Nodal proteins show a different affinity to TGF-β receptors; it was demonstrated that the presence of binding proteins that act as partners enhances the signal cascade, improving the interactions between Nodal and its receptors. It was established that a fundamental obligatory co-receptor for the TGF-β family member Nodal is the cell surface glycosylphosphatidylinositol (GPI)-linked glycoprotein Cripto-1 (CR-1). Co-immunoprecipitation experiments show the interaction between Nodal and CR-1, supporting the idea that CR-1 acts as an obliged coreceptor for Nodal that potentiates its signal cascade.
1. Introduction
Nodal signal is transduced from the membrane to the nucleus through cooperation of proteins which activation is regulated by Nodal itself. Indeed, SMAD heterodimers bind to transcription factors that regulate Lefty genes transcription. Lefty proteins are antagonists of Nodal morphogens, switching off Nodal signal cascade and thus acting as negative feedback regulators [1].
Evidence shows that Nodal has signal activity even in the absence of CR-1 interacting directly with its receptor [2]. CR-1, also known as Teratocarcinoma-derived growth factor1 (TDGF-1), was firstly identified in teratocarcinoma cells NTERA2 [3]. It belongs to the EGF-CFC ( Cripto-1/FRL1/Cryptic) family that encodes a group of structurally related proteins that serve as important factors during early embryogenesis in vertebrates such as Xenopus , Zebrafish , mice, and humans [4]. Prior to gastrulation event, in the epiblast, CR-1 expression is symmetrical and uniform. Then, it forms an asymmetrical proximal-distal gradient, shifting caudally towards the region of the nascent primitive streak, regulating A-P patterning [5][6].
As other members of the EGF-CFC family, CR-1 contains an NH2-terminal signal peptide, a modified EGF-like region, a conserved cysteine-rich domain (CFC motif), and a short hydrophobic COOH-terminus that contains additional sequences for GPI cleavage and attachment that generate a biologically active soluble form [7]. Site-directed mutagenesis experiments showed that CR-1 directly interacts with ALK4 through its CFC domain and accommodates Nodal protein by interacting through its EGF-like motif. The binding between CR-1 and ALK4, and in particular the substrate-determining loop (L45) of ALK4, is fundamental for Nodal-induced SMAD2 activation. In fact, alterations of this loop are sufficient to redirect the Nodal signal from SMAD2 to SMAD1 activation through the ALK4/CR-1 complex [8]. In addition, Watanabe et al. showed that the GPI-signal sequence of CR-1 is necessary to induce optimum Nodal signaling in trans as well as in cis , demonstrating that both transmembrane and soluble CR-1 forms are active to induce Nodal signal cascade [9].
Recently, an additional role of these signal cascades was demonstrated in the maintaining of embryonic stem cell pluripotency [10], and in the sustaining of the cancer stem cell (CSC) compartments ( Figure 1 ), which is discussed in the next section.
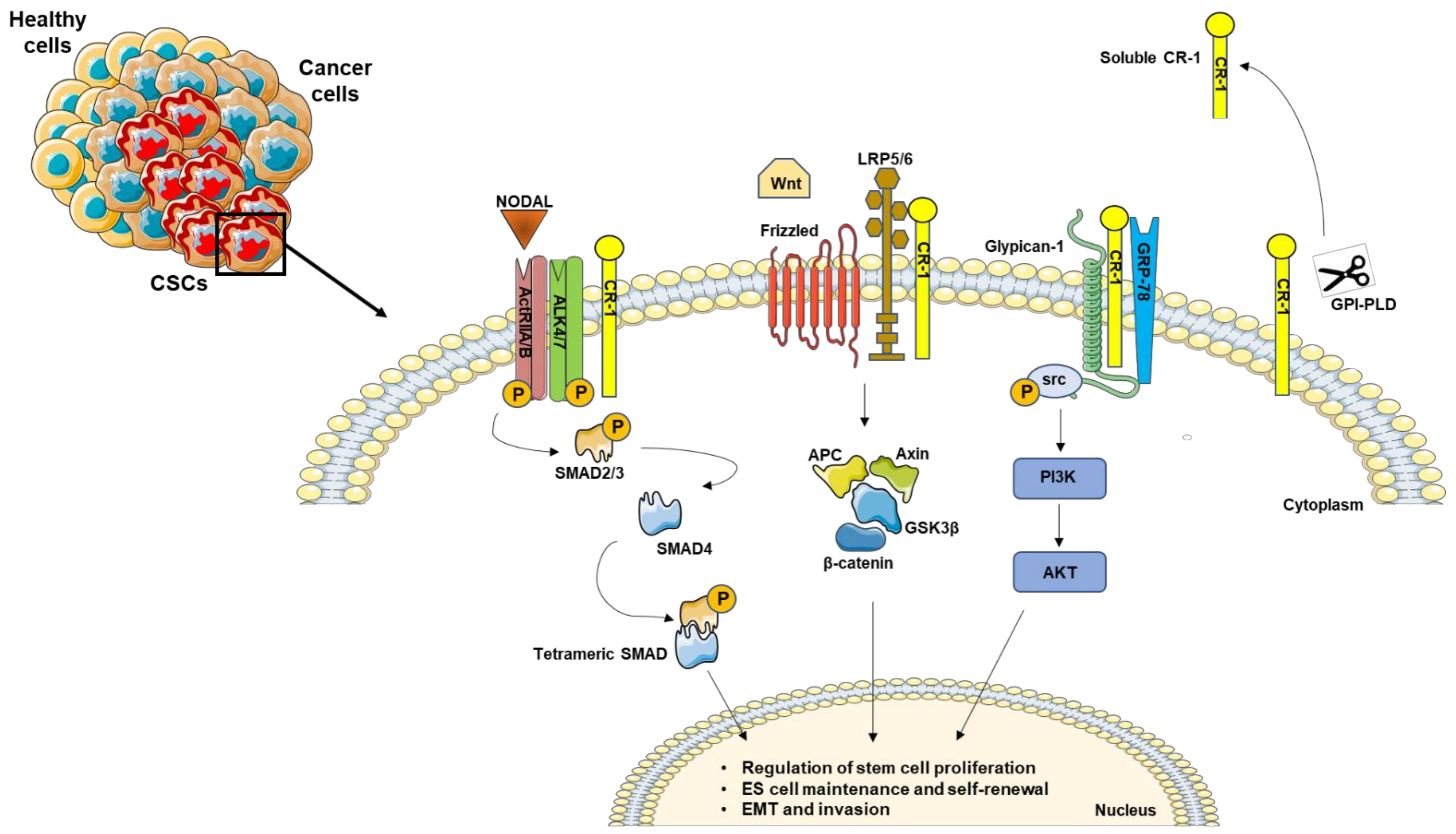 Figure 1. Schematic representation of Nodal/CR-1 signaling pathway in cancer stem cells. CR-1 acts as a cofactor for Nodal to activate downstream SMADs effectors. In the absence of Nodal, CR-1 can also signal in a non-canonical pathway through Frizzled and LRP4/5 complex, activating β-catenin signaling. In addition, CR-1 interacts with Glypican-1 and GRP78, triggering PIK3/AKT activation by Src phosphorylation. In addition, the GPI-specific phospholipase D (GPI-PLD) is able to cleave CR-1, generating a biological active soluble form.
Figure 1. Schematic representation of Nodal/CR-1 signaling pathway in cancer stem cells. CR-1 acts as a cofactor for Nodal to activate downstream SMADs effectors. In the absence of Nodal, CR-1 can also signal in a non-canonical pathway through Frizzled and LRP4/5 complex, activating β-catenin signaling. In addition, CR-1 interacts with Glypican-1 and GRP78, triggering PIK3/AKT activation by Src phosphorylation. In addition, the GPI-specific phospholipase D (GPI-PLD) is able to cleave CR-1, generating a biological active soluble form.
2. Nodal and Cripto-1 as Theranostic Targets
The deciphering of the multiple genetic lesions that drive cancer spread is addressed to develop new therapeutic approaches that may be combined with traditional strategies to overcome malignancies.
Most of the anti-cancer treatments widely used today were developed prior to 1975. At the time, scientists and researchers wanted to exploit the emphasized characteristics of cancer cells by interfering with behaviors shared with their healthy counterparts. In the past few decades, the knowledge about the hallmarks of cancer allowed the scientific community to understand strengths and weaknesses of many neoplastic diseases. The studies about the molecular signature that sustain cancer growth reveal, in some cases, the dependence of certain human cancer cells on a gene and/or an oncogene that drives the transforming process. This situation has been termed oncogene addiction and represents an important weakness of cancer cells; indeed, these genes have an irreplaceable role in sustaining cancer growth
[11].
On the other hand, chemoresistance is the “sword of Damocles” in cancer therapy as the new insights about the plasticity and the self-renewal capacity of CSCs define new scenarios that hinder the success of cancer therapies
[12]. In the past few decades, the major challenge of most scientists has been to identify the unique profile that characterize the subpopulation of CSCs searching for antigens and biomarkers to be used as attractive targets
[13]. Several studies have considered CR-1 and Nodal proteins as promising and useful prognostic as well as diagnostic biomarkers
[14]. It is well established that CR-1 levels in sera of patients with non-small cell lung cancers
[15], in testicular germ cell-tumors
[16], in HCC
[16], and in clear cell renal carcinomas
[17] is correlated with poor disease outcome. Liu et al.
[18] highlighted that CR-1
high cells exhibit highly invasive and metastatic phenotype in ESCC. In addition, CR-1
high cells in ESCC express high levels of stemness-related genes, demonstrating that CR-1 plays a fundamental role in the establishment of CSC niches. Soluble CR-1 is detected in plasma patients affected by BC and CRC. Recent evidence suggests that CR-1 might be a useful marker as it was found at an early stage of disease in patients with BC compared with benign lesion and healthy counterparts. With regards to CRC, CR-1 is clearly detectable in serum of patients affected by CRCs, even if it does not correlate with tumor size or staging/grading
[19]. Furthermore, a higher positivity for CR-1 has been observed in healthy colon tissues from high-risk individuals (one or more cases of CRC among first-degree relatives)
[20].
In this context, CR-1 and Nodal represent ideal candidates. A schematic representation of the most relevant findings about Nodal and CR-1 is provided in Figure 2.
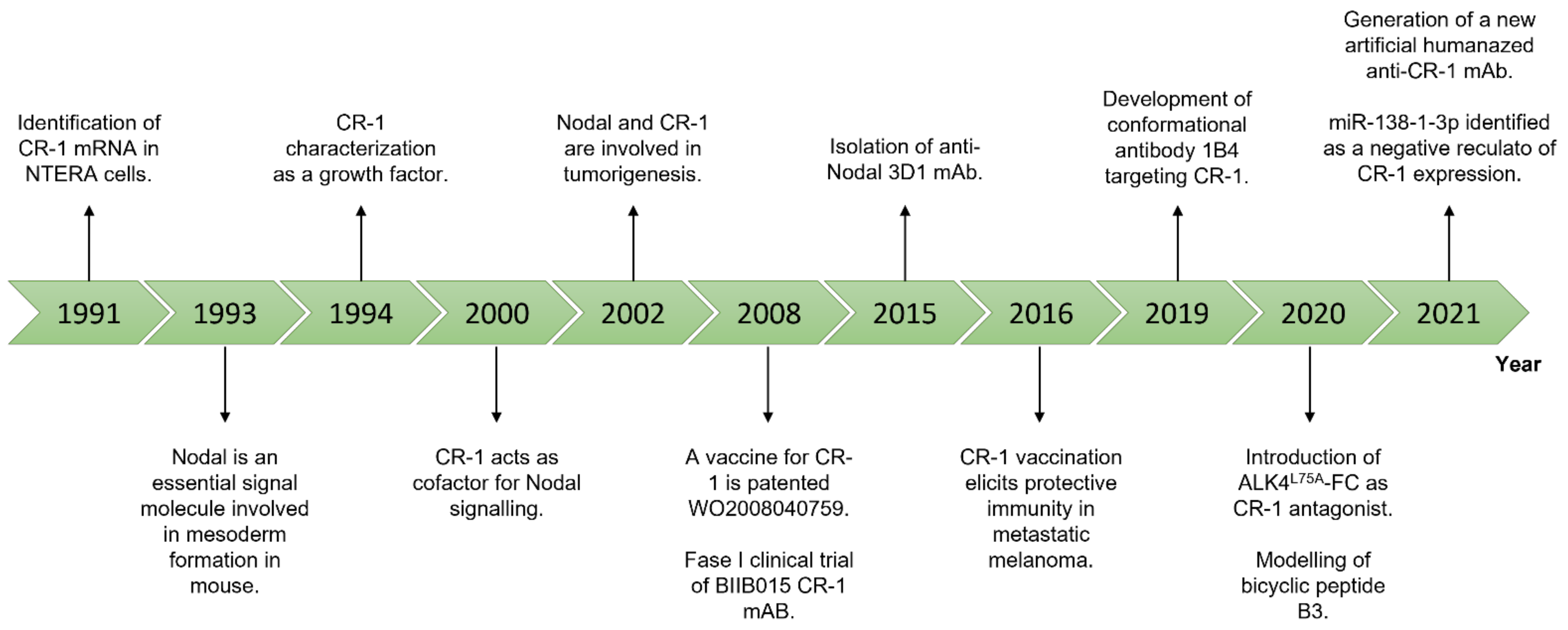 Figure 2.
Figure 2. Timeline of the most relevant findings about Nodal and CR-1.
3. Conclusions
The ability of cancer cells to take advantage of pro-survival stimuli and resist the organism’s attempts to eradicate them is a notion that research community acquired decades ago. Dozens of these pathways have been identified so far, including p53, NF-kB, PI3K, and MAPK as well as several growth factors and cell cycle regulatory pathways. Among these, a remarkable body of literature has evidenced the deep involvement of TGF-β family signaling in cancer. As key proteins in this pathway, Nodal and CR-1 represent ideal candidates for a “two-level” targeted therapy. The first level considers that they are both absent and low-expressed in adult tissues, which should reduce the adverse effects correlated to the interference with the two proteins. The second level is represented by the frequent overexpression of Nodal and/or CR-1 in the CSC compartments of several solid tumors, potentially allowing treatments to hit the most difficult and less targetable sub-population, which is thought to be the one that thwarts the complete tumor eradication. In addition, CR-1 and Nodal overexpression is correlated with a worse prognosis in several human cancers. For all these considerations, CR-1 and Nodal have the high potential to be both diagnostic and therapeutic targets consistent with the so-called “theranostic” approach.
The availability of different tools and strategies to target both CR-1 and Nodal render this an exciting field of study. mAbs are, so far, the best candidates due to their high specificity for the targets, allowing them to precisely hit cancer cells while simultaneously reducing the adverse effects. These mAbs can be produced by the modern biotech industry, or ideally an immune response can be elicited through cancer vaccines. These new therapeutic tools may be clinically effective alone or in combination with the conventional radio- or chemo-therapies to maximize the effects, targeting all the sub-populations of which a tumor is composed.
However, an important aspect should be considered: CR-1 and Nodal (or in general TGF-β signaling) are involved in maintaining the stemness of physiological stem cells. To the best of our knowledge, no studies have been conducted to address the impact that such treatments could have on the healthy stem cell compartments.

 Figure 1. Schematic representation of Nodal/CR-1 signaling pathway in cancer stem cells. CR-1 acts as a cofactor for Nodal to activate downstream SMADs effectors. In the absence of Nodal, CR-1 can also signal in a non-canonical pathway through Frizzled and LRP4/5 complex, activating β-catenin signaling. In addition, CR-1 interacts with Glypican-1 and GRP78, triggering PIK3/AKT activation by Src phosphorylation. In addition, the GPI-specific phospholipase D (GPI-PLD) is able to cleave CR-1, generating a biological active soluble form.
Figure 1. Schematic representation of Nodal/CR-1 signaling pathway in cancer stem cells. CR-1 acts as a cofactor for Nodal to activate downstream SMADs effectors. In the absence of Nodal, CR-1 can also signal in a non-canonical pathway through Frizzled and LRP4/5 complex, activating β-catenin signaling. In addition, CR-1 interacts with Glypican-1 and GRP78, triggering PIK3/AKT activation by Src phosphorylation. In addition, the GPI-specific phospholipase D (GPI-PLD) is able to cleave CR-1, generating a biological active soluble form. Figure 2. Timeline of the most relevant findings about Nodal and CR-1.
Figure 2. Timeline of the most relevant findings about Nodal and CR-1.