determinants of muscle development, cell identity and regeneration. Semin. Cell Dev. Biol. 2017, 72, 10–18.
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Med. Cell. Longev. 2009, 2, 270–278.
- Rasouli, H.; Farzaei, M.H.; Reza Khodarahmi, R. Polyphenols and their benefits: A review. Int. J. Food Prop. 2017, 2, 1700–1741.
- Singla, R.K.; Dubey, A.K.; Garg, A.; Sharma, R.K.; Fiorino, M.; Ameen, S.M.; Haddad, M.A.; Al-Hiary, M. Natural Polyphenols: Chemical Classification, Definition of Classes, Subcategories, and Structures. J. AOAC Int. 2019, 102, 1397–1400.
- Marino, M.; Del Bo’, C.; Martini, D.; Porrini, M.; Riso, P. A Review of Registered Clinical Trials on Dietary (Poly)Phenols: Past Efforts and Possible Future Directions. Foods 2020, 9, 1606.
- Salucci, S.; Falcieri, E. Polyphenols and their potential role in preventing skeletal muscle atrophy. Nutr. Res. 2020, 74, 10–22.
- Saibabu, V.; Fatima, Z.; Khan, L.A.; Hameed, S. Therapeutic Potential of Dietary Phenolic Acids. Adv. Pharmacol. Sci. 2015, 2015, 823539.
- Chang, W.T.; Huang, S.C.; Cheng, H.L.; Chen, S.C.; Hsu, C.L. Rutin and Gallic Acid Regulates Mitochondrial Functions via the SIRT1 Pathway in C2C12 Myotubes. Antioxidants 2021, 10, 286.
- Hong, K.B.; Lee, H.S.; Hong, J.S.; Kim, D.H.; Moon, J.M.; Park, Y. Effects of tannase-converted green tea extract on skeletal muscle development. BMC Complement. Med. Ther. 2020, 20, 47.
- Aslan, A.; Beyaz, S.; Gok, O.; Erman, O. The effect of ellagic acid on caspase-3/bcl-2/Nrf-2/NF-kB/TNF-α /COX-2 gene expression product apoptosis pathway: A new approach for muscle damage therapy. Mol. Biol. Rep. 2020, 47, 2573–2582.
- Khodaei, F.; Rashedinia, M.; Heidari, R.; Rezaei, M.; Khoshnoud, M.J. Ellagic acid improves muscle dysfunction in cuprizone-induced demyelinated mice via mitochondrial Sirt3 regulation. Life Sci. 2019, 237, 116954.
- Ryu, D.; Mouchiroud, L.; Andreux, P.; Katsyuba, E.; Moullan, N.; A Nicolet-Dit-Félix, A.; Williams, E.; Jha, P.; Sasso, G.L.; Huzard, D.; et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat. Med. 2016, 22, 879–888.
- Luan, P.; D’Amico, D.; Andreux, P.A.; Laurila, P.P.; Wohlwend, M.; Li, H.; Imamura de Lima, T.; Place, N.; Rinsch, C.; Zanou, N.; et al. Urolithin A improves muscle function by inducing mitophagy in muscular dystrophy. Sci. Transl. Med. 2021, 13, eabb0319.
- Ghosh, N.; Das, A.; Biswas, N.; Gnyawali, S.; Singh, K.; Gorain, M.; Polcyn, C.; Khanna, S.; Roy, S.; Sen, C.K. Urolithin A augments angiogenic pathways in skeletal muscle by bolstering NAD+ and SIRT1. Sci. Rep. 2020, 10, 20184.
- Rodriguez, J.; Pierre, N.; Naslain, D.; Bontemps, F.; Ferreira, D.; Priem, F.; Deldicque, L.; Francaux, M. Urolithin B, a newly identified regulator of skeletal muscle mass. J. Cachexia Sarcopenia Muscle 2017, 8, 583–597.
- Rodriguez, J.; Caille, O.; Ferreira, D.; Francaux, M. Pomegranate extract prevents skeletal muscle of mice against wasting induced by acute TNF-α injection. Mol. Nutr. Food Res. 2017, 61, 1600169.
- Chen, X.; Guo, Y.; Jia, G.; Zhao, H.; Liu, G.; Huang, Z. Ferulic acid regulates muscle fiber type formation through the Sirt1/AMPK signaling pathway. Food Funct. 2019, 10, 259–265.
- Shereen, M.; Samir, M.D.; Abeer, F.; Mostafa, M.D. Ferulic Acid Promotes Growth of Both Fast Glycolytic and Slow Oxidative Skeletal Muscles in Corticosteroid-Induced Rat Myopathy. Med. J. Cairo Univ. 2019, 44, 1703–1715.
- Wen, Y.; Ushio, H. Ferulic Acid Promotes Hypertrophic Growth of Fast Skeletal Muscle in Zebrafish Model. Nutrients 2017, 9, 1066.
- Ommati, M.M.; Farshad, O.; Mousavi, K.; Khalili, M.; Jamshidzadeh, A.; Heidari, R. Chlorogenic acid supplementation improves skeletal muscle mitochondrial function in a rat model of resistance training. Biologia 2020, 75, 1221–1230.
- Shen, Y.C.; Yen, J.C.; Liou, K.T. Ameliorative effects of caffeic acid phenethyl ester on an eccentric exercise-induced skeletal muscle injury by down-regulating NF-κb mediated inflammation. Pharmacology 2013, 91, 219–228.
- Dayangac-Erden, D.; Bora-Tatar, G.; Dalkara, S.; Demir, A.S.; Erdem-Yurter, H. Carboxylic acid derivatives of histone deacetylase inhibitors induce full length SMN2 transcripts: A promising target for spinal muscular atrophy therapeutics. Arch. Med. Sci. 2011, 7, 230–234.
- Jang, Y.J.; Son, H.J.; Kim, J.S.; Jung, C.H.; Ahn, J.; Hur, J.; Ha, T.Y. Coffee consumption promotes skeletal muscle hypertrophy and myoblast differentiation. Food Funct. 2018, 9, 1102–1111.
- Ilavenil, S.; Kim, d.; Srigopalram, S.; Arasu, M.V.; Lee, K.D.; Lee, J.C.; Lee, J.S.; Renganathan, S.; Choi, K.C. Potential Application of p-Coumaric Acid on Differentiation of C2C12 Skeletal Muscle and 3T3-L1 Preadipocytes-An in Vitro and in Silico Approach. Molecules 2016, 21, 997.
- Ríos, J.L.; Giner, R.M.; Marín, M.; Recio, M.C. A Pharmacological Update of Ellagic Acid. Planta Med. 2018, 84, 1068–1093.
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47.
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750.
- Rupasinghe, H. Special Issue “Flavonoids and Their Disease Prevention and Treatment Potential”: Recent Advances and Future Perspectives. Molecules 2020, 25, 4746.
- Owona, B.A.; Abia, W.A.; Moundipa, P.F. Natural compounds flavonoids as modulators of inflammasomes in chronic diseases. Int. Immunopharmacol. 2020, 84, 106498.
- Mansuri, M.L.; Parihar, P.; Solanki, I.; Parihar, M.S. Flavonoids in modulation of cell survival signalling pathways. Genes Nutr. 2014, 9, 400.
- Gutierrez-Salmean, G.; Chiarelli, T.P.; Nogueira, L.; Barboza, J.; Taub, P.R.; Hogan, M.C.; Henry, R.R.; Meaney, E.; Villarreal, F.; Ceballos, G.; et al. Effects of (−)-epicatechin on molecular modulators of skeletal muscle growth and differentiation. J. Nutr. Biochem. 2014, 25, 91–94.
- Si, H.; Wang, X.; Zhang, L.; Parnell, L.D.; Admed, B.; LeRoith, T.; Ansah, T.A.; Zhang, L.; Li, J.; Ordovás, J.M.; et al. Dietary epicatechin improves survival and delays skeletal muscle degeneration in aged mice. FASEB J. 2019, 33, 965–977.
- Lee, I.; Hüttemann, M.; Malek, M.H. (−)-Epicatechin Attenuates Degradation of Mouse Oxidative Muscle Following Hindlimb Suspension. J. Strength Cond. Res. 2016, 30, 1–10.
- Gonzalez-Ruiz, C.; Cordero-Anguiano, P.; Morales-Guadarrama, A.; Mondragón-Lozano, R.; Sánchez-Torres, S.; Salgado-Ceballos, H.; Villarreal, F.; Meaney, E.; Ceballos, G.; Nájera, N. (−)-Epicatechin reduces muscle waste after complete spinal cord transection in a murine model: Role of ubiquitin–proteasome system. Mol. Biol. Rep. 2020, 47, 8975–8985.
- McDonald, C.M.; Ramirez-Sanchez, I.; Oskarsson, B.; Joyce, N.; Aguilar, C.; Nicorici, A.; Dayan, J.; Goude, E.; Abresch, R.T.; Villarreal, F.; et al. (−)-Epicatechin induces mitochondrial biogenesis and markers of muscle regeneration in adults with Becker muscular dystrophy. Muscle Nerve 2021, 63, 239–249.
- Moreno-Ulloa, A.; Miranda-Cervantes, A.; Licea-Navarro, A.; Mansour, C.; Beltrán-Partida, E.; Donis-Maturano, L.; Delgado De la Herrán, H.C.; Villarreal, F.; Álvarez-Delgado, C. (−)-Epicatechin stimulates mitochondrial biogenesis and cell growth in C2C12 myotubes via the G-protein coupled estrogen receptor. Eur. J. Pharmacol. 2018, 822, 95–107.
- Lee, S.J.; Leem, Y.E.; Go, G.Y.; Choi, Y.; Song, Y.J.; Kim, I.; Kim, D.Y.; Kim, Y.K.; Seo, D.W.; Kang, J.S.; et al. Epicatechin elicits MyoD-dependent myoblast differentiation and myogenic conversion of fibroblasts. PLoS ONE 2017, 12, e0175271.
- Munguia, L.; Ramirez-Sanchez, I.; Meaney, E.; Villarreal, F.; Ceballos, G.; Najera, N. Flavonoids from dark chocolate and (−)-epicatechin ameliorate high-fat diet-induced decreases in mobility and muscle damage in aging mice. Food Biosci. 2020, 37, 100710.
- Hemdan, D.I.; Hirasaka, K.; Nakao, R.; Kohno, S.; Kagawa, S.; Abe, T.; Harada-Sukeno, A.; Okumura, Y.; Nakaya, Y.; Terao, J.; et al. Polyphenols prevent clinorotation-induced expression of atrogenes in mouse C2C12 skeletal myotubes. J. Med. Investig. 2009, 56, 26–32.
- Meador, B.M.; Mirza, K.A.; Tian, M.; Skelding, M.B.; Reaves, L.A.; Edens, N.K.; Tisdale, M.J.; Pereira, S.L. The Green Tea Polyphenol Epigallocatechin-3-Gallate (EGCg) Attenuates Skeletal Muscle Atrophy in a Rat Model of Sarcopenia. J. Frailty Aging 2015, 4, 209–215.
- Chang, Y.C.; Liu, H.W.; Chan, Y.C.; Hu, S.H.; Liu, M.Y.; Chang, S.J. The green tea polyphenol epigallocatechin-3-gallate attenuates age-associated muscle loss via regulation of miR-486-5p and myostatin. Arch. Biochem. Biophys. 2020, 692, 108511.
- Mirza, K.A.; Pereira, S.L.; Edens, N.K.; Tisdale, M.J. Attenuation of muscle wasting in murine C2C 12 myotubes by epigallocatechin-3-gallate. J. Cachexia Sarcopenia Muscle 2014, 5, 339–345.
- Wang, H.; Lai, Y.J.; Chan, Y.L.; Li, T.L.; Wu, C.J. Epigallocatechin-3-gallate effectively attenuates skeletal muscle atrophy caused by cancer cachexia. Cancer Lett. 2011, 305, 40–49.
- Woodman, K.G.; Coles, C.A.; Lamandé, S.R.; White, J.D. Nutraceuticals and Their Potential to Treat Duchenne Muscular Dystrophy: Separating the Credible from the Conjecture. Nutrients 2016, 8, 713.
- Alway, S.E.; Bennett, B.T.; Wilson, J.C.; Edens, N.K.; Pereira, S.L. Epigallocatechin-3-gallate improves plantaris muscle recovery after disuse in aged rats. Exp. Gerontol. 2014, 50, 82–94.
- Takahashi, H.; Suzuki, Y.; Mohamed, J.S.; Gotoh, T.; Pereira, S.L.; Alway, S.E. Epigallocatechin-3-gallate increases autophagy signaling in resting and unloaded plantaris muscles but selectively suppresses autophagy protein abundance in reloaded muscles of aged rats. Exp. Gerontol. 2017, 92, 56–66.
- Murata, M.; Shimizu, Y.; Marugame, Y.; Nezu, A.; Fujino, K.; Yamada, S.; Kumazoe, M.; Fujimura, Y.; Tachibana, H. EGCG down-regulates MuRF1 expression through 67-kDa laminin receptor and the receptor signaling is amplified by eriodictyol. J. Nat. Med. 2020, 74, 673–679.
- Onishi, S.; Ishino, M.; Kitazawa, H.; Yoto, A.; Shimba, Y.; Mochizuki, Y.; Unno, K.; Meguro, S.; Tokimitsu, I.; Miura, S. Green tea extracts ameliorate high-fat diet-induced muscle atrophy in senescence-accelerated mouse prone-8 mice. PLoS ONE 2018, 13, e0195753.
- Wimmer, R.J.; Russell, S.J.; Schneider, M.F. Green tea component EGCG, insulin and IGF-1 promote nuclear efflux of atrophy-associated transcription factor Foxo1 in skeletal muscle fibers. J. Nutr. Biochem. 2015, 26, 1559–1567.
- Kim, A.R.; Kim, K.M.; Byun, M.R.; Hwang, J.H.; Park, J.I.; Oh, H.T.; Kim, H.K.; Jeong, M.G.; Hwang, E.S.; Hong, J.H. Catechins activate muscle stem cells by Myf5 induction and stimulate muscle regeneration. Biochem. Biophys. Res. Commun. 2017, 489, 142–148.
- Haramizu, S.; Ota, N.; Hase, T.; Murase, T. Catechins suppress muscle inflammation and hasten performance recovery after exercise. Med. Sci. Sports Exerc. 2013, 45, 1694–1702.
- Ota, N.; Soga, S.; Haramizu, S.; Yokoi, Y.; Hase, T.; Murase, T. Tea catechins prevent contractile dysfunction in unloaded murine soleus muscle: A pilot study. Nutrition 2011, 27, 955–959.
- Biesemann, N.; Ried, J.S.; Ding-Pfennigdorff, D.; Dietrich, A.; Rudolph, C.; Hahn, S.; Hennerici, W.; Asbrand, C.; Leeuw, T.; Strübing, C. High throughput screening of mitochondrial bioenergetics in human differentiated myotubes identifies novel enhancers of muscle performance in aged mice. Sci. Rep. 2018, 8, 9408.
- Pellegrini, M.; Bulzomi, P.; Galluzzo, P.; Lecis, M.; Leone, S.; Pallottini, V.; Marino, M. Naringenin modulates skeletal muscle differentiation via estrogen receptor α and β signal pathway regulation. Genes Nutr. 2014, 9, 425.
- Mukai, R.; Horikawa, H.; Fujikura, Y.; Kawamura, T.; Nemoto, H.; Nikawa, T.; Terao, J. Prevention of disuse muscle atrophy by dietary ingestion of 8-prenylnaringenin in denervated mice. PLoS ONE 2012, 7, e45048.
- Mukai, R.; Horikawa, H.; Lin, P.Y.; Tsukumo, N.; Nikawa, T.; Kawamura, T.; Nemoto, H.; Terao, J. 8-Prenylnaringenin promotes recovery from immobilization-induced disuse muscle atrophy through activation of the Akt phosphorylation pathway in mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 311, R1022–R1031.
- Wang, D.; Yang, Y.; Zou, X.; Zhang, J.; Zheng, Z.; Wang, Z. Antioxidant Apigenin Relieves Age-Related Muscle Atrophy by Inhibiting Oxidative Stress and Hyperactive Mitophagy and Apoptosis in Skeletal Muscle of Mice. J. Gerontol. Ser. A 2020, 75, 2081–2088.
- Choi, W.H.; Son, H.J.; Jang, Y.J.; Ahn, J.; Jung, C.H.; Ha, T.Y. Apigenin Ameliorates the Obesity-Induced Skeletal Muscle Atrophy by Attenuating Mitochondrial Dysfunction in the Muscle of Obese Mice. Mol. Nutr. Food Res. 2017, 61.
- Choi, W.H.; Jang, Y.J.; Son, H.J.; Ahn, J.; Jung, C.H.; Ha, T.Y. Apigenin inhibits sciatic nerve denervation-induced muscle atrophy. Muscle Nerve 2018, 58, 314–318.
- Jang, Y.J.; Son, H.J.; Choi, Y.M.; Ahn, J.; Jung, C.H.; Ha, T.Y. Apigenin enhances skeletal muscle hypertrophy and myoblast differentiation by regulating Prmt7. Oncotarget 2017, 8, 78300–78311.
- Shiota, C.; Abe, T.; Kawai, N.; Ohno, A.; Teshima-Kondo, S.; Mori, H.; Terao, J.; Tanaka, E.; Nikawa, T. Flavones Inhibit LPS-Induced Atrogin-1/MAFbx Expression in Mouse C2C12 Skeletal Myotubes. J. Nutr. Sci. Vitaminol. 2015, 61, 188–194.
- Chen, T.; Li, B.; Xu, Y.; Meng, S.; Wang, Y.; Jiang, Y. Luteolin reduces cancer-induced skeletal and cardiac muscle atrophy in a Lewis lung cancer mouse model. Oncol. Rep. 2018, 40, 1129–1137.
- Hawila, N.; Hedya, S.; Elmaboud, A.M.; Abdin, A. Luteolin Attenuates Dexamethasone-Induced Skeletal Muscle Atrophy in Male Albino Rats. Med. J. Cairo Univ. 2019, 87, 3365–3374.
- Hirasaka, K.; Maeda, T.; Ikeda, C.; Haruna, M.; Kohno, S.; Abe, T.; Ochi, A.; Mukai, R.; Oarada, M.; Eshima-Kondo, S.; et al. Isoflavones derived from soy beans prevent MuRF1-mediated muscle atrophy in C2C12 myotubes through SIRT1 activation. J. Nutr. Sci. Vitaminol. 2013, 59, 317–324.
- Hirasaka, K.; Saito, S.; Yamaguchi, S.; Miyazaki, R.; Wang, Y.; Haruna, M.; Taniyama, S.; Higashitani, A.; Terao, J.; Nikawa, T.; et al. Dietary Supplementation with Isoflavones Prevents Muscle Wasting in Tumor-Bearing Mice. J. Nutr. Sci. Vitaminol. 2016, 62, 178–184.
- Tabata, S.; Aizawa, M.; Kinoshita, M.; Ito, Y.; Kawamura, Y.; Takebe, M.; Pan, W.; Sakuma, K. The influence of isoflavone for denervation-induced muscle atrophy. Eur. J. Nutr. 2019, 58, 291–300.
- Aoyama, S.; Jia, H.; Nakazawa, K.; Yamamura, J.; Saito, K.; Kato, H. Dietary Genistein Prevents Denervation-Induced Muscle Atrophy in Male Rodents via Effects on Estrogen Receptor-α. J. Nutr. 2016, 146, 1147–1154.
- Gan, M.; Yang, D.; Fan, Y.; Du, J.; Shen, L.; Li, Q.; Jiang, Y.; Tang, G.; Li, M.; Wang, J.; et al. Bidirectional regulation of genistein on the proliferation and differentiation of C2C12 myoblasts. Mol. Toxicol. 2020, 50, 1352–1358.
- Ogawa, M.; Kitano, T.; Kawata, N.; Sugihira, T.; Kitakaze, T.; Harada, N.; Yamaji, R. Daidzein down-regulates ubiquitin-specific protease 19 expression through estrogen receptor β and increases skeletal muscle mass in young female mice. J. Nutr. Biochem. 2017, 49, 63–70.
- Zhang, H.; Chi, M.; Chen, L.; Sun, X.; Wan, L.; Yang, Q.; Guo, C. Daidzein alleviates cisplatin-induced muscle atrophy by regulating Glut4/AMPK/FoxO pathway. Phytother. Res. 2021.
- Lee, S.J.; Vuong, T.A.; Go, G.Y.; Song, Y.J.; Lee, S.; Lee, S.Y.; Kim, S.W.; Lee, J.; Kim, Y.K.; Seo, D.W.; et al. An isoflavone compound daidzein elicits myoblast differentiation and myotube growth. J. Funct. Foods 2017, 38, 438–446.
- Khairallah, R.J.; O’Shea, K.M.; Ward, C.W.; Butteiger, D.N.; Mukherjea, R.; Krul, E.S. Chronic dietary supplementation with soy protein improves muscle function in rats. PLoS ONE 2017, 12, e0189246.
- Liu, L.; Hu, R.; You, H.; Li, J.; Liu, Y.; Li, Q.; Wu, X.; Huang, J.; Cai, X.; Wang, M.; et al. Formononetin ameliorates muscle atrophy by regulating myostatin-mediated PI3K/Akt/FoxO3a pathway and satellite cell function in chronic kidney disease. J. Cell. Mol. Med. 2021, 25, 1493–1506.
- Yoshioka, Y.; Kubota, Y.; Samukawa, Y.; Yamashita, Y.; Ashida, H. Glabridin inhibits dexamethasone-induced muscle atrophy. Arch. Biochem. Biophys. 2019, 664, 157–166.
- Le, N.H.; Kim, C.S.; Park, T.; Park, J.H.; Sung, M.K.; Lee, D.G.; Hong, S.M.; Choe, S.Y.; Goto, T.; Kawada, T.; et al. Quercetin protects against obesity-induced skeletal muscle inflammation and atrophy. Mediat. Inflamm. 2014, 2014, 834294.
- Kim, Y.; Kim, C.S.; Joe, Y.; Chung, H.T.; Ha, T.Y.; Yu, R. Quercetin Reduces Tumor Necrosis Factor Alpha-Induced Muscle Atrophy by Upregulation of Heme Oxygenase-1. J. Med. Food 2018, 21, 551–559.
- Levolger, S.; Engel, S.V.D.; Ambagtsheer, G.; Ijzermans, J.N.; de Bruin, R.W. Quercetin Supplementation Attenuates Muscle Wasting in Cancer-associated Cachexia in Mice. Nutr. Healthy Aging 2021, 6, 35–47.
- Chan, S.T.; Chuang, C.H.; Lin, Y.C.; Liao, J.W.; Lii, C.K.; Yeh, S.L. Quercetin enhances the antitumor effect of trichostatin A and suppresses muscle wasting in tumor-bearing mice. Food Funct. 2018, 9, 871–879.
- Chen, C.; Yang, J.S.; Lu, C.C.; Chiu, Y.J.; Chen, H.C.; Chung, M.I.; Wu, Y.T.; Chen, F.A. Effect of Quercetin on Dexamethasone-Induced C2C12 Skeletal Muscle Cell Injury. Molecules 2021, 25, 3267.
- Otsuka, Y.; Egawa, K.; Kanzaki, N.; Izumo, T.; Rogi, T.; Shibata, H. Quercetin glycosides prevent dexamethasone-induced muscle atrophy in mice. Biochem. Biophys. Rep. 2019, 18, 100618.
- Mukai, R.; Nakao, R.; Yamamoto, H.; Nikawa, T.; Takeda, E.; Terao, J. Quercetin prevents unloading-derived disused muscle atrophy by attenuating the induction of ubiquitin ligases in tail-suspension mice. J. Nat. Prod. 2010, 73, 1708–1710.
- Mukai, R.; Matsui, N.; Fujikura, Y.; Matsumoto, N.; Hou, D.X.; Kanzaki, N.; Shibata, H.; Horikawa, M.; Iwasa, K.; Hirasaka, K.; et al. Preventive effect of dietary quercetin on disuse muscle atrophy by targeting mitochondria in denervated mice. J. Nutr. Biochem. 2016, 31, 67–76.
- Kanzaki, N.; Takemoto, D.; Ono, Y.; Izumo, T.; Shibata, H.; Ye, X. Quercetin Glycosides Improve Motor Performance and Muscle Weight in Adult Mice. J. Nutr. Food Sci. 2019, 9.
- Davis, J.M.; Murphy, E.A.; Carmichael, M.D.; Davis, B. Quercetin increases brain and muscle mitochondrial biogenesis and exercise tolerance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R1071–R1077.
- Yoshimura, T.; Saitoh, K.; Sun, L.; Wang, Y.; Taniyama, S.; Yamaguchi, K.; Uchida, T.; Ohkubo, T.; Higashitani, A.; Nikawa, T.; et al. Morin suppresses cachexia-induced muscle wasting by binding to ribosomal protein S10 in carcinoma cells. Biochem. Biophys. Res. Commun. 2018, 506, 773–779.
- Ulla, A.; Uchida, T.; Miki, Y.; Sugiura, K.; Higashitani, A.; Kobayashi, T.; Ohno, A.; Nakao, R.; Hirasaka, K.; Sakakibara, I.; et al. Morin attenuates dexamethasone-mediated oxidative stress and atrophy in mouse C2C12 skeletal myotubes. Arch. Biochem. Biophys. 2021, 704, 108873.
- Murata, M.; Nonaka, H.; Komatsu, S.; Goto, M.; Morozumi, M.; Yamada, S.; Lin, I.C.; Yamashita, S.; Tachibana, H. Delphinidin Prevents Muscle Atrophy and Upregulates miR-23a Expression. J. Agric. Food Chem. 2017, 65, 45–50.
- Murata, M.; Kosaka, R.; Kurihara, K.; Yamashita, S.; Tachibana, H. Delphinidin prevents disuse muscle atrophy and reduces stress-related gene expression. Biosci. Biotechnol. Biochem. 2016, 80, 1636–1640.
- Saclier, M.; Bonfanti, C.; Antonini, S.; Angelini, G.; Mura, G.; Zanaglio, F.; Taglietti, V.; Romanello, V.; Sandri, M.; Tonelli, C.; et al. Nutritional intervention with cyanidin hinders the progression of muscular dystrophy. Cell Death Dis. 2020, 11, 127.
- Khan, M.K.; Zill-E-Huma; Dangles, O. A comprehensive review on flavanones, the major citrus polyphenols. J. Food Compos. Anal. 2014, 33, 85–104.
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food Sources, Bioavailability, Metabolism, and Bioactivity. Adv. Nutr. 2017, 8, 423–435.
- Křížová, L.; Dadáková, K.; Kašparovská, J.; Kašparovský, T. Isoflavones. Molecules 2019, 24, 1076.
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M.T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, Inflammation and Immunity. Nutrients 2016, 8, 167.
- Rajput, S.A.; Wang, X.Q.; Yan, H.C. Morin hydrate: A comprehensive review on novel natural dietary bioactive compound with versatile biological and pharmacological potential. Biomed. Pharmacother. 2021, 138, 111511.
- Mattioli, R.; Francioso, A.; Mosca, L.; Silva, P. Anthocyanins: A Comprehensive Review of Their Chemical Properties and Health Effects on Cardiovascular and Neurodegenerative Diseases. Molecules 2020, 25, 3809.
- Alam, M.A.; Islam, P.; Subhan, N.; Rahman, M.; Khan, F.; Burrows, G.E.; Nahar, L.; Sarker, S.D. Potential health benefits of anthocyanins in oxidative stress related disorders. Phytochem. Rev. 2021.




