
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Haim Sohmer | + 2979 word(s) | 2979 | 2021-07-12 04:32:45 | | | |
| 2 | Bruce Ren | -21 word(s) | 2958 | 2021-08-04 09:31:56 | | |
Video Upload Options
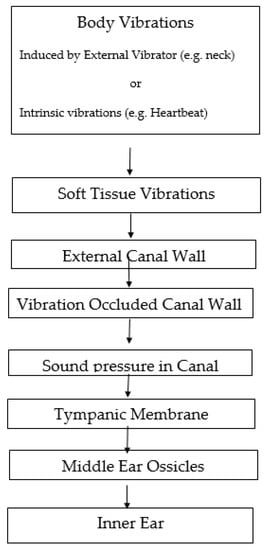
Soft tissue conduction is an additional mode of auditory stimulation which can be initiated either by applying an external vibrator to skin sites not overlying skull bone such as the neck (so it is not bone conduction) or by intrinsic body vibrations resulting, for example, from the heartbeat and vocalization. The soft tissue vibrations thereby induced are conducted by the soft tissues to all parts of the body, including the walls of the external auditory canal. In order for soft tissue conduction to elicit hearing, the soft tissue vibrations which are induced must penetrate into the cochlea in order to excite the inner ear hair cells and auditory nerve fibers. This final stage can be achieved either by an osseous bone conduction mechanism, or, more likely, by the occlusion effect: the vibrations of the walls of the occluded canal induce air pressures in the canal which drive the tympanic membrane and middle ear ossicles and activate the inner ear, acting by means of a more air conduction-like mechanism. In fact, when the clinician applies his stethoscope to the body surface of his patient in order to detect heart sounds or pulmonary air flow, he is detecting soft tissue vibrations.
1. Introduction
1.1. Air Conduction
1.2. Bone Conduction
1.3. Soft Tissue Conduction
1.4. Final Stage
2. Soft Tissue Conduction
2.1. Acoustic Impedance
2.2. Is Bone Conduction the Final Stage?
2.3. Occlusion Effect
References
- Stenfelt, S.; Goode, R.L. Bone-conducted sound: Physiological and clinical aspects. Otol. Neurotol. 2005, 26, 1245–1261.
- Håkansson, B.; Tjellström, A.; Rosenhall, U. Hearing thresholds with direct bone conduction versus conventional bone conduction. Scand. Audiol. 1984, 13, 3–13.
- Hakansson, B.; Tjellstrom, A.; Rosenhall, U. Acceleration levels at hearing threshold with direct bone conduction versus conventional bone conduction. Acta Oto-Laryngol. 1985, 100, 240–252.
- Stone, M.A.; Paul, A.M.; Axon, P.; Moore, B.C.J. A technique for estimating the occlusion effect for frequencies below 125 Hz. Ear Hear. 2014, 35, 49–55.
- Von Bekesy, G. The structure of the middle ear and the hearing of one’s own voice by bone conduction. J. Acoust. Soc. Am. 1949, 21, 217–232.
- Sohmer, H. Soft tissue conduction: Review, mechanisms, and implications. Trends Hear. 2017, 21, 1–8.
- Geal-Dor, M.; Adelman, C.; Chordekar, S.; Sohmer, H. Occlusion Effect in Response to Stimulation by Soft Tissue Conduction-Implications. Audiol. Res. 2020, 10, 69–76.
- Reinfeldt, S.; Ostli, P.; Håkansson, B.; Stenfelt, S. Hearing one’s own voice during phoneme vocalization--transmission by air and bone conduction. J. Acoust. Soc. Am. 2010, 128, 751–762.
- Sohmer, H.; Perez, R.; Sichel, J.Y.; Priner, R.; Freeman, S. The pathway enabling external sounds to reach and excite the fetal inner ear. Audiol. Neurootol. 2001, 6, 109–116.
- De Ridder, D.; Vanneste, S.; Menovsky, T. Pulsatile tinnitus due to a tortuous siphon-like internal carotid artery successfully treated by arterial remodeling. Case Rep. Otolaryngol. 2013, 2013, 938787.
- Chordekar, S.; Perez, R.; Adelman, C.; Sohmer, H.; Kishon-Rabin, L. Does hearing in response to soft-tissue stimulation involve skull vibrations? A within-subject comparison between skull vibration magnitudes and hearing thresholds. Hear. Res. 2018, 364, 59–67.
- Wever, E.G.; Lawrence, M. The function of the middle ear. In Physiological Acoustics; Princeton University Press: Princeton, NJ, USA, 1954; pp. 69–78.
- Baun, J. Interaction with soft tissue. In Physical Principles of General and Vascular Sonography; ProSono Publishing: San Francisco, CA, USA, 2004; pp. 28–41.
- Blakley, B.W.; Siddique, S. A qualitative explanation of the Weber test. Otolaryngol. Head Neck Surg. 1999, 120, 1–4.
- Gerhardt, K.J.; Huang, X.; Arrington, K.E.; Meixner, K.; Abrams, R.M.; Antonelli, P.J. Fetal sheep in utero hear through bone conduction. Am. J. Otolaryngol. 1996, 17, 374–379.
- Adelman, C.; Chordekar, S.; Perez, R.; Sohmer, H. Investigation of the mechanism of soft tissue conduction explains several perplexing auditory phenomena. J. Basic Clin. Physiol. Pharmacol. 2014, 25, 269–272.
- Priner, R.; Perez, R.; Freeman, S.; Sohmer, H. Mechanisms responsible for postnatal middle ear amniotic fluid clearance. Hear. Res. 2003, 175, 133–139.
- Karlsen, S.J.; Bull-Njaa, T.; Krokstad, A. Measurement of sound emission by endoscopic lithotripters: An in vitro study and theoretical estimation of risk of hearing loss in a fetus. J. Endourol. 2001, 15, 821–826.
- Opperman, L.A. Cranial sutures as intramembranous bone growth sites. Dev. Dyn. 2000, 219, 472–485.
- Ito, T.; Roosli, C.; Kim, C.J.; Sim, J.H.; Huber, A.M.; Probst, R. Bone conduction thresholds and skull vibration measured on the teeth during stimulation at different sites on the human head. Audiol. Neurootol. 2011, 16, 12–22.
- Watanabe, T.; Bertoli, S.; Probst, R. Transmission pathways of vibratory stimulation as measured by subjective thresholds and distortion-product otoacoustic emissions. Ear Hear. 2008, 29, 667–673.
- Geal-Dor, M.; Chordekar, S.; Adelman, C.; Sohmer, H. Bone conduction thresholds without bone vibrator application force. J. Am. Acad. Audiol. 2015, 26, 645–651.
- Fagelson, M.A.; Martin, F.N. The occlusion effect and ear canal sound pressure level. Am. J. Audiol. 1998, 7, 50–54.
- Goldstein, D.P.; Hayes, C.S. The occlusion effect in bone conduction hearing. J. Speech Hear. Res. 1965, 8, 137–148.
- Chordekar, S.; Perez, R.; Adelman, C.; Sohmer, H.; Kishon-Rabin, L. The Effect of Soft Tissue Stimulation on Skull Vibrations and Hearing Thresholds in Humans. Otol. Neurotol. 2021. Online ahead of print.
- Geal-Dor, M.; Chordekar, S.; Adelman, C.; Kaufmann-Yehezkely, M.; Sohmer, H. Audiogram in Response to Stimulation Delivered to Fluid Applied to the External Meatus. J. Audiol. Otol. 2020, 24, 79–84.




