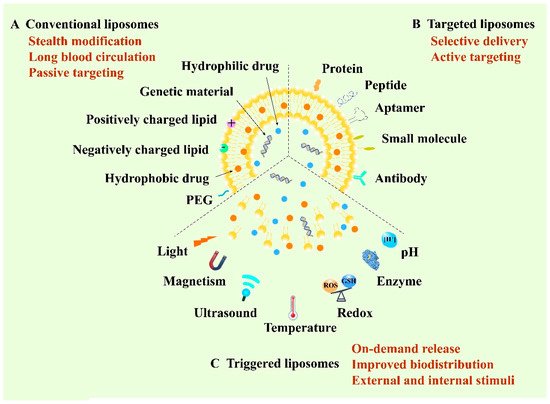
Liposome, structured by natural non-toxic phospholipids and cholesterol, is a spherical vesicle containing an aqueous inner compartment and a phospholipid bilayer, which is able to load both hydrophilic and hydrophobic cargos [
43,
44]. Structurally, the similar composition with the biological membrane allows cells to take up liposomes more easily and cause lower toxicity [
45]. Liposomes represent one of the most promising delivery vehicles for anticancer therapy owing to their biocompatibility, high loading capacity and modifiable sites [
46]. So far, they are the most widely developed nanomedicines and are also the most likely to be put onto the market [
47]. According to the development of liposomes, they can be classified into conventional liposomes, targeted liposomes and triggered liposomes. The first conventional liposomes mainly adopt the stealth strategy to escape capture by the reticuloendothelial system (RES) and to prolong blood circulation [
48]. Polyethylene glycol (PEG) is the most widely used material for the stealth modification of the liposomes, thus the PEGylated liposomes are also called stealth liposomes [
49]. Furthermore, stealth liposomes can achieve passive targeting by virtue of the EPR effect [
50]. In 1995, Doxil
®, as the first stealth liposome encapsulating doxorubicin (DOX), was approved by the FDA for the clinical application of cancer treatment [
51]. However, recent research has reported that PEGylated liposomes can elicit unexpected immune responses, such as the accelerated blood clearance (ABC) phenomenon [
52]. In this case, the long-circulating properties of PEGylated liposomes will decrease when administered repeatedly at certain intervals to the same animal [
53]. To achieve selective delivery, targeted liposomes were developed by modifying various active targeting ligands including antibodies, aptamers, small molecules and peptides to the surface of the membrane of liposomes to target the specific receptors of tumor cells [
54,
55]. Recently, stimuli-responsive liposomal systems have emerged as an attractive approach for the on-demand release of encapsulated drugs. This kind of liposome can respond to specific stimuli, either external stimuli such as light, temperature, ultrasound and magnetism, or internal stimuli such as pH, enzyme and redox [
56,
57]. Additionally, liposomes can co-load multiple drugs with different structures, ranging from small molecules to macromolecules such as proteins and genes, for combination administration to improve the therapeutic efficacy [
58]. Therefore, liposomes play a vital role in anticancer targeted drug delivery and are of great research and clinical interest (
Figure 1).
1.1. Encapsulation of Small Molecule Drugs with Liposome
IOX1 (5-carboxy-8-hydroxyquinoline), a kind of histone demethylase inhibitor, is used as an antibody-free small molecular drug for immunotherapy. Liu et al. fabricated IOXL by loading IOX1 into liposome with a similar formula of commercially available PEGylated liposomal DOX (PLD, LIBOD
®) [
9]. Subsequently, the mixed liposome IPLD was obtained by mixing IOXL with commercial PLD at an optimized molar ratio. DOX can induce immunogenic the cell death (ICD) of cancer cells and promote the transfer of tumor-associated antigens to dendritic cells (DCs), and thus activate DC maturation and the infiltration of T cells and memory T cells to the tumors. Synergistically, IOX1 could inhibit cancer cells’ P-glycoproteins (P-gp) through the JMJD1A/β-catenin/P-gp pathway and greatly enhance DOX-induced immune-stimulatory ICD. As a result, the IOX1 and DOX combination greatly promoted T cell infiltration and activity and significantly reduced tumor immunosuppressive factors. Therefore, long-term antitumor immunities were observed after the treatment of IPLD in murine colon cancer CT26 cells. In addition, the antitumor performance of IPLD was better than that of the combination of DOX with anti-PD-L1 antibody (αPD-L1) against subcutaneous (
s.c.) CT26 tumors in BALB/c mice without apparent adverse effects.
Mitochondria, one of the vital intracellular organelles, play a crucial role in cellular metabolism and serve as key regulators of cell death [
59]. Hence, mitochondrial targeting has been widely explored as a supplementary method to induce cancerous cells’ ablation [
60]. Owing to the negative charge of the mitochondrial membrane, lipophilic triphenylphosphonium (TPP) cation is commonly modified to various nanocarriers for mitochondrial targeting [
61]. Jiang et al. fabricated a novel mitochondrion-targeted liposome based on dendritic lipopeptide (DLP) modified with arginine residues, showing a 3.7-fold higher level of accumulation in the mitochondria of 4T1 cells than that of a TPP decorated nanoplatform [
10]. The encapsulated photosensitizer indocyanine green (ICG) was also delivered into the mitochondria of the tumor cell, resulting in complete tumor eradication in mice bearing 4T1 mammary tumors after photo-irradiation.
To enhance the efficacy of oxygen-dependent PDT, platinum NPs (nano-Pt), acting as catalase (CAT)-like nanoenzymes, can generate oxygen through catalysis of elevated H
2O
2 in cancer cells [
62,
63]. An example was designed by Liu et al.; they adopted a reverse phase evaporation strategy to improve the aqueous drug loading capacity of nano-Pt in the liposome; then, the clinical hydrophobic photosensitizer verteporfin (VP) was loaded into the lipid bilayer to confer PDT activity. Finally, murine macrophage cell membranes were hybridized into the liposomal membrane to endow the delivery system with biomimetic and targeting features. The resulting liposomal system, termed nano-Pt/VP@MLipo, exhibited a long circulation time and inflammatory endothelium targeting ability [
11]. After targeting to the tumor site, the self-supply of oxygen improved the VP-mediated PDT effect, which in turn triggered the release of nano-Pt via membrane permeabilization. Under light irradiation, nano-Pt/VP@MLipo showed remarkable tumor inhibition in 4T1 tumor-bearing BALB/c mice, which also inhibited the lung metastasis and extended the survival time without overt toxicity.
1.2. Encapsulation of Biological Macromolecules with Liposome
As an essential constituent of the electron transport chain in mitochondrion, the apoptotic protein cytochrome C (CytoC) can mediate the initiation of cell apoptosis after being transported to the cytoplasm of cancer cells [
64,
65]. Chen et al. designed a liposome-based nanoassembly (p53/C-rNC/L-FA) for intracellular site-specific delivery of an apoptotic protein CytoC and a plasmid DNA encoding tumor-suppressing p53 protein (p53 DNA) [
12]. p53/C-rNC/L-FA consisted of an acid-activated fusogenic liposomal membrane shell modified with folic acid (L-FA) and a DNA/protein complex core assembled by the p53 DNA, protamine and CytoC-encapsulated redox-responsive nanocapsule (C-rNC). With the favorable tumor-targeting capacity of FA, p53/C-rNC/L-FA achieved a high level of accumulation in the tumor that overexpresses the folate receptor in vivo. Owing to the arginine-rich nucleus-targeted protamine, the p53 DNA could efficiently accumulate in the nucleus and produce the p53 protein for tumor suppression, which, in combination with the pro-apoptotic effect of CytoC, could augment anticancer efficacy. In vivo antitumor activity of p53/C-rNC/L-FA showed great potential for inducing tumor cell apoptosis and inhibiting tumor growth in the orthotopic MCF-7 tumor mice model.
To overcome the tumor hypoxia, exogenous H
2O
2 and its decomposing catalase (CAT) were separately loaded into the PEG modified stealthy liposomes by Song et al. [
13]. In vivo results demonstrated that the combined treatment of CAT@liposome and H
2O
2@liposome could promote tumor oxygenation, which further reversed the polarization of immune-supportive M1-type tumor-associated macrophages (TAMs). Due to the relieved tumor hypoxia, the favorable antitumor immunities were formed, which remarkably enhanced the tumor suppression efficacy by radiotherapy (RT) to promote the infiltration of cytotoxic T lymphocytes (CTLs) and benefited the further application of the CTLA-4 checkpoint blockade to inhibit tumor growth. The striking radio-immunotherapy was achieved both in 4T1 tumor-bearing mice models and prostatic patient-derived xenograft (PDX) tumor models. Moreover, no additional toxic side effects were observed during the treatment.