
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jeyakumar Rajesh Banu | + 1685 word(s) | 1685 | 2021-08-10 09:39:21 | | | |
| 2 | Lindsay Dong | Meta information modification | 1685 | 2021-08-27 10:28:48 | | |
Video Upload Options
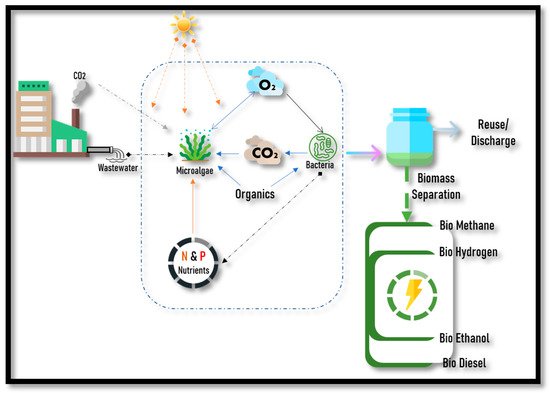
Using microalgae to treat wastewater as well as simultaneously produce biofuels is one of the approaches for a sustainable future. The manufacture of biofuels from microalgae is one of the next-generation biofuel solutions that has recently received a lot of interest, as it can remove nutrients from the wastewater whilst capturing carbon dioxide from the atmosphere. The resulting biomass are employed to generate biofuels, which can run fuel cell vehicles of zero emission, power combustion engines and power plants. By cultivating microalgae in wastewater, eutrophication can be prevented, thereby enhancing the quality of the effluent. Thus, by combining wastewater treatment and biofuel production, the cost of the biofuels, as well as the environmental hazards, can be minimized, as there is a supply of free and already available nutrients and water.
1. Introduction
2. Biofuel Production Pathways
2.1. Biodiesel
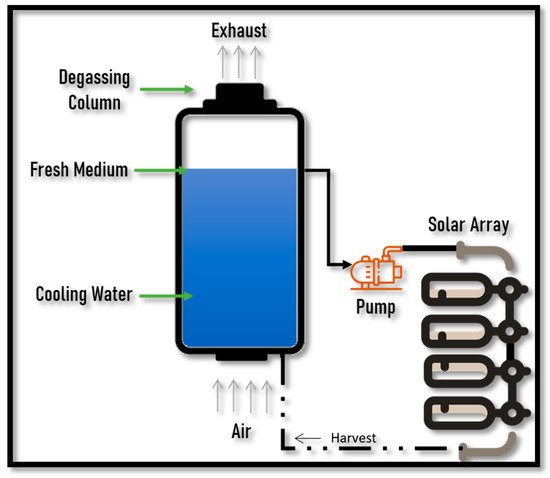
2.2. Biomethane
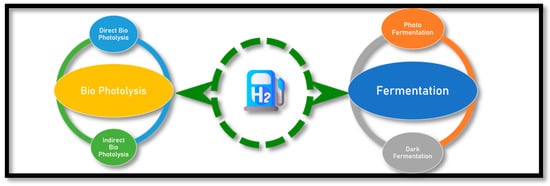
2.3. Biohydrogen
2.4. Bioethanol
3. Conclusions
References
- Steinman, A.D.; Vymazal, J. Algae and element cycling in wetlands. J. N. Am. Benthol. Soc. 1996, 15, 138–140.
- Larkum, A. Photosynthesis in Algae; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2003.
- Cock, J.M. Introduction to Marine Genomics; Springer: Berlin, Germany, 2010.
- Lee, R.E. Phycology, 4th ed.; Cambridge University Press: Cambridge, UK, 2008.
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577.
- Packer, M. Algal capture of carbon dioxide; biomass generation as a tool for greenhouse gas mitigation with reference to New Zealand energy strategy and policy. Energy Policy 2009, 37, 3428–3437.
- Ren, H.-Y.; Liu, B.-F.; Kong, F.; Zhao, L.; Ma, J.; Ren, N.-Q. Favorable energy conversion efficiency of coupling dark fermentation and microalgae production from food wastes. Energy Convers. Manag. 2018, 166, 156–162.
- Wendt, L.M.; Kinchin, C.; Wahlen, B.D.; Davis, R.; Dempster, T.A.; Gerken, H. Assessing the stability and techno-economic implications for wet storage of harvested microalgae to manage seasonal variability. Biotechnol. Biofuels 2019, 12, 80.
- Christenson, L.; Sims, R. Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnol. Adv. 2011, 29, 686–702.
- Pittman, J.K.; Dean, A.; Osundeko, O. The potential of sustainable algal biofuel production using wastewater resources. Bioresour. Technol. 2011, 102, 17–25.
- de Alva, M.S.; Luna-Pabello, V.M.; Cadena, E.; Ortíz, E. Green microalga Scenedesmusacutus grown on municipal wastewater to couple nutrient removal with lipid accumulation for biodiesel production. Bioresour. Technol. 2013, 146, 744–748.
- Prajapati, S.K.; Kaushik, P.; Malik, A.; Vijay, V.K. Phycoremediation coupled production of algal biomass, harvesting and anaerobic digestion: Possibilities and challenges. Biotechnol. Adv. 2013, 31, 1408–1425.
- Nayak, M.; Karemore, A.; Sen, R. Performance evaluation of microalgae for concomitant wastewater bioremediation, CO2 biofixation and lipid biosynthesis for biodiesel application. Algal Res. 2016, 16, 216–223.
- Prandini, J.M.; da Silva, M.L.B.; Mezzari, M.P.; Pirolli, M.; Michelon, W.; Soares, H.M. Enhancement of nutrient removal from swine wastewater digestate coupled to biogas purification by microalgae Scenedesmus spp. Bioresour. Technol. 2016, 202, 67–75.
- Van Wagenen, J.; Pape, M.L.; Angelidaki, I. Characterization of nutrient removal and microalgal biomass production on an industrial waste-stream by application of the deceleration-stat technique. Water Res. 2015, 75, 301–311.
- Gao, F.; Li, C.; Yang, Z.-H.; Zeng, G.-M.; Feng, L.; Liu, J.-Z.; Liu, M.; Cai, H.-W. Continuous microalgae cultivation in aquaculture wastewater by a membrane photobioreactor for biomass production and nutrients removal. Ecol. Eng. 2016, 92, 55–61.
- Green, F.B.; Bernstone, L.S.; Lundquist, T.J.; Oswald, W.J. Advanced integrated wastewater pond systems for nitrogen removal. Water Sci. Technol. 1996, 33.
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306.
- Johnson, M.B.; Wen, Z. Production of biodiesel fuel from the microalga Schizochytrium limacinum by direct transesterification of algal biomass. Energy Fuels 2009, 23, 5179–5183.
- Trindade, S.C. Nanotech biofuels and fuel additives. In Biofuel’s Engineering Process Technology; IntechOpen: London, UK, 2011.
- Wahlen, B.; Willis, R.M.; Seefeldt, L. Biodiesel production by simultaneous extraction and conversion of total lipids from microalgae, cyanobacteria, and wild mixed-cultures. Bioresour. Technol. 2011, 102, 2724–2730.
- Zabed, H.M.; Boyce, A.N.; Sahu, J.N.; Faruq, G. Evaluation of the quality of dried distiller’s grains with solubles for normal and high sugary corn genotypes during dry–grind ethanol production. J. Clean. Prod. 2017, 142, 4282–4293.
- Ayala-Parra, P.; Liu, Y.; Field, J.; Sierra-Alvarez, R. Nutrient recovery and biogas generation from the anaerobic digestion of waste biomass from algal biofuel production. Renew. Energy 2017, 108, 410–416.
- Zamalloa, C.; Boon, N.; Verstraete, W. Anaerobic digestibility of Scenedesmus obliquus and Phaeodactylum tricornutum under mesophilic and thermophilic conditions. Appl. Energy 2012, 92, 733–738.
- Saratale, R.G.; Kumar, G.; Banu, R.; Xia, A.; Periyasamy, S.; Saratale, G.D. A critical review on anaerobic digestion of microalgae and macroalgae and co-digestion of biomass for enhanced methane generation. Bioresour. Technol. 2018, 262, 319–332.
- Ward, A.; Lewis, D.; Green, F. Anaerobic digestion of algae biomass: A review. Algal Res. 2014, 5, 204–214.
- Park, J.; Craggs, R.; Shilton, A. Wastewater treatment high rate algal ponds for biofuel production. Bioresour. Technol. 2011, 102, 35–42.
- Sivakumar, G.; Xu, J.; Thompson, R.W.; Yang, Y.; Randol-Smith, P.; Weathers, P.J. Integrated green algal technology for bioremediation and biofuel. Bioresour. Technol. 2012, 107, 1–9.
- Xu, J.; Zhao, Y.; Zhao, G.; Zhang, H. Nutrient removal and biogas upgrading by integrating freshwater algae cultivation with piggery anaerobic digestate liquid treatment. Appl. Microbiol. Biotechnol. 2015, 99, 6493–6501.
- Ehimen, E.; Sun, Z.; Carrington, C.; Birch, E.; Eaton-Rye, J. Anaerobic digestion of microalgae residues resulting from the biodiesel production process. Appl. Energy 2011, 88, 3454–3463.
- Sforza, E.; Barbera, E.; Girotto, F.; Cossu, R.; Bertucco, A. Anaerobic digestion of lipid-extracted microalgae: Enhancing nutrient recovery towards a closed loop recycling. Biochem. Eng. J. 2017, 121, 139–146.
- Gonzalez, L.M.; Correa, D.F.; Ryan, S.; Jensen, P.D.; Pratt, S.; Schenk, P.M. Integrated biodiesel and biogas production from microalgae: Towards a sustainable closed loop through nutrient recycling. Renew. Sustain. Energy Rev. 2018, 82, 1137–1148.
- Wang, J.; Wan, W. Factors influencing fermentative hydrogen production: A review. Int. J. Hydrogen Energy 2009, 34, 799–811.
- Argun, H.; Kargi, F.; Kapdan, I.K.; Oztekin, R. Biohydrogen production by dark fermentation of wheat powder solution: Effects of C/N and C/P ratio on hydrogen yield and formation rate. Int. J. Hydrogen Energy 2008, 33, 1813–1819.
- Ferreira, A.F.; Marques, A.C.; Batista, A.P.; Marques, P.A.; Gouveia, L.; Silva, C. Biological hydrogen production by Anabaena sp.—Yield, energy and CO2 analysis including fermentative biomass recovery. Int. J. Hydrogen Energy 2012, 37, 179–190.
- Ruiz-Marin, A.; Canedo-López, Y.; Chávez-Fuentes, P. Biohydrogen production by Chlorella vulgaris and Scenedesmus obliquus immobilized cultivated in artificial wastewater under different light quality. AMB Express 2020, 10, 1–7.
- Nobre, B.; Villalobos, F.; Barragán-Huerta, B.E.; Oliveira, A.; Batista, A.P.; Marques, P.; Mendes, R.; Sovova, H.; Palavra, A.; Gouveia, L. A biorefinery from Nannochloropsis sp. microalga—Extraction of oils and pigments. Production of biohydrogen from the leftover biomass. Bioresour. Technol. 2013, 135, 128–136.
- Batyrova, K.; Hallenbeck, P.C. Hydrogen production by a Chlamydomonas reinhardtii strain with inducible expression of photosystem II. Int. J. Mol. Sci. 2017, 18, 647.
- Juantorena, A.; Sebastian, P.; Santoyo, E.; Gamboa, S.; Lastres, O.; Sánchez-Escamilla, D.; Bustos, A.; Eapen, D. Hydrogen production employing Spirulina maxima 2342: A chemical analysis. Int. J. Hydrogen Energy 2007, 32, 3133–3136.
- Batista, A.P.; Moura, P.; Marques, P.A.; Ortigueira, J.; Alves, L.; Gouveia, L. Scenedesmus obliquus as feedstock for biohydrogen production by Enterobacter aerogenes and Clostridium butyricum. Fuel 2014, 117, 537–543.
- Wieczorek, N.; Kucuker, M.A.; Kuchta, K. Fermentative hydrogen and methane production from microalgal biomass (Chlorella vulgaris) in a two-stage combined process. Appl. Energy 2014, 132, 108–117.
- Yang, Z.; Guo, R.; Xu, X.; Fan, X.; Li, X. Enhanced hydrogen production from lipid-extracted microalgal biomass residues through pretreatment. Int. J. Hydrogen Energy 2010, 35, 9618–9623.
- Hallenbeck, P. Fundamentals of the fermentative production of hydrogen. Water Sci. Technol. 2005, 52, 21–29.
- Kapdan, I.K.; Kargi, F. Bio-hydrogen production from waste materials. Enzym. Microb. Technol. 2006, 38, 569–582.
- Dale, B.E. Thinking clearly about biofuels: Ending the irrelevant ‘net energy’ debate and developing better performance metrics for alternative fuels. Biofuels Bioprod. Biorefin. 2007, 1, 14–17.
- Demirbaş, A. Conversion of biomass using glycerin to liquid fuel for blending gasoline as alternative engine fuel. Energy Convers. Manag. 2000, 41, 1741–1748.
- Naik, S.; Goud, V.V.; Rout, P.K.; Dalai, A.K. Production of first and second generation biofuels: A comprehensive review. Renew. Sustain. Energy Rev. 2010, 14, 578–597.
- Harun, R.; Singh, M.; Forde, G.M.; Danquah, M.K. Bioprocess engineering of microalgae to produce a variety of consumer products. Renew. Sustain. Energy Rev. 2010, 14, 1037–1047.
- Radakovits, R.; Jinkerson, R.; Darzins, A.; Posewitz, M.C. Genetic engineering of algae for enhanced biofuel production. Eukaryot. Cell 2010, 9, 486–501.
- González-Fernández, C.; Ballesteros, M. Linking microalgae and cyanobacteria culture conditions and key-enzymes for carbohydrate accumulation. Biotechnol. Adv. 2012, 30, 1655–1661.
- Ueno, Y.; Kurano, N.; Miyachi, S. Ethanol production by dark fermentation in the marine green alga, Chlorococcum littorale. J. Ferment. Bioeng. 1998, 86, 38–43.
- Hernández, D.; Riaño, B.; Coca, M.; González, M.C.G. Saccharification of carbohydrates in microalgal biomass by physical, chemical and enzymatic pre-treatments as a previous step for bioethanol production. Chem. Eng. J. 2015, 262, 939–945.
- Magneschi, L.; Catalanotti, C.; Subramanian, V.; Dubini, A.; Yang, W.; Mus, F.; Posewitz, M.C.; Seibert, M.; Perata, P.; Grossman, A.R. A mutant in the ADH1 gene of Chlamydomonas reinhardtii elicits metabolic restructuring during anaerobiosis. Plant Physiol. 2012, 158, 1293–1305.
- Dexter, J.; Armshaw, P.; Sheahan, C.; Pembroke, J.T. The state of autotrophic ethanol production in cyanobacteria. J. Appl. Microbiol. 2015, 119, 11–24.
- Saad, M.G.; Dosoky, N.S.; Zoromba, M.S.; Shafik, H.M. Algal Biofuels: Current Status and Key Challenges. Energies 2019, 12, 1920.




