
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Trinidad Ruiz-Téllez | + 3089 word(s) | 3089 | 2021-08-10 06:11:05 | | | |
| 2 | Catherine Yang | -5 word(s) | 3084 | 2021-08-24 02:47:34 | | |
Video Upload Options
Verbascum species (common mullein) have been widely used in Spanish folk medicine to treat pathologies related to the musculature, skeleton, and circulatory, digestive, and respiratory systems, as well as to treat infectious diseases and organ-sense illnesses.
1. Introduction
The genus Verbascum (Scrophulariaceae, Lamiales) comprises more than 300 Eurasiatic species. It is the largest genus of the family, and its origin is the center of the Eastern Mediterranean Basin. In the Iberian Peninsula, it is represented by 26 species [1]. In Spain, they are popularly named “gordolobos” (in English, common mullein), and the Spanish Inventory of Traditional Knowledge related to Biodiversity [2] has catalogued 10 species which have been used to treat a wide range of pathologies. These are Verbascum pulverulentum Vill. , V. sinuatum L., V. thapsus L., V. boerhavii L., V. creticum (L.) Cav. , V. dentifolium Delile, V. giganteum Willk. , V. lychnitis L., V. rotundifolium Ten. , and V. virgatum Stokes in With.
In order to realize the potential pharmacological application of these species, we must perform a deep analysis of their chemical compositions as a starting point to understand which phytochemicals could exert the medical actions described in the traditional knowledge. The chemical components of Verbascum spp. , and the biological actions attributed to these phytochemicals, can be found in the literature [3][4][5][6][7][8][9][10][11][12][13][14][15][16][17], with the correlation between the phytochemicals’ bioactivity and their traditional uses being a key point to validate their traditional ethnobotanical uses.
The aforementioned bibliographic prospection could be complemented by in silico approaches to demonstrate the phytochemicals’ affinities using molecular targets. The combination of bibliographic research and computer programming could provide a strong tool to approach the botanical bioactive compounds existing in Verbascum spp. with the medical uses collected by folk knowledge.
2. Use and application
2.1. Anti-Inflammatory Action of Verbascum
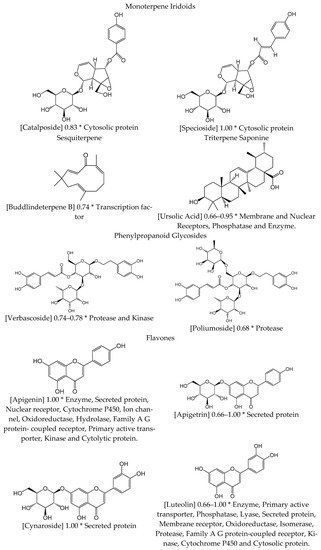
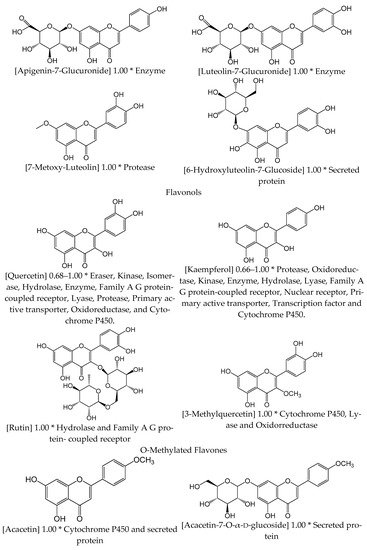
2.2. Circulatory System Diseases
| Uses | Vp | Vs | Vt | Vb | Vc | Vd | Vg | Vl | Vr | Vv | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Circulatory | Anti-hemorrhoidal | B/T | B/T | S/B/T | B/T | T | B | T | |||
| Leg treatment | B | ||||||||||
| Anti-hypertensive | I/B | I | B | ||||||||
| Digestive | Teeth pain, gumboil | B/T | B | B/T | T | ||||||
| Digestive | I/B/T | B/T | |||||||||
| Gastric ulcer/inflammation | B/T | I/B/T | B | ||||||||
| Liver inflammation | I/B | T | I/B/T | I/B | |||||||
| Gallstone | I | I | I/B | I | |||||||
| Anti-diarrhoea | T | I | T | ||||||||
| Constipation | B | E | |||||||||
| Respiratory | Hoarse, tonsillitis | B/T | I/T | I/B/T | |||||||
| Cold | B | I | I/B | I | B | ||||||
| Cough, asthma, bronchitis, hemoptysis | B | I/B/M | I/B | I | I | B | |||||
| Musculature & Skeleton | Anti-inflammatory (swelling) | B/T | I/B/T | ||||||||
| Contusion, broken bones | I/T | T | I/B/T | ||||||||
| Arthrosis, rheumatism | B/T | B/T | I | T | |||||||
| Skin | Eczema, exanthema | B/T | B/T | T | |||||||
| Cysts and zits | T | T | I/B/T | T | T | ||||||
| Wounds, ulcers, burns | B/T | M/T | I/B/M/ | T | T | ||||||
| Horsefly bite | M/T | ||||||||||
| Chilblain | B/T | B | B/T | B/T | B/T | ||||||
| Nail conditions | B/T | ||||||||||
| Sense | Conjunctivitis | M | M | M | |||||||
| Otitis | B/M | M | B | ||||||||
| Infectious parasitic diseases | Diphtheria | T | |||||||||
| Helminthiasis | B | ||||||||||
| Tuberculosis | I | ||||||||||
| Typhus | T | ||||||||||
| Mange | T |
2.3. Digestive Apparatus
2.4. Respiratory Diseases
2.5. Musculature and Skeleton
2.6. Skin and Sense Organs
2.7. Other Uses
References
- Benedí, C. Verbascum. Flora Iberica. In Flora Iberica; Benedí Gonzalez, C., Rico Hernández, E., Güemes Heras, J., Herrero Nieto, A., Eds.; CSIC: Madrid, Spain, 2009; Volume 13, pp. 49–97.
- Pardo de Santayana, M.; Morales, R.; Aceituno-Mata, L.; Molina, M. (Eds.) Inventario Español de Conocimientos Tradicionales Relativos a la Biodiversidad. Fase II (2); Ministerio de Agricultura y Pesca, Alimentación y Medio Ambiente: Madrid, Spain, 2018; ISBN 978-84-491-1472-4.
- De Pascual, T.J.; Diaz, F.; Grande, M. Components of Verbascum thapsus L. I. Triterpenes. An. Quim. 1978, 74, 311–314.
- De Pascual, T.J.; Diaz, F.; Grande, M. Components of Verbascum thapsus L. III. Contribution to the study of saponins. An. Quim. Ser. C 1980, 76, 107–110.
- Klimek, B. 6-O-p-coumaroylcatapol from Verbascum lychnitis. Planta Med. 1991, 57, 298.
- Warashina, T.; Miyase, T.; Ueno, A. Phenylethanoid and lignan glycosides from Verbascum thapsus. Phytochemistry 1992, 31, 961–965.
- Klimek, B. Flavonoid glucuronides from Verbascum lychnitis and V. nigrum. Acta Pol. Pharm. 1995, 52, 53–56.
- Tatli, I.I.; Akdemir, Z.Ş. Chemical constituents of Verbascum L. species. Fabad J. Pharm. Sci. 2004, 29, 93–107.
- Riaz, M.; Zia-Ul-Haq, M.; Jaafar, H.Z.E. Common mullein, pharmacological and chemical aspects. Braz. J. Pharmacogn. 2013, 23, 948–959.
- Bianco, A.; Guiso, M.; Iavarone, C.; Passacantilli, P.; Trogolo, C. 6-O-ß-D-Xylopyranosylaucubin from Ver bascum sinuatum. Phytochemistry 1980, 19, 571–573.
- Bianco, A.; Guiso, M.; Iavarone, C.; Passacantilli, P.; Trogolo, C. Sinuatol (6-O-a-L-rhamnopyranosyl-aucu bin) from Verbascum sinuatum. Planta Med. 1981, 41, 75–79.
- Bianco, A.; Guiso, M.; Iavarone, C.; Passacantilli, P.; Trogolo, C. 6-O-a-Sinuatosyl aucubin from Verbascum sinuatum. Phytochemistry 1981, 20, 465–468.
- Falsone, G.; Laryea, M.; Crea, A.; Finner, E. Iridoids from Verbascum sinuatum. J. Med. Plant Res. 1982, 44, 150–153.
- Hernandez-Hernandez, J. Determinacion de la estructura de los glicosidos de Verbascum lychnitis por espectrometria de masas. Quim. Ind. 1985, 31, 503–507.
- Souleles, C.; Geronikaki, A. Flavonoids from Verbascum thapsus. Sci. Pharm. 1989, 57, 59–61.
- Mehrotra, R.; Ahmed, B.; Vishwakarma, R.; Thakur, R. Verbacoside: A new luteolin glycoside from Verbascum thapsus. J. Nat. Prod. 1989, 52, 640–643.
- Warashina, T.; Miyase, T.; Ueno, A. Iridoid glycosides from Verbascum thapsus L. Chem. Pharm. Bull. 1991, 39, 3261–3264.
- Gupta, S.C.; Kunnumakkara, A.B.; Aggarwal, S.; Aggarwal, B.B. Inflammation, a Double-Edge Sword for Cancer and Other Age-Related Diseases. Front. Immunol. 2018, 9, 2160.
- De Caterina, R.; Zampolli, A. From Asthma to Atherosclerosis—5-Lipoxygenase, Leukotrienes, and Inflammation. N. Engl. J. Med. 2004, 350, 4–7.
- Werz, O. Inhibition of 5-lipoxygenase product synthesis by natural compounds of plant origin. Planta Med. 2007, 73, 1331–1357.
- Wisastra, R.; Dekker, F.J. Inflammation, cancer and oxidative lipoxygenase activity are intimately linked. Cancers 2014, 6, 1500–1521.
- Smith, W.L.; Murphy, R.C. The Eicosanoids: Cyclooxygenase, Lipoxygenase and Epoxygenase Pathways. In Biochemistry of Lipids, Lipoproteins and Membranes, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 259–296. ISBN 9780444634382.
- Calder, P.C. Eicosanoids. Essays Biochem. 2020, 64, 423–441.
- Shahidi, F.; Yeo, J.D. Bioactivities of phenolics by focusing on suppression of chronic diseases: A review. Int. J. Mol. Sci. 2018, 19, 1573.
- Bedard, K.; Krause, K.H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313.
- Xia, F.; Wang, C.; Jin, Y.; Liu, Q.; Meng, Q.; Liu, K.; Sun, H. Luteolin protects HUVECs from TNF-α-induced oxidative stress and inflammation via its effects on the Nox4/ROS-NF-κB and MAPK pathways. J. Atheroscler. Thromb. 2014, 21, 768–783.
- Hu, J.; Ma, W.; Li, N.; Wang, K.J. Antioxidant and anti-inflammatory flavonoids from the flowers of chuju, a medical cultivar of chrysanthemum morifolim ramat. J. Mex. Chem. Soc. 2017, 61, 282–289.
- Zhao, N.; Dong, Q.; Fu, X.X.; Du, L.L.; Cheng, X.; Du, Y.M.; Liao, Y.H. Acacetin blocks Kv1.3 channels and inhibits human T cell activation. Cell. Physiol. Biochem. 2014, 34, 1359–1372.
- Guo, H.; Li, M.; Xu, L.J. Apigetrin treatment attenuates LPS-induced acute otitis media though suppressing inflammation and oxidative stress. Biomed. Pharmacother. 2019, 109, 1978–1987.
- Lee, S.A.; Park, B.R.; Moon, S.M.; Shin, S.H.; Kim, J.S.; Kim, D.K.; Kim, C.S. Cynaroside protects human periodontal ligament cells from lipopolysaccharide-induced damage and inflammation through suppression of NF-κB activation. Arch. Oral Biol. 2020, 120, 104944.
- Lin, H.; Song, P.; Zhao, Y.; Xue, L.J.; Liu, Y.; Chu, C.Q. Targeting Th17 cells with small molecules and small interference RNA. Mediat. Inflamm. 2015, 2015, 290657.
- Cium, L.; Milaciu, M.V.; Runcan, O.; Vesa, C.; Negrean, V.; Pern, M.; Donca, V.I. The Effects of Flavonoids in Cardiovascular Diseases. Molecules 2020, 25, 4320.
- Gallego, E. Estudio Etnobotánico del Occidente Alistano; CSIC, Diputación de Zamora; Instituto de Estudios Zamoranos “Florián de Ocampo”: Zamora, Spain, 2009.
- Li, W.; Du, L.; Li, M. Alkaloids and flavonoids as α(1)-adrenergic receptor antagonists. Curr. Med. Chem. 2011, 18, 4923–4932.
- Sahni, S.K.; Baboota, S. Rutin: Therapeutic potential and recent advances in drug delivery. Drug Eval. 2013, 22, 1063–1079.
- Lorenz, P.; Conrad, J.; Stintzing, F.C. Metabolic fate of depsides and alkaloid constituents in aqueous extracts from Mercurialis perennis L. during fermentation. Chem. Biodivers. 2013, 10, 1706–1723.
- Li, D.; Ren, D.; Luo, Y.; Yang, X. Protective effects of ursolic acid against hepatotoxicity and endothelial dysfunction in mice with chronic high choline diet consumption. Chem. Biol. Interact. 2016, 258, 102–107.
- Hyndman, D.; Bauman, D.R.; Heredia, V.V.; Penning, T.M. The aldo-keto reductase superfamily homepage. Chem. Biol. Interact. 2003, 143, 621–631.
- Ianiro, G.; Pecere, S.; Giorgio, V.; Gasbarrini, A.; Cammarota, G. Digestive Enzyme Supplementation in Gastrointestinal Diseases. Curr. Drug Metab. 2016, 17, 187–193.
- Babamoradi, N.; Yousefi, S.; Ziarati, P. Optimization of ultrasound-assisted extraction of functional polysaccharides from common mullein (Verbascum thapsus L.) flowers. J. Food Process Eng. 2018, 41, e12851.
- Bylka, W.; Witkowska-Banaszczak, E.; Studzińska-Sroka, E.; Matławska, I. Phytotherapy of respiratory tract diseases. Wiad. Lek. 2012, 65, 124–131.
- Kumar, A.; Choudhir, G.; Shukla, S.K.; Sharma, M.; Tyagi, P.; Bhushan, A.; Rathore, M. Identification of phytochemical inhibitors against main protease of -19 using molecular modeling approaches. J. Biomol. Struct. Dyn. 2020.
- Mitra, D.; Verma, D.; Mahakur, B.; Kamboj, A.; Srivastava, R.; Gupta, S.; Pandey, A.; Arora, B.; Pant, K.; Panneerselvam, P.; et al. Molecular docking and simulation studies of natural compounds of Vitex negundo L. against papain-like protease (PLpro) of SARS CoV-2 (coronavirus) to conquer the pandemic situation in the world. J. Biomol. Struct. Dyn. 2021, 18, 1–22.
- Vardhan, S.; Sahoo, S.K. In silico ADMET and molecular docking study on searching potential inhibitors from limonoids and triterpenoids for COVID-19. Comput. Biol. Med. 2020, 124, 103936.
- Ryu, Y.B.; Jeong, H.J.; Kim, J.H.; Kim, Y.M.; Park, J.Y.; Kim, D.; Naguyen, T.T.H.; Park, S.J.; Chang, J.S.; Park, K.H.; et al. Biflavonoids from Torreya nucifera displaying SARS-CoV 3CLpro inhibition. Bioorg. Med. Chem. 2010, 18, 7940–7947.
- Kunnumakkara, A.B.; Rana, K.V.; Parama, D.; Banik, K.; Girisa, S.; Sahu, H.; Thakur, K.K.; Dutta, U.; Garodia, P.; Gupta, S.C.; et al. COVID-19, cytokines, inflammation, and spices: How are they related? Life Sci. 2020, 119201.
- Lai, C.C.; Shih, T.P.; Ko, W.C.; Tang, H.J.; Hsueh, P.R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int. J. Antimicrob. Agents 2020, 55, 105924.
- Ludwig, S.; Zarbock, A. Coronaviruses and SARS-CoV-2: A Brief Overview. Anesth. Analg. 2020, 131, 93–96.
- Wu, D.; Wu, T.; Liu, Q.; Yang, Z. The SARS-CoV-2 outbreak: What we know. Int. J. Infect. Dis. 2020, 44–48.
- Peter, A.E.; Sandeep, B.V.; Rao, B.G.; Kalpana, V.L. Calming the Storm: Natural Immunosuppressants as Adjuvants to Target the Cytokine Storm in COVID-19. Front. Pharmacol. 2021, 11, 2305.
- Jo, S.; Kim, S.; Shin, D.H.; Kim, M.S. Inhibition of SARS-CoV 3CL protease by flavonoids. J. Enzyme Inhib. Med. Chem. 2020, 35, 145–151.
- Saakre, M.; Mathew, D.; Ravisankar, V. Perspectives on plant flavonoid quercetin-based drugs for novel SARS-CoV-2. Beni Suef Univ. J. Basic Appl. Sci. 2021, 10, 21.
- Russo, M.; Moccia, S.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Roles of flavonoids against coronavirus infection. Chem. Biol. Interact. 2020, 328, 109211.
- Gasparotto-Junior, A.; Lima-Tolouei, S.E.; dos Reis Lívero, F.A.; Gasparotto, F.; Boeing, T.; de Souza, P. Natural Agents Modulating ACE-2: A Review of Compounds with Potential against SARS-CoV-2 Infections. Curr. Pharm. Des. 2021, 27, 1588–1596.
- Wang, W.; Sun, C.; Mao, L.; Ma, P.; Liu, F.; Yang, J.; Gao, Y. The biological activities, chemical stability, metabolism and delivery systems of quercetin: A review. Trends Food Sci. Technol. 2016, 56, 21–38.
- Almeida, A.F.; Borge, G.I.A.; Piskula, M.; Tudose, A.; Tudoreanu, L.; Valentová, K.; Williamson, G.; Santos, C.N. Bioavailability of Quercetin in Humans with a Focus on Interindividual Variation. Compr. Rev. Food Sci. Food Saf. 2018, 17, 714–731.
- Williamson, G.; Kerimi, A. Testing of natural products in clinical trials targeting the SARS-CoV-2 (Covid-19) viral spike protein-angiotensin converting enzyme-2 (ACE2) interaction. Biochem. Pharmacol. 2020, 178, 114123.
- Di Pierro, F.; Khan, A.; Bertuccioli, A.; Maffioli, P.; Derosa, G.; Khan, S.; Khan, B.A.; Nigar, R.; Ujjan, I.; Devraian, B.R. Quercetin Phytosome® as a potential candidate for managing COVID-19. Minerva Gastroenterol. 2021, 67, 190–195.
- Malik, N.A. Revised definition of pain by ‘International Association for the Study of Pain’: Concepts, challenges and compromises. Anaesth. Pain Intensive Care 2020, 24, 481–483.
- Gerdle, B.; Ghafouri, B. Proteomic studies of common chronic pain conditions—A systematic review and associated network analyses. Exp. Rev. Proteom. 2020, 17, 483–506.
- Holmquist, G.L. Opioid metabolism and effects of cytochrome P450. Pain Med. 2009, 10, S20–S29.
- Stoyanov, S.; Fleming, T.; Konrade, I.; Haag, G.; Humpert, P.; Rabbani, N.; Thornalley, P.; Brownlee, M.; Nawroth, P.; Bierhaus, A. The Glyoxalase I (GLO-1) system as modulator of pain in early diabetic neuropathy. Diabetol. Stoffwechs. 2008, 3, A36.
- Persson, L. Prenatal nutrition, socioenvironmental conditions, and child development. Lancet Glob. Health 2017, 5, 127–128.
- Indiana, M.; de Souza, F.H.V.; Eduardo, J.; Dantas Nascimento, P.G.B. Protein Kinases and Pain. In Protein Kinases; InTech: Rijeka, Croatia, 2012.
- Ma, H.; Qin, S.; Zhao, S. Osteoarthritis is Prevented in Rats by Verbascoside via Nuclear Factor kappa B (NF-kB) Pathway Downregulation. Med. Sci. Monit. 2020, 26, e921276.
- Gutiérrez-Rebolledo, G.A.; Garduño-Siciliano, L.; Chávez-Rueda, A.K.; Siordia-Reyes, A.G.; Zamilpa, A.; Jiménez-Arellanes, M.A. In vivo anti-arthritic and antioxidant effects from the standardized ethanolic extract of Moussonia deppeana. Rev. Bras. Farmacogn. 2018, 28, 198–206.
- Haleagrahara, N.; Miranda-Hernandez, S.; Alim, M.A.; Hayes, L.; Bird, G.; Ketheesan, N. Therapeutic effect of quercetin in collagen-induced arthritis. Biomed. Pharmacother. 2017, 90, 38–46.
- Pan, D.; Li, N.; Liu, Y.; Xu, Q.; Liu, Q.; You, Y.; Wei, Z.; Jiang, Y.; Liu, M.; Guo, T.; et al. Kaempferol inhibits the migration and invasion of rheumatoid arthritis fibroblast-like synoviocytes by blocking activation of the MAPK pathway. Int. Immunopharmacol. 2018, 55, 174–182.
- Nguyen, T.; Chen, X.; Chai, J.; Li, R.; Han, X.; Chen, X.; Liu, S.; Chen, M.; Xu, X. Antipyretic, anti-inflammatory and analgesic activities of Periplaneta americana extract and underlying mechanisms. Biomed. Pharmacother. 2020, 123, 109753.




