
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wei Wang | + 4606 word(s) | 4606 | 2021-08-06 16:17:47 | | | |
| 2 | Conner Chen | Meta information modification | 4606 | 2021-08-11 03:54:40 | | |
Video Upload Options
The construction of nanosized drug delivery systems possesses tremendous potential due to their ability to improve the solubility of poorly soluble drugs and to reduce metabolism by dissolving them in their hydrophobic or hydrophilic compartment. In addition, nanomedicine holds the advantages of passive targeting ability due to an enhanced permeability and retention (EPR) effect, a large surface-to-volume ratio for drug loading, a tunable size for modification, a prolonged plasma half-life and a different biodistribution profile compared to conventional chemotherapy. Typical nano-based delivery vehicles include liposome, micelle, dendrimer, inorganic vector, nanogel and nanoemulsion, while novel nanocarriers also contain biomimetic reconstituted high-density lipoprotein (rHDL), exosome and the hybrid nanoparticle, which come from the mixture of nanomaterials. Each of these nanotools displays its unique physiochemical properties and possesses the ability for further modification of active targeting ligands.
1. Liposome
Liposome, structured by natural non-toxic phospholipids and cholesterol, is a spherical vesicle containing an aqueous inner compartment and a phospholipid bilayer, which is able to load both hydrophilic and hydrophobic cargos [1][2]. Structurally, the similar composition with the biological membrane allows cells to take up liposomes more easily and cause lower toxicity [3]. Liposomes represent one of the most promising delivery vehicles for anticancer therapy owing to their biocompatibility, high loading capacity and modifiable sites [4]. So far, they are the most widely developed nanomedicines and are also the most likely to be put onto the market [5]. According to the development of liposomes, they can be classified into conventional liposomes, targeted liposomes and triggered liposomes. The first conventional liposomes mainly adopt the stealth strategy to escape capture by the reticuloendothelial system (RES) and to prolong blood circulation [6]. Polyethylene glycol (PEG) is the most widely used material for the stealth modification of the liposomes, thus the PEGylated liposomes are also called stealth liposomes [7]. Furthermore, stealth liposomes can achieve passive targeting by virtue of the EPR effect [8]. In 1995, Doxil®, as the first stealth liposome encapsulating doxorubicin (DOX), was approved by the FDA for the clinical application of cancer treatment [9]. However, recent research has reported that PEGylated liposomes can elicit unexpected immune responses, such as the accelerated blood clearance (ABC) phenomenon [10]. In this case, the long-circulating properties of PEGylated liposomes will decrease when administered repeatedly at certain intervals to the same animal [11]. To achieve selective delivery, targeted liposomes were developed by modifying various active targeting ligands including antibodies, aptamers, small molecules and peptides to the surface of the membrane of liposomes to target the specific receptors of tumor cells [12][13]. Recently, stimuli-responsive liposomal systems have emerged as an attractive approach for the on-demand release of encapsulated drugs. This kind of liposome can respond to specific stimuli, either external stimuli such as light, temperature, ultrasound and magnetism, or internal stimuli such as pH, enzyme and redox [14][15]. Additionally, liposomes can co-load multiple drugs with different structures, ranging from small molecules to macromolecules such as proteins and genes, for combination administration to improve the therapeutic efficacy [16]. Therefore, liposomes play a vital role in anticancer targeted drug delivery and are of great research and clinical interest (Figure 1).
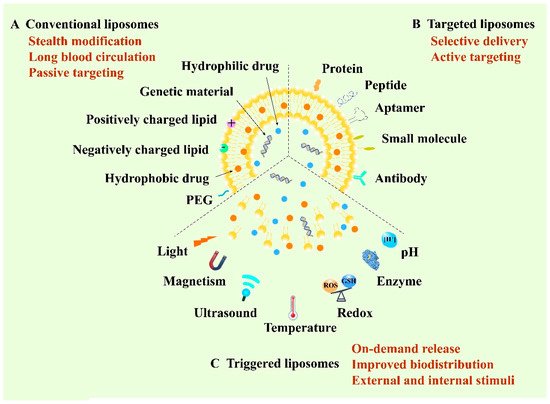
Figure 1. Schematic illustration of various liposome-based delivery systems and their representative properties and typical functions.
1.1. Encapsulation of Small Molecule Drugs with Liposome
IOX1 (5-carboxy-8-hydroxyquinoline), a kind of histone demethylase inhibitor, is used as an antibody-free small molecular drug for immunotherapy. Liu et al. fabricated IOXL by loading IOX1 into liposome with a similar formula of commercially available PEGylated liposomal DOX (PLD, LIBOD®) [17]. Subsequently, the mixed liposome IPLD was obtained by mixing IOXL with commercial PLD at an optimized molar ratio. DOX can induce immunogenic the cell death (ICD) of cancer cells and promote the transfer of tumor-associated antigens to dendritic cells (DCs), and thus activate DC maturation and the infiltration of T cells and memory T cells to the tumors. Synergistically, IOX1 could inhibit cancer cells’ P-glycoproteins (P-gp) through the JMJD1A/β-catenin/P-gp pathway and greatly enhance DOX-induced immune-stimulatory ICD. As a result, the IOX1 and DOX combination greatly promoted T cell infiltration and activity and significantly reduced tumor immunosuppressive factors. Therefore, long-term antitumor immunities were observed after the treatment of IPLD in murine colon cancer CT26 cells. In addition, the antitumor performance of IPLD was better than that of the combination of DOX with anti-PD-L1 antibody (αPD-L1) against subcutaneous (s.c.) CT26 tumors in BALB/c mice without apparent adverse effects.
Mitochondria, one of the vital intracellular organelles, play a crucial role in cellular metabolism and serve as key regulators of cell death [18]. Hence, mitochondrial targeting has been widely explored as a supplementary method to induce cancerous cells’ ablation [19]. Owing to the negative charge of the mitochondrial membrane, lipophilic triphenylphosphonium (TPP) cation is commonly modified to various nanocarriers for mitochondrial targeting [20]. Jiang et al. fabricated a novel mitochondrion-targeted liposome based on dendritic lipopeptide (DLP) modified with arginine residues, showing a 3.7-fold higher level of accumulation in the mitochondria of 4T1 cells than that of a TPP decorated nanoplatform [21]. The encapsulated photosensitizer indocyanine green (ICG) was also delivered into the mitochondria of the tumor cell, resulting in complete tumor eradication in mice bearing 4T1 mammary tumors after photo-irradiation.
To enhance the efficacy of oxygen-dependent PDT, platinum NPs (nano-Pt), acting as catalase (CAT)-like nanoenzymes, can generate oxygen through catalysis of elevated H2O2 in cancer cells [22][23]. An example was designed by Liu et al.; they adopted a reverse phase evaporation strategy to improve the aqueous drug loading capacity of nano-Pt in the liposome; then, the clinical hydrophobic photosensitizer verteporfin (VP) was loaded into the lipid bilayer to confer PDT activity. Finally, murine macrophage cell membranes were hybridized into the liposomal membrane to endow the delivery system with biomimetic and targeting features. The resulting liposomal system, termed nano-Pt/VP@MLipo, exhibited a long circulation time and inflammatory endothelium targeting ability [24]. After targeting to the tumor site, the self-supply of oxygen improved the VP-mediated PDT effect, which in turn triggered the release of nano-Pt via membrane permeabilization. Under light irradiation, nano-Pt/VP@MLipo showed remarkable tumor inhibition in 4T1 tumor-bearing BALB/c mice, which also inhibited the lung metastasis and extended the survival time without overt toxicity.
1.2. Encapsulation of Biological Macromolecules with Liposome
As an essential constituent of the electron transport chain in mitochondrion, the apoptotic protein cytochrome C (CytoC) can mediate the initiation of cell apoptosis after being transported to the cytoplasm of cancer cells [25][26]. Chen et al. designed a liposome-based nanoassembly (p53/C-rNC/L-FA) for intracellular site-specific delivery of an apoptotic protein CytoC and a plasmid DNA encoding tumor-suppressing p53 protein (p53 DNA) [27]. p53/C-rNC/L-FA consisted of an acid-activated fusogenic liposomal membrane shell modified with folic acid (L-FA) and a DNA/protein complex core assembled by the p53 DNA, protamine and CytoC-encapsulated redox-responsive nanocapsule (C-rNC). With the favorable tumor-targeting capacity of FA, p53/C-rNC/L-FA achieved a high level of accumulation in the tumor that overexpresses the folate receptor in vivo. Owing to the arginine-rich nucleus-targeted protamine, the p53 DNA could efficiently accumulate in the nucleus and produce the p53 protein for tumor suppression, which, in combination with the pro-apoptotic effect of CytoC, could augment anticancer efficacy. In vivo antitumor activity of p53/C-rNC/L-FA showed great potential for inducing tumor cell apoptosis and inhibiting tumor growth in the orthotopic MCF-7 tumor mice model.
To overcome the tumor hypoxia, exogenous H2O2 and its decomposing catalase (CAT) were separately loaded into the PEG modified stealthy liposomes by Song et al. [28]. In vivo results demonstrated that the combined treatment of CAT@liposome and H2O2@liposome could promote tumor oxygenation, which further reversed the polarization of immune-supportive M1-type tumor-associated macrophages (TAMs). Due to the relieved tumor hypoxia, the favorable antitumor immunities were formed, which remarkably enhanced the tumor suppression efficacy by radiotherapy (RT) to promote the infiltration of cytotoxic T lymphocytes (CTLs) and benefited the further application of the CTLA-4 checkpoint blockade to inhibit tumor growth. The striking radio-immunotherapy was achieved both in 4T1 tumor-bearing mice models and prostatic patient-derived xenograft (PDX) tumor models. Moreover, no additional toxic side effects were observed during the treatment.
2. Reconstituted High-Density Lipoprotein (rHDL)
High-density lipoprotein (HDL) is an important member of the lipoprotein family, which possesses multiple functions. HDL is a type of serum protein that is mainly composed of apolipoprotein A-I (apoA-I), phospholipids, free cholesterol, cholesteryl ester (CE), triglyceride (TG), and other components [29][30][31]. There are many distinctions among different types of HDL, which can be classified according to hydration density, particle size, charge and composition of apoA. In addition to apolipoprotein, there are other types of proteins in HDL, such as lecithin cholesterol acyltransferase (LCAT), cholesteryl ester transfer protein (CETP), apolipoprotein M (apoM) and paraoxonase (PON) [32][33]. Interestingly, many studies have shown that, with the increase of the proportion of proteins in HDL, the molecular weight of HDL will also increase. HDL is expected to become a promising delivery candidate for anticancer therapy owing to its good biocompatibility, fewer side effects, lower toxicity and the characteristics of reversing cholesterol transport, anti-oxidation and anti-inflammation [34]. However, the complex composition, the strict extraction process and the low yield greatly limit the application scope of HDL. Hence, to overcome the above tricky problems and retain the excellent properties of HDL, reconstituted high-density lipoprotein (rHDL) is manually synthesized using various common materials in vitro.
rHDL can be synthesized and its properties are similar to HDL [35]. In terms of composition, they both contain apoA-I, which accounts for about 65% of HDL and 75% of rHDL. rHDL has two configurations, spherical rHDL (s-rHDL) and discoid rHDL (d-rHDL) [36]. Compared with HDL’s complex constituents, rHDL generally contains apoA-I, phospholipids, free cholesterol, CE, TG, or sometimes only includes the first three components (Figure 2A). In addition, the content of each component can be flexibly adjusted according to the experimental purpose. Scavenger receptor class B type I (SR-BI) is overexpressed in tumor cells and participates in the cholesterol metabolism of tumor cells [37][38]. ApoA-I can specifically target SR-BI and mediate the anti-inflammatory ability of macrophages by regulating the production of cytokines in macrophages [39]. Therefore, SR-BI can be used as a specific target for the treatment of common tumors such as breast cancer, ovarian cancer and prostate cancer [40][41] (Figure 2B). Hence, rHDL can be regarded as an outstanding encapsulated carrier to deliver various drugs with the following unique advantages (Figure 2C). First of all, rHDL-based NPs possess a small particle size and SR-BI targeting ability so that they can actively orientate to the tumor site. Secondly, they have good biocompatibility and low toxicity and side effects. In addition, no obvious drug leakage is identified in the rHDL encapsulated delivery system, which is suitable for enhancing the stability and solubility of some insoluble drugs, changing the pharmacokinetic parameters, and prolonging the circulation time [42]. Besides, rHDL itself can also exert a direct effect on the tumor cells; the main component apoA-I exhibits anti-neoplastic biological effects indirectly via alterations in the functions of macrophages and other immune cells in vivo [43]. Therefore, to overcome the physicochemical limitations of free drugs, including poor solubility and instability, decreasing immune clearance, off-target deposition and other disadvantages, exploring the bio-derived NPs like rHDL is necessary [44]. To summarize, rHDL is a new type of biomimetic nanocarrier that has been emerging in recent years. rHDL has many excellent characteristics such as the capability to overcome several biological barriers in cancer therapy, intrinsic targeting ability, endosomal escape ability, high biological safety and so on. However, the complex preparation process may restrict its wide clinical application and scale-up preparation. Therefore, more and more studies need to be carried out to simplify the preparation method of rHDL, making it easy to clinically translate.
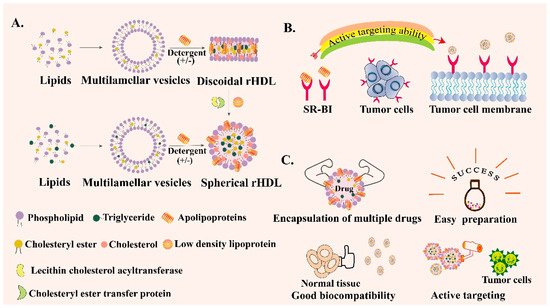
Figure 2. Schematic introduction of reconstituted high-density lipoprotein (rHDL). (A) Preparation process of spherical rHDL and discoid rHDL. (B) Active targeting ability of rHDL through scavenger receptor class B type I (SR-BI)-mediated specific targeting. (C) Various advantages of rHDL as a drug delivery vehicle.
2.1. Encapsulation of Small Molecule Drugs with rHDL
Cell-penetrating peptides (CPPs), cationic or amphipathic in nature, can facilitate efficient translocation across the cell membrane. However, the application of CPPs is badly restricted because of the extensive penetration in vivo and the poor selectivity and permeability before reaching target sites [45]. Gambogic acid (GA) can activate impaired apoptosis pathways in cancerous cells through a complex mechanism involving the participation of multi-molecular targets. In addition, GA can serve as a p53 inducer to stimulate cancer cell apoptosis [46]. Ding et al. designed nano-encapsulated NPs, which consisted of GA-loaded lipid NP (LNP/GA) as a lipid core, then they inserted apoA-I and a pH-responsive CPP (STRR6H4) onto the surface of the LNP to form GA-encapsulated and CPP-anchored rHDL (cp-rHDL/GA) [47]. In vitro and in vivo results based on the HepG2 xenograft mice model showed that the new design had an outstanding antitumor effect with remarkable cytotoxicity and apoptotic effect via triggering the p53 pathway, exhibiting an approximately 5-fold increase in IC50 compared to free GA, indicating the promising future of this biomimetic nanoplatform in drug encapsulation.
Valrubicin is a kind of chemotherapeutic drug that is mainly used for bladder cancer treatment [48]. Sabins et al. developed a novel targeting approach through the encapsulation of valrubicin into a superparamagnetic iron oxide nanoparticle (SPION) containing rHDL to form rHDL-SPION-valrubicin hybrid NPs by the self-assemble method [49]. The experimental results showed that the solubility and bioavailability of valrubicin were greatly enhanced after encapsulation into rHDL NPs. The cytotoxicity test toward PC-3 cells demonstrated that rHDL-SPION-valrubicin NPs were up to 4.6 and 31 times more effective at the respective valrubicin concentrations of 42.4 µg/mL and 85 µg/mL than valrubicin alone, implying that rHDL-based targeted drug delivery through magnetic navigation could effectively enhance the therapeutic efficacy of prostate cancer treatment.
2.2. Encapsulation of Biological Macromolecules with rHDL
Small interfering RNA (siRNA) has shown enormous potential in cancer treatment because it can silence specific gene expressions, such as vascular endothelial growth factor (VEGF), of which expression can be up-regulated in response to hypoxia in ischemic tumor tissues [50]. With this aim in mind, Ding et al. developed nanocomplexes consisting of fluorescent-tagged apoA-I and cholesterol-conjugated siRNA (Chol-siRNA) to form Chol-siRNA-loaded rHDL NPs (rHDL/Chol-siRNA), providing an effective approach to transfer Chol-siRNA across the membrane directly into the cytoplasm via the SR-BI-mediated non-endocytotic mechanism, which bypassed endo-lysosomal trapping [51]. In addition, the new nanocomplexes based on rHDL significantly enhanced antitumor efficacy against MCF-7 human breast cancer in vivo, efficiently decreased VEGF expression level and inhibited the formation of intratumoral microvessels in the tumor tissue.
Except for siRNA, the deregulation of microRNAs (miRNAs) also involves in tumor angiogenesis and thus offers an opportunity for exploring new therapeutic approaches to modulate angiogenesis [52]. However, the lack of effective miRNA delivery strategies limits its practical application. To overcome this obstacle, Chen et al. designed miR-204-5p inhibitor (miR204-5p-inh) encapsulated rHDL NPs to suppress tumor growth in the HeyA8-MDR ovarian tumor-bearing mice model [53]. This novel NP could arrive at the tumor site by binding with SR-BI. The in vivo and in vitro results revealed that miR inhibitors could be delivered by rHDL NPs to silence the expression of the oncogene miR-204-5p at tumor sites, opening rational avenues for solving issues in tumor anti-angiogenic therapy.
3. Micelle
Micelles refer to a large number of molecular-ordered aggregates that begin to form when the concentration of surfactant reaches a concentration above the critical micelle concentration (CMC) in an aqueous solution [54]. In micelles, the hydrophobic group of the surfactant molecule aggregates to form the core, and the hydrophilic polar group forms the outer layer of the micelles [55]. The compounds that form micelles are generally amphiphilic molecules, so the micelles are not only soluble in polar solvents, such as water, but also non-polar solvents in the form of reverse micelles [56]. Because of the above characteristics, micelles can be used as a drug delivery carrier. Plenty of antitumor drugs have poor water solubility and bad stability, which limit their broad application in cancer therapy. However, the hydrophobic blocks that constitute the core compartment of polymeric micelles can load those hydrophobic drugs effectively. Generally, there are two methods to encapsulate drugs in polymer micelles; the first is to encapsulate the hydrophobic drug in the hydrophobic region of the micelle through physical action, and the other is to connect the drug to the hydrophobic core through chemical bonds, different encapsulation methods can be designed according to the properties and experimental purpose of certain drugs [57][58].
Micelles have various shapes including spherical, layered, rod-shaped, worm-like, and hexagonal shapes [59]. According to the length of the hydrophobic block, micelles can be divided into two types. If the length of the hydrophilic end is longer than the specific hydrophobic end, a star micelle will be formed, otherwise, a crew-cut micelle will be formed [60]. Most of the copolymer micellar hydrophilic blocks are biocompatible copolymers such as PEG, polyethylene oxide (PEO), and polyvidone (PVP). Hydrophobic blocks are biodegradable copolymers such as polylactic acid (PLA), poly (lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL). There are also non-degradable copolymers, such as polystyrene (PS), and triblock hydrophilic–hydrophobic copolymers, such as micelle materials like poloxamer (PEO-PPO-PEO) and PEG-PLGA-PEG [61]. Currently, different kinds of micelles are designed for intelligent tumor-targeted drug delivery according to the properties of the tumor microenvironment such as low pH, hypoxia, high tissue osmotic pressure and the overexpression of matrix metalloproteinases. Therefore, the micelle-based nano drug delivery system is also a promising tool in anticancer targeted therapy.
In summary, micelles are easy to prepare and can encapsulate multiple drugs while protecting them against degradation and destruction. They are characterized by high stability, multiple available routes of administrations and the possibility of adjusting the controlled drug release with environment correspondence. Furthermore, compared to other nanomaterials, micelles have the feature of biodegradability, low immunogenicity and non-toxicity, which can be used as a safe carrier in drug delivery. Genexol-PM is a polymeric micellar formulation of paclitaxel without cremophor EL. Keam et al. conducted a Phase II Study that investigated the efficacy and safety of Genexol-PM plus cisplatin as induction chemotherapy (IC) in patients with locally advanced head and neck squamous cell carcinoma (LA-HNSCC) [62]. The results showed that IC with Genexol-PM and cisplatin exhibited the modest tumor response with well-tolerated toxicity profiles for patients with LA-HNSCC. Considering the high clinical transformation potential of micelles, it is very necessary to speed up the research on micelles.
3.1. Encapsulation of Small Molecule Drugs with Micelle
Artesunate (ART), a semi-synthetic derivative of artemisinin, has been reported by many studies to have antitumor effects on various cancer cell lines [63][64]. However, the applications of ART are limited due to its poor water solubility and stability, low bioavailability and short half-life in blood circulation. Therefore, it is desirable to design an ART depot with sustained release and targeted delivery properties. Hao et al. prepared a micelle composed of pH-sensitive PEOz as hydrophilic corona and polylactide/PBAE conjugation with ART as the hydrophobic core [65]. The outside copolymer exhibited proton sponge properties at a pH below its pKb (~6.5) owing to the presence of amine groups, which resulted in its pH sensitivity [66]. The micelles showed enhanced cellular internalization and a stronger inhibitory effect on CT-26 cells in vitro. In addition, the antitumor effect based on the colon carcinoma mice model also showed that the ART-based micelle group could decrease the most tumor volumes compared to the other groups.
S-nitrosoglutathione (GSNO) is a prodrug that can generate nitric oxide (NO) under the presence of glutathione (GSH) [67]. Many studies have revealed that NO has the potential to overcome multidrug resistance (MDR) with low side effects [68]. However, GSNO is unstable and its stability can be easily affected by ambient temperature, pH and other factors. Besides, NO is released rapidly in water, resulting in the limited application range of GSNO. The poly(propylene sulfide) (PPS) block is able to converse from hydrophobic to hydrophilic under the condition of excess ROS in the tumor sites, which enables the on-demand delivery of therapeutic agents [69]. Therefore, in order to retain the advantages of GSNO, while minimizing its disadvantages, Wu et al. chemically conjugated GSNO to an amphiphilic block copolymer PPS-PEG (GSNO-PPS-PEG), and then encapsulated the anticancer drug DOX in the region of PPS to form a spherical micelle [70]. An in vitro release study showed that the micelle was ROS- and GSH-responsive. To investigate the potential of PEG-PPS-GSNO in overcoming the MDR of cancer, the toxicity of free DOX and PEG-PPS-GSNO@DOX toward HepG2/ADR cells were tested and compared in vitro. As the experiment’s results showed, the micelle group exhibited the strongest cytotoxicity compared to the other groups, indicating that this nano drug delivery system (DDS) could serve as an effective co-delivery platform of NO and DOX to selectively kill chemo-resistant cancer cells.
Retinoblastoma (Rb) is a malignant tumor of the retina in infants and is also the most common childhood malignancy. Melphalan (MEL) can be regarded as an effective chemotherapeutic agent for the treatment of Rb. Li et al. adopted micelles as an ideal drug delivery vehicle to solve the MEL inherent cardiopulmonary toxicity and realize precision drug delivery [71]. N-acetylheparosan (AH) is a natural heparin-like polysaccharide in mammals with a long circulation effect and good biocompatibility which was linked by d-α-tocopherol acid succinate (VES) via cystamine (CYS) to synthesize reduction-responsive N-acetylheparosan-CYS-Vitamin E succinate (AHV) copolymers. Furthermore, MEL-loaded AHV (MEL/AHV) micelles were prepared with the properties of small particle size, high drug loading content, and obvious reduction-triggered release behavior. In vitro antitumor effects based on WERI-Rb-1 Rb cells showed that the MEL/AHV micelles could significantly enhance the cytotoxicity against Rb tumor cells, suggesting that MEL/AHV micelles might be a potential delivery system for the enhanced delivery of melphalan to Rb cells.
3.2. Encapsulation of Biological Macromolecules with Micelle
The desmoplastic and hypoxic microenvironment of pancreatic cancer can induce the aberrant expression of miRNAs and hypoxia-inducible factor-1 (HIF-1), which are responsible for gemcitabine (GEM) resistance [72]. MiR-519c could bind to HIF-1 mRNA and then inhibit HIF-1 expression, slow down the tumor metabolism and decrease profibrotic and angiogenic responses. Xin et al. used 2′-O-methyl phosphorothioate (2′-OMe-PS) to modify miRNA and formed OMe-PS-miR-519c, then they conjugated the complex to a kind of redox-sensitive polymeric micelle via stable disulfide bonds [73]. The effect of the micelle on the MIA PaCa-2R cells was determined in vitro, suggesting that miR-519c inhibited the tumor cells’ proliferation by inducing G2 phase cell cycle arrest and sensitizing GEM to kill hypoxic GEM-resistant tumor. Moreover, in vivo and in vitro results based on the desmoplastic pancreatic cancer mice model showed that the multifunctional nano-micelle could effectively deliver GEM and miR-519c to the tumor site and reverse the GEM resistance remarkably, which resulted in synergistic inhibition of pancreatic cancer.
Many studies have shown that the epigenetic regulation drug histone deacetylase inhibitor (HDACi) was able to reverse exhausted T cells by changing the epigenetic transcription program, which can solve the challenge of immune escape and be beneficial to PD-1/PD-L1 blockade therapy. Lu et al. developed a kind of micelle called PDDS to encapsulate the siRNA of PD-L1 to form pH-sensitive siRNA-PD-L1-loaded micelles (siRNA@PPDS) [74]. In vitro and in vivo results revealed that the complexes could not only be spontaneously released into the cytoplasm under the acidic and lipase conditions in the lysosome, thereby inducing B16-F10 melanoma tumor cells’ apoptosis, but also silence the expression of PD-L1 protein in a dose-dependent manner. Furthermore, the micelles also showed outstanding inhibition ability of pulmonary metastasis.
Encouragingly, in addition to the micelle-related pre-clinical studies mentioned above, many polymeric micelles have been developed as antineoplastic drugs, such as Genexol-PM® and Paclical®. Besides, many other polymer micelle-based nanomedicines, including NK012, NK105, SP1049C and NC-4016, are under clinical evaluation for treating several types of tumors, indicating their bright future in clinical translation [75].
4. Dendrimer
Dendrimers are novel polymeric materials with a 3D structure that has extensively branched. Dendrimers have multiple functional groups on the surface which makes them easy to be decorated and provides a new train of thought to its application. The dendrimers generally consist of a central core that includes a single atom or group of atoms, building blocks containing many layers of repeating units known as generations, and numerous functional groups on the surface, which play a key role in their properties [76] (Figure 3). Compared with traditional linear polymers, dendrimers have a variety of new physical properties because of their unique structure with numbers of chains whose ends are combined with a high degree of branching, these characteristics make them attractive for biological applications and drug delivery. Dendrimers possess many charming properties such as good water-solubility, nanoscale uniform size, symmetrical shapes, high densities of peripheral functionalities, available internal cavities, good biocompatibility, biosafety, stability and large drug loading capacity [77]. In addition, the structure of dendrimers is convenient to synthesize and the products with fixed molecular weight due to their step-by-step controlled synthesis are easy to obtain. They can be synthesized using divergent methods or convergent methods [78]. Dendrimers have been demonstrated as an ideal carrier for the targeted delivery of therapeutic and diagnostic agents, such as the delivery of small molecular chemotherapeutic drugs, macromolecular biological drugs, genes, proteins and enzymes [79]. In addition, dendrimers can be designed to realize controlled release by electrostatic interactions, conjugation of the drug to dendrimers, encapsulation of drugs within the dendritic architecture [80]. Moreover, the nanostructure of dendrimers facilitates the passive targeting of drugs to tumor tissues via the EPR effect. Besides, because of their immunogenicity, dendrimers can also be used as a carrier for gene delivery.
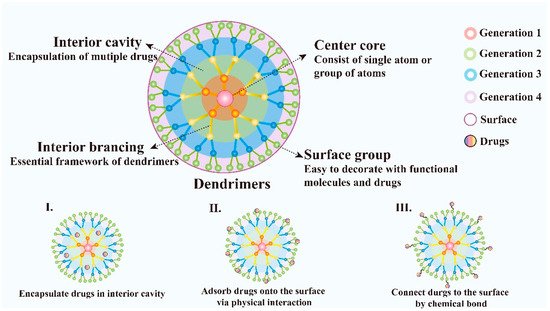
Figure 3. Schematic diagram of dendrimer architecture. Distinct functions of each dendrimer component and various ways of dendrimers to deliver drugs are also illustrated.
To conclude, dendrimers are different from linear polymers by architecture with tailor-made surface groups. Their properties are mainly determined by the functional groups on their surface with many modified methods. They have the advantages of biocompatibility, easy elimination from the body and appropriate particle size to express the EPR effect. The limitation of dendrimers is also the complicated synthesis process, which requires many chemical reactions. Besides, the cytotoxicity to normal cells resulting from the physiological stability of cationic groups of dendrimers may exert certain security threats. Fortunately, dendrimers have been applied in many fields since they came out, VivaGel® is the first dendrimer-based commercial medical product and many others are now in clinical trials, indicating its promising future in anticancer therapy [81].
4.1. Encapsulation of Small Molecule Drugs with Dendrimer
Targeted delivery of small molecular chemotherapeutics to the tumor site is an effective means to achieve antitumor purposes. For example, DOXIL® can effectively accumulate in the tumor tissues but fail to penetrate through the tumor, thus designing a DDS is essential to achieving the predicted goal [82]. Sun et al. made a cluster-bomb-like nanocarrier that synergized based on dendrimers. A sixth-generation nontoxic degradable polyaminoester dendrimer with a diameter of 5 nm was synthesized with pH-dependent 2-(N,N-diethylamino)ethyl termini. A fusogenic phospholipid DOPE (1,2-dioleoyl-sn-glycerol-3-phosphoethanol-amine) was chosen to coat the dendrimers, but this DOPE was expected to peel off by fusion once inside the tumor tissue. A PEGylated lipid DSPE-PEG and cholesterol were added to make the nano-assembly stealthy and stable in the blood [83]. The dendrimers’ core contained many tertiary amines and they were hydrophobic at a neutral pH but could become water-soluble at a lower pH condition. Hence, hydrophobic DOX could be encapsulated inside the dendrimers at a neutral pH and released once at a lower pH. The antitumor efficacy was tested based on human ovarian cancer cells SKOV-3 in vitro and the dendrimers were found to ship the drugs into the cytosol and circumvent the multidrug resistance. The in vivo therapeutic efficacy of this DDS was compared with the micellar PCL-PEG/DOX using subcutaneous drug-sensitive/resistant MCF-7 and MCF-7/ADR breast tumor-bearing mice, which showed that the DDS based on dendrimers had a superior antitumor effect.
References
- Franco, M.S.; Gomes, E.R.; Roque, M.C.; Oliveira, M.C. Triggered Drug Release From Liposomes: Exploiting the Outer and Inner Tumor Environment. Front. Oncol. 2021, 11, 623760.
- Lakshmi, B.A.; Reddy, A.S.; Sangubotla, R.; Hong, J.W.; Kim, S. Ruthenium(II)-curcumin liposome nanoparticles: Synthesis, characterization, and their effects against cervical cancer. Colloids Surf. B Biointerfaces 2021, 204, 111773.
- Zylberberg, C.; Matosevic, S. Pharmaceutical liposomal drug delivery: A review of new delivery systems and a look at the regulatory landscape. Drug Deliv. 2016, 23, 3319–3329.
- Alwattar, J.K.; Mneimneh, A.T.; Abla, K.K.; Mehanna, M.M.; Allam, A.N. Smart Stimuli-Responsive Liposomal Nanohybrid Systems: A Critical Review of Theranostic Behavior in Cancer. Pharmaceutics 2021, 13, 355.
- Lu, W.; Yao, J.; Zhu, X.; Qi, Y. Nanomedicines: Redefining traditional medicine. Biomed. Pharm. 2021, 134, 111103.
- Han, B.; Yang, Y.; Chen, J.; Tang, H.; Sun, Y.; Zhang, Z.; Wang, Z.; Li, Y.; Li, Y.; Luan, X.; et al. Preparation, Characterization, and Pharmacokinetic Study of a Novel Long-Acting Targeted Paclitaxel Liposome with Antitumor Activity. Int. J. Nanomed. 2020, 15, 553–571.
- Elamir, A.; Ajith, S.; Sawaftah, N.A.; Abuwatfa, W.; Mukhopadhyay, D.; Paul, V.; Al-Sayah, M.H.; Awad, N.; Husseini, G.A. Ultrasound-triggered herceptin liposomes for breast cancer therapy. Sci. Rep. 2021, 11, 7545.
- Darwitan, A.; Tan, Y.F.; Wong, Y.S.; Nedumaran, A.M.; Czarny, B.; Venkatraman, S. Targeting efficiency of nanoliposomes on atherosclerotic foam cells: Polyethylene glycol-to-ligand ratio effects. Expert Opin. Drug Deliv. 2020, 17, 1165–1176.
- Bogner, J.R.; Kronawitter, U.; Rolinski, B.; Truebenbach, K.; Goebel, F.D. Liposomal doxorubicin in the treatment of advanced AIDS-related Kaposi sarcoma. J. Acquir. Immune Defic. Syndr. 1994, 7, 463–468.
- Mohamed, M.; Abu Lila, A.S.; Shimizu, T.; Alaaeldin, E.; Hussein, A.; Sarhan, H.A.; Szebeni, J.; Ishida, T. PEGylated liposomes: Immunological responses. Sci. Technol. Adv. Mater. 2019, 20, 710–724.
- Ichihara, M.; Shimizu, T.; Imoto, A.; Hashiguchi, Y.; Uehara, Y.; Ishida, T.; Kiwada, H. Anti-PEG IgM Response against PEGylated Liposomes in Mice and Rats. Pharmaceutics 2010, 3, 1–11.
- Rommasi, F.; Esfandiari, N. Liposomal Nanomedicine: Applications for Drug Delivery in Cancer Therapy. Nanoscale Res. Lett. 2021, 16, 95.
- Mojarad-Jabali, S.; Farshbaf, M.; Walker, P.R.; Hemmati, S.; Fatahi, Y.; Zakeri-Milani, P.; Sarfraz, M.; Valizadeh, H. An update on actively targeted liposomes in advanced drug delivery to glioma. Int. J. Pharm. 2021, 602, 120645.
- Lee, Y.; Thompson, D.H. Stimuli-responsive liposomes for drug delivery. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, 10.
- Wang, Y.; Kohane, D.S. External triggering and triggered targeting strategies for drug delivery. Nat. Rev. Mater. 2017, 2, 17020.
- Haggag, Y.; Abu Ras, B.; El-Tanani, Y.; Tambuwala, M.M.; McCarron, P.; Isreb, M.; El-Tanani, M. Co-delivery of a RanGTP inhibitory peptide and doxorubicin using dual-loaded liposomal carriers to combat chemotherapeutic resistance in breast cancer cells. Expert Opin. Drug Deliv. 2020, 17, 1655–1669.
- Liu, J.; Zhao, Z.; Qiu, N.; Zhou, Q.; Wang, G.; Jiang, H.; Piao, Y.; Zhou, Z.; Tang, J.; Shen, Y. Co-delivery of IOX1 and doxorubicin for antibody-independent cancer chemo-immunotherapy. Nat. Commun. 2021, 12, 2425.
- Nandi, I.; Aroeti, L.; Ramachandran, R.P.; Kassa, E.G.; Zlotkin-Rivkin, E.; Aroeti, B. Type III secreted effectors that target mitochondria. Cell. Microbiol. 2021, e13352.
- Xia, J.; Qian, M.; Yao, Q.; Meng, Z.; Cui, H.; Zhang, L.; Li, Y.; Wu, S.; Wang, J.; Chen, Q.; et al. Synthetic infrared nano-photosensitizers with hierarchical zoom-in target-delivery functionalities for precision photodynamic therapy. J. Control. Release 2021, 334, 263–274.
- Chen, Y.; Ai, W.; Guo, X.; Li, Y.; Ma, Y.; Chen, L.; Zhang, H.; Wang, T.; Zhang, X.; Wang, Z. Mitochondria-Targeted Polydopamine Nanocomposite with AIE Photosensitizer for Image-Guided Photodynamic and Photothermal Tumor Ablation. Small 2019, 15, e1902352.
- Jiang, L.; Zhou, S.; Zhang, X.; Li, C.; Ji, S.; Mao, H.; Jiang, X. Mitochondrion-specific dendritic lipopeptide liposomes for targeted sub-cellular delivery. Nat. Commun. 2021, 12, 2390.
- Liu, Y.; Qin, Y.; Zhang, Q.; Zou, W.; Jin, L.; Guo, R. Arginine-rich peptide/platinum hybrid colloid nanoparticle cluster: A single nanozyme mimicking multi-enzymatic cascade systems in peroxisome. J. Colloid. Interface Sci. 2021, 600, 37–48.
- You, Q.; Zhang, K.; Liu, J.; Liu, C.; Wang, H.; Wang, M.; Ye, S.; Gao, H.; Lv, L.; Wang, C.; et al. Persistent Regulation of Tumor Hypoxia Microenvironment via a Bioinspired Pt-Based Oxygen Nanogenerator for Multimodal Imaging-Guided Synergistic Phototherapy. Adv. Sci. 2020, 7, 1903341.
- Liu, X.-L.; Dong, X.; Yang, S.-C.; Lai, X.; Liu, H.-J.; Gao, Y.; Feng, H.-Y.; Zhu, M.-H.; Yuan, Y.; Lu, Q.; et al. Biomimetic Liposomal Nanoplatinum for Targeted Cancer Chemophototherapy. Adv. Sci. 2021, 8, 2003679.
- Bazylianska, V.; Kalpage, H.A.; Wan, J.; Vaishnav, A.; Mahapatra, G.; Turner, A.A.; Chowdhury, D.D.; Kim, K.; Morse, P.T.; Lee, I.; et al. Lysine 53 Acetylation of Cytochrome c in Prostate Cancer: Warburg Metabolism and Evasion of Apoptosis. Cells 2021, 10, 802.
- Xiao, S.; Wang, X.; Xu, L.; Miao, D.; Li, T.; Su, G.; Zhao, Y. Novel ginsenoside derivatives have shown their effects on PC-3 cells by inducing G1-phase arrest and reactive oxygen species-mediate cell apoptosis. Bioorg. Chem. 2021, 112, 104864.
- Chen, X.; Zhu, Q.; Xu, X.; Shen, S.; Zhang, Y.; Mo, R. Sequentially Site-Specific Delivery of Apoptotic Protein and Tumor-Suppressor Gene for Combination Cancer Therapy. Small 2019, 15, e1902998.
- Song, X.; Xu, J.; Liang, C.; Chao, Y.; Jin, Q.; Wang, C.; Chen, M.; Liu, Z. Self-Supplied Tumor Oxygenation through Separated Liposomal Delivery of H2O2 and Catalase for Enhanced Radio-Immunotherapy of Cancer. Nano Lett. 2018, 18, 6360–6368.
- Pownall, H.J.; Rosales, C.; Gillard, B.K.; Ferrari, M. Native and Reconstituted Plasma Lipoproteins in Nanomedicine: Physicochemical Determinants of Nanoparticle Structure, Stability, and Metabolism. Methodist Debakey Cardiovasc. J. 2016, 12, 146–150.
- Vickers, K.C.; Palmisano, B.T.; Shoucri, B.M.; Shamburek, R.D.; Remaley, A.T. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat. Cell Biol. 2011, 13, 423–433.
- Guerin, M.; Le Goff, W.; Lassel, T.S.; Van Tol, A.; Steiner, G.; Chapman, M.J. Atherogenic role of elevated CE transfer from HDL to VLDL(1) and dense LDL in type 2 diabetes: Impact of the degree of triglyceridemia. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 282–288.
- Zhang, P.; Gao, J.; Pu, C.; Feng, G.; Wang, L.; Huang, L.; Zhang, Y. ApoM/HDL-C and apoM/apoA-I ratios are indicators of diabetic nephropathy in healthy controls and type 2 diabetes mellitus. Clin. Chim. Acta 2017, 466, 31–37.
- Ertek, S. High-density Lipoprotein (HDL) Dysfunction and the Future of HDL. Curr. Vasc. Pharmacol. 2018, 16, 490–498.
- Riggs, K.A.; Rohatgi, A. HDL and Reverse Cholesterol Transport Biomarkers. Methodist Debakey Cardiovasc. J. 2019, 15, 39–46.
- Kuai, R.; Li, D.; Chen, Y.E.; Moon, J.J.; Schwendeman, A. High-Density Lipoproteins: Nature’s Multifunctional Nanoparticles. ACS Nano 2016, 10, 3015–3041.
- Jiang, C.; Qi, Z.; Tang, Y.; Jia, H.; Li, Z.; Zhang, W.; Liu, J. Rational Design of Lovastatin-Loaded Spherical Reconstituted High Density Lipoprotein for Efficient and Safe Anti-Atherosclerotic Therapy. Mol. Pharm. 2019, 16, 3284–3291.
- Twiddy, A.L.; Cox, M.E.; Wasan, K.M. Knockdown of scavenger receptor class B type I reduces prostate specific antigen secretion and viability of prostate cancer cells. Prostate 2012, 72, 955–965.
- Babitt, J.; Trigatti, B.; Rigotti, A.; Smart, E.J.; Anderson, R.G.; Xu, S.; Krieger, M. Murine SR-BI, a high density lipoprotein receptor that mediates selective lipid uptake, is N-glycosylated and fatty acylated and colocalizes with plasma membrane caveolae. J. Biol. Chem. 1997, 272, 13242–13249.
- Witkowski, A.; Carta, S.; Lu, R.; Yokoyama, S.; Rubartelli, A.; Cavigiolio, G. Oxidation of methionine residues in human apolipoprotein A-I generates a potent pro-inflammatory molecule. J. Biol. Chem. 2019, 294, 3634–3646.
- Monroy-Iglesias, M.J.; Russell, B.; Crawley, D.; Allen, N.E.; Travis, R.C.; Perez-Cornago, A.; Van Hemelrijck, M.; Beckmann, K. Metabolic syndrome biomarkers and prostate cancer risk in the UK Biobank. Int. J. Cancer 2021, 148, 825–834.
- Revilla, G.; Cedo, L.; Tondo, M.; Moral, A.; Perez, J.I.; Corcoy, R.; Lerma, E.; Fuste, V.; Reddy, S.T.; Blanco-Vaca, F.; et al. LDL, HDL and endocrine-related cancer: From pathogenic mechanisms to therapies. Semin. Cancer Biol. 2020, 73, 134–157.
- Pedersbaek, D.; Kraemer, M.K.; Kempen, P.J.; Ashley, J.; Braesch-Andersen, S.; Andresen, T.L.; Simonsen, J.B. The Composition of Reconstituted High-Density Lipoproteins (rHDL) Dictates the Degree of rHDL Cargo- and Size-Remodeling via Direct Interactions with Endogenous Lipoproteins. Bioconjug. Chem. 2019, 30, 2634–2646.
- Mei, Y.; Tang, L.; Xiao, Q.; Zhang, Z.; Zhang, Z.; Zang, J.; Zhou, J.; Wang, Y.; Wang, W.; Ren, M. Reconstituted high density lipoprotein (rHDL), a versatile drug delivery nanoplatform for tumor targeted therapy. J. Mater. Chem. B 2021, 9, 612–633.
- Yoo, J.W.; Irvine, D.J.; Discher, D.E.; Mitragotri, S. Bio-inspired, bioengineered and biomimetic drug delivery carriers. Nat. Rev. Drug Discov. 2011, 10, 521–535.
- Zorko, M.; Langel, U. Cell-penetrating peptides: Mechanism and kinetics of cargo delivery. Adv. Drug Deliv. Rev. 2005, 57, 529–545.
- Pandey, M.K.; Sung, B.; Ahn, K.S.; Kunnumakkara, A.B.; Chaturvedi, M.M.; Aggarwal, B.B. Gambogic acid, a novel ligand for transferrin receptor, potentiates TNF-induced apoptosis through modulation of the nuclear factor-kappaB signaling pathway. Blood 2007, 110, 3517–3525.
- Ding, Y.; Wang, Y.; Opoku-Damoah, Y.; Wang, C.; Shen, L.; Yin, L.; Zhou, J. Dual-functional bio-derived nanoparticulates for apoptotic antitumor therapy. Biomaterials 2015, 72, 90–103.
- Tse, J.; Singla, N.; Ghandour, R.; Lotan, Y.; Margulis, V. Current advances in BCG-unresponsive non-muscle invasive bladder cancer. Expert Opin. Investig. Drugs 2019, 28, 757–770.
- Sabnis, S.; Sabnis, N.A.; Raut, S.; Lacko, A.G. Superparamagnetic reconstituted high-density lipoprotein nanocarriers for magnetically guided drug delivery. Int. J. Nanomed. 2017, 12, 1453–1464.
- Ryther, R.C.; Flynt, A.S.; Phillips, J.A., 3rd; Patton, J.G. siRNA therapeutics: Big potential from small RNAs. Gene Ther. 2005, 12, 5–11.
- Ding, Y.; Wang, Y.; Zhou, J.; Gu, X.; Wang, W.; Liu, C.; Bao, X.; Wang, C.; Li, Y.; Zhang, Q. Direct cytosolic siRNA delivery by reconstituted high density lipoprotein for target-specific therapy of tumor angiogenesis. Biomaterials 2014, 35, 7214–7227.
- Li, Y.; Cai, B.; Shen, L.; Dong, Y.; Lu, Q.; Sun, S.; Liu, S.; Ma, S.; Ma, P.X.; Chen, J. MiRNA-29b suppresses tumor growth through simultaneously inhibiting angiogenesis and tumorigenesis by targeting Akt3. Cancer Lett. 2017, 397, 111–119.
- Chen, X.; Mangala, L.S.; Mooberry, L.; Bayraktar, E.; Dasari, S.K.; Ma, S.; Ivan, C.; Court, K.A.; Rodriguez-Aguayo, C.; Bayraktar, R.; et al. Identifying and targeting angiogenesis-related microRNAs in ovarian cancer. Oncogene 2019, 38, 6095–6108.
- Haftka, J.J.; Scherpenisse, P.; Oetter, G.; Hodges, G.; Eadsforth, C.V.; Kotthoff, M.; Hermens, J.L. Critical micelle concentration values for different surfactants measured with solid-phase microextraction fibers. Environ. Toxicol. Chem. 2016, 35, 2173–2781.
- Cheng, F.R.; Su, T.; Cao, J.; Luo, X.L.; Li, L.; Pu, Y.; He, B. Environment-stimulated nanocarriers enabling multi-active sites for high drug encapsulation as an “on demand” drug release system. J. Mater. Chem. B 2018, 6, 2258–2273.
- Ghosh, S.; Ray, A.; Pramanik, N. Self-assembly of surfactants: An overview on general aspects of amphiphiles. Biophys. Chem. 2020, 265, 106429.
- Croy, S.R.; Kwon, G.S. Polymeric micelles for drug delivery. Curr. Pharm. Des. 2006, 12, 4669–4684.
- Irby, D.; Du, C.; Li, F. Lipid-Drug Conjugate for Enhancing Drug Delivery. Mol. Pharm. 2017, 14, 1325–1338.
- Danov, K.D.; Kralchevsky, P.A.; Stoyanov, S.D.; Cook, J.L.; Stott, I.P. Analytical modeling of micelle growth. 1. Chain-conformation free energy of binary mixed spherical, wormlike and lamellar micelles. J. Colloid. Interface Sci. 2019, 547, 245–255.
- Zhao, J.; Wang, Y.; Luan, L. Star-shaped polycaprolactone-polyethyleneglycol copolymer micelle-like nanoparticles for picropodophyllin delivery. J. Biomed. Nanotechnol. 2014, 10, 1627–1634.
- Isoglu, I.A.; Ozsoy, Y.; Isoglu, S.D. Advances in Micelle-based Drug Delivery: Cross-linked Systems. Curr. Top. Med. Chem. 2017, 17, 1469–1489.
- Keam, B.; Lee, K.W.; Lee, S.H.; Kim, J.S.; Kim, J.H.; Wu, H.G.; Eom, K.Y.; Kim, S.; Ahn, S.H.; Chung, E.J.; et al. A Phase II Study of Genexol-PM and Cisplatin as Induction Chemotherapy in Locally Advanced Head and Neck Squamous Cell Carcinoma. Oncologist 2019, 24, 751-e231.
- Du, J.H.; Zhang, H.D.; Ma, Z.J.; Ji, K.M. Artesunate induces oncosis-like cell death in vitro and has antitumor activity against pancreatic cancer xenografts in vivo. Cancer Chemother. Pharmacol. 2010, 65, 895–902.
- Efferth, T.; Kaina, B. Toxicity of the antimalarial artemisinin and its dervatives. Crit. Rev. Toxicol. 2010, 40, 405–421.
- Hao, D.L.; Xie, R.; De, G.J.; Yi, H.; Zang, C.; Yang, M.Y.; Liu, L.; Ma, H.; Cai, W.Y.; Zhao, Q.H.; et al. pH-Responsive Artesunate Polymer Prodrugs with Enhanced Ablation Effect on Rodent Xenograft Colon Cancer. Int. J. Nanomed. 2020, 15, 1771–1786.
- Clamme, J.P.; Krishnamoorthy, G.; Mely, Y. Intracellular dynamics of the gene delivery vehicle polyethylenimine during transfection: Investigation by two-photon fluorescence correlation spectroscopy. Biochim. Biophys. Acta 2003, 1617, 52–61.
- Manda-Handzlik, A.; Bystrzycka, W.; Cieloch, A.; Glodkowska-Mrowka, E.; Jankowska-Steifer, E.; Heropolitanska-Pliszka, E.; Skrobot, A.; Muchowicz, A.; Ciepiela, O.; Wachowska, M.; et al. Nitric oxide and peroxynitrite trigger and enhance release of neutrophil extracellular traps. Cell. Mol. Life Sci. 2020, 77, 3059–3075.
- Lin, Y.J.; Chen, C.C.; Nguyen, D.; Su, H.R.; Lin, K.J.; Chen, H.L.; Hu, Y.J.; Lai, P.L.; Sung, H.W. Biomimetic Engineering of a Scavenger-Free Nitric Oxide-Generating/Delivering System to Enhance Radiation Therapy. Small 2020, 16, e2000655.
- Napoli, A.; Valentini, M.; Tirelli, N.; Muller, M.; Hubbell, J.A. Oxidation-responsive polymeric vesicles. Nat. Mater. 2004, 3, 183–189.
- Wu, W.; Chen, M.; Luo, T.; Fan, Y.; Zhang, J.; Zhang, Y.; Zhang, Q.; Sapin-Minet, A.; Gaucher, C.; Xia, X. ROS and GSH-responsive S-nitrosoglutathione functionalized polymeric nanoparticles to overcome multidrug resistance in cancer. Acta Biomater. 2020, 103, 259–271.
- Li, J.; Wang, J.; Zhang, X.; Xia, X.; Zhang, C. Biodegradable reduction-responsive polymeric micelles for enhanced delivery of melphalan to retinoblastoma cells. Int. J. Biol. Macromol. 2019, 141, 997–1003.
- Mimeault, M.; Batra, S.K. Hypoxia-inducing factors as master regulators of stemness properties and altered metabolism of cancer- and metastasis-initiating cells. J. Cell. Mol. Med. 2013, 17, 30–54.
- Xin, X.; Kumar, V.; Lin, F.; Kumar, V.; Bhattarai, R.; Bhatt, V.R.; Tan, C.; Mahato, R.I. Redox-responsive nanoplatform for codelivery of miR-519c and gemcitabine for pancreatic cancer therapy. Sci. Adv. 2020, 6, eabd6764.
- Lu, F.; Hou, L.; Wang, S.; Yu, Y.; Zhang, Y.; Sun, L.; Wang, C.; Ma, Z.; Yang, F. Lysosome activable polymeric vorinostat encapsulating PD-L1KD for a combination of HDACi and immunotherapy. Drug Deliv. 2021, 28, 963–972.
- Cao, J.; Huang, D.; Peppas, N.A. Advanced engineered nanoparticulate platforms to address key biological barriers for delivering chemotherapeutic agents to target sites. Adv. Drug Deliv. Rev. 2020, 167, 170–188.
- Wu, L.P.; Ficker, M.; Christensen, J.B.; Trohopoulos, P.N.; Moghimi, S.M. Dendrimers in Medicine: Therapeutic Concepts and Pharmaceutical Challenges. Bioconjug. Chem. 2015, 26, 1198–1211.
- Parat, A.; Bordeianu, C.; Dib, H.; Garofalo, A.; Walter, A.; Begin-Colin, S.; Felder-Flesch, D. Dendrimer-nanoparticle conjugates in nanomedicine. Nanomedicine 2015, 10, 977–992.
- Zamboulis, A.; Nakiou, E.A.; Christodoulou, E.; Bikiaris, D.N.; Kontonasaki, E.; Liverani, L.; Boccaccini, A.R. Polyglycerol Hyperbranched Polyesters: Synthesis, Properties and Pharmaceutical and Biomedical Applications. Int. J. Mol. Sci. 2019, 20, 6210.
- Singh, P.; Gupta, U.; Asthana, A.; Jain, N.K. Folate and folate-PEG-PAMAM dendrimers: Synthesis, characterization, and targeted anticancer drug delivery potential in tumor bearing mice. Bioconjug. Chem. 2008, 19, 2239–2252.
- Wang, F.; Cai, X.; Su, Y.; Hu, J.; Wu, Q.; Zhang, H.; Xiao, J.; Cheng, Y. Reducing cytotoxicity while improving anti-cancer drug loading capacity of polypropylenimine dendrimers by surface acetylation. Acta Biomater. 2012, 8, 4304–4313.
- Sinokrot, H.; Smerat, T.; Najjar, A.; Karaman, R. Advanced Prodrug Strategies in Nucleoside and Non-Nucleoside Antiviral Agents: A Review of the Recent Five Years. Molecules 2017, 22, 1736.
- Yuan, F.; Leunig, M.; Huang, S.K.; Berk, D.A.; Papahadjopoulos, D.; Jain, R.K. Microvascular permeability and interstitial penetration of sterically stabilized (stealth) liposomes in a human tumor xenograft. Cancer Res. 1994, 54, 3352–3356.
- Sun, Q.; Sun, X.; Ma, X.; Zhou, Z.; Jin, E.; Zhang, B.; Shen, Y.; Van Kirk, E.A.; Murdoch, W.J.; Lott, J.R.; et al. Integration of nanoassembly functions for an effective delivery cascade for cancer drugs. Adv. Mater. 2014, 26, 7615–7621.





