Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tamba Solomon Lebbie | + 2917 word(s) | 2917 | 2021-08-16 10:15:19 | | | |
| 2 | Rita Xu | Meta information modification | 2917 | 2021-08-31 05:48:52 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Lebbie, T. E-Waste in Africa. Encyclopedia. Available online: https://encyclopedia.pub/entry/13680 (accessed on 07 February 2026).
Lebbie T. E-Waste in Africa. Encyclopedia. Available at: https://encyclopedia.pub/entry/13680. Accessed February 07, 2026.
Lebbie, Tamba. "E-Waste in Africa" Encyclopedia, https://encyclopedia.pub/entry/13680 (accessed February 07, 2026).
Lebbie, T. (2021, August 30). E-Waste in Africa. In Encyclopedia. https://encyclopedia.pub/entry/13680
Lebbie, Tamba. "E-Waste in Africa." Encyclopedia. Web. 30 August, 2021.
Copy Citation
Waste electronic and electrical equipment (e-waste) consists of used and discarded electrical and electronic items ranging from refrigerators to cell phones and printed circuit boards.
e-waste
children
Africa
air pollution
cognitive function
1. Introduction
How to safely dispose of waste products coming from human activity is a global problem. Often in countries without adequate regulation, wastes are simply discarded. Most countries put wastes into landfills, but without adequate control landfills stink, leachate coming from the landfill can contaminate water and methane escaping from degradation of organic material contributes to climate change. In addition, landfills often attract vermin than can cause and spread disease. In many poor countries, people—including children—scavenge landfills for items of value, increasing risk of injury, infection and exposure to dangerous substances. Incineration is an alternative to landfills, but if incinerators are not operated with appropriate controls, they release toxic metals and dioxins that have the potential to cause significant harm to human health [1][2].
The harm to the environment is made worse when the discarded material is not readily degradable, as is the case with plastics that now contaminate land, lakes, streams and even the ocean. About eight million metric tons of plastics enter the oceans each year [3]. Hazardous wastes, defined as wastes that are ignitable, corrosive, reactive or toxic, add to the problem and are often intermixed with non-hazardous solid wastes. Hazardous wastes can cause significant harm to human health if not properly contained and disposed, including neuropsychological harm to children [4] and elevations in cancer and birth defects [5].
The term electronic and electrical waste (e-waste) refers to “electrical or electronic equipment, which is waste, including all components, subassemblies, and consumables, which are part of the equipment at the time the equipment becomes waste [6]”. E-waste includes large, discarded appliances, such as refrigerators, air conditioners and washing machines, as well as small personal items, including computers, televisions, mobile phones, and many other devices that are operated by electrical currents or batteries [7][8]. E-waste contains plastics, ceramics, metals, glass, toxic chemicals, such as organic flame retardants and polychlorinated biphenyls (PCBs), and various other potentially hazardous compounds [9][10].
In 2019, the world generated an estimated 53.6 million metric tons (Mt) of e-waste, or an average of 7.3 kg per person [11]. Furthermore, only 17.4% of this was officially documented as properly collected and recycled. Africa generated a total of 2.9 Mt of e-waste in 2019, or 2.5 kg per capita, the lowest regional rate in the world.
The value of the raw materials in global e-waste is estimated to be 57 billion USD, with iron, copper and gold contributing the most [11]. In some African countries, recycling and dismantling electronic devices has become a major source of employment and income. For example, in Ghana in 2010, an estimated 10,000 to 15,000 people were involved in refurbishing old and second-hand computers while another 20,300 to 33,600 were estimated to be working in recycling and e-waste management [12]. More recently this number has been estimated at more than 40,000 [13]. In 2010, an estimated 201,600 people across Ghana, including families and children, were dependent on e-waste recycling and management for support [12]. This number has almost certainly grown over time.
E-waste includes many substances that are dangerous to the health of humans and the environment if released in an unsound manner. E-waste recorded in 2019 contained as much as 50 tons of mercury and 71 kt of brominated flame retardants [11]. The improper dumping and recycling of e-waste in several African countries serves as a major source for the release of harmful substances. These harmful substances can pollute soil, water, air, dust, and food sources [14][15]. Numerous studies have reported contamination of e-waste workers and local residents with toxic metals, dioxins and furans, brominated flame retardants (BFRs), PCBs, polyaromatic hydrocarbons (PAHs), per- and polyfluoroalkyl substances (PFAS), particulate matter and other air pollutants, phthalates and other chemicals in plastics, and the chemical mixtures at these sites [16][17][18][19][20][21][22]. There are other chemicals present in e-waste for which little information is available. Growing research has found associations between e-waste recycling and a range of adverse health effects, including negative birth outcomes, impaired neurological and behavioral development, impaired thyroid function, and increased risk of chronic diseases later in life [23].
While there is significant exposure risk to all individuals who are involved in informal e-waste recycling [24] children are particularly vulnerable to exposure from hazardous chemicals released during informal or unregulated e-waste recycling activities due to their developing organs and immune system, rapid growth, and developmental vulnerabilities. Child labor has been documented at informal e-waste recycling areas across the world. Children as young as five years of age have been observed engaging in e-waste activities in Ghana [12]. Children breathe more air and ingest more food and water relative to their size than adults. As a result, children have higher intakes of pollutants relative to their size than adults. In addition, children’s bodies metabolize and eliminate toxic substances differently compared to adults, making them less able to break down and eliminate some hazardous substances. Children are also closer to the ground, where some toxicants may be at their highest concentration, and are more likely to put their hands, objects, and soil into their mouths, increasing their risk of ingesting contaminants [25][26][27]. Steps taken to protect children will also serve to protect adults.
2. What Chemicals Are Present in E-Waste?
Tsydenova and Bengtsson [28] reviewed the distribution of toxic chemicals in different items commonly found in e-waste. Their research indicated that common electronic items and their components, such as batteries, switches, relays, and printed circuit boards, may contain antimony, barium, beryllium, cadmium, copper, gold, lead, lithium, mercury, nickel, silver, palladium, and zinc. Items are also known to contain a variety of organic chemicals and rare earth metals, many of which have not been studied for health effects. Plastics may constitute as much as 30% of e-waste by weight [29] and BFRs are added to most plastics to reduce flammability. BFRs are often found in computers and other electronics for the same reason. As many plastics contain chlorine, combustion of plastic results in the formation of both chlorinated and brominated dioxins and furans. Figure 1 shows the sources of some of these chemicals.
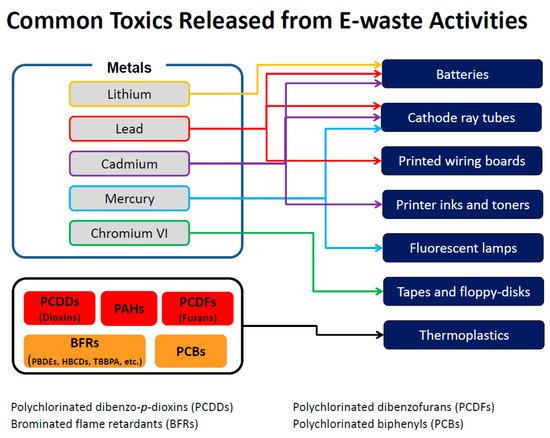
Figure 1. Sources and chemicals released from some specific e-waste sites. Adapted from: Frazzoli, et al. [30].
Landfills in African countries containing e-waste have shown elevations of many different potentially hazardous metals [31]. Soils around an informal e-waste recycling site in Nigeria have displayed elevated levels of copper, lead, zinc, manganese, nickel, antimony, chromium, cadmium [32]. Alabi et al. [33] compared levels of metals in soils and plants at e-waste sites in China and Nigeria and found that both sites had significant elevations of lead, copper, chromium, nickel, cadmium, and manganese relative to Dutch and Chinese standards. Several reports have investigated blood concentrations of metals in the blood of residents living near e-waste recycling sites as compared to control areas. Li et al. [34] reported that residents living near Taizhou, when studied two years after the site was closed, had elevated levels of chromium, arsenic, cobalt, nickel, silver, tin, mercury, lanthanum, and cerium as compared to residents at a control site. In Ghana, workers at an e-waste recycling site displayed significantly higher concentrations of blood lead, cadmium, chromium, and urinary nickel when compared to non-e-waste workers [35]. Many of these metals are toxic, especially to children.
3. How Does E-Waste Pollute the Environment?
E-waste is recognized as a resource as it contains valuable materials [18]. However, in developing countries many e-waste recyclers use primitive methods, such as mechanical shredding, manual dismantling and sorting and open burning, to isolate these valuable materials [16][36]. Plastics are burned, often at low temperatures, to dispose of computer casings and to retrieve metals from electronic chips and other components leading to the formation of dioxins [37]. Old tires may be burned to generate the heat to melt wires and incineration is used to extract valuable materials [38]. Since there are often inadequate stack emission controls, incineration can also release harmful heavy metals into the environment [37]. Strong acids are used to extract metals from printed circuit boards [39].
These methods result in severe air pollution containing many toxic substances around e-waste recycling areas. Particulates and other air pollutants are inhaled by workers and nearby residents. In addition, chemicals escape and may contaminate dust, soil, and water in communities around dump sites leading to additional routes of exposure, such as dermal contact and consumption of contaminated food and water [40]. E-waste contains as many as 1000 different harmful substances that have been identified as either components of e-waste or involved in the processing systems engaged by informal recyclers [41]. Fetuses, infants and children are at particular risk as their bodies are undergoing vital development. Exposure to such toxicants results in the disruption of these development processes and may result in long-term health impacts. Physical injuries, including burns, cuts and scrapes and musculoskeletal injuries, are also a major problem among e-waste workers in African countries, due to lack of safety measures, training, and education [42]. Some of the hazardous substances found in e-waste and their associated health effects are discussed in detail below. As very few health effects studies have been completed at African sites, the findings are supported by additional research completed at other international e-waste sites.
4. Health Effects Associated with Chemicals Found in E-Waste
Lead: Lead is a well-known neurotoxin [43]. Even relatively low lead exposure in children can result in a reduction in total intelligence quotient (IQ) and several behavioral abnormalities, including decrease in attention span and increase in frustration and disruptive behavior. At higher exposures, lead can cause anemia, coma and even death. There is no concentration of lead that does not have adverse effects on neurobehavior [44]. Lead exposure at e-waste recycling sites has been associated with altered physical development, increased bone resorption and childhood temperament abnormalities [45][46]. The chain of activities at e-waste sites and the long duration of exposures to lead are major contributing factors to the elevated blood lead levels in African countries [47].
Manganese: Manganese is an essential nutrient, but both deficiency and excessive exposure can cause disease. Excessive prenatal manganese levels may result in lower birth weight [48] and adverse effects on child neurodevelopment [49]. Rodriquez-Barranco et al. [50], in a meta-analysis of 17 publications, concluded that a 50% increase in manganese concentrations in hair was associated with a 0.7 IQ deficit in children between 6–13 years of age. Manganese exposure from e-waste has been associated with reduced lung function and elevations in levels of malondialdehyde and superoxide dismutase in children aged 8–13 years [51].
Mercury: E-waste workers are at risk from exposure to both mercury and methylmercury. Methylmercury is formed when elemental or inorganic mercury is deposited in the environment. Methylmercury is a potent neurotoxicant and the major route of exposure is consumption of contaminated fish. The release of mercury compounds near e-waste sites can result in serious contamination of an important food source. Sensitive populations, such as pregnant women and children, are at high risk of severe health effects of mercury inhalation due to its impact on the renal and central nervous systems [52]. Metal artisans, who are at risk of chronic mercury exposure, have displayed prevalence of cough, chest pain, dyspnea, interstitial pneumonitis, and impaired pulmonary function [53]. High blood mercury levels in children aged 3–6 years were associated with elevated 8-hydroxydeoxyguanosine (8-OHdG), an indication of oxidative DNA damage at an e-waste site [54].
Nickel: Nickel is a neurotoxic, immunotoxic, nephrotoxic, and genotoxic agent [55]. Some nickel compounds are known human carcinogens while metallic nickel is listed as a possible human carcinogen [56]. Allergic reactions are common effects of nickel exposure, such as skin rashes, dermatitis, and hand eczema. Inhalation of nickel exposure can trigger asthma attacks. Occupational exposure to dust containing nickel has been linked to reduced lung function, nasal sinus, chronic bronchitis, and lung cancer [56]. E-waste workers are vulnerable to these health outcomes. Ni et al. [57] suggested a positive association between nickel and neonatal umbilical cord blood plasma 8-OHdG concentrations. School children residing in an e-waste recycling area have shown significant body accumulation of nickel. Lower forced vital capacity, decrease in catalase activities and significant increase in superoxide dismutase activities and malondialdehyde levels has been associated with nickel exposure in boys aged 8–9 years at an e-waste site [51].
Arsenic: Arsenic is highly toxic to humans. Studies of children in China have found that elevated arsenic in drinking water is associated with a reduction in childhood IQ [58][59]. Parajuli et al. [60] found that cord blood levels of arsenic were associated with reduced neurodevelopmental indicators in newborns. Liu et al. [61] reported that women exposed to elevated arsenic levels in soil during pregnancy were associated with elevated rates of developmental delays in their children. High urinary arsenic has been associated with decrease in IQ in children between 6–13 years of age [62]. Arsenic is also a potent carcinogen [63] and a major cause of cardiovascular disease [64]. While these diseases may not appear during childhood, exposure during childhood may increase risk of disease later in life. Very little research on arsenic has been conducted at e-waste sites.
Cadmium: Cadmium is a known human carcinogen and can have adverse effects on cognitive function. Cadmium has been associated with adverse birth outcomes, reduced cognitive development and IQ and elevated withdrawal, social and attention problems in children [65][66][67][68]. Cadmium exposure in adults is associated with elevated risk of peripheral artery disease [64], but this has not been studied in children. At e-waste sites in China, cadmium exposure has been linked to increased risk of sex-specific adverse birth outcomes and altered mitochondrial respiration [69][70].
Chromium: Chromium (VI) is a genotoxic carcinogen, whereas chromium (III) is an essential micronutrient [71]. Children exposed to chromium at e-waste sites have shown lymphocyte DNA damage [72] and reduced weight and chest circumference as compared to unexposed children [73]. Preschool age children from an e-waste site have shown reduced lung function and lower concentrations of hemoglobin in comparison to unexposed children [74].
Polybrominated diphenyl ethers (PBDEs): PBDEs are a group of BFRs. BFRs can be detected at elevated levels in humans and the environment in areas far away from the points of production, are resistant to degradation and can bioaccumulate [75]. Studies of the health effects of PBDEs on humans have shown significant change in thyroid stimulating hormone in children [76], decrease in full-scale IQ following prenatal exposure [77] and changes in childhood body mass index (BMI) [78] Animal studies indicate that early life exposure to PBDEs promotes obesity later in life [79], and human studies support this conclusion [80]. Elevated concentrations of PBDEs have been found in breastmilk, soil, plants and animals at e-waste sites in China [81][82]. Workers at e-waste sites have shown altered thyroid function associated with elevated serum PBDE levels [82]. Exposure to PBDEs at e-waste sites has also been correlated with reduced head circumference and neonatal BMI, decrease in Apgar1 score, changes to human semen quality and thyroid and endocrine system function [83][84][85][86][87].
Dioxins, furans, PCBs: Dioxins, furans, and PCBs are known human carcinogens [88][89] Dioxins and furans at e-waste sites are primarily products of combustion. Dioxins, furans and PCBs have been found at elevated levels in soil and air at e-waste sites [90]. There is strong evidence that individuals working at e-waste sites and their children have elevated blood, hair, serum and tissue levels of dioxins, furans, polybrominated biphenyls (PBBs) and PCBs associated with e-waste recycling activities in Ghana [22][91] and China [92][93][94]. PCBs can cause adverse alterations to the nervous system, skin, thyroid and sex steroid hormonal systems, liver, kidney, cardiovascular system, and pancreas [95][96][97]. Children exposed to dioxins, furans and PCBs are at significant risk of respiratory infections and decrease in lung function [98][99]. Exposure to dioxins, furans and PCBs at e-waste sites has been linked to changes in immune system function and significant changes to thyroid and endocrine system function [84][100][101][102].
Perfluoroalkyl substances (PFAS): There are more than 5000 PFAS widely used as stain resistant and water repellent chemicals on fabrics, household products and as fire-fighting foam [103]. These chemicals have been found in drinking water, food, and indoor and outdoor air [104]. Exposure can result in elevated rates of some cancers [105], altered immune function with a reduced response to immunization [106] and disruption of thyroid hormone function [107]. There is little available research on PFAS compounds at e-waste sites [108] reported that mothers from an e-waste site were exposed to higher levels of perfluorooctanoic acid (PFOA), a common PFAS, and that prenatal exposure was associated with adverse birth outcomes and decreased physical development in children.
Polycyclic aromatic hydrocarbons (PAHs): PAHs at e-waste sites are products of incomplete combustion. Workers and residents at e-waste sites encounter PAHs primarily through inhalation and dermal exposure. PAH exposure has been associated with cancers in adults [109][110], DNA damage in newborns [109], respiratory and cardiovascular effects in schoolchildren [111][112] and adverse neuro- and physical development outcomes in children [113][114]. Elevated levels of PAH metabolites have been found in the urine of e-waste workers in Ghana [17]. Cancer risk from inhalation of PAHs near an e-waste recycling area in South China was reported to be 1.6 times that of a control area [115]. Carcinogenic PAH congeners have been positively associated with BMI and child physical growth indicators, such as reduced head circumference and Apgar1 score, at e-waste sites [116][117]. PAHs were observed in preschool children at an e-waste recycling site in China and linked to exacerbated vascular endothelial inflammation [118].
Particulate air pollution: The joint effects of ambient and household air pollution caused an estimated 543,000 deaths in children under five years and seven million premature deaths across the globe in 2016 [119]. Children in African countries are exposed to a double burden of toxicants from air pollution. While children are exposed to ambient air pollution, including from e-waste activities, they also spend a lot of time at home, indoors, where they may be exposed to toxic pollutants from the incomplete combustion of polluting fuels and technologies used for cooking, heating, and lighting. African countries have some of the highest burdens of disease and disability-adjusted life-years among children due to household air pollution [119].
References
- Garcia-Perez, J.; Fernandez-Navarro, P.; Castello, A.; Lopez-Cima, M.F.; Ramis, R.; Boldo, E.; Lopez-Abente, G. Cancer mortality in towns in the vicinity of incinerators and installations for the recovery or disposal of hazardous waste. Environ. Int. 2012, 51, 31–44.
- Hu, S.-W.; Sju, C.M. Health effects of waste incineration: A review of epidemiologic studies. J. Air Waste Manag. Assoc. 2001, 51, 1100–1109.
- Gourmelon, G. Global Plastic Production Rises, Recycling Lags; Vital Signs, Worldwatch Institute: Washington, DC, USA, 2015; pp. 91–95.
- Wright, R.O.; Amarasiriwardeena, C.; Woolf, A.D.; Jim, R.; Bellinger, D.C. Neuropsychological Correlates of Hair Arsenic, Manganese, and Cadmium Levels in School-Age Children Residing Near a Hazardous Waste Site. NeuroToxicology 2000, 27, 210–216.
- Elliott, P.; Briggs, D.; Morris, S.; de Hoogh, C.; Hurt, C.; Jensen, T.K.; Maitland, I.; Richardson, S.; Wakefield, J.; Jarup, L. Risk of adverse birth outcomes in populations living near landfill sites. BMJ 2001, 323, 363–368.
- UNEP (United Nations Environment Programme). Technical Guidelines on Transboundary Movements of Electrical and Electronic Waste and Used Electrical and Electronic Equipment, in Particular Regarding the Distinction between Waste and Non-Waste under the Basel Convention. 2019. Available online: http://www.basel.int/Implementation/TechnicalMatters/DevelopmentofTechnicalGuidelines/TechnicalGuidelines/tabid/8025/Default.aspx (accessed on 20 December 2020).
- Kaya, M. Recovery of metals and nonmetals from electronic waste by physical and chemical recycling processes. Waste Manag. 2016, 57, 64–90.
- Ongondo, F.O.; Williams, I.D.; Cherrett, T.J. How are WEEE doing? A global review of the management of electrical and electronic wastes. Waste Manag. 2011, 31, 714–730.
- Carpenter, D.O. Occurrence and human health risk of emerging organic contaminants in e-waste. Compr. Anal. Chem. 2015, 67, 347–362.
- Iqbal, M.; Syed, J.H.; Breivik, K.; Chaudhry, M.J.I.; Li, J.; Zhang, G.; Malik, R.N. E-waste driven pollution in Pakistan: The first evidence of environmental and human exposure to flame retardants (frs) in Karachi city. Environ. Sci. Technol. 2017, 51, 13895–13905.
- Forti, V.; Balde, C.P.; Kuehr, R.; Bel, G. The Global E-Waste Monitor 2020. 2020. Available online: https://www.iswa.org/home/news/news-detail/article/-21c8325490/109/ (accessed on 11 September 2020).
- Prakash, S.; Manhart, A.; Amoyaw-Osie, Y.; Agyekum, O.O. Socio-Economic Assessment and Feasibility Study on Sustainable E-Waste Management in Ghana. Commissioned by Inspectorate of the Ministry of Housing, Spatial Planning and the Environment of the Netherlands and the Dutch Association for the Disposal of Metal and Electrical Products. Available online: https://www.oeko.de/oekodoc/1057/2010-105-en.pdf (accessed on 6 March 2020).
- Oteng-Ababio, M.; van der Velden, M.; Taylor, M.B. Building policy coherence for sound waste electrical and electronic equipment management in a developing country. J. Environ. Dev. 2020, 29, 306–328.
- Deng, W.J.; Louie, P.K.K.; Liu, W.K.; Bid, X.H.; Fu, J.M.; Wong, M.H. Atmospheric levels and cytotoxicity of PAHs and heavy metal in TSP and PM2.5 at an electronic waste recycling site in southeast China. Atmos. Environ. 2006, 40, 56945–56955.
- Leung, A.O.W.; Duzgoren-Aydin, N.S.; Cheung, K.C.; Wong, M.H. Heavy metals concentrations of surface dust from e-waste recycling and its human health implications in Southeast China. Environ. Sci. Technol. 2008, 42, 2674–2680.
- Asante, K.A.; Agusa, T.; Biney, C.A.; Agyekum, W.A.; Bello, M.; Otsuka, M.; Itai, T.; Takahashi, S.; Tanabe, S. Multi-trace element levels and arsenic speciation in urine of e-waste recycling workers from Agbogbloshie, Accra in Ghana. Sci. Total Environ. 2012, 424, 63–73.
- Feldt, T.; Fobil, J.N.; Willsiepe, J.; Wilhelm, M.; Till, H.; Zoufaly, A.; Burchard, G.; Goen, T. High levels of PAH-metabolites in urine of e-waste recycling workers from Agbogbloshie, Ghana. Sci. Total Environ. 2014, 466–467, 369–376.
- Heacock, M.; Kelly, C.B.; Suk, W.A. E-waste: The growing global problem and next steps. Rev. Environ. Health 2016, 31, 131–135.
- Kaifie, A.; Schettgen, T.; Bertram, J.; Löhndorf, K.; Waldschmidt, S.; Felten, M.K.; Kraus, T.; Fobil, J.N.; Küpper, T. Informal e-waste recycling and plasma levels of non-dioxin-like polychlorinated biphenyls (NDL-PCBs)—A cross-sectional study at Agbogbloshie, Ghana. Sci. Total Environ. 2020, 723, 138073.
- Srigboh, R.K.; Basu, N.; Stephens, J.; Asampong, E.; Perkins, M.; Neitzel, R.L.; Fobil, J. Multiple elemental exposures amongst workers at the Agbogbloshie electronic waste (e-waste) site in Ghana. Chemosphere 2016, 164, 68–74.
- Tue, N.M.; Goto, A.; Takahashi, S.; Itai, T.; Asante, K.A.; Kunisue, T.; Tanabe, S. Release of chlorinated, brominated, and mixed halogenated dioxin-related compounds to soils from open burning of e-waste in Agbogbloshie (Accra, Ghana). J. Hazard. Mater. 2016, 302, 151–157.
- Wittsiepe, J.; Fobil, J.N.; Till, H.; Burchard, G.D.; Wilhelm, M.; Feldt, T. Levels of polychlorinated dibenzo-p-dioxins, dibenzofurans (PCDD/Fs) and biphenyls (PCBs) in blood of informal e-waste recycling workers from Agbogbloshie, Ghana, and controls. Environ. Int. 2015, 79, 65–73.
- Grant, K.; Goldizen, F.; Sly, P.; Bruné, M.; Neira, M.; Berg, M.V.; Norman, R. Health consequences of exposure to e-waste: A systematic review. Lancet Glob. Health 2013, 1, e350–e361.
- Ohajinwa, C.M.; van Bodegom, P.M.; Osibanjo, O.; Xie, Q.; Chen, J.; Vijver, M.G.; Peijnenburg, W. Health risks of polybrominated diphenyl ethers (PBDES) and metals at informal electronic waste recycling sites. Int. J. Environ. Res. Public Health 2019, 16, 906.
- Chen, A.; Dietrich, K.N.; Huo, X.; Ho, S.M. Developmental neurotoxicants in e-waste: An emerging health concern. Environ. Health Perspect. 2011, 119, 431–438.
- Heacock, M.; Kelly, C.B.; Asante, K.A.; Birnbaum, L.S.; Bergman, A.L.; Bruné, M.N.; Buka, I.; Carpenter, D.O.; Chen, A.; Huo, X.; et al. E-waste and harm to vulnerable populations: A growing global problem. Environ. Health Perspect. 2015, 124, 550–555.
- Suk, W.A.; Ruchirawat, K.M.; Balakristnan, K.; Berger, M.; Carpenter, D.; Damstra, T.; de Garbino, P.J.; Koh, D.; Landrigan, P.J.; Makalinao, I.; et al. Environmental threats to children’s health in Southeast Asia and the Western Pacific. Environ. Health Perspect. 2003, 111, 1340–1347.
- Tsydenova, O.; Bengtsson, M. Chemical hazards associated with treatment of waste electrical and electronic equipment. Waste Manag. 2011, 31, 45–58.
- Schlummer, M.; Gruber, A.; Wolz, G.; van Eldik, R. Characterization of polymer fractions from waste electrical and electronic equipment (WEEE) and implications for waste management. Chemosphere 2007, 67, 1866–1876.
- Frazzoli, C.; Orisakwe, O.E.; Dragone, R.; Mantovani, A. Diagnostic health risk assessment of electronic waste on the general population in developing countries scenarios. Environ. Impact Assess. Rev. 2010, 30, 388–399.
- Kiddee, P.; Naidu, R.; Wong, M.H. Metals and polybrominated diphenyl ethers leaching from electronic waste in simulated landfills. J. Hazard. Mater. 2013, 252–253, 243–249.
- Isimekhai, K.A.; Garelick, H.; Watt, J.; Purchase, D. Heavy metals distribution and risk assessment in soil from an informal e-waste recycling site in Lagos State, Nigeria. Environ. Sci. Pollut. Res. Int. 2017, 24, 17206–17219.
- Alabi, O.A.; Bakare, A.A.; Xu, X.; Li, B.; Zhang, Y.; Huo, X. Comparative evaluation of environmental contamination and DNA damage induced by electronic waste in Nigeria and China. Sci. Total Environ. 2012, 423, 62–72.
- Li, Z.; Li, X.; Qian, Y.; Guo, C.; Liu, H.; Wang, Z.; Wei, Y. The sustaining effects of e-waste on HPA axis reactivity and oxidative stress and their association with metals of blood. Sci. Total Environ. 2019, 739, 139964.
- Wittsiepe, J.; Feldt, T.; Till, H.; Burchard, G.; Wilhelm, M.; Fobil, J.N. Pilot study on the internal exposure to heavy metals of informal-level electronic waste workers in Agbogbloshie, Accra, Ghana. Environ. Sci. Pollut. Res. Int. 2017, 24, 3097–3107.
- Asante, K.A.; Pwamang, J.A.; Amoyaw-Osei, Y.; Ampofo, J.A. E-waste interventions in Ghana. Rev. Environ. Health 2016, 31, 145–148.
- Perkins, D.N.; Brune Drisse, M.N.; Nxele, T.; Sly, P.D. E-waste: A global hazard. Ann. Glob. Health 2014, 80, 286–295.
- Okwu, P.I.; Onyeje, I.N. Extraction of valuable substances from e-waste. Am. J. Eng. Res. 2014, 3, 299–304.
- Nnorom, I.C.; Osibanjo, O. Electronic waste (e-waste): Material flows and management practices in Nigeria. Waste Manag. 2008, 28, 1472–1479.
- Daum, K.; Stoler, J.; Grant, R. Toward a more sustainable trajectory for e-waste policy: A review of a decade of e-waste research in Accra, Ghana. Int. J. Environ. Res. Public Health 2017, 14, 135.
- Needhidasan, S.; Samuel, M.; Chidambaram, R. Electronic waste—An emerging threat to the environment of urban India. J. Environ. Health Sci. Eng. 2014, 12, 36.
- Ohajinwa, C.M.; van Bodegom, P.M.; Vijver, M.G.; Olumide, A.O.; Osibanjo, O.; Peijenburg, W.J. Prevalence and injury patterns among electronic waste workers in the informal sector in Nigeria. Inj. Prev. 2018, 24, 185–192.
- Needleman, H.L. Deficits in psychological and classroom performance of children with elevated dentine lead levels. N. Engl. J. Med. 1979, 300, 689–695.
- Lanphear, B.P.; Horning, R.; Khoury, J.; Yolton, K.; Baghurst, P.; Bellinger, D.C.; Canfield, R.L.; Dietrich, K.N.; Bornschein, R.; Greene, T.; et al. Low-level environmental lead exposure and children’s intellectual function: An international pooled analysis. Environ. Health Perspect. 2005, 113, 894–899.
- Liu, J.; Xu, X.; Wu, K.; Piao, Z.; Huang, J.; Cuo, Y.; Li, W.; Zhang, Y.; Chen, A.; Huo, X. Association between lead exposure from electronic waste recycling and child temperament alterations. Neurotoxicology 2011, 32, 458–464.
- Yang, H.; Huo, X.; Tekeen, T.A.; Zheng, Q.; Zheng, M.; Xu, X. Effects of lead and cadmium exposure from electronic waste on child physical growth. Environ. Sci. Pollut. Res. Int. 2013, 20, 4441–4447.
- Orisakwe, O.E.; Frazzoli, C.; Ilo, C.E.; Oritesemuelebi, B. Public health burden of e-waste in Africa. J. Health Pollut. 2019, 9, 190610.
- Zota, A.R.; Ettinger, A.S.; Bouchard, M.; Amarasiriwardena, C.J.; Schwartz, J.; Hu, H.; Wright, R.O. Maternal blood manganese levels and infant birth weight. Epidemiology 2009, 20, 367–373.
- Claus, H.B.; Ettinger, A.S.; Schwart, J.; Tellez-Rojo, M.M.; Lamadrid-Figueroa, H.; Hernandez-Avila, M.; Schnaas, L.; Amarasirwardena, C.; Bellinger, D.C.; Hu, H.; et al. Early postnatal blood manganese levels and children’s neurodevelopment. Epidemiology 2010, 21, 433–439.
- Carpenter, D.O.; Arcaro, K.; Spink, D.C. Understanding the human health effects of chemical mixtures. Environ. Health Perspect. 2002, 110, 25–42.
- Zheng, G.; Xu, X.M.; Bi, B.; Wu, K.; Yekeen, T.A.; Huo, X. Association between lung function in school children and exposure to three transition metals from an e-waste recycling area. J. Expo. Sci. Environ. Epidemiol. 2013, 23, 67–72.
- Diaz, S.M.; Palma, R.M.; Munoz, M.N.; Becerra-Arias, C.; Fernández Niño, J.A. Factors associated with high mercury levels in women and girls from the Mojana Region, Colombia, 2013–2015. Int. J. Environ. Res. Public Health 2020, 17, 1827.
- Monger, A.; Wangdi, K. Lead and mercury exposure and related health problems in metal artisan workplaces and high-risk household contacts in Thimphu, Bhutan. Sci. World J. 2020, 9267181.
- Xu, X.; Liao, W.; Lin, Y.; Dai, Y.; Shi, Z.; Huo, X. Blood concentrations of lead, cadmium, mercury and their association with biomarkers of DNA oxidative damage in preschool children living in an e-waste recycling area. Environ. Geochem. Health 2018, 40, 1481–1494.
- Das, K.K.; Das, S.N.; Dhundasi, S.A. Nickel, its adverse health effects, and oxidative stress. Indian J. Med. Res. 2008, 128, 412–425.
- ATSDR (Agency for Toxic Substances and Disease Registry). Public Health Statement for Nickel. 2005. Available online: https://www.atsdr.cdc.gov/phs/phs.asp?id=243&tid=44 (accessed on 8 May 2020).
- Ni, W.; Huang, Y.; Wang, X.; Zhang, J.; Wu, K. Associations of neonatal lead, cadmium, chromium and nickel co-exposure with DNA oxidative damage in an electronic waste recycling town. Sci. Total Environ. 2014, 472, 354–362.
- Dong, J.; Su, S.Y. The association between arsenic and children’s intelligence: A meta-analysis. Biol. Trace Elem. Res. 2009, 129, 88–93.
- Wang, S.X.; Wang, Z.H.; Cheng, X.T.; Li, J.; Sang, Z.P.; Zhang, X.D.; Han, L.; Qiao, X.; Wu, Z.; Wang, Z. Arsenic and fluoride exposure in drinking water: Children’s IQ and growth in Shanyin County, Shanxi Province, China. Environ. Health Perspect. 2007, 115, 643–647.
- Parajuli, R.P.; Fujiwara, T.; Umezaki, M.; Watanabe, C. Association of cord blood levels of lead, arsenic and zinc with neurodevelopmental indicators in newborns: A birth cohort study in Chitwan Valley, Nepal. Environ. Res. 2013, 121, 45–51.
- Liu, Y.; McDermott, S.; Lawson, A.; Aelion, C.M. The relationship between mental retardation and developmental delays in children and level in soil samples of arsenic, mercury and lead in soil samples taken near their mother’s residence during pregnancy. Int. J. Hyg. Environ. Health 2010, 213, 116–123.
- Rodriquez-Barrancco, M.; Lacasan, M.; Aguilar-Garduno, C.; Alguacil, J.; Gil, F.; González-Alzaga, B.; Rojas-García, A. Association of arsenic, cadmium, and manganese exposure with neurodevelopment and behavioural disorders in children: A systematic review and meta-analysis. Sci. Total Environ. 2013, 454–455, 562–577.
- Smith, A.H.; Goycolea, M.; Haque, R.; Biggs, M.L. Marked increase in bladder and lung cancer mortality in a region of Northern Chile due to arsenic in drinking water. Am. J. Epidemiol. 1998, 147, 660–669.
- Navas-Acien, A.; Silbergeld, E.K.; Sharrett, A.R.; Calderon-Aranda, E.; Selvin, E.; Guailar, E. Metals in urine and peripheral arterial disease. Environ. Health Perspect. 2005, 113, 164–169.
- Bao, Q.S.; Lu, C.Y.; Song, H.; Wang, M.; Ling, W.; Chen, W.Q.; Deng, X.Q.; Hao, Y.T.; Rao, S. Behavioral development of school-aged children who live around a multi-metal sulphide mine in Guangdong province, China: A cross-sectional study. BMC Public Health 2009, 9, 217.
- Caserta, D.; Granziano, A.; LoMonte, G.; Bordi, G.; Moscarini, M. Heavy metals and placental fetal-maternal barrier: A mini review on the major concerns. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 2198–2206.
- Kippler, M.; Bottai, M.; Georgiou, V.; Konra, K.; Chalkiadaki, G.; Kampouri, M.; Kyriklaki, A.; Vafeiadi, M.; Fthenou, E.; Vassilaki, M.; et al. Impact of prenatal exposure to cadmium on cognitive development at preschool age and the importance of selenium and iodine. Eur. J. Epidemiol. 2016, 31, 1123–1134.
- Tian, L.; Zhao, Y.C.; Wang, X.C.; Gu, J.L.; Sun, Z.I.; Zhang, Y.; Wang, J. Effects of gestational cadmium exposure on pregnancy outcome and development in the offspring at age 4.5 years. Biol. Trace Elem. Res. 2009, 132, 51–59.
- Xu, L.; Ge, J.; Huo, X.; Zhang, Y.; Lau, A.T.Y.; Xu, X. Differential proteomic expression of human placenta and fetal development following e-waste lead and cadmium exposure in utero. Sci. Total Environ. 2016, 550, 1163–1170.
- Zhang, Y.; Xu, X.; Chen, A.; Davuljigari, C.B.; Zheng, X.; Kim, S.S.; Dietrich, K.N.; Ho, S.M.; Reponen, T.; Huo, X. Maternal urinary cadmium levels during pregnancy associated with risk of sex-dependent birth outcomes from an e-waste pollution site in China. Reprod. Toxicol. 2018, 75, 49–55.
- Pellerin, C.; Booker, S.M. Reflections on hexavalent chromium: Health hazards of an industrial heavyweight. Environ. Health Perspect. 2000, 108, A402–A407.
- Li, Y.; Xu, X.; Liu, J.; Wu, K.; Gu, C.; Shao, G.; Chen, S.; Chen, G.; Huo, X. The hazard of chromium exposure to neonates in Guiyu of China. Sci. Total Environ. 2008, 403, 99–104.
- Xu, X.; Uekeen, T.A.; Liu, J.; Zhuang, B.; Li, W.; Huo, X. Chromium exposure among children from an electronic waste recycling town of China. Environ. Sci. Pollut. Res. Int. 2013, 22, 1778–1785.
- Zeng, X.; Xu, X.; Boezen, H.M.; Vonk, J.M.; Wu, W.; Huo, X. Decreased lung function with mediation of blood parameters linked to e-waste lead and cadmium exposure in preschool children. Environ. Pollut. 2017, 230, 838–848.
- Rahman, F.; Langford, K.H.; Scrimshaw, M.D.; Lester, J.N. Polybrominated diphenyl ether (PBDE) flame retardants. Sci. Total Environ. 2001, 275, 1–17.
- Jacobson, M.H.; Barr, D.B.; Marcus, M.; Muri, A.B.; Lyles, R.H.; Howards, P.P.; Pardo, L.; Darrow, L.A. Serum diphenyl ether concentrations and thyroid function in young children. Environ. Res. 2016, 149, 222–230.
- Chen, A.; Yoltan, K.; Rauch, S.A.; Webster, G.M.; Hornung, R.; Sjodin, A.; Dietrich, K.N.; Lanphear, B.P. Prenatal polybrominated diphenyl ether exposures and neurodevelopment in US children through 5 years of age: The HOME study. Environ. Health Perspect. 2014, 122, 856–862.
- Erkin-Cakmak, A.; Harley, K.G.; Chevrier, J.; Bradman, A.; Kogut, K.; Huen, K.; Eskenazi, B. In utero and childhood polybrominated diphenyl ether exposures and body mass at age 7 years: The CHAMACOS study. Environ. Health Perspect. 2015, 123, 636–642.
- Hoppe, A.A.; Carey, G.B. Polybrominated diphenyl ethers as endocrine disruptors of adipocyte metabolism. Obesity 2007, 15, 2942–2950.
- Dishaw, L.V.; Macaulay, L.J.; Roberts, S.C.; Stapleton, H.M. Exposures, mechanisms, and impact of endocrine-active flame retardants. Curr. Opin. Pharmacol. 2014, 19, 125–133.
- Liu, H.; Zhou, Q.; Wang, Y.; Zhang, Q.; Cai, Z.; Jiang, G. E-waste recycling induced polybrominated diphenyl ethers, polychlorinated biphenyls, polychlorinated dibenzo-p-dioxins and dibenzo-furans pollution in the ambient environment. Environ. Int. 2008, 34, 67–72.
- Yuan, J.; Chen, L.; Chen, D.; Guo, H.; Bi, X.; Ju, Y.; Jiang, P.; Shi, J.; Yu, Z.; Yang, J.; et al. Elevated serum polybrominated diphenyl ethers and thyroid-stimulating hormone associated with lymphocytic micronuclei in Chinese workers from an e-waste dismantling site. Environ. Sci. Technol. 2008, 42, 2195–2200.
- Li, M.; Huo, X.; Pan, Y.; Cai, H.; Dai, Y.; Xu, X. Proteomic evaluation of human umbilical cord tissue exposed to polybrominated diphenyl ethers in an e-waste recycling area. Environ. Int. 2018, 111, 362–371.
- Xu, L.; Huo, X.; Zhang, Y.; Li, W.; Zhang, J.; Xu, X. Polybrominated diphenyl ethers in human placenta associated with neonatal physiological development at a typical e-waste recycling area in China. Environ. Pollut. 2015, 196, 414–422.
- Xu, P.; Lou, X.; Ding, G.; Shen, H.; Wu, L.; Chen, Z.; Han, J.; Han, G.; Wang, X. Association of PCB, PBDE and PCDD/F body burdens with hormone levels for children in an e-waste dismantling area of Zhejiang province, China. Sci. Total Environ. 2014, 499, 55–61.
- Xu, P.; Lou, X.; Ding, G.; Shen, H.; Wu, L.; Chen, Z.; Han, J.; Wang, X. Effects of PCBs and PBDEs on thyroid hormone, lymphocyte proliferation, hematology and kidney injury markers in residents of an e-waste dismantling area in Zhejiang, China. Sci. Total Environ. 2015, 536, 215–222.
- Yu, Y.J.; Lin, B.G.; Liang, W.B.; Li, L.Z.; Hong, Y.D.; Chen, X.C.; Xu, X.Y.; Xiang, M.D.; Huang, S. Associations between PBDEs exposure from house dust and human semen quality at an e-waste area in South China: A pilot study. Chemosphere 2018, 198, 266–273.
- IARC (International Agency for Research on Cancer). Chemical Agents and Related Occupations: IARC Monographs on the Evaluation of Carcinogenic Risk to Humans; IARC: Lyon, France, 2012; Volume 100F, pp. 225–248. ISBN 978-92-832-1323-9.
- IARC (International Agency for Research on Cancer). Polychlorinated Biphenyls and Polybrominated Biphenyls; IARC Press: Lyon, France, 2016.
- Tue, N.M.; Takahashi, S.; Subramanian, A.; Sakai, S.; Tanabe, S. Environmental contamination and human exposure to dioxin-related compounds in e-waste recycling sites of developing countries. Environ. Sci. Process. Impacts 2013, 15, 1326–1331.
- Fujimori, T.; Itai, T.; Goto, A.; Asante, K.A.; Otsuka, M.; Takahashi, S.; Tanabe, S. Interplay of metals and bromine with dioxin-related compounds concentrated in e-waste open burning soil from Agbogbloshie in Accra, Ghana. Environ. Pollut. 2016, 209, 155–163.
- Zhao, G.; Wang, Z.; Dong, M.H.; Rao, K.; Luo, J.; Wang, D.; Zha, J.; Huang, S.; Xu, Y.; Ma, M. PBBs, PBDEs and PCBs levels in hair of residents around e-waste disassembly sites in Zhejiang Province, China, and their potential sources. Sci. Total Environ. 2008, 397, 46–57.
- Zhao, G.; Wang, Z.; Zhou, H.; Zhao, Q. Burden for PBBs, PBDEs, and PCBs in tissues of the cancer patients in the e-waste disassembly sites in Zhejiang, China. Sci. Total Environ. 2009, 407, 4831–4837.
- Shen, H.; Ding, G.; Han, G.; Wang, X.; Xu, X.; Han, J.; Lou, X.; Xu, C.; Cai, D.; Song, Y.; et al. Distribution of PCDD/Fs, PCBs, PBDEs and organochlorine residues in children’s blood from Zhejiang, China. Chemosphere 2010, 80, 170–175.
- ATSDR (Agency for Toxic Substances and Disease Registry). Toxicological Profile for Polychlorinated Biphenyls. US Department of Health and Human Services. 2000. Available online: https://www.atsdr.cdc.gov/toxprofiles/tp.asp?id=142&tid=26 (accessed on 8 May 2020).
- Carpenter, D.O. Exposure to and health effects of volatile PCBs. Rev. Environ. Health 2015, 30, 81–92.
- Carpenter, D.O. Polychlorinated biphenyls: Routes of exposure and effects on human health. Rev. Environ. Health 2006, 21, 1–23.
- Stolevik, S.B.; Nygaard, U.C.; Namork, E.; Haugen, M.; Kvalem, H.E.; Meltzer, H.M.; Alexander, J.; van Delft, J.; Loveren, H.; Lovik, M.; et al. Prenatal exposure to polychlorinated biphenyls and dioxins is associated with increased risk of wheeze and infections in infants. Food Chem. Toxicol. 2011, 49, 1843–1848.
- Tusscher, G.W.; de Weerdt, J.; Roos, C.M.; Griffioen, R.W.; De Jongh, F.H.; Westra, M.; van der Slikke, J.W.; Oosting, J.; Olie, K.; Koppe, J.G. Decreased lung function associated with perinatal exposure to Dutch background levels of dioxins. Acta Paediatr. 2001, 90, 1292–1298.
- Eguchi, A.; Nomiyama, K.; Minh Tue, N.; Trang, P.T.; Hung Viet, P.; Takahashi, S.; Tanabe, S. Residue profiles of organohalogen compounds in human serum from e-waste recycling sites in north Vietnam: Association with thyroid hormone levels. Environ. Res. 2015, 137, 440–449.
- Li, R.; Yang, Q.; Qiu, X.; Li, K.; Li, G.; Zhu, P.; Zhu, T. Reactive oxygen species alteration of immune cells in local residents at an electronic waste recycling site in northern China. Environ. Sci. Technol. 2013, 47, 3344–3352.
- Lv, Q.X.; Wang, W.; Li, X.H.; Yu, L.; Zhang, Y.; Tian, Y. Polychlorinated biphenyls and polybrominated biphenyl ethers in adipose tissue and matched serum from an e-waste recycling area (Wenling, China). Environ. Pollut. 2015, 199, 219–226.
- Yu, N.; Wen, H.; Wang, X.; Yamazaki, E.; Taniyasu, S.; Yamashita, N.; Yu, H.; Wei, S. Nontarget discovery of per- and polyfluoroalkyl substances in atmospheric particulate matter and gaseous phase using cryogenic air sampler. Environ. Sci. Technol. 2020, 54, 3103–3113.
- Poothong, S.; Papadopoulou, E.; Padilla-Sanchez, J.A.; Thomsen, C.; Haug, L.S. Multiple pathways of human exposure to poly-and perfluoroalkyl substances (PFAS): From external exposure to human blood. Environ. Int. 2020, 134, 105–244.
- Vieira, V.M.; Hoffman, K.; Shin, H.M.; Weinberg, J.M.; Webster, T.F.; Fletcher, T. Perfluorooctanoic acid exposure and cancer outcomes in a contaminated community: A geographic analysis. Environ. Health Perspect. 2013, 121, 318–323.
- Looker, C.; Luster, M.I.; Callafat, A.M.; Johnson, V.J.; Burleson, G.R.; Fletcher, T. Influenza vaccine response in adults exposed to perfluorooctanoate and perfluorooctanesulfonate. Toxicol. Sci. 2014, 138, 76–88.
- Wang, Y.; Rogan, W.J.; Chen, P.C.; Lien, G.W.; Chen, H.Y.; Tseng, Y.C.; Longnecker, M.P.; Wang, S.L. Association between maternal serum perfluoroalkyl substances during pregnancy and maternal and cord thyroid hormones: Taiwan maternal and infant cohort study. Environ. Health Perspect. 2014, 122, 5290534.
- Wu, K.; Xu, X.; Peng, L.; Liu, J.; Guo, Y.; Huo, X. Association between maternal exposure to perfluorooctanoic acid (PFOA) from electronic waste recycling and neonatal health outcomes. Environ. Int. 2012, 48, 1–8.
- Chen, S.C.; Liao, C.M. Health risk assessment on human exposed to environmental polycyclic aromatic hydrocarbons pollution sources. Sci. Total Environ. 2006, 366, 112–123.
- Shen, H.; Tao, S.; Liu, J.; Huang, Y.; Chen, H.; Li, W.M.; Zhang, Y.; Chen, Y.; Su, S.; Lin, B.; et al. Global lung cancer risk from PAH exposure highly depends on emission sources and individual susceptibility. Sci. Rep. 2014, 4, 6561.
- Barraza-Villarreal, A.; Escamilla-Nunez, M.C.; Schilmann, A.; Hernandez-Cadena, L.; Li, Z.; Romanoff, L.; Sjodin, A.; Rio-Navarro, B.E.; Diaz-Sanchez, D.; Diaz-Barriga, F.; et al. Lung function, airway inflammation, and polycyclic aromatic hydrocarbons exposure in Mexican schoolchildren: A pilot study. J. Occup. Environ. Med. 2014, 56, 415–419.
- Liu, H.; Xu, C.; Jiang, Z.Y.; Gu, A. Association of polycyclic aromatic hydrocarbons and asthma among children 6-19 years: NHANES 2001–2008 and NHANES 2011–2012. Respir. Med. 2016, 110, 20–27.
- Edwards, S.C.; Jedrychlowki, W.; Butscher, M.; Camann, D.; Kieltyka, A.; Mroz, E.; Flak, E.; Li, Z.; Wang, S.; Rauh, V.; et al. Prenatal exposure to airborne polycyclic aromatic hydrocarbons and children’s intelligence at 5 years of age in a prospective cohort study in Poland. Environ. Health Perspect. 2010, 118, 1326–1331.
- Perera, F.P.; Tang, D.; Wang, S.; Vishnevetsky, J.; Zhang, Z.; Diaz, D.; Ccamann, D.; Rauh, V. Prenatal polycyclic aromatic hydrocarbon (PAH) exposure and child behavior at age 6–7 years. Environ. Health Perspect. 2012, 120, 921–926.
- Wang, J.; Chen, S.; Tian, M.; Zheng, X.; Gonzales, L.; Ohura, T.; Mai, B.; Massey Simonich, S.L. Inhalation cancer risk associated with exposure to complex polycyclic aromatic hydrocarbon mixtures in an electronic waste and urban area in South China. Environ. Sci. Technol. 2012, 46, 9745–9752.
- Huo, X.; Wu, Y.; Xu, L.; Zeng, X.; Qin, Q.; Xu, X. Maternal urinary metabolites of PAHs and its association with adverse birth outcomes in an intensive e-waste recycling area. Environ. Pollut. 2019, 245, 453–461.
- Xu, X.; Liu, J.; Huang, C.; Lu, F.; Chiung, Y.M.; Huo, X. Association of polycyclic aromatic hydrocarbons (PAHs) and lead co-exposure with child physical growth and development in an e-waste recycling town. Chemosphere 2015, 139, 295–302.
- Zheng, X.; Huo, X.; Zhang, Y.; Wang, Q.; Zhang, Y.; Xu, X. Cardiovascular endothelial inflammation by chronic coexposure to lead (Pb) and polycyclic aromatic hydrocarbons from preschool children in an e-waste recycling area. Environ. Pollut. 2019, 246, 587–596.
- WHO (World Health Organization). Air Pollution and Child Health: Prescribing Clean Air Summary; World Health Organization: Geneva, Switzerland, 2018; Available online: https://apps.who.int/iris/handle/10665/275545 (accessed on 3 June 2020).
More
Information
Subjects:
Environmental Sciences
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.4K
Revisions:
2 times
(View History)
Update Date:
05 Sep 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




