Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Takeo Mukai | + 3470 word(s) | 3470 | 2021-08-03 05:08:43 | | | |
| 2 | Vivi Li | Meta information modification | 3470 | 2021-08-03 08:58:50 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Mukai, T. MSC and Neurological Diseases. Encyclopedia. Available online: https://encyclopedia.pub/entry/12675 (accessed on 07 February 2026).
Mukai T. MSC and Neurological Diseases. Encyclopedia. Available at: https://encyclopedia.pub/entry/12675. Accessed February 07, 2026.
Mukai, Takeo. "MSC and Neurological Diseases" Encyclopedia, https://encyclopedia.pub/entry/12675 (accessed February 07, 2026).
Mukai, T. (2021, August 03). MSC and Neurological Diseases. In Encyclopedia. https://encyclopedia.pub/entry/12675
Mukai, Takeo. "MSC and Neurological Diseases." Encyclopedia. Web. 03 August, 2021.
Copy Citation
Several studies have shown that mesenchymal stromal/stem cells (MSCs) exert their neuroprotective and neurorestorative efficacy via the secretion of neurotrophic factors. Based on these studies, many clinical trials using MSCs for the treatment of neurological disorders have been conducted, and results regarding their feasibility and efficacy have been reported.
mesenchymal stromal cell
umbilical cord
neurological disorders
cerebral palsy
neurotrophic factor
1. Introduction
Mesenchymal stromal/stem cells (MSCs) have been the focus of new cell therapy development due to their potential to treat neurological disorders [1]. MSCs were first discussed in 1991 when they were introduced by Caplan as mesenchymal cells in bone marrow [2]. Now, MSCs have been isolated from several sources, including bone marrow (BM), the umbilical cord (UC), umbilical cord blood (UCB), dental pulp (DP), and adipose tissue (AD) [1][3][4][5][6][7][8].
The characteristics of MSCs are defined by a set of criteria that form the basis for their clinical use (Figure 1). The International Society for Cellular Therapy proposed the criteria for defining human MSCs [9][10]. Firstly, the MSCs must be plastic-adherent when maintained in standard culture conditions. Secondly, they must express CD105, CD73, and CD90, but not CD45, CD34, CD14 or CD11b, CD79α or CD19, and HLA-DR surface molecules. Thirdly, MSCs must be able to differentiate into adipocytes, chondroblasts, and osteoblasts in vitro. Immunomodulatory effects are the most important and popular property of MSCs in their clinical use [11]. MSCs lack HLA-class II expression and do not express the co-stimulatory surface antigens CD80 or CD86, which activate T-cells [12]. As a result, these cells are able to escape from activated T-cells. MSC-mediated immunomodulation results from the MSC secretome, which includes components such as indoleamine 2,3-dioxygenase (IDO), PGE2, galectin-1, and HLA-G5 [13]. With these anti-inflammatory properties, MSCs could be useful therapeutic candidates for use in the treatment of neurological disorders accompanying inflammation.
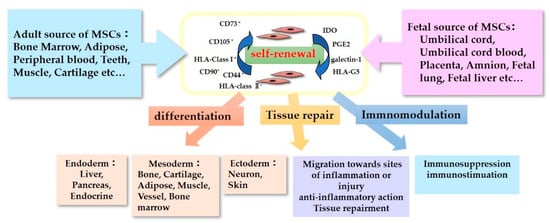
Figure 1. Characteristics of Mesenchymal stromal/stem cells (MSCs).
The tissue repair properties of MSCs are also important to their neurorestorative effect. The neurorestorative and neuroprotective effects of MCSs regarding tissue repair can be divided into two main mechanisms: (1) neurogenic differentiation and eternal cell replacement and (2) the secretion of neurotrophic factors [14]. Regarding the former, we have observed in our experiments that MSCs do not engraft and differentiate into neural cells, and they disappear within two weeks of administration in non-immunocompromised mouse models [15]. In contrast, we found that UC-MSCs that secrete neurotrophic factors such as brain-derived neurotrophic factor (BDNF) and hepatocyte growth factor (HGF) but not nerve growth factor (NGF) attenuate brain injury [15][16]. This MSC paracrine effect is expected to contribute toward their use in therapeutics for neurological injuries. Many studies using neurological disorder models have reported improvements in the studied conditions after the administration of MSCs, and clinical studies using MSCs to treat neurological disorders have been already conducted.
2. Clinical Application of MSCs for Neurological Disorders
Based on the mechanisms suggested by the basic experiments mentioned above, several clinical trials using MSCs for neurological disorders have been conducted, and the recent clinical reports are summarized in this review.
Most of these clinical studies were performed with adult participants, while trials focusing on cerebral palsy were performed with children. Regarding the origin of MSCs; BM, UC, UCB, and AD sources have all been used. In addition, DPSCs are used for clinical trials for neurological disorders [17][18]. As for the administration of MSCs, most clinical studies adopt IV and/or IT. These clinical trials have mainly reported on the feasibility and efficacy of MSC therapies for neurological disorders, with some reporting adverse events, such as fever, vomiting, and headaches, while severe adverse events have not been observed.
2.1. Ischemic Stroke
The recent clinical reports using MSCs for the treatment of ischemic stroke are summarized in Table 1. Some studies have reported on the safety and feasibility of BM-MSCs in patients with ischemic stroke injury [19][20][21][22]. In these clinical trials, patients received intravenous injections of BM-MSCs, and an improvement in neurological functioning was observed, while no treatment-related adverse events were seen. Qiao et al. highlighted the safety and feasibility of the co-transplantation of neural stem/progenitor cells (NSPCs) and UC-MSCs in patients who had suffered from an ischemic stroke [23]. In the study, no tumorigenesis was found during a two-year follow-up, and the neurological functions, disability levels, and daily living abilities of the patients had improved. Jiang et al. reported on the safety and efficacy of UC-MSCs delivered via a catheter to a near-lesion site for treating an infarction in the middle cerebral artery territory [24]. UC-MSCs were infused via catheterization in the M1 segment of the middle cerebral artery. Cell delivery was performed successfully in all the patients, and no major accidents were observed. After this cellular therapy, two of the three ischemic stroke patients demonstrated improved muscle strength. These reports suggest that the transplantation of MSCs in subjects with ischemic stroke is safe and may promote neurological improvement. On the other hand, Nagpal et al. conducted a clinical trial using DPSC for stroke called TOOTH (The Open study of dental pulp stem cell Therapy in Humans) and are investigating the use of autologous stem cell therapy for stroke survivors with chronic disability [18].
Table 1. Summary of recent clinical trials using MSCs for ischemic stroke.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Chung et al. [19] | Ischemic stroke (Phase 3) |
BM | 39 | 15 | 68 (28–83) |
IV | 1 × 106/kg | 1 | Lower extremity motor functional recovery after 3 months |
No |
| Bhasin et al. [20] | Ischemic stroke | BM | 6 | 6 | 42.8 (20–60) |
IV | 5–6 × 107 cells | 1 | Improvement in the activities of daily living (ADL) after 156 and 208 weeks | No |
| Qiao et al. [23] | Ischemic stroke (Phase 1/2) |
UC | 6 | 0 | 56.17 (3–85) |
IVIV + IC | IV:MSC 0.5 × 106/kg IC:MSC 5 × 106 cells NSPC 6 × 106 cells at one-week interval |
4 or 1 + 3 |
Improvement in the neurological functions and ADL after 3, 12, 24 months | Fever, dizziness |
| Jiang et al. [24] | Ischemic and hemorrhagic stroke | UC | 4 | 0 | 40–59 | IA (intra-arterial) via catheterization | 2 × 107 cells | 1 | Motor functional recovery and improvement in the ADL after 3 and 6 months | No |
| Bhasin et al. [21] | Ischemic stroke | BM | 20 MSC6 MNC14 |
20 | 45.1 | IV | 5–6 × 107 cells | 1 | Improvement in the ADL after 8 and 24 weeks |
No |
| Honmou et al. [22] | Ischemic stroke (Phase 1) |
BM | 12 | 0 | 59.2 (41–73) |
IV | 0.6–1.8 × 108 cells | 1 | Incremental daily rate of change in the disability scales during 12 months |
Fever, nausea, itching |
IV, intravenous injection; IT, intrathecal injection; IC, intracranial; BM, bone marrow; UC, umbilical cord.
2.2. Spinal Cord Injury
In most of the clinical trials involving MSC treatment for spinal cord injury (SCI), MSCs were administered via intrathecal or direct infusion to the injured lesion (Table 2). Vaquero et al. reported that patients administered BM-MSCs showed variable clinical improvements in sensitivity, motor power, spasms, spasticity, neuropathic pain, sexual function, and/or sphincter dysfunction, regardless of the level/degree of injury, age, or time elapsed since the SCI [25]. Hur showed the effects and safety of the intrathecal transplantation of autologous AD-MSCs in patients with SCI. Over the 8 months of follow-up, patients who received intrathecal transplantation of autologous AD-MSCs for SCI treatment did not experience any serious adverse events, and several patients showed mild improvements in neurological function [26]. Transplanting collagen scaffolds with human UC-MSCs has also been reported to have therapeutic potential as a treatment for SCI. Collagen scaffolds with human UC-MSCs were transplanted into the injury site directly, and the recovery of sensory and motor functions was observed in both patients [27]. Oh et al. reported on the injection of autologous BM-MSCs into the intramedullary area and subdural space and concluded that this single MSCs application was safe, but it had a very weak therapeutic effect compared with multiple MSC injections [28]. Therefore, further clinical trials to enhance the effect of MSCs are necessary in the future.
Table 2. Summary of recent clinical trials using MSCs for SCI.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Xiao et al. [27] | Spinal cord injury (Phase 1) |
UCB | 2 | 0 | 28, 30 | Transplantation into the lesion with collagen scaffolds | 4 × 107 cells | 1 | Motor functional recovery after 3, 6, 12 months Sensory functional recovery after 2, 4, 12 months |
No |
| Vaquero et al. [25] | Spinal cord injury (Phase 2) |
BM | 11 | 0 | 44.91 (28–62) |
IT | 100 × 106 cells at 3 months interval | 3 | Motor, sensory and bladder–bowel functional recovery after 4, 7, 10 months |
No |
| Vaquero et al. [29] | Post-traumatic syringomyelia (Phase 2) |
BM | 6 | 0 | 39 (30–50) |
Direct injection into the lesion | 300 × 106 cells | 1 | Achieving reduction of syrinx and valiable clinical improvements after 6 months | No |
| Vaquero et al. [30] | Spinal cord injury (Phase 2) |
BM | 10 | 0 | 42.2 (34–59) |
IT | 30 × 106 cells at 3-months interval | 4 | Motor, sensory and bladder–bowel functional recovery after 3, 6, 9, 12 months | Headache, puncture pain |
| Satti et al. [31] | Spinal cord injury (Phase 1) |
BM | 9 | 0 | 31.6 (24–38) |
IT | 1.2 × 106/kg at 4 weeks interval | 2 or 3 | Only safety assessment | No |
| Oh et al. [28] | Spinal cord injury (Phase 3) |
BM | 16 | 0 | 40.9 (18–65) |
Direct injection into the lesion + IT | 1.6 × 107 cells 3.2 × 107 cells |
1 | Very weak therapeutic efficacy after 6 months | Sensory deterioration, muscle rigidity, tingling sense |
| Hur et al. [26] | Spinal cord injury (Phase 1) |
AD | 14 | 0 | 41.9 (20–66) |
IT | 3 × 107 at 1-month interval | 3 | Motor and sensory functional recovery after 8 months | Nausea, vomit, headache |
| Mendonça et al. [32] | Spinal cord injury (Phase 1) |
BM | 14 | 0 | 35.7 (23–61) |
Direct injection into the lesion | 5 × 106 cells/cm3 per lesion volume | 1 | Motor, sensory, and bladder–bowel functional recovery after 6 months | Low-intensity pain at the incision site, cerebrospinal fluid leak |
| Cheng et al. [33] | Spinal cord injury (Phase 2) |
UC | 10 | 34 | 35.3 (19–57) |
Direct injection into the lesion | 2 × 107 cells at 10 days interval | 2 | Motor, sensory, and bladder functional recovery after 6 months Superior efficacy than that of rehabilitation therapy |
Radiating neuralgia |
| Dai et al. [34] | Spinal cord injury (Phase 1/2) |
BM | 20 | 20 | 22–54 | Direct injection into the lesion | 20 × 106 cells | 1 | Motor, sensory, and bladder functional recovery after 6 months | Fever, headache, pain |
| Karamouzian et al. [35] | Spinal cord injury (Phase 1/2) |
BM | 11 | 20 | 33.2 (23–48) |
IT | 0.7–1.2 × 106 cells | 1 | Possible efficacy in the motor and sensory function | No |
IT, intrathecal injection; BM, bone marrow; AD, adipose; UC, umbilical cord; UCB, umbilical cord blood.
2.3. Cerebral Palsy
Recently, MSCs have been emerging for use in potential new therapeutic treatments for children with cerebral palsy. The recent clinical reports using MSCs for the treatment of cerebral palsy are summarized in Table 3. Huang et al. reported on a randomized, placebo-controlled trial of UCB-MSC infusion in children with cerebral palsy [36]. The infusion group was comprised of 27 patients, each of whom received four infusions of UCB-MSCs and basic rehabilitation treatment, whereas another 27 patients were assigned to the control group and received 0.9% normal saline and basic rehabilitation treatment. The changes in the gross motor and comprehensive functional scale in the UCB-MSC infusion group were significantly higher than those in control group at 3-, 6-, 12-, and 24-months post treatment. Liu et al. investigated whether BM-MSCs and BM-mononuclear cells (BM-MNCs) had any difference in curative effect regarding their use in the treatment of cerebral palsy. Their results indicated that BM-MSC transplantation for the treatment of cerebral palsy is safe and can improve gross and fine motor function significantly when compared with the results of BM-MNC treatment [37]. Cerebral palsy and its associated conditions can cause significant economic burdens to families. Therefore, clinical trials that may lead to new cell therapy strategies should be further investigated.
Table 3. Summary of recent clinical trials using MSCs for cerebral palsy.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Gu et al. [38] | Cerebral palsy (Phase 1/2) |
UC | 19 | 20 | 4.29 | IV | 4.5–5.5 × 107 cells at 7-day intervals |
4 | Gross motor and comprehensive functional recovery and improvement in the ADL after 3, 6, 12 months | No |
| Ahn et al. [39] | Intraventricular hemorrhage (Phase 1) |
UCB | 9 | 0 | 11.6 (7–15) (days) |
Intraventricular | 5 × 106/kg or 1 × 107/kg |
1 | Only safety assessment | No |
| Huang et al. [36] | Cerebral palsy (Phase 1/2) |
UCB | 27 | 27 | 7.4 (3–12) |
IV | 5 × 107 cells at 7-day intervals | 4 | Gross motor and comprehensive functional recovery after 3, 6, 12, 24 months | No |
| Liu et al. [37] | Cerebral palsy (Phase 1/2) |
BM | MSC 33 MNC34 |
35 | 7–132 (months) |
IT | 1 × 106/kg at 3–4-day intervals | 4 | Motor functional recovery after 12 months | No |
| Wang et al. [40] | Cerebral palsy (Phase 4) |
UC | 16 (8 twins) | 0 | 6.29 (3–12) |
IT | 1–2 × 106 cells at 3–5-day intervals | 4 | Motor functional recovery after 1 and 6 months | No |
| Wang X et al. [41] | Cerebral palsy | BM | 46 | 0 | 6–180 (months) |
IT Intra-Parenchymal |
2 × 107 cells 4 × 107 cells at 5-day intervals |
2 + 1 or 4 |
Gross motor functional recovery after 1, 6, 18 months | No |
IV, intravenous injection; IT, intrathecal injection; BM, bone marrow; UC, umbilical cord; UCB, umbilical cord blood.
2.4. Amyotrophic Lateral Sclerosis (ALS)
ALS is a fatal neurodegenerative disease characterized by the degeneration of motor neurons in the brain and spinal cord, resulting in progressive muscle weakness and respiratory failure. The recent clinical reports using MSCs for the treatment of ALS are summarized in Table 4. Berry et al. highlighted the safety and efficacy of neurotrophic factor (NTF)-secreting MSCs (NurOwn®, autologous bone marrow-derived MSCs, induced to secrete NTFs) delivered by combined intrathecal and intramuscular administration to participants with ALS in a phase 2 randomized controlled trial [42]. The rate of disease progression (Revised ALS Functional Rating Scale (ALSFRS-R) slope change) in the overall study population was similar in the treated and placebo participants, while in a prespecified rapid progressor subgroup, the rate of disease progression improved at early time points. Furthermore, CSF neurotrophic factors increased, and associated inflammatory biomarker levels decreased in the treated participants post-NTF-secreting MSC transplantation. Another report showed that intrathecal and intramuscular administration of BM-MSC secreting neurotrophic factors in patients with ALS is safe and may provide clinical benefits [43]. Syková et al. demonstrated that the intrathecal application of BM-MSCs in ALS patients is a safe procedure and that this treatment could slow down the progression of the disease; a reduction in ALSFRS decline at three months after application was observed which, in some cases, persisted for six months [44]. Oh et al. reported that two repeated intrathecal injections of autologous BM-MSCs was a safe and feasible treatment for ALS patients throughout the duration of a 12-month follow-up period [45]. These results support the possibility that the use of MSCs in ALS patients could slow down the progression of the disease.
Table 4. Summary of recent clinical trials using MSCs for ALS.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Berry et al. [42] | ALS (Phase 2) |
BM-NTF | 36 | 12 | 51.1 (26–71) |
IM + IT | IM: 48 × 106 cells IT: 125 × 106 cells |
1 | Improvement in the rate of disease progression after 6 months |
Headache, fever, back pain, injection site bruising |
| Syková et al. [44] | ALS (Phase 1/2) |
BM | 26 | 0 | 51.2 (33–64) |
IT | 15 ± 4.5 × 106 cells | 1 | Slowing down of the diseaseprogression after 3, 6, 9 months | Headache |
| Staff et al. [46] | ALS (Phase 1) |
AD | 27 | 0 | 36–75 | IT | 1 × 107, 5 × 107, 5 × 107 × 2, 1 × 108, 1 × 108 × 2 | 1 or 2 | Only safety assessment | Temporary back and leg pain in the highest dose |
| Petrou et al. [43] | ALS (Phase 1/2) |
BM-NTF | 26 | 0 | 48.1, 50.8 (23–65) |
IM IT IM + IT |
IM: 2.4–4.8 × 107 cells IT: 1.0–2.0 × 106 /kg |
1 | Improvement in the rate of disease progression after 6 months | Fever, vomiting, headache |
| Rushkevich et al. [47] | ALS | BM-MSC and neural induced MSC | 10 | 15 | 54.5, 55.0 (37–66) |
IV + IT | 0.5–1.5 × 106/kg 5.0–9.7 × 106 cells at 5–7-month intervals |
1 or 2 | Slowing down of the disease progression after 12 months |
Fever, headache |
| Oh et al. [45] | ALS (Phase 1) |
BM | 8 | 0 | 45.7 (29–62) |
IT | 1 × 106/kg at 26-day intervals |
2 | No efficacy after 6 months | Fever, pain, headache |
| Kim et al. [48] | ALS | BM | 37 | 0 | 52.7, 48.8 | IT | 1 × 106/kg at one-month intervals |
2 | Trophic factors associated with a positive response to treat | No |
| Mazzini et al. [49] | ALS (Phase 1) | BM | 19 | 0 | 20–75 | Direct injection into spinal cord | 7–152 × 106 cells | 1 | No long-term adverse effect after nearly 9 years | No |
IV, intravenous injection; IT, intrathecal injection; IM, intramuscular injection; BM, bone marrow; AD, adipose.
2.5. Multiple Sclerosis
Multiple sclerosis is a chronic immune-mediated inflammatory disease in which the immune system progressively destroys the myelin sheath in the central nervous system. This disease can last from a few months to many years. The recent clinical reports using MSCs for the treatment of multiple sclerosis are summarized in Table 5. Petrou et al. evaluated the optimal safe and effective clinical transplantation of MSCs in patients with active and progressive multiple sclerosis [50]. In the study, patients were randomized into three groups and treated intrathecally (IT) or intravenously (IV) with autologous BM-MSCs or sham injections. Significantly fewer patients experienced treatment failure in the MSC-IT and MSC-IV groups compared with those in the sham-treatment group. During the 1-year follow-up period, no evidence of disease activity was observed in 58.6% and 40.6% of patients treated with MSC-IT and MSC-IV, respectively, compared with 9.7% in the sham-treated group. MSC-IT transplantation induced additional benefits regarding the relapse rate, and the researchers concluded that the IT administration was more efficacious than the IV administration regarding several parameters of the disease. Furthermore, a safety and feasibility study was completed, focusing on the use of UC-MSCs for treating multiple sclerosis. Twenty subjects were enrolled in the study, and symptom improvements were most notable a month after treatment [51]. Infusion with MSCs is considered safe and feasible in patients with multiple sclerosis. However, larger studies investigating the number of doses and route of administration are needed to assess potential therapeutic benefits of this technique.
Table 5. Summary of recent clinical trials using MSCs for multiple sclerosis.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Petrou et al. [50] | Multiple sclerosis (Phase 2) |
BM | 16, 16 |
16 | 47.6 (37.9–57.3) |
IT or IV |
1 × 106/kg at 6-month intervals | 1 or 2 | Improvement in the course of the disease and comprehensive functional recovery after 3, 6, 12 months. IT is more efficacious than IV |
No |
| Fernández et al. [52] | Multiple sclerosis (phase 1/2) |
AD | 10, 9 |
11 | 44.8 47.8 46.3 |
IV | 1 × 106/kg or 4 × 106/kg |
1 | Partial efficacy in the imaging studies and evoked potentials after 12 months | urinary infection |
| Riordan et al. [51] | Multiple sclerosis (phase 1/2) |
UC | 20 | 0 | 41.15 | IV | 20 × 106 cells at 1–4-day intervals | 7 | Comprehensive functional recovery after one month | Headache, fatigue |
| Harris et al. [53] | Multiple sclerosis (Phase 1) |
BM MSC -derived neural progenitors | 20 | 0 | 27–65 | IT | 5.3–10 × 106 cells at 3-month intervals | 3 | Motor, bladder and comprehensive functional recovery after 3 months | headache, fever |
| Dahbour et al. [54] | Multiple sclerosis (Phase 1/2) |
BM MSC-CM |
10 | 0 | 34.9 (18–54) |
IT | 93–168 × 106 cells CM:13–20 mL at 1-month intervals |
1 + 1 | Comprehensive functional recovery after 12 months | Pain, headache, fever |
| Llufriu et al. [55] | Multiple sclerosis (Phase 2) |
BM | 9 | 0 | 36.8 (23–48) |
IV | 1–2 × 106/kg | 1 | Improvement in the imaging studies after 6 months | No |
| Li et al. [56] | Multiple sclerosis | UC | 13 | 10 | 41.7, 39.4 | IV | 4 × 106 cells/kg at 2-week intervals | 3 | Improvement in the overall symptoms and fewer incidences of relapse during 12 months |
No |
| Bonab et al. [57] | Multiple sclerosis (Phase 2) |
BM | 25 | 0 | 34.7 (23–50) |
IT | 2.95 × 107 cells | 1 | Improvement or stabilization in the course of the disease during 12 months |
Fever, nausea, weakness in the lower limbs, headache |
| Lee et al. [58] | Multiple sclerosis (Phase 2) |
BM | 16 | 17 | 56.1, 55.8 | IA (intra-arterial) + IV | IA: 4 × 107 cells IV: 4 × 107 cells at 30-day intervals |
1 + 3 | Efficacy in preventing the progression of neurological deficits during 12 months | Small ischemic lesions |
| Connick et al. [59] | Multiple sclerosis (Phase 2) |
BM | 10 | 0 | 48.8 (40–53) |
IV | 1.6 × 106/kg | 1 | Visual functional recovery after 10 months | Macular rash, self-limiting infections |
IV, intravenous injection; IT, intrathecal injection; BM, bone marrow; AD, adipose; UC, umbilical cord.
2.6. Parkinson’s Disease
Parkinson’s disease is the common and progressive neurodegenerative disease with major symptoms such as bradykinesia, impaired posture, and tremor. Some studies have reported on the safety and feasibility of MSCs in patients with Parkinson’s disease (Table 6). Canesi et al. demonstrated the feasibility of BM-MSC in Parkinson’s disease patients. One year after cell infusion, all treated patients were alive, except one, who died 9 months after the infusion for reasons not related to cell administration or to disease progression (accidental fall), and in all treated patients, motor function rating scales remained stable for at least six months during the one-year follow-up [60]. On the other hand, Carstens et al. showed the efficacy of AD-MSCs in two patients with Parkinson’s disease. After the administration of AD-MSCs, subjective functional recovery after 2 weeks and up to 5 years are observed [61].
Table 6. Summary of recent clinical trials using MSCs for Parkinson’s disease.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Canesi et al. [60] | Progressive supranuclear palsy (Phase 1) |
BM | 5 | 0 | 60–68 | IA (intra-arterial) via catheterization | 1.7 (1.2–2.0) × 106/kg | 1 | Clinical stabilization for at least 6 months during the one-year follow-up | Transient left hemiparesis |
| Carstens et al. [61] | Parkinson’s disease (Case studies) |
AD MSC-derived stromal vascular fraction | 2 | 0 | 72, 50 | Facial and nasal transplantation | 6.0 × 107 cells | 1 | Subjective functional recovery after 2 weeks and up to 5 years | No |
BM, bone marrow; AD, adipose.
2.7. Traumatic Brain Injury
Traumatic brain injury is one of the major serious diseases that threaten human life and health, causing traffic accidents, collisions with hard objects, and falls from high places. With improving medical technology, the survival rate of patients with traumatic brain injury has increased significantly. However, the prognosis for patients with severe TBI remains poor, such as disturbance of consciousness and motor disorder. The recent clinical reports using MSCs for the treatment of traumatic brain injury are summarized in Table 7. Wang et al. showed the results of a phase 2 clinical trial using UC-MSCs for traumatic brain injury patients [62]. Forty patients with sequelae of traumatic brain injury were randomly assigned to the stem cell treatment group or the control group, and UC-MSCs administration improved the neurological function and self-care in patients after 6 months. On the other hand, Tian et al. explored the clinical therapeutic effects and safety of autologous BM-MSCs therapy for traumatic brain injury by lumbar puncture [63]. The results showed improvement in the function of brain in the form of post-therapeutic improvements in consciousness and motor functions. In addition, they showed the age of patients and the time elapsed between injury and therapy had effects on the outcomes of the cellular therapy, and no correlation was found between the number of cell injections and improvements.
Table 7. Summary of recent clinical trials using MSCs for traumatic brain injury.
| Reference | Disease | Source | Number | Mean Age (Range), Year | Route of Administration | Number of Cells | Number of Treatments | Results | Adverse Events | |
|---|---|---|---|---|---|---|---|---|---|---|
| Trial | Control | |||||||||
| Wang et al. [62] | Traumatic brain injury (Phase 2) |
UC | 20 | 20 | 27.5 ± 9.4 28.6 ± 10.1 |
IT | 6.0 × 107 cells | 4 | Comprehensive functional recovery and improvement in the ADL after 6 months |
Mild dizziness, headache |
| Tian et al. [63] | Traumatic brain injury | BM | 97 | 0 | - | IT | 3.0–5.0 × 106 cells | 1 | Improvement of consciousness and motor function after 14 days | No |
IT, intrathecal injection; BM, bone marrow; UC, Umbilical cord.
References
- Nagamura-Inoue, T.; Mukai, T. Umbilical Cord is a Rich Source of Mesenchymal Stromal Cells for Cell Therapy. Curr. Stem Cell Res. Ther. 2016, 11, 634–642.
- Caplan, A.I. Mesenchymal stem cells. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 1991, 9, 641–650.
- Gnecchi, M.; Melo, L.G. Bone marrow-derived mesenchymal stem cells: Isolation, expansion, characterization, viral transduction, and production of conditioned medium. Methods Mol. Biol. 2009, 482, 281–294.
- Bieback, K.; Kluter, H. Mesenchymal stromal cells from umbilical cord blood. Curr. Stem Cell Res. Ther. 2007, 2, 310–323.
- In’t Anker, P.S.; Scherjon, S.A.; Kleijburg-van der Keur, C.; de Groot-Swings, G.M.; Claas, F.H.; Fibbe, W.E.; Kanhai, H.H. Isolation of mesenchymal stem cells of fetal or maternal origin from human placenta. Stem Cells 2004, 22, 1338–1345.
- Gruber, H.E.; Deepe, R.; Hoelscher, G.L.; Ingram, J.A.; Norton, H.J.; Scannell, B.; Loeffler, B.J.; Zinchenko, N.; Hanley, E.N.; Tapp, H. Human adipose-derived mesenchymal stem cells: Direction to a phenotype sharing similarities with the disc, gene expression profiling, and coculture with human annulus cells. Tissue Eng. Part A 2010, 16, 2843–2860.
- Chen, T.F.; Chen, K.W.; Chien, Y.; Lai, Y.H.; Hsieh, S.T.; Ma, H.Y.; Wang, K.C.; Shiau, C.Y. Dental Pulp Stem Cell-Derived Factors Alleviate Subarachnoid Hemorrhage-Induced Neuroinflammation and Ischemic Neurological Deficits. Int. J. Mol. Sci. 2019, 20, 3747.
- Aliaghaei, A.; Boroujeni, M.E.; Ahmadi, H.; Bayat, A.H.; Tavirani, M.R.; Abdollahifar, M.A.; Pooyafar, M.H.; Mansouri, V. Dental pulp stem cell transplantation ameliorates motor function and prevents cerebellar atrophy in rat model of cerebellar ataxia. Cell Tissue Res. 2019, 376, 179–187.
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317.
- Horwitz, E.M.; Le Blanc, K.; Dominici, M.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Deans, R.J.; Krause, D.S.; Keating, A.; International Society for Cellular Therapy. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 2005, 7, 393–395.
- Nagamura-Inoue, T.; He, H. Umbilical cord-derived mesenchymal stem cells: Their advantages and potential clinical utility. World J. Stem Cells 2014, 6, 195–202.
- Weiss, M.L.; Anderson, C.; Medicetty, S.; Seshareddy, K.B.; Weiss, R.J.; VanderWerff, I.; Troyer, D.; McIntosh, K.R. Immune properties of human umbilical cord Wharton’s jelly-derived cells. Stem Cells 2008, 26, 2865–2874.
- Seshareddy, K.; Troyer, D.; Weiss, M.L. Method to isolate mesenchymal-like cells from Wharton’s Jelly of umbilical cord. Methods Cell Biol. 2008, 86, 101–119.
- Mukai, T.; Tojo, A.; Nagamura-Inoue, T. Mesenchymal stromal cells as a potential therapeutic for neurological disorders. Regen. Ther. 2018, 9, 32–37.
- Mukai, T.; Mori, Y.; Shimazu, T.; Takahashi, A.; Tsunoda, H.; Yamaguchi, S.; Kiryu, S.; Tojo, A.; Nagamura-Inoue, T. Intravenous injection of umbilical cord-derived mesenchymal stromal cells attenuates reactive gliosis and hypomyelination in a neonatal intraventricular hemorrhage model. Neuroscience 2017, 355, 175–187.
- Mukai, T.; Tojo, A.; Nagamura-Inoue, T. Umbilical Cord-Derived Mesenchymal Stromal Cells Contribute to Neuroprotection in Neonatal Cortical Neurons Damaged by Oxygen-Glucose Deprivation. Front. Neurol. 2018, 9, 466.
- Luzuriaga, J.; Polo, Y.; Pastor-Alonso, O.; Pardo-Rodríguez, B.; Larrañaga, A.; Unda, F.; Sarasua, J.R.; Pineda, J.R.; Ibarretxe, G. Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies. Int. J. Mol. Sci. 2021, 22, 3564.
- Nagpal, A.; Kremer, K.L.; Hamilton-Bruce, M.A.; Kaidonis, X.; Milton, A.G.; Levi, C.; Shi, S.; Carey, L.; Hillier, S.; Rose, M.; et al. TOOTH (The Open study Of dental pulp stem cell Therapy in Humans): Study protocol for evaluating safety and feasibility of autologous human adult dental pulp stem cell therapy in patients with chronic disability after stroke. Int. J. Stroke Off. J. Int. Stroke Soc. 2016, 11, 575–585.
- Chung, J.W.; Chang, W.H.; Bang, O.Y.; Moon, G.J.; Kim, S.J.; Kim, S.K.; Lee, J.S.; Sohn, S.I.; Kim, Y.H. Efficacy and Safety of Intravenous Mesenchymal Stem Cells for Ischemic Stroke. Neurology 2021, 96, e1012–e1023.
- Bhasin, A.; Kumaran, S.S.; Bhatia, R.; Mohanty, S.; Srivastava, M.V.P. Safety and Feasibility of Autologous Mesenchymal Stem Cell Transplantation in Chronic Stroke in Indian patients. A four-year follow up. J. Stem Cells Regen. Med. 2017, 13, 14–19.
- Bhasin, A.; Srivastava, M.V.; Mohanty, S.; Bhatia, R.; Kumaran, S.S.; Bose, S. Stem cell therapy: A clinical trial of stroke. Clin. Neurol. Neurosurg. 2013, 115, 1003–1008.
- Honmou, O.; Houkin, K.; Matsunaga, T.; Niitsu, Y.; Ishiai, S.; Onodera, R.; Waxman, S.G.; Kocsis, J.D. Intravenous administration of auto serum-expanded autologous mesenchymal stem cells in stroke. Brain 2011, 134, 1790–1807.
- Qiao, L.Y.; Huang, F.J.; Zhao, M.; Xie, J.H.; Shi, J.; Wang, J.; Lin, X.Z.; Zuo, H.; Wang, Y.L.; Geng, T.C. A two-year follow-up study of cotransplantation with neural stem/progenitor cells and mesenchymal stromal cells in ischemic stroke patients. Cell Transplant. 2014, 23, S65–S72.
- Jiang, Y.; Zhu, W.; Zhu, J.; Wu, L.; Xu, G.; Liu, X. Feasibility of delivering mesenchymal stem cells via catheter to the proximal end of the lesion artery in patients with stroke in the territory of the middle cerebral artery. Cell Transplant. 2013, 22, 2291–2298.
- Vaquero, J.; Zurita, M.; Rico, M.A.; Aguayo, C.; Bonilla, C.; Marin, E.; Tapiador, N.; Sevilla, M.; Vazquez, D.; Carballido, J.; et al. Intrathecal administration of autologous mesenchymal stromal cells for spinal cord injury: Safety and efficacy of the 100/3 guideline. Cytotherapy 2018, 20, 806–819.
- Hur, J.W.; Cho, T.H.; Park, D.H.; Lee, J.B.; Park, J.Y.; Chung, Y.G. Intrathecal transplantation of autologous adipose-derived mesenchymal stem cells for treating spinal cord injury: A human trial. J. Spinal Cord Med. 2016, 39, 655–664.
- Xiao, Z.; Tang, F.; Zhao, Y.; Han, G.; Yin, N.; Li, X.; Chen, B.; Han, S.; Jiang, X.; Yun, C.; et al. Significant Improvement of Acute Complete Spinal Cord Injury Patients Diagnosed by a Combined Criteria Implanted with NeuroRegen Scaffolds and Mesenchymal Stem Cells. Cell Transplant. 2018, 27, 907–915.
- Oh, S.K.; Choi, K.H.; Yoo, J.Y.; Kim, D.Y.; Kim, S.J.; Jeon, S.R. A Phase III Clinical Trial Showing Limited Efficacy of Autologous Mesenchymal Stem Cell Therapy for Spinal Cord Injury. Neurosurgery 2016, 78, 436–447.
- Vaquero, J.; Zurita, M.; Rico, M.A.; Aguayo, C.; Fernandez, C.; Rodriguez-Boto, G.; Marin, E.; Tapiador, N.; Sevilla, M.; Carballido, J.; et al. Cell therapy with autologous mesenchymal stromal cells in post-traumatic syringomyelia. Cytotherapy 2018, 20, 796–805.
- Vaquero, J.; Zurita, M.; Rico, M.A.; Bonilla, C.; Aguayo, C.; Fernández, C.; Tapiador, N.; Sevilla, M.; Morejón, C.; Montilla, J.; et al. Repeated subarachnoid administrations of autologous mesenchymal stromal cells supported in autologous plasma improve quality of life in patients suffering incomplete spinal cord injury. Cytotherapy 2017, 19, 349–359.
- Satti, H.S.; Waheed, A.; Ahmed, P.; Ahmed, K.; Akram, Z.; Aziz, T.; Satti, T.M.; Shahbaz, N.; Khan, M.A.; Malik, S.A. Autologous mesenchymal stromal cell transplantation for spinal cord injury: A Phase I pilot study. Cytotherapy 2016, 18, 518–522.
- Mendonça, M.V.; Larocca, T.F.; de Freitas Souza, B.S.; Villarreal, C.F.; Silva, L.F.; Matos, A.C.; Novaes, M.A.; Bahia, C.M.; de Oliveira Melo Martinez, A.C.; Kaneto, C.M.; et al. Safety and neurological assessments after autologous transplantation of bone marrow mesenchymal stem cells in subjects with chronic spinal cord injury. Stem Cell Res. Ther. 2014, 5, 126.
- Cheng, H.; Liu, X.; Hua, R.; Dai, G.; Wang, X.; Gao, J.; An, Y. Clinical observation of umbilical cord mesenchymal stem cell transplantation in treatment for sequelae of thoracolumbar spinal cord injury. J. Transl. Med. 2014, 12, 253.
- Dai, G.; Liu, X.; Zhang, Z.; Yang, Z.; Dai, Y.; Xu, R. Transplantation of autologous bone marrow mesenchymal stem cells in the treatment of complete and chronic cervical spinal cord injury. Brain Res. 2013, 1533, 73–79.
- Karamouzian, S.; Nematollahi-Mahani, S.N.; Nakhaee, N.; Eskandary, H. Clinical safety and primary efficacy of bone marrow mesenchymal cell transplantation in subacute spinal cord injured patients. Clin. Neurol. Neurosurg. 2012, 114, 935–939.
- Huang, L.; Zhang, C.; Gu, J.; Wu, W.; Shen, Z.; Zhou, X.; Lu, H. A Randomized, Placebo-Controlled Trial of Human Umbilical Cord Blood Mesenchymal Stem Cell Infusion for Children With Cerebral Palsy. Cell Transplant. 2018, 27, 325–334.
- Liu, X.; Fu, X.; Dai, G.; Wang, X.; Zhang, Z.; Cheng, H.; Zheng, P.; An, Y. Comparative analysis of curative effect of bone marrow mesenchymal stem cell and bone marrow mononuclear cell transplantation for spastic cerebral palsy. J. Transl. Med. 2017, 15, 48.
- Gu, J.; Huang, L.; Zhang, C.; Wang, Y.; Zhang, R.; Tu, Z.; Wang, H.; Zhou, X.; Xiao, Z.; Liu, Z.; et al. Therapeutic evidence of umbilical cord-derived mesenchymal stem cell transplantation for cerebral palsy: A randomized, controlled trial. Stem Cell Res. Ther. 2020, 11, 43.
- Ahn, S.Y.; Chang, Y.S.; Sung, S.I.; Park, W.S. Mesenchymal Stem Cells for Severe Intraventricular Hemorrhage in Preterm Infants: Phase I Dose-Escalation Clinical Trial. Stem Cells Transl. Med. 2018, 7, 847–856.
- Wang, X.; Hu, H.; Hua, R.; Yang, J.; Zheng, P.; Niu, X.; Cheng, H.; Dai, G.; Liu, X.; Zhang, Z.; et al. Effect of umbilical cord mesenchymal stromal cells on motor functions of identical twins with cerebral palsy: Pilot study on the correlation of efficacy and hereditary factors. Cytotherapy 2015, 17, 224–231.
- Wang, X.; Cheng, H.; Hua, R.; Yang, J.; Dai, G.; Zhang, Z.; Wang, R.; Qin, C.; An, Y. Effects of bone marrow mesenchymal stromal cells on gross motor function measure scores of children with cerebral palsy: A preliminary clinical study. Cytotherapy 2013, 15, 1549–1562.
- Berry, J.D.; Cudkowicz, M.E.; Windebank, A.J.; Staff, N.P.; Owegi, M.; Nicholson, K.; McKenna-Yasek, D.; Levy, Y.S.; Abramov, N.; Kaspi, H.; et al. NurOwn, phase 2, randomized, clinical trial in patients with ALS: Safety, clinical, and biomarker results. Neurology 2019, 93, e2294–e2305.
- Petrou, P.; Gothelf, Y.; Argov, Z.; Gotkine, M.; Levy, Y.S.; Kassis, I.; Vaknin-Dembinsky, A.; Ben-Hur, T.; Offen, D.; Abramsky, O.; et al. Safety and Clinical Effects of Mesenchymal Stem Cells Secreting Neurotrophic Factor Transplantation in Patients With Amyotrophic Lateral Sclerosis: Results of Phase 1/2 and 2a Clinical Trials. JAMA Neurol. 2016, 73, 337–344.
- Syková, E.; Rychmach, P.; Drahorádová, I.; Konrádová, Š.; Růžičková, K.; Voříšek, I.; Forostyak, S.; Homola, A.; Bojar, M. Transplantation of Mesenchymal Stromal Cells in Patients With Amyotrophic Lateral Sclerosis: Results of Phase I/IIa Clinical Trial. Cell Transplant. 2017, 26, 647–658.
- Oh, K.W.; Moon, C.; Kim, H.Y.; Oh, S.I.; Park, J.; Lee, J.H.; Chang, I.Y.; Kim, K.S.; Kim, S.H. Phase I trial of repeated intrathecal autologous bone marrow-derived mesenchymal stromal cells in amyotrophic lateral sclerosis. Stem Cells Transl. Med. 2015, 4, 590–597.
- Staff, N.P.; Madigan, N.N.; Morris, J.; Jentoft, M.; Sorenson, E.J.; Butler, G.; Gastineau, D.; Dietz, A.; Windebank, A.J. Safety of intrathecal autologous adipose-derived mesenchymal stromal cells in patients with ALS. Neurology 2016, 87, 2230–2234.
- Rushkevich, Y.N.; Kosmacheva, S.M.; Zabrodets, G.V.; Ignatenko, S.I.; Goncharova, N.V.; Severin, I.N.; Likhachev, S.A.; Potapnev, M.P. The Use of Autologous Mesenchymal Stem Cells for Cell Therapy of Patients with Amyotrophic Lateral Sclerosis in Belarus. Bull. Exp. Biol. Med. 2015, 159, 576–581.
- Kim, H.Y.; Kim, H.; Oh, K.W.; Oh, S.I.; Koh, S.H.; Baik, W.; Noh, M.Y.; Kim, K.S.; Kim, S.H. Biological markers of mesenchymal stromal cells as predictors of response to autologous stem cell transplantation in patients with amyotrophic lateral sclerosis: An investigator-initiated trial and in vivo study. Stem Cells 2014, 32, 2724–2731.
- Mazzini, L.; Mareschi, K.; Ferrero, I.; Miglioretti, M.; Stecco, A.; Servo, S.; Carriero, A.; Monaco, F.; Fagioli, F. Mesenchymal stromal cell transplantation in amyotrophic lateral sclerosis: A long-term safety study. Cytotherapy 2012, 14, 56–60.
- Petrou, P.; Kassis, I.; Levin, N.; Paul, F.; Backner, Y.; Benoliel, T.; Oertel, F.C.; Scheel, M.; Hallimi, M.; Yaghmour, N.; et al. Beneficial effects of autologous mesenchymal stem cell transplantation in active progressive multiple sclerosis. Brain 2020, 143, 3574–3588.
- Riordan, N.H.; Morales, I.; Fernández, G.; Allen, N.; Fearnot, N.E.; Leckrone, M.E.; Markovich, D.J.; Mansfield, D.; Avila, D.; Patel, A.N.; et al. Clinical feasibility of umbilical cord tissue-derived mesenchymal stem cells in the treatment of multiple sclerosis. J. Transl. Med. 2018, 16, 57.
- Fernández, O.; Izquierdo, G.; Fernández, V.; Leyva, L.; Reyes, V.; Guerrero, M.; León, A.; Arnaiz, C.; Navarro, G.; Páramo, M.D.; et al. Adipose-derived mesenchymal stem cells (AdMSC) for the treatment of secondary-progressive multiple sclerosis: A triple blinded, placebo controlled, randomized phase I/II safety and feasibility study. PLoS ONE 2018, 13, e0195891.
- Harris, V.K.; Stark, J.; Vyshkina, T.; Blackshear, L.; Joo, G.; Stefanova, V.; Sara, G.; Sadiq, S.A. Phase I Trial of Intrathecal Mesenchymal Stem Cell-derived Neural Progenitors in Progressive Multiple Sclerosis. EBioMedicine 2018, 29, 23–30.
- Dahbour, S.; Jamali, F.; Alhattab, D.; Al-Radaideh, A.; Ababneh, O.; Al-Ryalat, N.; Al-Bdour, M.; Hourani, B.; Msallam, M.; Rasheed, M.; et al. Mesenchymal stem cells and conditioned media in the treatment of multiple sclerosis patients: Clinical, ophthalmological and radiological assessments of safety and efficacy. CNS Neurosci. Ther. 2017, 23, 866–874.
- Llufriu, S.; Sepúlveda, M.; Blanco, Y.; Marín, P.; Moreno, B.; Berenguer, J.; Gabilondo, I.; Martínez-Heras, E.; Sola-Valls, N.; Arnaiz, J.A.; et al. Randomized placebo-controlled phase II trial of autologous mesenchymal stem cells in multiple sclerosis. PLoS ONE 2014, 9, e113936.
- Li, J.F.; Zhang, D.J.; Geng, T.; Chen, L.; Huang, H.; Yin, H.L.; Zhang, Y.Z.; Lou, J.Y.; Cao, B.; Wang, Y.L. The potential of human umbilical cord-derived mesenchymal stem cells as a novel cellular therapy for multiple sclerosis. Cell Transplant. 2014, 23, S113–S122.
- Bonab, M.M.; Sahraian, M.A.; Aghsaie, A.; Karvigh, S.A.; Hosseinian, S.M.; Nikbin, B.; Lotfi, J.; Khorramnia, S.; Motamed, M.R.; Togha, M.; et al. Autologous mesenchymal stem cell therapy in progressive multiple sclerosis: An open label study. Curr. Stem Cell Res. Ther. 2012, 7, 407–414.
- Lee, P.H.; Lee, J.E.; Kim, H.S.; Song, S.K.; Lee, H.S.; Nam, H.S.; Cheong, J.W.; Jeong, Y.; Park, H.J.; Kim, D.J.; et al. A randomized trial of mesenchymal stem cells in multiple system atrophy. Ann. Neurol. 2012, 72, 32–40.
- Connick, P.; Kolappan, M.; Crawley, C.; Webber, D.J.; Patani, R.; Michell, A.W.; Du, M.Q.; Luan, S.L.; Altmann, D.R.; Thompson, A.J.; et al. Autologous mesenchymal stem cells for the treatment of secondary progressive multiple sclerosis: An open-label phase 2a proof-of-concept study. Lancet Neurol 2012, 11, 150–156.
- Canesi, M.; Giordano, R.; Lazzari, L.; Isalberti, M.; Isaias, I.U.; Benti, R.; Rampini, P.; Marotta, G.; Colombo, A.; Cereda, E.; et al. Finding a new therapeutic approach for no-option Parkinsonisms: Mesenchymal stromal cells for progressive supranuclear palsy. J. Transl. Med. 2016, 14, 127.
- Carstens, M.; Haq, I.; Martinez-Cerrato, J.; Dos-Anjos, S.; Bertram, K.; Correa, D. Sustained clinical improvement of Parkinson’s disease in two patients with facially-transplanted adipose-derived stromal vascular fraction cells. J. Clin. Neurosci. 2020, 81, 47–51.
- Wang, S.; Cheng, H.; Dai, G.; Wang, X.; Hua, R.; Liu, X.; Wang, P.; Chen, G.; Yue, W.; An, Y. Umbilical cord mesenchymal stem cell transplantation significantly improves neurological function in patients with sequelae of traumatic brain injury. Brain Res. 2013, 1532, 76–84.
- Tian, C.; Wang, X.; Wang, X.; Wang, L.; Wang, X.; Wu, S.; Wan, Z. Autologous bone marrow mesenchymal stem cell therapy in the subacute stage of traumatic brain injury by lumbar puncture. Exp. Clin. Transplant. 2013, 11, 176–181.
More
Information
Subjects:
Neurosciences
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.0K
Revisions:
2 times
(View History)
Update Date:
03 Aug 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




