
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alessia Ciogli | + 1953 word(s) | 1953 | 2021-07-05 11:22:32 | | | |
| 2 | Lindsay Dong | Meta information modification | 1953 | 2021-07-28 11:13:02 | | |
Video Upload Options
Electrostatic Repulsion Reversed-Phase Liquid Chromatography (ERRP) allows the elution of bases in protonated form without peak distorsion (as commonly happen in reversed phase) due to the presence of positive charges in the chromatographic system.
1. Introduction
An efficient resolution of basic compounds by reversed phase HPLC (RP-HPLC) often requires fine tuning of unconventional mobile phases. In fact, their difficult separation is mainly due to the interaction of basic nitrogen with the residual, non-negligible acid silanols found on the stationary phase. Indeed, considering this, at most operating pHs, basic sites are protonated while silanols are in anionic form, and the strong resulting electrostatic interaction increases retention times of analyte and produces a peak shape distortion, including tailing, fronting, and splitting. In addition, taking into account that a large number of compounds of pharmaceutical interest are bases, and that biopharmaceutical and biological samples (e.g., peptides) also have multibasic sites, the HPLC analysis of basic samples has received particular attention [1][2][3]. Reversed phase HPLC remains the first choice of work, offering a set of possible solutions.
Regardless of the anchoring chemistry (often not stated in detail) to prepare silica-based stationary phases for RP-HPLC, it should be noted that about 50% of original silanols present on the silica surface remain unreacted after derivatization with organo-silane. Surface coverage of a typical C18 ligand span between 4 and 4.5 micromoles/m2, while the surface concentration of silanols on bare silica is about 8.0 micromoles/m2 [4]. Moreover, the additional “end-capping” procedure, with smaller silylating agents, fails to inactivate the residual silanols [5]. In the last 20 years, with the diffusion of ultra-high-pressure liquid chromatography (UHPLC), a lot of attention has been paid to innovative silica bulks with a reduced number of silanols. High-purity silica and hybrid silica particles were routinely used to develop stationary phases. The reduced amount of metals on the surface and the organic moieties in the siliceous skeleton enhance the peak performance, reducing the unwanted secondary selector/selectand interactions and increasing mechanical resistance. Hybrid silica, in particular, causes a reduction of nearly one-third of the number of silanol sites [6][7][8][9][10]. Unfortunately, in resolving bases, the use of recently developed silica bulks alone does not circumvent the problem. Therefore, working at a low pH (usually pH < 3.5) in the mobile phase represents one of two suitable solutions that allow the use of the standard RP commercially available columns. In these elution conditions, silanols are uncharged and basic solutes protonated. Sample peaks are sharp and their retention times are shortened. Alternately, the addition of amines to the eluent, preferably tertiary ones, can efficiently shield residual silanols, achieving similar effects. In this case, amines are positively charged at acidic pH and can interact electrostatically with the surface anionic silanols, while the hydrophobic moiety of the molecule can associate with the alkyl fragment of the stationary phase. Despite the two approaches having limitations (in some cases very low retention and consequent loss of selectivity are achieved; additives and acid mobile phases are not perfectly well-matched with MS detection), they have gained renewed interest due to the mixed-mode electrostatic repulsive interaction combined with the hydrophobic one. The two approaches, which have, to date, only focused on the suppression of silanols to enhance hydrophobic interaction and peak shape, were recently revised to investigate the effect of electrostatic repulsion. The modulation of the amount of fixed (herein called static) or adsorbed (herein called dynamic) positive charges on the stationary phase can efficiently contribute to retention of bases. Looking to the literature, a similar interaction has been provided in electrostatic repulsion hydrophilic interaction chromatography, named ERLIC. This elution mode involves a combination of the ion-exchange stationary phase with the same charge of analytes and the high organic solvent concentration as classical hydrophilic interaction liquid chromatography (HILIC) conditions [11][12]. In reversed phase liquid chromatography, the expression “electrostatic repulsion reversed phase” (ERRP) was introduced by Gritti in 2013 by studying the absorption behaviors of ionizable compounds in the presence of static positive charges on the C18 stationary phase [13].
2. Static ERRP-HPLC
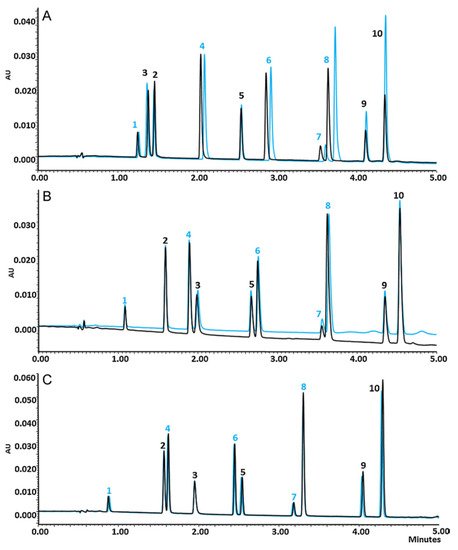
The designing of alternative stationary phases to resolve basic compounds is an ambitious goal. Being the most intuitive and immediate solution, mixed-mode ion-exchange/reversed phase stationary phases (weak anion exchange/strong anion exchange, WAX/SAX, as well as weak cation exchange/strong cation exchange, WCX/SCX, and RP) were optimized to be used in these fields, exploiting the interaction due to the opposite charges if cation exchange supports were employed or the shielding of silanols using anionic exchange supports. For example, D.V. McCalley and coworkers reported the use of mixed-mode RP/embedded ion-exchange stationary phases. In this work, the elution of a set of bases (e.g., codeine, amphetamine and benzylamine) on different mixed-mode/RP stationary phases has been investigated: Primesep phases containing the strong carboxylate functionality (totally ionized over the normal pH range of RP working conditions) and a Hypersil Duet made of a mixture of 50% discrete C18 silica particles and 50% discrete sulfonic acid cation-exchange functionalized particles [14]. Different amounts of acetonitrile/phosphate buffer and different pHs were evaluated choosing low pH values (pH < 3) as optimal to obtain favorable retention. Premisep phases give good peak shapes due to the shielding of silanols by embedded ionic groups. Moreover, an additional and significant advantage of these phases lies in the increase in loadability for basic compounds. This behavior is amenable in the purification step of basic compounds.
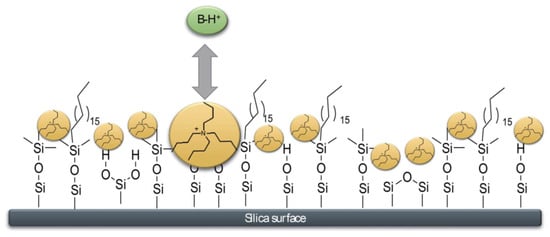
3. Dynamic ERRP-HPLC
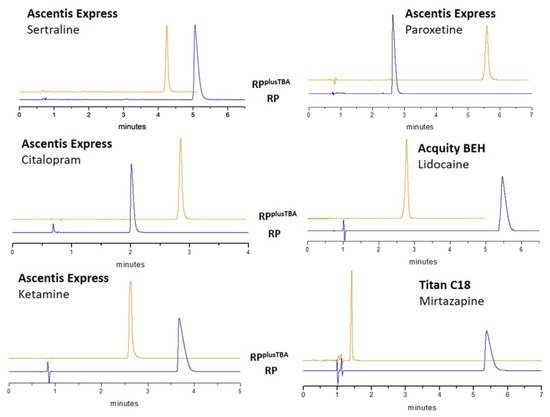
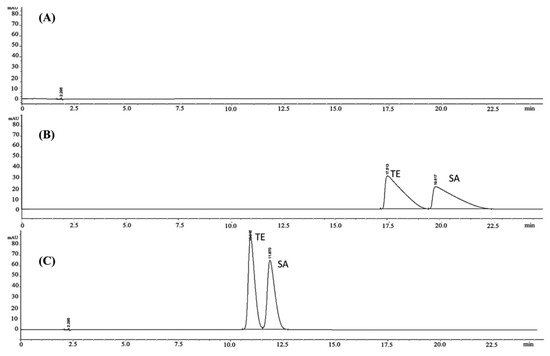
4. Conclusions
References
- Manallack, D.T. The acid–base profile of a contemporary set of drugs: Implications for drug discovery. SAR QSAR Environ. Res. 2009, 20, 611–655.
- Manallack, D.T. The pKa Distribution of Drugs: Application to Drug Discovery. Perspect. Med. Chem. 2007, 1, 25–38.
- McCalley, D.T. The challenges of the analysis of basic compounds by high performance liquid chromatography: Some possi-ble approaches for improved separations. J. Chromatogr. A 2010, 1217, 858–880.
- Galea, C.; Mangelings, D.; Vander-Heyden, Y. Characterization and classification of stationary phases in HPLC and SFC–a review. Anal. Chim. Acta 2015, 886, 1–15.
- Poole, C.F. Chromatographic test methods for characterizing alkylsiloxane-bonded silica columns for reversed-phase liquid chromatography. J. Chromatogr. B 2018, 1092, 207–219.
- Wyndham, K.D.; O’Gara, J.E.; Walter, T.H.; Glose, K.H.; Lawrence, N.L.; Alden, B.A.; Izzo, G.S.; Hudalla, C.J.; Iraneta, P.C. Characterization and Evaluation of C18HPLC Stationary Phases Based on Ethyl-Bridged Hybrid Organic/Inorganic Particles. Anal. Chem. 2003, 75, 6781–6788.
- Pesek, J.J.; Matyska, M.T. Hydride-based silica stationary phases for HPLC: Fundamental properties and applications. J. Sep. Sci. 2005, 28, 1845–1854.
- Grumbach, E.S.; Diehl, D.M.; Neue, U.D. The application of novel 1.7micron ethylene bridged hybrid particles for hydro-philic interaction chromatography. J. Sep. Sci. 2008, 31, 1511–1518.
- Claessens, H.A. Trends and progress in the characterization of stationary phases for reversed-phase liquid chromatography. TrAC Trends Anal. Chem. 2001, 20, 563–583.
- Alpert, A.J. Electrostatic Repulsion Hydrophilic Interaction Chromatography for Isocratic Separation of Charged Solutes and Selective Isolation of Phosphopeptides. Anal. Chem. 2008, 80, 62–76.
- Hao, P.; Ren, Y.; Dutta, B.; Sze, S.K. Comparative evaluation of electrostatic repulsion–hydrophilic interaction chromatography (ERLIC) and high-pH reversed phase (Hp-RP) chromatography in profiling of rat kidney proteome. J. Proteom. 2013, 82, 254–262.
- Gritti, F.; Guiochon, G. Adsorption behaviors of neutral and ionizable compounds on hybrid stationary phases in the absence (BEH-C18) and the presence (CSH-C18) of immobile surface charges. J. Chromatogr. A 2013, 1282, 58–71.
- Davies, N.H.; Euerby, M.R.; McCalley, D.V. A study of retention and overloading of basic compounds with mixed-mode reversed-phase/cation-exchange columns in high performance liquid chromatography. J. Chromatogr. A 2007, 1138, 65–72.
- Iraneta, P.C.; Wyndham, K.D.; McCabe, D.R.; Walter, T.H. A Review of Waters Hybrid Particle Technology; Part 3; Waters Corporation: Milford, CT, USA, 2010.
- Liu, Q.; Liang, T.; Li, K.; Ke, Y.; Jin, Y.; Liang, X. Preparation of a stationary phase with quaternary ammonium embedded group for selective separation of alkaloids based on ion-exclusion interaction. J. Sep. Sci. 2012, 35, 2685–2692.
- Progent, F.; Taverna, M.; Banco, A.; Tchapla, A.; Smadja, C. Chromatographic behaviour of peptides on a mixed-mode sta-tionary phase with an embedded charged group by capillary electrochromatography and high-performance liquid chroma-tography. J. Chromatogr. A 2006, 1136, 221–225.
- Li, Y.; Yang, J.; Jin, J.; Sun, X.; Wang, L.; Chen, J. New reversed-phase/anion-exchange/hydrophilic interaction mixed-mode stationary phase based on dendritic polymer-modified porous silica. J. Chromatogr. A 2014, 1337, 133–139.
- Qiao, L.; Wang, S.; Li, H.; Shan, Y.; Dou, A.; Shi, X.; Xu, G. A novel surface-confined glucaminium-based ionic liquid sta-tionary phase for hydrophilic interaction/anion-exchange mixed-mode chromatography. J. Chromatogr. A 2014, 1360, 240–247.
- Wang, Q.; Baker, G.A.; Baker, S.N.; Colón, L.A. Surface confined ionic liquid as a stationary phase for HPLC. Analyst 2006, 131, 1000–1005.
- Li, Y.; Feng, Y.; Chen, T.; Zhang, H. Imidazoline type stationary phase for hydrophilic interaction chromatography and re-versed-phase liquid chromatography. J Chromatogr. A 2011, 1218, 5987–5994.
- Abbood, A.; Smadja, C.; Herrenknecht, C.; Alahmad, Y.; Tchapla, A.; Taverna, M. Retention mechanism of peptides on a sta-tionary phase embedded with a quaternary ammonium group: A liquid chromatography study. J. Chromatogr. A 2009, 1216, 3244–3251.
- Makarov, A.; LoBrutto, R.; Kazakevich, Y. Liophilic Mobile Phase Additives in Reversed Phase HPLC. J. Liq. Chromatogr. Relat. Technol. 2008, 31, 1533–1567.
- Bartha, A.; Vigh, G.; Billiet, A.H.; de Galan, L. Studies in reversed-phase ion-pair chromatography: IV. The role of the chain length of the pairing ion. J. Chromatogr. A 1984, 303, 29–38.
- Bartha, A.; Vigh, G. Studies in reversed-phase ion-pair chromatography: V. Simultaneous effects of the eluent concentration of the inorganic counter ion and the surface concentration of the pairing ion. J. Chromatogr. A 1987, 396, 503–509.
- Sokolowski, A. Zone formation in Ion-Pair HPLC. I. Effects of adsorption of organic ions on established column equilibria. Chromatographia 1986, 22, 168–176.
- Sokolowski, A. Zone formation in Ion-Pair HPLC. II. System Peak retention and effects of desorption of organic ions on es-tablished column equilibria. Chromatographia 1986, 22, 177–182.
- Manetto, S.; Mazzoccanti, G.; Ciogli, A.; Villani, C.; Gasparrini, F. Ultra-high performance separation of basic compounds on reversed-phase columns packed with fully/superficially porous silica and hybrid particles by using ultraviolet transparent hydrophobic cationic additives. J. Sep. Sci. 2020, 43, 1653–1662.
- Ubeda-Torres, M.T.; Ortiz-Bolsico, C.; García-Alvarez-Coque, M.C.; Ruiz-Angel, M.J. Gaining insight in the behavior of im-idazolium-based ionic liquids as additives in reversed-phase liquid chromatography for the analysis of basic compounds. J. Chromatogr. A 2015, 1380, 96–103.
- Calabuig-Hernández, S.; García-Alvarez-Coque, M.C.; Ruiz-Angel, M.J. Performance of amines as silanol suppressors in reversed-phase liquid chromatography. J. Chromatogr. A 2016, 1465, 98–106.
- Mai, X.-L.; Choi, Y.; Truong, Q.-K.; Nguyen, T.-N.-V.; Han, S.B.; Kim, K.H. Alternative chromatographic method for the assay test of terbutaline and salbutamol using ionic liquid assisted aqueous mobile phase. Anal. Sci. Technol. 2020, 33, 169–176.
- Mazzoccanti, G.; Manetto, S.; Bassan, M.; Foschini, A.; Orlandin, A.; Ricci, A.; Cabri, W.; Ismail, O.H.; Catani, M.; Cavazzi-ni, A.; et al. Boosting basic-peptide separation through dynamic electrostatic-repulsion reversed-phase (d-ERRP) liq-uid chromatography. RSC Adv. 2020, 10, 12604–12610.
- Buszewska-Forajta, M.; Markuszewski, M.J.; Kaliszan, R. Free silanols and ionic liquids as their suppressors in liquid chro-matography. J. Chromatogr. A 2018, 1559, 17–43.
- Berthod, A.; Ruiz-Angel, M.J.; Huguet, S. Nonmolecular solvents in separation methods: Dual nature of room temperature ionic liquids. Anal. Chem. 2005, 77, 4071–4080.




